Submitted:
23 April 2026
Posted:
24 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Information Sources and Search Strategy
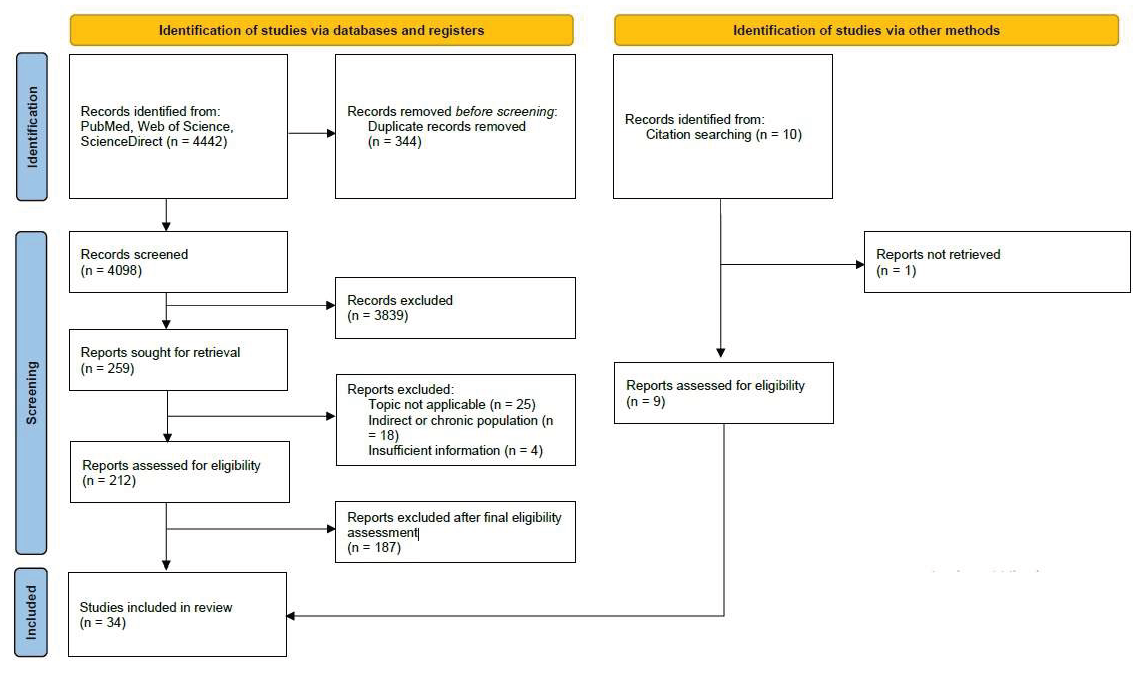
2.3. Study Selection
2.4. Data Extraction
2.5. Evidence Synthesis and Mapping
2.6. Transparency and Data Availability
3. Results
3.1. Characteristics of the Included Studies
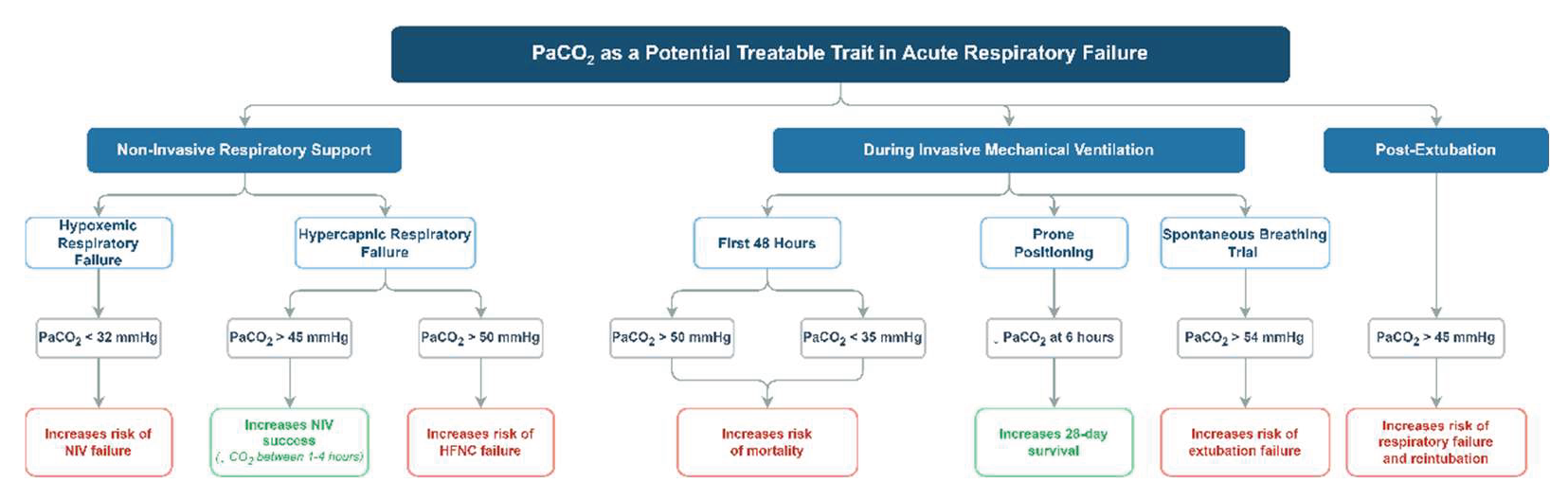
3.2. Mapping of Evidence by Clinical Domain
3.2.1. Pre-Intubation and Early Acute Presentation
3.2.2. Non-Invasive Respiratory Support
3.2.3. Invasive Mechanical Ventilation and CO2 Dysfunction in the ICU
3.2.4. Ventilatory Weaning and Post-Extubation
4. Discussion
4.1. PaCO2 in Hypoxemic Respiratory Failure
4.2. PaCO2 in Hypercapnic Respiratory Failure
4.3. PaCO2 in Patients on Invasive Mechanical Ventilation
4.4. PaCO2 in Extubation and Post-Extubation Outcomes
4.5. Limitations
4.6. Clinical Implications and Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kempker, J.A.; Abril, M.K.; Chen, Y.; Kramer, M.R.; Waller, L.A.; Martin, G.S. The Epidemiology of Respiratory Failure in the United States 2002–2017: A Serial Cross-Sectional Study. Crit. Care Explor. 2020, 2, e0128, . [CrossRef]
- Chen, L.; Rackley, C.R. Diagnosis and Epidemiology of Acute Respiratory Failure. Crit. Care Clin. 2024, 40, 221–233, . [CrossRef]
- McDonald, V.M.; Fingleton, J.; Agusti, A.; Hiles, S.A.; Clark, V.L.; Holland, A.E.; Marks, G.B.; Bardin, P.P.; Beasley, R.; Pavord, I.D.; et al. Treatable Traits: A New Paradigm for 21st Century Management of Chronic Airway Diseases: Treatable Traits Down Under International Workshop Report. Eur. Respir. J. 2019, 53, 1802058, . [CrossRef]
- Agusti, A.; Bel, E.; Thomas, M.; Vogelmeier, C.; Brusselle, G.; Holgate, S.; Humbert, M.; Jones, P.; Gibson, P.G.; Vestbo, J.; et al. Treatable Traits: Toward Precision Medicine of Chronic Airway Diseases. Eur. Respir. J. 2016, 47, 410–419, . [CrossRef]
- Agustí, A.; Bafadhel, M.; Beasley, R.; Bel, E.H.; Faner, R.; Gibson, P.G.; Louis, R.; McDonald, V.M.; Sterk, P.J.; Thomas, M.; et al. Precision Medicine in Airway Diseases: Moving to Clinical Practice. Eur. Respir. J. 2017, 50, 1701655, . [CrossRef]
- Jonkman, A.H.; de Vries, H.J.; Heunks, L.M.A. Physiology of the Respiratory Drive in ICU Patients: Implications for Diagnosis and Treatment. Crit. Care 2020, 24, 104, . [CrossRef]
- Osorio-Rodríguez, E.; Correa-Guerrero, J.; Rodelo-Barrios, D.; Bonilla-Llanos, M.; Rebolledo-Maldonado, C.; Patiño-Patiño, J.; Viera-Torres, J.; Arias-Gómez, M.; Gracia-Ordoñez, M.; González-Betancur, D.; et al. Hypercapnia as a Double-Edged Modulator of Innate Immunity and Alveolar Epithelial Repair: A PRISMA-ScR Scoping Review. Int. J. Mol. Sci. 2025, 26, 9622, . [CrossRef]
- Kato, T.; Kasai, T.; Suda, S.; Sato, A.; Ishiwata, S.; Yatsu, S.; Matsumoto, H.; Shitara, J.; Shimizu, M.; Murata, A.; et al. Prognostic Effects of Arterial Carbon Dioxide Levels in Patients Hospitalized into the Cardiac Intensive Care Unit for Acute Heart Failure. Eur. Hear. Journal. Acute Cardiovasc. Care 2021, 10, 497–502, . [CrossRef]
- Carrillo-Aleman, L.; Carrasco-Gónzalez, E.; Araújo, M.J.; Guia, M.; Alonso-Fernández, N.; Renedo-Villarroya, A.; López-Gómez, L.; Higon-Cañigral, A.; Sanchez-Nieto, J.M.; Carrillo-Alcaraz, A. Is Hypocapnia a Risk Factor for Non-Invasive Ventilation Failure in Cardiogenic Acute Pulmonary Edema? J. Crit. Care 2022, 69, 153991, . [CrossRef]
- Xu, X.; Ma, M.; Min, Y.; Hu, W.; Bai, L.; Duan, J. PaCO2 Is Nonlinearly Associated with NIV Failure in Patients with Hypoxemic Respiratory Failure. BMC Pulm. Med. 2024, 24, 228, . [CrossRef]
- Tiruvoipati, R.; Pilcher, D.; Buscher, H.; Botha, J.; Bailey, M. Effects of Hypercapnia and Hypercapnic Acidosis on Hospital Mortality in Mechanically Ventilated Patients*. Crit. Care Med. 2017, 45, e649–e656, . [CrossRef]
- Nin, N.; Muriel, A.; Peñuelas, O.; Brochard, L.; Lorente, J.A.; Ferguson, N.D.; Raymondos, K.; Ríos, F.; Violi, D.A.; Thille, A.W.; et al. Severe Hypercapnia and Outcome of Mechanically Ventilated Patients with Moderate or Severe Acute Respiratory Distress Syndrome. Intensive Care Med. 2017, 43, 200–208, . [CrossRef]
- Ferrer, M.; Sellarés, J.; Valencia, M.; Carrillo, A.; Gonzalez, G.; Badia, J.R.; Nicolas, J.M.; Torres, A. Non-Invasive Ventilation after Extubation in Hypercapnic Patients with Chronic Respiratory Disorders: Randomised Controlled Trial. Lancet 2009, 374, 1082–1088, . [CrossRef]
- Algarin-Lara, H.; Osorio-Rodríguez, E.; Patiño-Patiño, J.; Solano-Ropero, J.; Rodado-Villa, R. Hipercapnia Refractaria En Paciente Con Síndrome de Obesidad-Hipoventilación Maligno Y COVID-19. Reporte de Caso Y Propuesta de Manejo. Acta Colomb. Cuid. Intensivo 2022, 22, S117–S122, . [CrossRef]
- Peters, M.D.J.; Marnie, C.; Tricco, A.C.; Pollock, D.; Munn, Z.; Alexander, L.; McInerney, P.; Godfrey, C.M.; Khalil, H. Updated Methodological Guidance for the Conduct of Scoping Reviews. JBI Evid. Synth. 2020, 18, 2119–2126, . [CrossRef]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473, . [CrossRef]
- Piper, A.J.; Sullivan, C.E. Effects of Short-Term NIPPV in the Treatment of Patients With Severe Obstructive Sleep Apnea and Hypercapnia. Chest 1994, 105, 434–440, . [CrossRef]
- Rabec, C.; Merati, M.; Baudouin, N.; Foucher, P.; Ulukavac, T.; Reybet-Degat, O. [Management of Obesity and Respiratory Insufficiency. The Value of Dual-Level Pressure Nasal Ventilation]. Rev. Mal. Respir. 1998, 15, 269–278.
- Hilbert, G.; Gruson, D.; Portel, L.; Gbikpi-Benissan, G.; Cardinaud, J. Noninvasive Pressure Support Ventilation in COPD Patients with Postextubation Hypercapnic Respiratory Insufficiency. Eur. Respir. J. 1998, 11, 1349–1353, . [CrossRef]
- Plant, P.; Owen, J.; Elliott, M. Early Use of Non-Invasive Ventilation for Acute Exacerbations of Chronic Obstructive Pulmonary Disease on General Respiratory Wards: A Multicentre Randomised Controlled Trial. Lancet 2000, 355, 1931–1935, . [CrossRef]
- Gattinoni, L.; Vagginelli, F.; Carlesso, E.; Taccone, P.; Conte, V.; Chiumello, D.; Valenza, F.; Caironi, P.; Pesenti, A. Decrease in Paco2 with Prone Position Is Predictive of Improved Outcome in Acute Respiratory Distress Syndrome*. Crit. Care Med. 2003, 31, 2727–2733, . [CrossRef]
- Nava, S.; Carbone, G.; DiBattista, N.; Bellone, A.; Baiardi, P.; Cosentini, R.; Marenco, M.; Giostra, F.; Borasi, G.; Groff, P. Noninvasive Ventilation in Cardiogenic Pulmonary Edema. Am. J. Respir. Crit. Care Med. 2003, 168, 1432–1437, . [CrossRef]
- Díaz, G.G.; Alcaraz, A.C.; Talavera, J.C.P.; Pèrez, P.J.; Rodriguez, A.E.; Cordoba, F.G.; Hill, N.S. Noninvasive Positive-Pressure Ventilation To Treat Hypercapnic Coma Secondary to Respiratory Failure. Chest 2005, 127, 952–960, . [CrossRef]
- Ferrer, M.; Valencia, M.; Nicolas, J.M.; Bernadich, O.; Badia, J.R.; Torres, A. Early Noninvasive Ventilation Averts Extubation Failure in Patients at Risk. Am. J. Respir. Crit. Care Med. 2006, 173, 164–170, . [CrossRef]
- González, Á.O.; Romero, G.P.-B.; Ormaechea, I.F.; Flores, R.C.; de Frutos, N.C.; Mangado, N.G. Evolution of Patients With Chronic Obstructive Pulmonary Disease, Obesity Hypoventilation Syndrome, or Congestive Heart Failure Undergoing Noninvasive Ventilation in a Respiratory Monitoring Unit. Arch. Bronconeumol. ((English Ed. 2006, 42, 423–429, . [CrossRef]
- Duarte, A.G.; Justino, E.; Bigler, T.; Grady, J. Outcomes of Morbidly Obese Patients Requiring Mechanical Ventilation for Acute Respiratory Failure*. Crit. Care Med. 2007, 35, 732–737, . [CrossRef]
- Sellares, J.; Ferrer, M.; Cano, E.; Loureiro, H.; Valencia, M.; Torres, A. Predictors of Prolonged Weaning and Survival during Ventilator Weaning in a Respiratory ICU. Intensive Care Med. 2011, 37, 775–784, . [CrossRef]
- Girault, C.; Bubenheim, M.; Abroug, F.; Diehl, J.L.; Elatrous, S.; Beuret, P.; Richecoeur, J.; L’Her, E.; Hilbert, G.; Capellier, G.; et al. Noninvasive Ventilation and Weaning in Patients with Chronic Hypercapnic Respiratory Failure. Am. J. Respir. Crit. Care Med. 2011, 184, 672–679, . [CrossRef]
- Carrillo, A.; Ferrer, M.; Gonzalez-Diaz, G.; Lopez-Martinez, A.; Llamas, N.; Alcazar, M.; Capilla, L.; Torres, A. Noninvasive Ventilation in Acute Hypercapnic Respiratory Failure Caused by Obesity Hypoventilation Syndrome and Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2012, 186, 1279–1285, . [CrossRef]
- Marik, P.E.; Desai, H. Characteristics of Patients With the “Malignant Obesity Hypoventilation Syndrome” Admitted to an ICU. J. Intensive Care Med. 2013, 28, 124–130, . [CrossRef]
- Pu, L.; Zhu, B.; Jiang, L.; Du, B.; Zhu, X.; Li, A.; Li, G.; He, Z.; Chen, W.; Ma, P.; et al. Weaning Critically Ill Patients from Mechanical Ventilation: A Prospective Cohort Study. J. Crit. Care 2015, 30, 862.e7-862.e13, . [CrossRef]
- Çiftci, F.; Çiledağ, A.; Erol, S.; Öz, M.; Acar, D.; Kaya, A. Evaluation of the Feasibility of Average Volume-Assured Pressure Support Ventilation in the Treatment of Acute Hypercapnic Respiratory Failure Associated with Chronic Obstructive Pulmonary Disease: A Pilot Study. J. Crit. Care 2017, 39, 232–237, . [CrossRef]
- Fuller, B.M.; Mohr, N.M.; Drewry, A.M.; Ferguson, I.T.; Trzeciak, S.; Kollef, M.H.; Roberts, B.W. Partial Pressure of Arterial Carbon Dioxide and Survival to Hospital Discharge among Patients Requiring Acute Mechanical Ventilation: A Cohort Study. J. Crit. Care 2017, 41, 29–35, . [CrossRef]
- Sellares, J.; Ferrer, M.; Anton, A.; Loureiro, H.; Bencosme, C.; Alonso, R.; Martinez-Olondris, P.; Sayas, J.; Peñacoba, P.; Torres, A. Discontinuing Noninvasive Ventilation in Severe Chronic Obstructive Pulmonary Disease Exacerbations: A Randomised Controlled Trial. Eur. Respir. J. 2017, 50, 1601448, . [CrossRef]
- Bry, C.; Jaffré, S.; Guyomarc’h, B.; Corne, F.; Chollet, S.; Magnan, A.; Blanc, F.-X. Noninvasive Ventilation in Obese Subjects After Acute Respiratory Failure. Respir. Care 2018, 63, 28–35, . [CrossRef]
- Thille, A.W.; Muller, G.; Gacouin, A.; Coudroy, R.; Decavèle, M.; Sonneville, R.; Beloncle, F.; Girault, C.; Dangers, L.; Lautrette, A.; et al. Effect of Postextubation High-Flow Nasal Oxygen With Noninvasive Ventilation vs High-Flow Nasal Oxygen Alone on Reintubation Among Patients at High Risk of Extubation Failure. JAMA 2019, 322, 1465, . [CrossRef]
- Li, X.-Y.; Tang, X.; Wang, R.; Yuan, X.; Zhao, Y.; Wang, L.; Li, H.-C.; Chu, H.-W.; Li, J.; Mao, W.-P.; et al. High-Flow Nasal Cannula for Chronic Obstructive Pulmonary Disease with Acute Compensated Hypercapnic Respiratory Failure: A Randomized, Controlled Trial. Int. J. Chron. Obstruct. Pulmon. Dis. 2020, Volume 15, 3051–3061, . [CrossRef]
- Madotto, F.; Rezoagli, E.; McNicholas, B.A.; Pham, T.; Slutsky, A.S.; Bellani, G.; Laffey, J.G. Patterns and Impact of Arterial CO2 Management in Patients With Acute Respiratory Distress Syndrome. Chest 2020, 158, 1967–1982, . [CrossRef]
- Tsonas, A.M.; Botta, M.; Horn, J.; Morales-Quinteros, L.; Artigas, A.; Schultz, M.J.; Paulus, F.; Neto, A.S. Clinical Characteristics, Physiological Features, and Outcomes Associated with Hypercapnia in Patients with Acute Hypoxemic Respiratory Failure due to COVID–19---Insights from the PRoVENT–COVID Study. J. Crit. Care 2022, 69, 154022, . [CrossRef]
- Braunsteiner, J.; Castro, L.; Wiessner, C.; Grensemann, J.; Schroeder, M.; Burdelski, C.; Sensen, B.; Kluge, S.; Fischer, M. Association Between Dyscapnia, Ventilatory Variables, and Mortality in Patients With Acute Respiratory Distress Syndrome—A Retrospective Cohort Study. J. Intensive Care Med. 2024, 39, 1099–1108, . [CrossRef]
- Robba, C.; Battaglini, D.; Abbas, A.; Sarrió, E.; Cinotti, R.; Asehnoune, K.; Taccone, F.S.; Rocco, P.R.; Schultz, M.J.; Citerio, G.; et al. Clinical Practice and Effect of Carbon Dioxide on Outcomes in Mechanically Ventilated Acute Brain-Injured Patients: A Secondary Analysis of the ENIO Study. Intensive Care Med. 2024, 50, 234–246, . [CrossRef]
- Tan, D.; Wang, B.; Cao, P.; Wang, Y.; Sun, J.; Geng, P.; Walline, J.H.; Wang, Y.; Wang, C. High Flow Nasal Cannula Oxygen Therapy versus Non-Invasive Ventilation for Acute Exacerbations of Chronic Obstructive Pulmonary Disease with Acute-Moderate Hypercapnic Respiratory Failure: A Randomized Controlled Non-Inferiority Trial. Crit. Care 2024, 28, 250, . [CrossRef]
- Villar, J.; González-Martín, J.M.; Fernández, C.; Soler, J.A.; Ambrós, A.; Pita-García, L.; Fernández, L.; Ferrando, C.; Arocas, B.; González-Vaquero, M.; et al. Predicting the Length of Mechanical Ventilation in Acute Respiratory Disease Syndrome Using Machine Learning: The PIONEER Study. J. Clin. Med. 2024, 13, 1811, . [CrossRef]
- Rusnak, J.; Schupp, T.; Weidner, K.; Ruka, M.; Egner-Walter, S.; Schmitt, A.; Akin, M.; Mashayekhi, K.; Ayoub, M.; Behnes, M.; et al. Partial Arterial Carbon Dioxide and Oxygen Pressure in Patients with Cardiogenic Shock. Intern. Emerg. Med. 2025, 20, 1077–1086, . [CrossRef]
- Brochard, L.; Slutsky, A.; Pesenti, A. Mechanical Ventilation to Minimize Progression of Lung Injury in Acute Respiratory Failure. Am. J. Respir. Crit. Care Med. 2017, 195, 438–442, . [CrossRef]
- Helms, J.; Catoire, P.; Abensur Vuillaume, L.; Bannelier, H.; Douillet, D.; Dupuis, C.; Federici, L.; Jezequel, M.; Jozwiak, M.; Kuteifan, K.; et al. Oxygen Therapy in Acute Hypoxemic Respiratory Failure: Guidelines from the SRLF-SFMU Consensus Conference. Ann. Intensive Care 2024, 14, 140, . [CrossRef]
- Seow, D.; Khor, Y.H.; Khung, S.-W.; Smallwood, D.M.; Ng, Y.; Pascoe, A.; Smallwood, N. High-Flow Nasal Oxygen Therapy Compared with Conventional Oxygen Therapy in Hospitalised Patients with Respiratory Illness: A Systematic Review and Meta-Analysis. BMJ Open Respir. Res. 2024, 11, e002342, . [CrossRef]
- Marjanovic, N.; Piton, M.; Lamarre, J.; Alleyrat, C.; Couvreur, R.; Guenezan, J.; Mimoz, O.; Frat, J.-P. High-Flow Nasal Cannula Oxygen versus Noninvasive Ventilation for the Management of Acute Cardiogenic Pulmonary Edema: A Randomized Controlled Pilot Study. Eur. J. Emerg. Med. 2024, 31, 267–275, . [CrossRef]
- Marjanovic, N.; Couvreur, R.; Lamarre, J.; Piton, M.; Guenezan, J.; Mimoz, O. High-Flow Nasal Cannula Oxygen Therapy versus Noninvasive Ventilation in Acute Respiratory Failure Related to Suspected or Confirmed Acute Heart Failure: A Systematic Review with Meta-Analysis. Eur. J. Emerg. Med. 2024, 31, 388–397, . [CrossRef]
- Masip, J.; Peacock, W.F.; Price, S.; Cullen, L.; Martin-Sanchez, F.J.; Seferovic, P.; Maisel, A.S.; Miro, O.; Filippatos, G.; Vrints, C.; et al. Indications and Practical Approach to Non-Invasive Ventilation in Acute Heart Failure. Eur. Heart J. 2018, 39, 17–25, . [CrossRef]
- Berbenetz, N.; Wang, Y.; Brown, J.; Godfrey, C.; Ahmad, M.; Vital, F.M.; Lambiase, P.; Banerjee, A.; Bakhai, A.; Chong, M. Non-Invasive Positive Pressure Ventilation (CPAP or Bilevel NPPV) for Cardiogenic Pulmonary Oedema. Cochrane Database Syst. Rev. 2019, 2019, . [CrossRef]
- Rochwerg, B.; Brochard, L.; Elliott, M.W.; Hess, D.; Hill, N.S.; Nava, S.; Navalesi, P.; Antonelli, M.; Brozek, J.; Conti, G.; et al. Official ERS/ATS Clinical Practice Guidelines: Noninvasive Ventilation for Acute Respiratory Failure. Eur. Respir. J. 2017, 50, 1602426, . [CrossRef]
- Carteaux, G.; Millán-Guilarte, T.; De Prost, N.; Razazi, K.; Abid, S.; Thille, A.W.; Schortgen, F.; Brochard, L.; Brun-Buisson, C.; Mekontso Dessap, A. Failure of Noninvasive Ventilation for De Novo Acute Hypoxemic Respiratory Failure. Crit. Care Med. 2016, 44, 282–290, . [CrossRef]
- Matthay, M.A. Saving Lives with High-Flow Nasal Oxygen. N. Engl. J. Med. 2015, 372, 2225–2226, . [CrossRef]
- Yoshida, T.; Uchiyama, A.; Matsuura, N.; Mashimo, T.; Fujino, Y. Spontaneous Breathing during Lung-Protective Ventilation in an Experimental Acute Lung Injury Model. Crit. Care Med. 2012, 40, 1578–1585, . [CrossRef]
- Osadnik, C.R.; Tee, V.S.; Carson-Chahhoud, K. V; Picot, J.; Wedzicha, J.A.; Smith, B.J. Non-Invasive Ventilation for the Management of Acute Hypercapnic Respiratory Failure due to Exacerbation of Chronic Obstructive Pulmonary Disease. Cochrane Database Syst. Rev. 2017, 2017, . [CrossRef]
- Contou, D.; Fragnoli, C.; Córdoba-Izquierdo, A.; Boissier, F.; Brun-Buisson, C.; Thille, A.W. Severe but Not Mild Hypercapnia Affects the Outcome in Patients with Severe Cardiogenic Pulmonary Edema Treated by Non-Invasive Ventilation. Ann. Intensive Care 2015, 5, 14, . [CrossRef]
- Masa, J.F.; Utrabo, I.; Gomez de Terreros, J.; Aburto, M.; Esteban, C.; Prats, E.; Núñez, B.; Ortega-González, Á.; Jara-Palomares, L.; Martin-Vicente, M.J.; et al. Noninvasive Ventilation for Severely Acidotic Patients in Respiratory Intermediate Care Units. BMC Pulm. Med. 2016, 16, 97, . [CrossRef]
- Scala, R.; Naldi, M.; Archinucci, I.; Coniglio, G.; Nava, S. Noninvasive Positive Pressure Ventilation in Patients With Acute Exacerbations of COPD and Varying Levels of Consciousness. Chest 2005, 128, 1657–1666, . [CrossRef]
- Xu, C.; Yang, F.; Wang, Q.; Gao, W. Comparison of High Flow Nasal Therapy with Non-Invasive Ventilation and Conventional Oxygen Therapy for Acute Hypercapnic Respiratory Failure: A Meta-Analysis of Randomized Controlled Trials. Int. J. Chron. Obstruct. Pulmon. Dis. 2023, Volume 18, 955–973, . [CrossRef]
- Gendreau, S.; Geri, G.; Pham, T.; Vieillard-Baron, A.; Mekontso Dessap, A. The Role of Acute Hypercapnia on Mortality and Short-Term Physiology in Patients Mechanically Ventilated for ARDS: A Systematic Review and Meta-Analysis. Intensive Care Med. 2022, 48, 517–534, . [CrossRef]
- Serpa Neto, A.; Deliberato, R.O.; Johnson, A.E.W.; Bos, L.D.; Amorim, P.; Pereira, S.M.; Cazati, D.C.; Cordioli, R.L.; Correa, T.D.; Pollard, T.J.; et al. Mechanical Power of Ventilation Is Associated with Mortality in Critically Ill Patients: An Analysis of Patients in Two Observational Cohorts. Intensive Care Med. 2018, 44, 1914–1922, . [CrossRef]
- Coppola, S.; Caccioppola, A.; Froio, S.; Formenti, P.; De Giorgis, V.; Galanti, V.; Consonni, D.; Chiumello, D. Effect of Mechanical Power on Intensive Care Mortality in ARDS Patients. Crit. Care 2020, 24, 246, . [CrossRef]
- Monteiro, A.C.C.; Vangala, S.; Wick, K.D.; Delucchi, K.L.; Siegel, E.R.; Thompson, B.T.; Liu, K.D.; Sapru, A.; Sinha, P.; Matthay, M.A. The Prognostic Value of Early Measures of the Ventilatory Ratio in the ARDS ROSE Trial. Crit. Care 2022, 26, 297, . [CrossRef]
- Carney, N.; Totten, A.M.; O’Reilly, C.; Ullman, J.S.; Hawryluk, G.W.J.; Bell, M.J.; Bratton, S.L.; Chesnut, R.; Harris, O.A.; Kissoon, N.; et al. Guidelines for the Management of Severe Traumatic Brain Injury, Fourth Edition. Neurosurgery 2017, 80, 6–15, . [CrossRef]
- Grasselli, G.; Calfee, C.S.; Camporota, L.; Poole, D.; Amato, M.B.P.; Antonelli, M.; Arabi, Y.M.; Baroncelli, F.; Beitler, J.R.; Bellani, G.; et al. ESICM Guidelines on Acute Respiratory Distress Syndrome: Definition, Phenotyping and Respiratory Support Strategies. Intensive Care Med. 2023, 49, 727–759, . [CrossRef]
- Qadir, N.; Sahetya, S.; Munshi, L.; Summers, C.; Abrams, D.; Beitler, J.; Bellani, G.; Brower, R.G.; Burry, L.; Chen, J.-T.; et al. An Update on Management of Adult Patients with Acute Respiratory Distress Syndrome: An Official American Thoracic Society Clinical Practice Guideline. Am. J. Respir. Crit. Care Med. 2024, 209, 24–36, . [CrossRef]
- Charron, C.; Repesse, X.; Bouferrache, K.; Bodson, L.; Castro, S.; Page, B.; Jardin, F.; Vieillard-Baron, A. PaCO2 and Alveolar Dead Space Are More Relevant than PaO2/FiO2 Ratio in Monitoring the Respiratory Response to Prone Position in ARDS Patients: A Physiological Study. Crit. Care 2011, 15, R175, . [CrossRef]
- Nava, S.; Gregoretti, C.; Fanfulla, F.; Squadrone, E.; Grassi, M.; Carlucci, A.; Beltrame, F.; Navalesi, P. Noninvasive Ventilation to Prevent Respiratory Failure after Extubation in High-Risk Patients*. Crit. Care Med. 2005, 33, 2465–2470, . [CrossRef]
- McNamee, J.J.; Gillies, M.A.; Barrett, N.A.; Perkins, G.D.; Tunnicliffe, W.; Young, D.; Bentley, A.; Harrison, D.A.; Brodie, D.; Boyle, A.J.; et al. Effect of Lower Tidal Volume Ventilation Facilitated by Extracorporeal Carbon Dioxide Removal vs Standard Care Ventilation on 90-Day Mortality in Patients With Acute Hypoxemic Respiratory Failure. JAMA 2021, 326, 1013, . [CrossRef]
- Zhou, Z.; Li, Z.; Liu, C.; Wang, F.; Zhang, L.; Fu, P. Extracorporeal Carbon Dioxide Removal for Patients with Acute Respiratory Failure: A Systematic Review and Meta-Analysis. Ann. Med. 2023, 55, 746–759, . [CrossRef]
- Stommel, A.-M.; Herkner, H.; Kienbacher, C.L.; Wildner, B.; Hermann, A.; Staudinger, T. Effects of Extracorporeal CO2 Removal on Gas Exchange and Ventilator Settings: A Systematic Review and Meta-Analysis. Crit. Care 2024, 28, 146, . [CrossRef]
| Citation | Year | Country | Type of study |
| Piper AJ [17] | 1994 | Australia | Retrospective observational study |
| Rabec C [18] | 1998 | France | Prospective observational study |
| Hilbert G [19] | 1998 | France | Observational case–control study |
| Plant PK [20] | 2000 | United Kingdom | Multicenter randomized controlled trial |
| Gattinoni L [21] | 2003 | Italy | Retrospective analysis of a randomized controlled trial |
| Nava S [22] | 2003 | Italy | Multicenter randomized controlled trial |
| Diaz GG [23] | 2005 | Spain | Prospective, open-label, uncontrolled observational study |
| Ferrer M [24] | 2006 | Spain | Prospective observational study |
| Ortega González [25] | 2006 | Spain | Prospective observational study |
| Duarte AG [26] | 2007 | USA | Retrospective observational study |
| Ferrer M [13] | 2009 | Spain | Randomized clinical trial |
| Sellarés J [27] | 2011 | Spain | Prospective cohort study |
| Girault C [28] | 2011 | France | Randomized clinical trial (multicenter) |
| Carrillo A [29] | 2012 | Spain | Prospective observational study |
| Marik PE [30] | 2013 | USA | Retrospective observational study |
| Pu L [31] | 2015 | China | Multicenter prospective cohort study |
| Tiruvoipati R [11] | 2017 | Australia/New Zealand | Retrospective multicenter observational study |
| Nin N [12] | 2017 | Multinational* | Prospective cohort study |
| Çiftci F [32] | 2017 | Turkey | Prospective observational study |
| Fuller BM [33] | 2017 | USA | Prospective observational study |
| Sellarés J [34] | 2017 | Spain | Randomized controlled trial |
| Bry C [35] | 2018 | France | Retrospective observational study |
| Thille AW [36] | 2019 | France | Randomized clinical trial |
| Li X [37] | 2020 | China | Randomized clinical trial |
| Madotto F [38] | 2020 | Multinational* | Multicenter observational study |
| Kato T [8] | 2021 | Japan | Retrospective observational study |
| Carrillo-Alemán L [9] | 2022 | Spain | Retrospective observational study |
| Tsonas AM [39] | 2022 | Netherlands | Retrospective multicenter observational study |
| Xu X [10] | 2024 | China | Retrospective observational study |
| Braunsteiner J [40] | 2024 | Germany | Retrospective observational study |
| Robba C [41] | 2024 | Multinational* | Multicenter prospective observational study |
| Tan D [42] | 2024 | China | Randomized controlled trial |
| Villar J [43] | 2024 | Multinational* | Prospective cohort study |
| Rusnak J [44] | 2025 | Germany | Prospective observational study |
| Citation | Design | n | Context | PaCO₂ definition/strata | Key outcome |
| Pre-intubation and early acute presentation | |||||
| Kato T et al., 2021 [8] |
Retrospective observational | 435 | Acute heart failure | PaCO₂ as continuous; cut-offs 31 and 40 mmHg | PaCO₂ <31 associated with higher mortality (HR 1.71) |
| Carrillo-Alemán L et al., 2022 [9] |
Retrospective observational | 1,138 | Cardiogenic pulmonary edema (NIV) | Hypocapnia/eucapnia/hypercapnia (hypercapnia threshold NR) | Hypocapnia: higher NIV failure and in-hospital mortality |
| Xu X et al., 2024 [10] |
Retrospective observational | 1,029 | Hypoxemic respiratory failure (NIV) | Hypocapnia ≤32 mmHg | Hypocapnia: higher NIV failure (adjusted HR 1.23) |
| Rusnak J et al., 2025 [44] |
Prospective observational | 238 | Cardiogenic shock | Hypocapnia ≤33 mmHg; hypercapnia >48.13 mmHg | Hypocapnia: higher 30-day mortality; hypercapnia not associated |
| Non-invasive respiratory support: NIV/HFNC | |||||
| NIV in hypercapnic acute respiratory failure (COPD / OHS / OSA–OHS) | |||||
| Piper AJ et al., 1994 [17] |
Retrospective observational | 13 | Obesity, BMI >35; nocturnal nasal ventilation | PaCO₂ >45 mmHg | NIV success ~64–69% |
| Rabec C et al., 1998 [18] |
Prospective observational | 41 | Sleep apnea + respiratory acidosis (NIV) | pH <7.35; PaCO₂ >45 mmHg | Intubation avoided in 95% |
| Diaz GG et al., 2005 [23] |
Prospective observational (open-label) | 681 | Hypercapnic coma (NIV) | PaCO₂ >45 mmHg | NIV success 80% |
| Ortega González et al., 2006 [25] | Prospective observational (open-label) | 53 | COPD, OHS, AHF (NIV) | pH >7.25; PaCO₂ >45 mmHg | At 3 h: pH ↑ and PaCO₂ ↓ |
| Duarte AG et al., 2007 [26] |
Retrospective observational | 50 | Morbid obesity, BMI >35 kg/m² (NIV) | PaCO₂ >50 mmHg | NIV success 64% |
| Çiftci F et al., 2007 [32] |
Prospective observational | 106 | Hypercapnic respiratory failure (assured volume PS) | pH <7.35; PaCO₂ >45 mmHg | NIV success 76.4% |
| Carrillo A et al., 2012 [29] |
Prospective observational | 716 | ARF episodes due to OHS and COPD | pH <7.35; PaCO₂ >45 mmHg | NIV success 88.4% |
| Marik PE et al., 2013 [30] |
Prospective observational | 61 | OHS, BMI >40 kg/m² (BiPAP) | PaCO₂ >45 mmHg | 37.7% progressed to IMV |
| Sellarés J et al., 2017 [34] |
Randomized clinical trial | 120 | NIV prolongation after ARF resolution | pH <7.35; PaCO₂ >45 mmHg | ARF recurrence 13% |
| Bry C et al., 2018 [35] |
Retrospective observational | 53 | BMI >30; long-term NIV after ARF hospitalization | PaCO₂ >45 mmHg | NIV success 90% |
| HFNC vs NIV trials/strategies in hypercapnic COPD | |||||
| Plant PK et al., 2000 [20] |
Multicenter randomized clinical trial | 236 | AECOPD: NIV | pH 7.25–7.35; PaCO₂ >45 mmHg | 15% progressed to IMV |
| Li X et al., 2020 [37] |
Randomized clinical trial | 320 | COPD: HFNC | pH >7.35; PaCO₂ >45 mmHg | 19% progressed to IMV |
| Tan D et al., 2024 [42] |
Randomized clinical trial | 225 | COPD: HFNC vs NIV | pH 7.25–7.35; PaCO₂ >50 mmHg | IMV: 25.7% (HFNC) vs 14.3% (NIV) |
| IMV and ICU CO₂ derangements | |||||
| Gattinoni L et al., 2003 [21] |
Retrospective analysis of an RCT | 225 | ALI/ARDS (prone) | Prone responders: PaCO₂ decrease ≥ 1 mmHg after 6 h | Responders had higher 28-day survival |
| Tiruvoipati R et al., 2017 [11] |
Retrospective multicenter observational | 252,812 | IMV patients | Hypercapnia >45 mmHg; hypercapnic acidosis: pH <7.35 | Hypercapnia/hypercapnic acidosis associated with higher mortality |
| Nin N et al., 2017 [12] |
Prospective non-interventional cohort | 889 | ARDS <48 h | Hypercapnia >40 mmHg; >50 mmHg emphasized | PaCO₂ >50 is associated with higher mortality (first 48 h) |
| Fuller BM et al., 2017 [33] |
Prospective observational | 1,491 | IMV (first 48 h) | Hypocapnia <35 mmHg; hypercapnia >45 mmHg | Higher survival with hypercapnia vs hypocapnia |
| Madotto F et al., 2020 [38] |
Multicenter observational | 2,813 | Early ARDS | Hypocapnia <35 mmHg; hypercapnia >45 mmHg | Mortality: hypercapnia 36%; hypocapnia 38.1% |
| Tsonas AM et al., 2022 [39] |
Retrospective multicenter observational | 824 | COVID-19 on IMV | Hypercapnia >45 mmHg | Longer MV duration and ICU/hospital LOS |
| Braunsteiner J et al., 2024 [40] |
Retrospective observational | 435 | Mechanical power + dyscapnia | Hypocapnia <35 mmHg; hypercapnia >50 mmHg | Hypocapnia is associated with higher mortality; hypercapnia>50 is not associated |
| Robba C et al., 2024 [41] |
Multicenter prospective observational | 1,476 | Acute brain injury | Hypocapnia <35 mmHg; hypercapnia >45 mmHg | Both hypo- and hypercapnia associated with higher mortality |
| Villar J et al., 2024 [43] |
Prospective cohort | 253 | Moderate-to-severe ARDS | Hypercapnia >45 mmHg; early PaCO₂ changes | Early PaCO₂ changes predict MV duration >14 days |
| Weaning and post-extubation | |||||
| Predictors of prolonged weaning/extubation failure | |||||
| Ferrer M et al., 2006 [24] |
Prospective observational | 162 | Weaning | Hypercapnia >45 mmHg | Higher orotracheal re-intubation |
| Sellarés J et al., 2011 [27] |
Prospective cohort | 181 | SBT/weaning | Hypercapnia >45 mmHg; ≥54 mmHg | Associated with prolonged weaning and extubation failure |
| Pu L et al., 2015 [31] |
Multicenter prospective cohort | 343 | First SBT | Hypercapnia >50 mmHg | Associated with prolonged mechanical ventilation |
| Post-extubation interventions (NIV/HFNC) in hypercapnic patients | |||||
| Hilbert G et al., 1998 [19] |
Case–control observational | 30 | COPD post-extubation | PaCO₂ ↑ 20% + pH <7.35 | NIV reduced re-intubation |
| Nava S et al., 2003 [22] |
Multicenter randomized clinical trial | 122 | NIV ≥8 h/day for 48 h | Hypercapnia >45 mmHg | NIV prevented post-extubation ventilatory failure |
| Ferrer M et al., 2009 [13] |
Randomized clinical trial | 164 | NIV vs standard oxygen | Hypercapnia >45 mmHg | NIV reduced ventilatory failure and 90-day mortality |
| Girault C et al., 2011 [28] |
Randomized clinical trial | 388 | NIV as a bridge to wean from IMV | Hypercapnia >45 mmHg or PaCO₂ ↑ >10% vs pre-extubation | NIV reduced re-intubation |
| Thille AW et al., 2019 [36] |
Prospective observational | NR | Post-extubation HFNC or NIV | Hypercapnia >45 mmHg | Lower re-intubation rate |
| Clinical context | PaCO₂ pattern | Main role | Practical message |
| Acute hypoxemia / pre-intubation | Low or low-normal | Prognostic / stratification | Hypocapnia may suggest higher respiratory drive and greater risk of non-invasive support failure, but it should not be used alone to determine support modality. |
| Cardiogenic pulmonary edema / acute heart failure / cardiogenic shock | Often low | Prognostic / monitoring | Hypocapnia has been associated with worse outcomes in some studies, but it is not, by itself, a treatment target. |
| Acute hypercapnic respiratory failure (COPD/OHS) | >45 mmHg, often with acidemia | Modality selection / monitoring | This is the clearest setting in which PaCO₂ helps identify patients likely to benefit from NIV; early improvement may also support response assessment. |
| IMV | Hypocapnia, hypercapnia, or hypercapnic acidosis | Monitoring / prognostic | Dyscapnia is associated with outcomes, but often reflects disease severity, ventilatory strategy, or dead space rather than an independent therapeutic target. |
| ARDS / lung-protective ventilation | Hypercapnia may be tolerated | Contextual physiological variable | Direct correction of PaCO₂ may conflict with lung-protective ventilation, and safe thresholds remain uncertain. |
| Prone positioning | Trend more important than isolated value | Response monitoring | A decrease in PaCO₂ may support physiological response assessment, but should not be interpreted as a stand-alone treatment target. |
| Neurocritical care | Avoid marked hypocapnia | Safety / monitoring | PaCO₂ is particularly important to avoid extremes, especially excessive hypocapnia. |
| Weaning and post-extubation | Persistent hypercapnia | Stratification / support selection | Hypercapnia may identify patients at higher risk of extubation failure and help support non-invasive strategies in selected cases. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




