Submitted:
23 April 2026
Posted:
24 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Participants and Sampling
2.2.1. Sample Size
2.3. Data Collection and Measurements
2.4. Statistical Analysis
2.5. Ethical Considerations
3. Results
3.1. Participant Characteristics and Prevalence of Sarcopenia
| Characteristics | n | % |
| Frailty status (Fried criteria) | ||
| Frail | 8 | 9.1 |
| Non-frail | 80 | 90.9 |
| Sarcopenia status(AWGS 2019) | ||
| No sarcopenia | 52 | 59.1 |
| Sarcopenia | 21 | 23.9 |
| Severe sarcopenia | 15 | 17.0 |
| Coexistence of frailty and sarcopenia | 7 | 7.9 |
3.2. Factors Associated with Sarcopenia
| Characteristics |
Sarcopenia (n=36) n (%) |
No Sarcopenia (n=52) n (%) |
p-value | |
| Age group | <0.05 | |||
| 60-65 years | 5 (13.9) | 23 (44.2) | ||
| 66-70 years | 11 (30.6) | 19 (36.5) | ||
| > 70 years | 20 (55.5) | 10 (19.3) | ||
| Gender | >0.05 | |||
| Male | 8 (22.2) | 11 (21.2) | ||
| Female | 28 (77.8) | 41 (78.8) | ||
| Occupation | <0.05 | |||
| Farmer | 30 (83.3) | 32 (61.5) | ||
| Others (Civil servant, freelance...) | 6 (16.7) | 20 (38.5) | ||
| Living conditions | >0.05 | |||
| Living alone | 6 (16.7) | 5 (9.6) | ||
| Living with relatives | 30 (83.3) | 47 (90.4) | ||
| Body Mass Index (BMI) | >0.05 | |||
| Underweight (< 18.5) | 8 (22.2) | 13 (25.0) | ||
| Normal (≥ 18,5) | 28 (77.8) | 39 (75.0) | ||
| Lifestyle Habits | ||||
| Smoking (Yes) | 3 (8.3) | 4 (7.7) | >0.05 | |
| Alcohol consumption (Yes) | 7 (19.4) | 9 (17.3) | >0.05 | |
| Commonmedicalconditions | ||||
| Hypertension | 9 (25.0) | 17 (32.7) | >0.05 | |
| Diabetes Mellitus | 3 (8.3) | 8 (15.4) | >0.05 | |
| Chronic Kidney Disease | 3 (8.3) | 8 (15.4) | >0.05 | |
| Clinicalcharacteristics of the Musculoskeletalsystem | ||||
| Severity of knee osteoarthritis (Grade 3-4) | 28 (77.8) | 38 (73.1) | >0.05 | |
| Number of medications used (≥ 3) | 15 (41.7) | 22 (42.3) | >0.05 | |
| Pathological Characteristics |
Sarcopenia (n=36) n (%) |
No Sarcopenia (n=52) n (%) |
p-value |
| Hypertension | >0.05 | ||
| Yes | 9 (25.0) | 17 (32.7) | |
| No | 27 (75.0) | 35 (67.3) | |
| Diabetes Mellitus | >0.05 | ||
| Yes | 3 (8.3) | 8 (15.4) | |
| No | 33 (91.7) | 44 (84.6) | |
| Chronic Kidney Disease | >0.05 | ||
| Yes | 3 (8.3) | 8 (15.4) | |
| No | 33 (91.7) | 44 (84.6) | |
| Severity of knee osteoarthritis | >0.05 | ||
| Grade 3-4 (Severe) | 28 (77.8) | 38 (73.1) | |
| Grade 1-2 (Mild/Moderate) | 8 (22.2) | 14 (26.9) | |
| Chronic back pain | >0.05 | ||
| Severe pain (VAS 7-10) | 27 (75,0) | 39 (75.0) | |
| Mild/moderate pain (VAS <7) | 9 (25.0) | 13 (25.0) | |
| History of falls | <0.001 | ||
| Yes | 13 (36.1) | 6 (11.5) | |
| No | 23 (63.9) | 46 (88.5) |
| Independent variables | Category | AOR | 95%CI | p-value |
| Age group | 60 - 65 years (Ref.) | 1.00 | - | - |
| 66 - 70 years | 2.4 | 0.7 – 8.4 | >0.05 | |
| > 70 years | 9.00 | 2.40 – 33.60 | <0.01 | |
| History of Falls | No (Ref.) | 1.00 | - | - |
| Yes | 6.33 | 2.77 – 14.45 | <0.001 | |
|
Low Education (Under Primary school) |
No (Ref.) | 1.00 | - | - |
| Yes | 2.86 | 1.46 – 5.61 | <0.01 | |
| Sleep Quality | Good (Ref.) | 1.00 | - | - |
| Poor (High PSQI) | 1.16 | 1.02 – 1.32 | 0.03 | |
| Living Situation | With Family (Ref.) | 1.00 | - | - |
| Living alone | 0.4 | 0.1 – 1.9 | >0.05 | |
| Occupation | Other occupation (Ref.) | 1.00 | - | - |
| Farmer | 0.4 | 0.1 – 1.2 | >0.05 | |
| Chronic spinal pain | None/Mild (Ref.) | 1.00 | - | - |
| Severe pain | 0.4 | 0.1 – 2.1 | >0.05 |
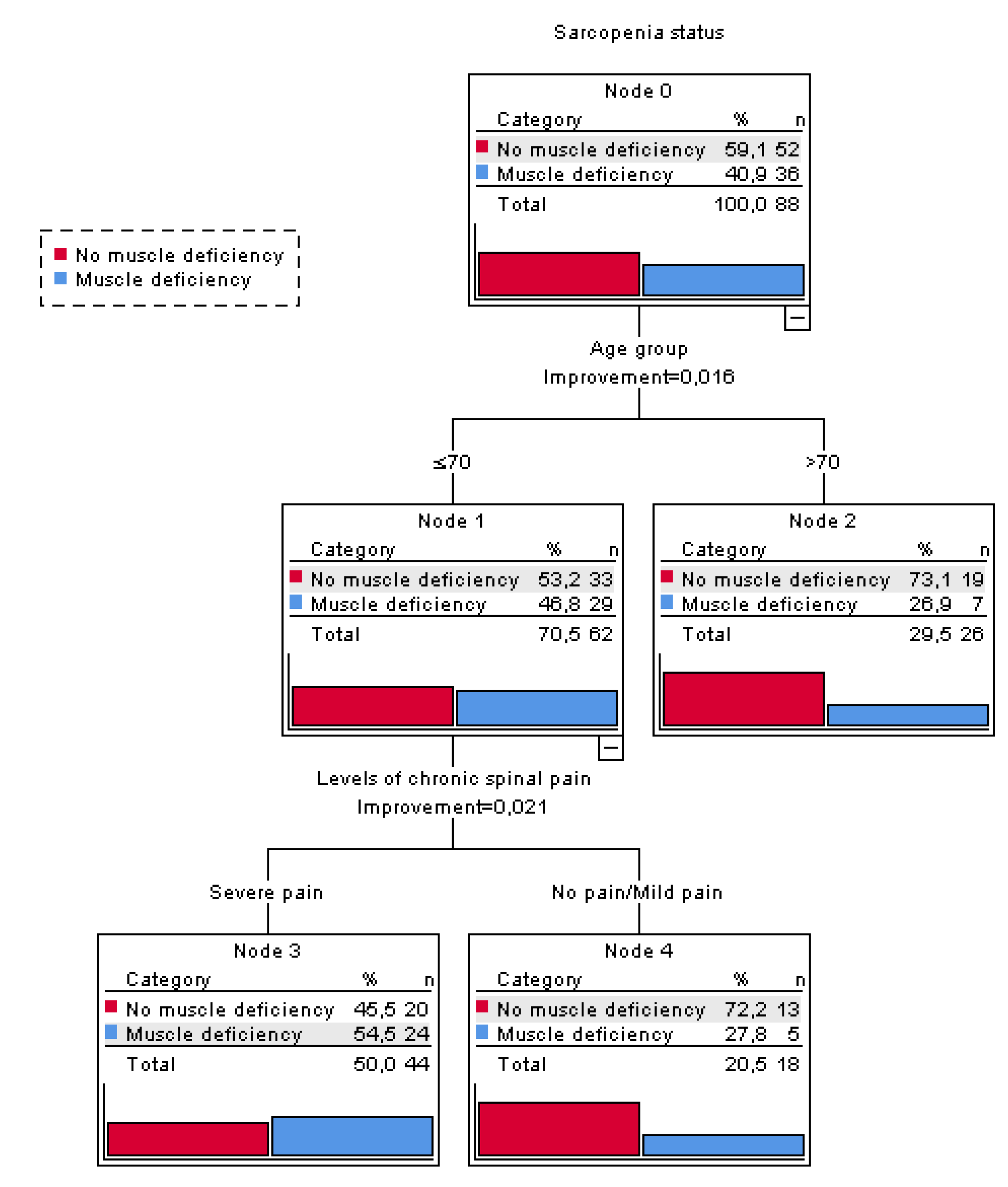
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AOR | Adjusted odds ratio |
| AWGS | Asian Working Group for Sarcopenia |
| BIA | Bioelectrical impedance analysis |
| BMI | Body mass index |
| CHAID | Chi-square Automatic Interaction Detection |
| CI | Confidence interval |
| KOA | Knee osteoarthritis |
| PSQI | Pittsburgh Sleep Quality Index |
| SD | Standard deviation |
| SE | Standard error |
| VAS | Visual Analogue Scale |
References
- United Nations Population Fund (UNFPA) Viet Nam; General Statistics Office of Viet Nam. The 2019 Viet Nam Population and Housing Census: Completed Results; Statistical Publishing House: Hanoi, 2021. [Google Scholar]
- Tran, G.H.; Ngo, L.P.; Do, A.T.; et al. Prevalence of sarcopenia and associated factors among older adults in Phong Dien district, Can Tho city, Vietnam, 2024. Can Tho Journal of Medicine and Pharmacy 2025, (83), 184–190. [Google Scholar] [CrossRef]
- Chen, L.K.; Woo, J.; Assantachai, P.; et al. Asian Working Group for Sarcopenia: 2019 Consensus Update on Sarcopenia Diagnosis and Treatment. Journal of the American Medical Directors Association 2020, 21(3), 300–307.e2. [Google Scholar] [CrossRef] [PubMed]
- Laskou, F.; Fuggle, N.R.; Patel, H.P.; et al. Associations of osteoporosis and sarcopenia with frailty and multimorbidity among participants of the Hertfordshire Cohort Study. J Cachexia Sarcopenia Muscle 2022, 13, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Sakai, Y.; Matsui, H.; Ito, S.; et al. Sarcopenia in elderly patients with chronic low back pain. Osteoporos Sarcopenia 2017, 3, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Du, Z.; Song, L.; et al. Factors, characteristics and influences of the changes of muscle activation patterns for patients with knee osteoarthritis: a review. J Orthop Surg Res. 2025, 20, 112. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.T.; Nguyen, L.H.; Nguyen, T.X.; et al. Frailty Prevalence and Association with Health-Related Quality of Life Impairment among Rural Community-Dwelling Older Adults in Vietnam. Int J Environ Res Public Health 2019, 16(20), 3834. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.D.; Nguyen, T.V.; Nguyen, T.D.; et al. Risk of falls in the older adult at Thai Binh Medical University Hospital and its related factors in 2024. Front Public Health 2025, 13, 1609745. [Google Scholar] [CrossRef]
- Song, Y.Y.; Lu, Y. Decision tree methods: applications for classification and prediction. Shanghai Arch Psychiatry 2015, 27(2), 130–135. [Google Scholar] [CrossRef] [PubMed]
- Loh, W.Y. Classification and regression trees. Wiley Interdiscip Rev Data Min Knowl Discov. 2011, 1(1), 14–23. [Google Scholar] [CrossRef]
- Ngo, H.L.; Huynh, T.H.; Nguyen, T.H.; Pham, H.L.; Lam, P.T.; Pham, H.K. Study of the rate of frailty and some related factors in elderly inpatients at Can Tho University of Medicine and Pharmacy Hospital. CTJUMP 2023, (67), 56–63. [Google Scholar] [CrossRef]
- Nguyen, T.N.; Nguyen, T.N.; Nguyen, A.T.; Nguyen, T.X.; Nguyen, H.T.T.; Nguyen, T.T.H.; et al. Prevalence of sarcopenia and its associated factors in patients attending geriatric clinics in Vietnam: a cross-sectional study. BMJ Open. 2020, 10(9), e037630. [Google Scholar] [CrossRef] [PubMed]
- Therakomen, V.; Petchlorlian, A.; Lakananurak, N. Prevalence and risk factors of primary sarcopenia in community-dwelling outpatient elderly: a cross-sectional study. Sci Rep. 2020, 10, 19551. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Park, C.J.; Jung, M.G.; Choi, Y.H.; Park, K.S.; Koh, H.S. High prevalence of sarcopenia in Asian female patients awaiting primary total knee arthroplasty: Application of updated diagnostic tools from the Asian Working Group for Sarcopenia. J Orthop Surg (Hong Kong) 2022, 30(2), 10225536221113034. [Google Scholar] [CrossRef] [PubMed]
- Weng, S.E.; Huang, Y.W.; Tseng, Y.C.; Peng, H.R.; Lai, H.Y.; Akishita, M.; et al. The Evolving Landscape of Sarcopenia in Asia: A systematic review and meta-analysis following the 2019 Asian Working Group for Sarcopenia (AWGS) diagnostic criteria. Arch Gerontol Geriatr. 2025, 128, 105596. [Google Scholar] [CrossRef] [PubMed]
- Tandon, S.; et al. Kinesiophobia and its association with muscle atrophy in patients with chronic musculoskeletal pain: A systematic review. J Rehabil Med. 2020, 52(4), jrm00045. [Google Scholar] [CrossRef]
- Visser, M.; et al. Relationship of Interleukin-6 and Tumor Necrosis Factor-alpha with Muscle Mass and Muscle Strength in Elderly Men and Women: The Health ABC Study. J Gerontol A Biol Sci Med Sci. 2002, 57(5), M326–M332. [Google Scholar] [CrossRef] [PubMed]
- Nygaard, H.; Kamper, R.S.; Ekmann, A.; et al. Co-Occurrence of Sarcopenia and Frailty in Acutely Admitted Older Medical Patients: Results from the Copenhagen PROTECT Study. J Frailty Aging 2024, 13, 91–97. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




