Submitted:
22 April 2026
Posted:
23 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
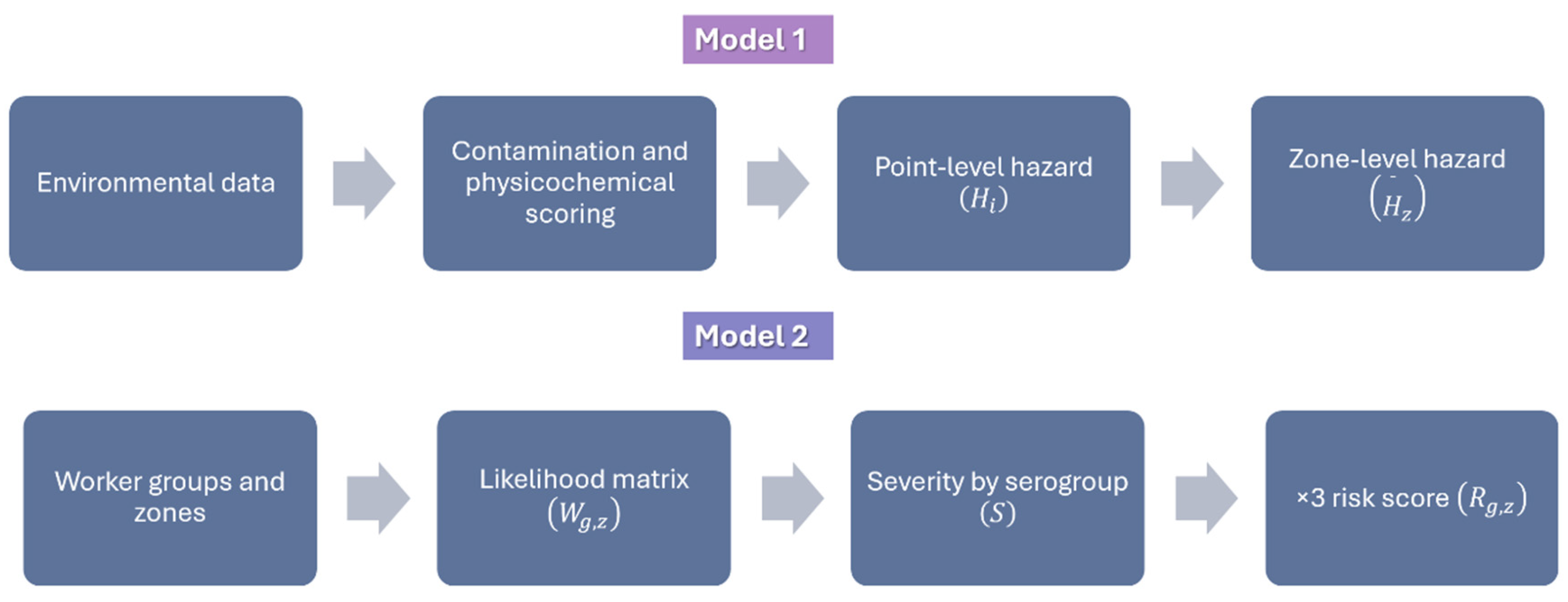
2.1. Study Framework and Environmental Data
2.2. Statistical Analysis and Facility-Level Risk Assessment
2.3. Contamination Scoring
2.4. Physicochemical Deviation Scoring
2.5. Point-Level Environmental Hazard
2.6. Zone-Level Hazard
2.7. Worker Groups
2.8. Zone Presence Weight
2.9. WHO-Style 3 × 3 Likelihood–Severity Occupational Risk Model
2.10. Quantitative Exposure Modelling (QMRA)
2.11. Personal Protective Equipment (PPE) Mapping
2.12. Process Overview
3. Results
3.1. Sampling Profile and Investigated Points
3.2. Culture-Based Legionella spp. Concentration Bands (≥1000 and ≥10,000 CFU/L)
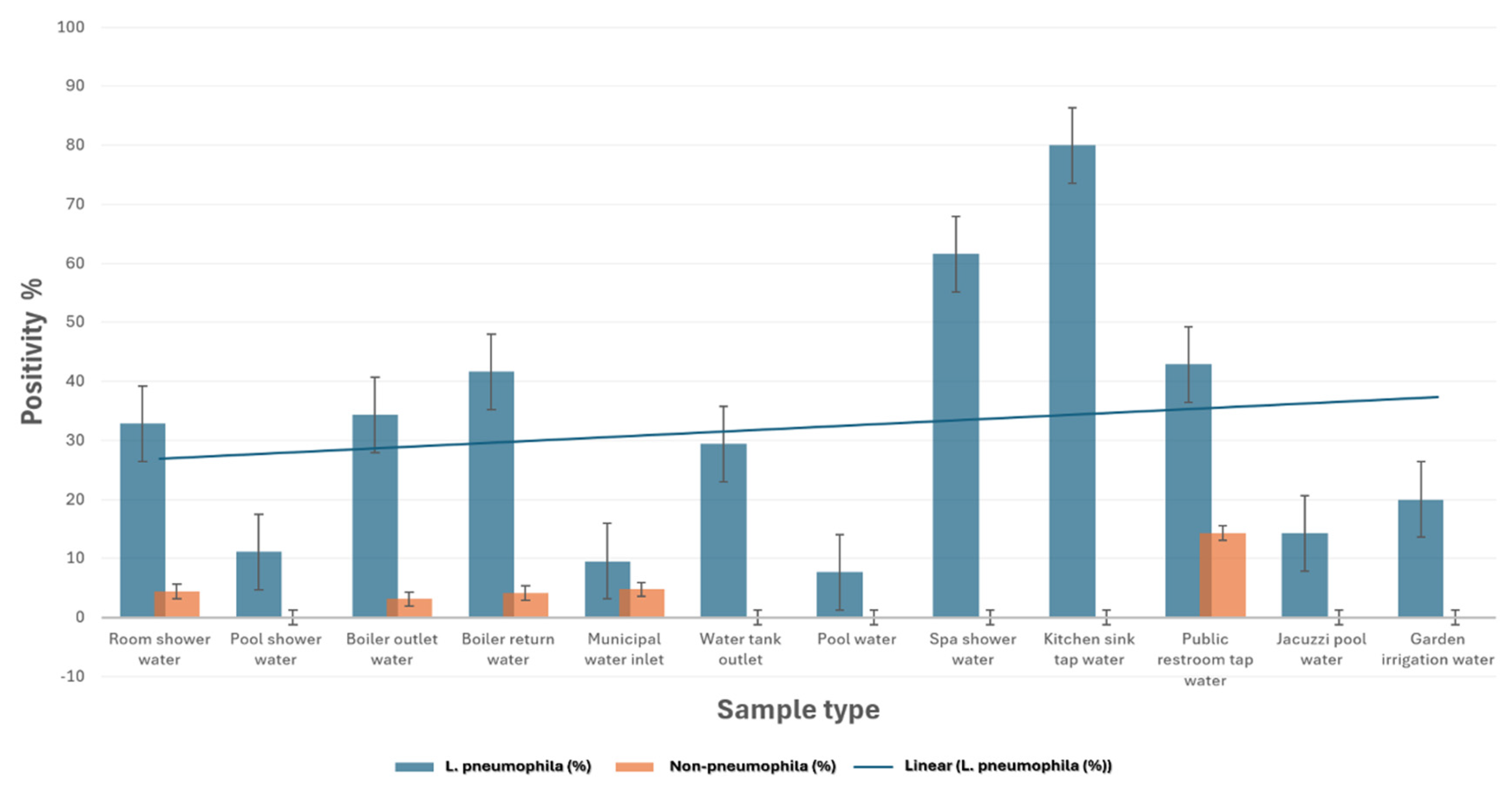
3.3. Culture-Based L. pneumophila Positivity (≥50 CFU/L) by Sample Type
3.4. Association Analyses (RR) for L. pneumophila Positivity (≥50 CFU/L)
3.5. Environmental Hazard Scoring Outputs (Equations 1–6)
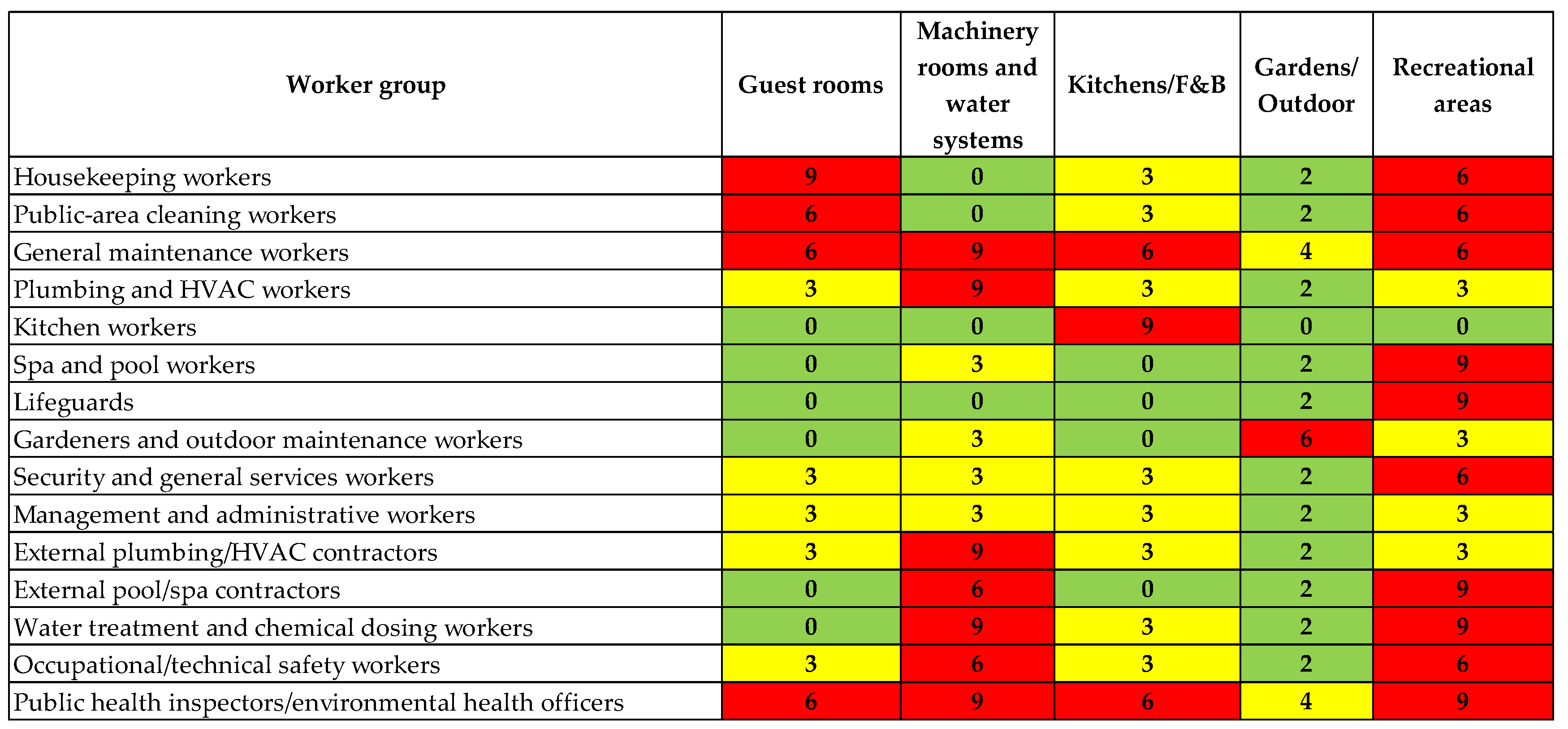
3.6. WHO-Style 3×3 Occupational Risk by Worker Group and Functional Zone
3.7. Occupational Exposure Stratification through Modeled Dose Estimation
3.8. Task-Based PPE Recommendations for High-Exposure Activities
4. Discussion
4.1. Principal Findings
4.2. Environmental Determinants of L. pneumophila Positivity
4.3. Zone-Level Hazard Patterns and Operational Implications
4.4. Occupational Exposure Profiles: Who Is at Risk and Where
4.5. Bridging the Gap: From Hazard Scoring to Modeled Dose Estimation
4.6. Integration with Water Safety Plans and the Hierarchy of Controls
4.7. Strengths, Limitations, and Interpretation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TALD | Travel-associated Legionnaires’ disease |
| WHO | World Health Organization |
| NPHO | National Public Health Organization |
| OSHA | Occupational Safety and Health Administration |
| PPE | Personal protective equipment |
| RR | Risk ratio |
| CI | Confidence interval |
| CFU/L | Colony-forming units per liter |
| SG | Serogroup |
| HVAC | Heating, ventilation, and air conditioning |
| F&B | Food and beverage |
| CDC | Centers for Disease Control and Prevention |
| ECDC | European Centre for Disease Prevention and Control |
References
- Accommodation Site List for Legionnaires’ Disease. Available online: https://www.ecdc.europa.eu/en/legionnaires-disease/threats-and-outbreaks/accommodation-site (accessed on 3 January 2026).
- Smith, S.S.; Ritger, K.; Samala, U.; Black, S.R.; Okodua, M.; Miller, L.; Kozak-Muiznieks, N.A.; Hicks, L.A.; Steinheimer, C.; Ewaidah, S.; et al. Legionellosis Outbreak Associated With a Hotel Fountain. Open Forum Infect. Dis. 2015, 2, ofv164. [Google Scholar] [CrossRef]
- Benin, A.L.; Benson, R.F.; Arnold, K.E.; Fiore, A.E.; Cook, P.G.; Williams, L.K.; Fields, B.; Besser, R.E. An Outbreak of Travel-Associated Legionnaires Disease and Pontiac Fever: The Need for Enhanced Surveillance of Travel-Associated Legionellosis in the United States. Journal of Infectious Diseases 2002, 185, 237–243. [Google Scholar] [CrossRef]
- Euser, S.M.; Pelgrim, M.; Den Boer, J.W. Legionnaires’ Disease and Pontiac Fever after Using a Private Outdoor Whirlpool Spa. Scand. J. Infect. Dis. 2010, 42, 910–916. [Google Scholar] [CrossRef] [PubMed]
- Lee, S. An Overview of the European Technical Guidelines for the Prevention, Control and Investigation of Infections Caused by Legionella Species. Perspect. Public Health 2018, 138, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Beauté, J.; Sandin, S.; de Jong, B.; Hallström, L.P.; Robesyn, E.; Giesecke, J.; Sparén, P. Factors Associated with Legionnaires’ Disease Recurrence in Hotel and Holiday Rental Accommodation Sites. Eurosurveillance 2019, 24. [Google Scholar] [CrossRef] [PubMed]
- Beauté, J.; Zucs, P.; de Jong, B. Risk for Travel-Associated Legionnaires’ Disease, Europe, 2009. Emerg. Infect. Dis. 2012, 18, 1811–1816. [Google Scholar] [CrossRef]
- Buchholz, U.; Brodhun, B.; Lehfeld, A.S. Incidence of Legionnaires’ Disease among Travelers Visiting Hotels in Germany, 2015–2019. Emerg. Infect. Dis. 2024, 30, 13. [Google Scholar] [CrossRef]
- Legionella in the Workplace—OSHwiki | European Agency for Safety and Health at Work. Available online: https://oshwiki.osha.europa.eu/en/themes/legionella-workplace (accessed on 3 January 2026).
- Dondero, T.J.; Rendtorff, R.C.; Mallison, G.F.; Weeks, R.M.; Levy, J.S.; Wong, E.W.; Schaffner, W. An Outbreak of Legionnaires’ Disease Associated with a Contaminated Air-Conditioning Cooling Tower. N. Engl. J. Med. 1980, 302, 365–370. [Google Scholar] [CrossRef]
- Wallis, L.; Robinson, P. Soil as a Source of Legionella Pneumophila Serogroup 1 (Lp1). Aust. N. Z. J. Public Health 2005, 29, 518–520. [Google Scholar] [CrossRef]
- Blatny, J.M.; Reif, B.A.P.; Skogan, G.; Andreassen, O.; Høiby, E.A.; Ask, E.; Waagen, V.; Aanonsen, D.; Aaberge, I.S.; Caugant, D.A. Tracking Airborne Legionella and Legionella Pneumophila at a Biological Treatment Plant. Environ. Sci. Technol. 2008, 42, 7360–7367. [Google Scholar] [CrossRef]
- Benin, A.L.; Benson, R.F.; Besser, R.E. Trends in Legionnaires Disease, 1980-1998: Declining Mortality and New Patterns of Diagnosis. Clin. Infect. Dis. 2002, 35, 1039–1046. [Google Scholar] [CrossRef]
- Mohamed, H.M.; Zielinski, L.; Diedhiou, A.; Clemmons, N.; Smith, J.C.; Rinsky, J.L.; Ritter, T.; Willby, M.; Burton, N.; Feldmann, K.; et al. Cluster of Legionellosis Cases Associated with Manufacturing Process, South Carolina, USA, 2022—Volume 31, Number 1—January 2025—Emerging Infectious Diseases Journal—CDC. Emerg. Infect. Dis. 2025, 31, 41–49. [Google Scholar] [CrossRef]
- Morano, L.H.; Morawski, B.M.; Herzig, C.T.A.; Edens, C.; Barskey, A.E.; Luckhaupt, S.E. Legionnaires’ Disease in Transportation, Construction and Other Occupations in 39 US Jurisdictions, 2014–2016. Occup. Environ. Med. 2024, 81, 163–166. [Google Scholar] [CrossRef]
- Scanlon, M.M.; Gordon, J.L.; Tonozzi, A.A.; Griffin, S.C. Reducing the Risk of Healthcare Associated Infections from Legionella and Other Waterborne Pathogens Using a Water Management for Construction (WMC) Infection Control Risk Assessment (ICRA) Tool. Infect. Dis. Rep. 2022, 14, 341–359. [Google Scholar] [CrossRef] [PubMed]
- Bongiovanni, A.; Colazingari, V.; Messineo, A.; Del Cimmuto, A.; De Giusti, M.; La Torre, G. Can Legionellosis Be Considered an Occupational Risk in the Healthcare Sector? A Systematic Review and Meta-Analysis. Public Health 2023, 214, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.E.; Stout, J.E.; Yu, V.L. Controlling Legionella in Hospital Drinking Water: An Evidence-Based Review of Disinfection Methods. Infect. Control Hosp. Epidemiol. 2011, 32, 166–173. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, E.; Humphreys, H. Surveillance of Hospital Water and Primary Prevention of Nosocomial Legionellosis: What Is the Evidence? Journal of Hospital Infection 2005, 59, 273–279. [Google Scholar] [CrossRef]
- Lin, Y.E.; Stout, J.E.; Yu, V.L. Prevention of Hospital-Acquired Legionellosis. Curr. Opin. Infect. Dis. 2011, 24, 350–356. [Google Scholar] [CrossRef]
- Correia, A.M.; Ferreira, J.S.; Borges, V.; Nunes, A.; Gomes, B.; Capucho, R.; Gonçalves, J.; Antunes, D.M.; Almeida, S.; Mendes, A.; et al. Probable Person-to-Person Transmission of Legionnaires’ Disease. N. Engl. J. Med. 2016, 374, 497–498. [Google Scholar] [CrossRef]
- Weiss, D.; Boyd, C.; Rakeman, J.L.; Greene, S.K.; Fitzhenry, R.; McProud, T.; Musser, K.; Huang, L.; Kornblum, J.; Nazarian, E.J.; et al. A Large Community Outbreak of Legionnaires’ Disease Associated With a Cooling Tower in New York City, 2015. Public Health Rep. 2017, 132, 241–250. [Google Scholar] [CrossRef]
- Chochlakis, D.; Sandalakis, V.; Ntoukakis, A.; Daskalaki, M.O.; Loppinet, T.; Thalassinaki, N.; Makridaki, R.; Panoulis, C.; Psaroulaki, A. Multi-Criterion Analysis of the Effect of Physico-Chemical Microbiological Agents on Legionella Detection in Hotel Water Distribution Systems in Crete. Front. Cell. Infect. Microbiol. 2023, 13. [Google Scholar] [CrossRef]
- Managing Legionella in Hot and Cold Water Systems—HSE. Available online: https://www.hse.gov.uk/healthservices/legionella.htm (accessed on 3 January 2026).
- Yao, X.H.; Shen, F.; Hao, J.; Huang, L.; Keng, B. A Review of Legionella Transmission Risk in Built Environments: Sources, Regulations, Sampling, and Detection. Front. Public Health 2024, 12, 1415157. [Google Scholar] [CrossRef] [PubMed]
- Carlson, K.M.; Boczek, L.A.; Chae, S.; Ryu, H. Legionellosis and Recent Advances in Technologies for Legionella Control in Premise Plumbing Systems: A Review. Water 2020, Vol. 12 12, 676. [Google Scholar] [CrossRef] [PubMed]
- Papadakis, A.; Chochlakis, D.; Sandalakis, V.; Keramarou, M.; Tselentis, Y.; Psaroulaki, A. Legionella Spp. Risk Assessment in Recreational and Garden Areas of Hotels. International Journal of Environmental Research and Public Health 2018, Vol. 15 15, 598. [Google Scholar] [CrossRef] [PubMed]
- Laganà, P.; Gioffrè, M.E.; Delia, S.A.; Facciolà, A. Legionella Spp. in Thermal Facilities: A Public Health Issue in the One Health Vision. Water 2023, 15, 689. [Google Scholar] [CrossRef]
- Marras, L.; Bertolino, G.; Sanna, A.; Carraro, V.; Coroneo, V. Legionella Spp. Monitoring in the Water Supply Systems of Accommodation Facilities in Sardinia, Italy: A Two-Year Retrospective Analysis. International Journal of Environmental Research and Public Health 2023, Vol. 20 20, 6722. [Google Scholar] [CrossRef]
- Legionella and Legionnaires’ Disease: A Policy Overview | Safety and Health at Work EU-OSHA. Available online: https://osha.europa.eu/en/publications/legionella-and-legionnaires-disease-policy-overview (accessed on 18 March 2026).
- Factsheet 100—Legionella and Legionnaires’ Disease: European Policies and Good Practices | Safety and Health at Work EU-OSHA. Available online: https://osha.europa.eu/en/publications/factsheet-100-legionella-and-legionnaires-disease-european-policies-and-good-practices (accessed on 18 March 2026).
- Benkel, D.H.; McClure, E.M.; Woolard, D.; Rullan, J. V.; Miller, G.B.; Jenkins, S.R.; Hershey, J.H.; Benson, R.F.; Pruckler, J.M.; Brown, E.W.; et al. Outbreak of Legionnaires’ Disease Associated with a Display Whirlpool Spa. Int. J. Epidemiol. 2000, 29, 1092–1098. [Google Scholar] [CrossRef]
- Kyritsi, M.A.; Mouchtouri, V.A.; Katsioulis, A.; Kostara, E.; Nakoulas, V.; Hatzinikou, M.; Hadjichristodoulou, C. Legionella Colonization of Hotel Water Systems in Touristic Places of Greece: Association with System Characteristics and Physicochemical Parameters. International Journal of Environmental Research and Public Health 2018, 15, 2707. [Google Scholar] [CrossRef]
- Borella, P.; Montagna, M.T.; Stampi, S.; Stancanelli, G.; Romano-Spica, V.; Triassi, M.; Marchesi, I.; Bargellini, A.; Tatò, D.; Napoli, C.; et al. Legionella Contamination in Hot Water of Italian Hotels. Appl. Environ. Microbiol. 2005, 71, 5805. [Google Scholar] [CrossRef]
- Füchslin, H.P.; Kötzsch, S.; Keserue, H.A.; Egli, T. Rapid and Quantitative Detection of Legionella Pneumophila Applying Immunomagnetic Separation and Flow Cytometry. Cytometry Part A 2010, 77A, 264–274. [Google Scholar] [CrossRef]
- Papadakis, A.A.; Tsirigotakis, I.; Katranitsa, S.; Donousis, C.; Papalexis, P.; Keramydas, D.; Chaidoutis, E.; Georgakopoulou, V.E.; Spandidos, D.A.; Constantinidis, T.C. Assessing the Impact of the COVID-19 Pandemic Health Protocols on the Hygiene Status of Swimming Pools of Hotel Units. Medicine International 2023, 3, 32. [Google Scholar] [CrossRef] [PubMed]
- Papadakis, A.; Chochlakis, D.; Koufakis, E.; Carayanni, V.; Psaroulaki, A. Recreational Water Safety in Hotels: Lessons from the COVID-19 Pandemic and the Way Forward for a Safe Aquatic Environment. Tourism and Hospitality 2024, 5, 1167–1181. [Google Scholar] [CrossRef]
- Hassanein, F.; Masoud, I.M.; Fekry, M.M.; Abdel-Latif, M.S.; Abdel-Salam, H.; Salem, M.; Shehata, A.I. Environmental Health Aspects and Microbial Infections of the Recreational Water. BMC Public Health 2023, 23, 302. [Google Scholar] [CrossRef] [PubMed]
- De Giglio, O.; Fasano, F.; Diella, G.; Lopuzzo, M.; Napoli, C.; Apollonio, F.; Brigida, S.; Calia, C.; Campanale, C.; Marzella, A.; et al. Legionella and Legionellosis in Touristic-Recreational Facilities: Influence of Climate Factors and Geostatistical Analysis in Southern Italy (2001–2017). Environ. Res. 2019, 178. [Google Scholar] [CrossRef]
- De Giglio, O.; Napoli, C.; Diella, G.; Fasano, F.; Lopuzzo, M.; Apollonio, F.; D’Ambrosio, M.; Campanale, C.; Triggiano, F.; Caggiano, G.; et al. Integrated Approach for Legionellosis Risk Analysis in Touristic-Recreational Facilities. Environ. Res. 2021, 202. [Google Scholar] [CrossRef]
- Jjemba, P.K.; Johnson, W.; Bukhari, Z.; LeChevallier, M.W. Occurrence and Control of Legionella in Recycled Water Systems. Pathogens 2015, 4, 470–502. [Google Scholar] [CrossRef]
- Kirschner, A.K.T. Determination of Viable Legionellae in Engineered Water Systems: Do We Find What We Are Looking For? Water Res. 2016, 93, 276–288. [Google Scholar] [CrossRef]
- Armstrong, T.W.; Haas, C.N. Legionnaires’ Disease: Evaluation of a Quantitative Microbial Risk Assessment Model. J. Water Health 2008, 6, 149–166. [Google Scholar] [CrossRef]
- Federigi, I.; De Giglio, O.; Diella, G.; Triggiano, F.; Apollonio, F.; D’ambrosio, M.; Cioni, L.; Verani, M.; Montagna, M.T.; Carducci, A. Quantitative Microbial Risk Assessment Applied to Legionella Contamination on Long-Distance Public Transport. International Journal of Environmental Research and Public Health 2022, 19, 1960. [Google Scholar] [CrossRef]
- De Giglio, O.; Fasano, F.; Diella, G.; Spagnuolo, V.; Triggiano, F.; Lopuzzo, M.; Apollonio, F.; Leone, C.M.; Montagna, M.T. Machine Learning vs. Regression Models to Predict the Risk of Legionella Contamination in a Hospital Water Network. Ann. Ig. 2025, 37, 128–140. [Google Scholar] [CrossRef]
- Martinelli, M.; Calaresu, E.; Musumeci, R.; Giubbi, C.; Perdoni, F.; Frugoni, S.; Castriciano, S.; Scaturro, M.; Ricci, M.L.; Cocuzza, C.E. Evaluation of an Environmental Transport Medium for Legionella Pneumophila Recovery. Int. J. Environ. Res. Public Health 2021, 18. [Google Scholar] [CrossRef]
- Eble, D.; Gehrig, V.; Schubert-Ullrich, P.; Köppel, R.; Füchslin, H.P. Comparison of the Culture Method with Multiplex PCR for the Confirmation of Legionella Spp. and Legionella Pneumophila. J. Appl. Microbiol. 2021, 131, 2600–2609. [Google Scholar] [CrossRef]
- Monteiro, S.N.; Robalo, A.M.; Santos, R.J. Evaluation of LegiolertTM for the Detection of Legionella Pneumophila and Comparison with Spread-Plate Culture and QPCR Methods. Curr. Microbiol. 2021, 78, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- Hirsh, M.B.; Baron, J.L.; Mietzner, S.M.; Rihs, J.D.; Yassin, M.H.; Stout, J.E. Evaluation of Recommended Water Sample Collection Methods and the Impact of Holding Time on Legionella Recovery and Variability from Healthcare Building Water Systems. Microorganisms 2020, 8, 1770. [Google Scholar] [CrossRef] [PubMed]
- Ditommaso, S.; Giacomuzzi, M.; Ricciardi, E.; Zotti, C.M. Cultural and Molecular Evidence of Legionella Spp. Colonization in Dental Unit Waterlines: Which Is the Best Method for Risk Assessment? International Journal of Environmental Research and Public Health 2016, 13, 211. [Google Scholar] [CrossRef] [PubMed]
- Prussin, A.J.; Schwake, D.O.; Marr, L.C. Ten Questions Concerning the Aerosolization and Transmission of Legionella in the Built Environment. Build. Environ. 2017, 123, 684–695. [Google Scholar] [CrossRef]
- Schoen, M.E.; Ashbolt, N.J. An In-Premise Model for Legionella Exposure during Showering Events. Water Res. 2011, 45, 5826–5836. [Google Scholar] [CrossRef]
- Donohue, M.J.; Mistry, J.H.; Tucker, N.; Vesper, S.J. Hot Water Plumbing in Residences and Office Buildings Have Distinctive Risk of Legionella Pneumophila Contamination. Int. J. Hyg. Environ. Health 2022, 245. [Google Scholar] [CrossRef]
- Falkinham, J.O. Living with Legionella and Other Waterborne Pathogens. Microorganisms 2020, 8, 1–10. [Google Scholar] [CrossRef]
- Hayward, C.; Ross, K.E.; Brown, M.H.; Bentham, R.; Whiley, H. The Presence of Opportunistic Premise Plumbing Pathogens in Residential Buildings: A Literature Review. Water (Switzerland) 2022, 14. [Google Scholar] [CrossRef]
- Burillo, A.; Pedro-Botet, M.L.; Bouza, E. Microbiology and Epidemiology of Legionnaire’s Disease. Infect. Dis. Clin. North Am. 2017, 31, 7–27. [Google Scholar] [CrossRef] [PubMed]
- Di Onofrio, V.; Pagano, M.; Santulli, M.; Rossi, A.; Liguori, R.; Di Dio, M.; Liguori, G. Contamination of Hotel Water Distribution Systems by Legionella Species: Environmental Surveillance in Campania Region, South Italy. Microorganisms 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Papadakis, A.; Koufakis, E.; Nakoulas, V.; Kourentis, L.; Manouras, T.; Kokkinomagoula, A.; Ntoula, A.; Malliarou, M.; Gerakoudis, K.; Tsilipounidaki, K.; et al. Beyond Microbiological Analysis: The Essential Role of Risk Assessment in Travel-Associated Legionnaires’ Disease Outbreak Investigations. Pathogens 2025, 14. [Google Scholar] [CrossRef] [PubMed]
- Paranjape, K.; Bédard, É; Whyte, L.G.; Ronholm, J.; Prévost, M.; Faucher, S.P. Presence of Legionella Spp. in Cooling Towers: The Role of Microbial Diversity, Pseudomonas, and Continuous Chlorine Application. Water Res. 2019, 169, 115252. [Google Scholar] [CrossRef]
- Papadakis, A.; Keramarou, M.; Chochlakis, D.; Sandalakis, V.; Mouchtouri, V.A.; Psaroulaki, A. Legionella Spp. Colonization in Water Systems of Hotels Linked with Travel-Associated Legionnaires’ Disease. Water 2021, 13. [Google Scholar] [CrossRef]
- Papadakis, A.; Koufakis, E.; Chaidoutis, E.A.; Chochlakis, D.; Psaroulaki, A. Comparative Risk Assessment of Legionella Spp. Colonization in Water Distribution Systems Across Hotels, Passenger Ships, and Healthcare Facilities During the COVID-19 Era. Water 2025, 17. [Google Scholar] [CrossRef]
- Papadakis, A.; Koufakis, E.; Raptakis, N.; Pitsoulis, G.; Kamekis, A.; Chochlakis, D.; Psaroulaki, A.; Lagiou, A. Risk Communication and Infodemic Misframing in Legionella Spp. Environmental Surveillance: An Infodemiology Case Study. Microorganisms 2026, 14, 536. [Google Scholar] [CrossRef]
- Saran, S.; Gurjar, M.; Baronia, A.; Sivapurapu, V.; Ghosh, P.S.; Raju, G.M.; Maurya, I. Heating, Ventilation and Air Conditioning (HVAC) in Intensive Care Unit. Crit. Care 2020, 24. [Google Scholar] [CrossRef]
- Fujii, J.; Arima, K.; Miyamoto, H.; Yoshida, S.I. Legionella Infection in Occupational and Environmental Health. J. UOEH 1996, 18, 61–76. [Google Scholar] [CrossRef]
- Principe, L.; Tomao, P.; Visca, P. Legionellosis in the Occupational Setting. Environ. Res. 2017, 152, 485–495. [Google Scholar] [CrossRef]
- Molina, J.J.; Bennassar, M.; Palacio, E.; Crespi, S. Impact of Prolonged Hotel Closures during the COVID-19 Pandemic on Legionella Infection Risks. Front. Microbiol. 2023, 14, 1136668. [Google Scholar] [CrossRef]
- Water Safety Plan Manual: Step-By-Step Risk Management for Drinking-Water Suppliers. 2023.
- Water Safety Plan Manual: Step-by-Step Risk Management for Drinking-Water Suppliers, Second Edition. Available online: https://www.who.int/publications/i/item/9789240067691 (accessed on 19 April 2026).
- Water Sanitation and Health. Available online: https://www.who.int/teams/environment-climate-change-and-health/water-sanitation-and-health/water-safety-and-quality/water-safety-planning (accessed on 19 April 2026).
- European Technical Guidelines for the Prevention, Control and Investigation, of Infections Caused by Legionella Species. 2017.
- Steps to Develop a Water Management Program | Control Legionella | CDC. Available online: https://www.cdc.gov/control-legionella/php/wmp/wmp-steps.html (accessed on 19 April 2026).
- Abdalla, A.I.; Saidan, M.N.; Al Alami, N.; Al-Naimat, H.; Abdalla, A.I. Comparative Assessment of Legionella Pneumophila Prevalence among Hospitals and Hotels Water Systems. 2020. [Google Scholar] [CrossRef]
- World Health Organization. Chapter 11—Laboratory Aspects of Legionella. In Legionella and the prevention of legionellosis; 2007. [Google Scholar]
- Niosh Preventing Occupational Exposure to Legionella., doi:10.26616/NIOSHPUB2019131.
- Barimani, M.J. Legionella: An Uncommon Cause of Community-Acquired Pneumonia. JAAPA 2022, 35, 38–42. [Google Scholar] [CrossRef]
- Gea-Izquierdo, E.; Gil-de-Miguel, Á; Rodríguez-Caravaca, G. Legionella Pneumophila Risk from Air–Water Cooling Units Regarding Pipe Material and Type of Water. Microorganisms 2023, 11. [Google Scholar] [CrossRef]
| Department | Job Position | Main Potential Sources of Exposure |
|---|---|---|
| Engineering / Maintenance | General maintenance technician | Hot and cold-water systems, guest room showers, mechanical rooms, pumps, valves |
| Engineering / Maintenance | Plumbing technician | Pipework repairs, showerheads and taps, removal of scale, stagnant sections of the network |
| Engineering / Maintenance | HVAC / cooling systems technician | Cooling towers, evaporative condensers, humidifiers, air-handling units (where present) |
| Engineering / Maintenance | Pool and spa systems technician | Swimming pools, spa pools, whirlpools, filters, backwash systems, dosing equipment |
| Spa / Wellness / Pool | Spa therapist / spa attendant | Spa pools, whirlpools, hydro-massage facilities, steam rooms, experience showers |
| Spa / Wellness / Pool | Pool attendant / lifeguard | Swimming pools, children’s pools, poolside showers, water features |
| Spa / Wellness / Pool | Spa receptionist / spa assistant | Presence in spa area with aerosol-generating facilities |
| Housekeeping / Floors | Room attendant (housekeeper) | Guest room showers and bathrooms, flushing of outlets during cleaning |
| Housekeeping / Floors | Public area cleaner | Public toilets, changing rooms, spa/fitness showers, staff showers |
| Housekeeping / Floors | Floor supervisor | Periodic presence in rooms and corridors with access to bathrooms |
| Food & Beverage (F&B) | Kitchen staff (chefs, cooks) | Hot water outlets in kitchens, dishwashing areas, steam and aerosols |
| Food & Beverage (F&B) | Stewarding / dishwashing staff | Dishwashers, sinks, hot water sprays and aerosols |
| Food & Beverage (F&B) | Waiters, bartenders | Ice machines, beverage dispensers connected to the water supply |
| Gardens / Exterior maintenance | Gardener / landscaping staff | Irrigation sprinklers, outdoor misting systems, decorative ponds and fountains |
| Gardens / Exterior maintenance | Exterior maintenance worker | Outdoor decorative fountains, waterfalls, façade cleaning systems |
| Front office / Administration | Receptionists / front-desk agents | General indoor air, lobby air-conditioning systems |
| Front office / Administration | Management, back-office staff | Offices supplied by central HVAC |
| Security / General services | Security guards | Patrols in all areas, including technical rooms and roofs with cooling systems |
| Security / General services | General services / handyman | Minor repairs in bathrooms, public areas, back-of-house facilities |
| External contractors | External pool & spa maintenance | Targeted work on pools, spa systems, filters, dosing equipment |
| External contractors | External HVAC / refrigeration technicians | Cooling towers, chillers, evaporative cooling systems |
| External contractors | Cleaning companies (outsourced housekeeping, etc.) | Cleaning of rooms and wet areas, similar tasks to in-house housekeeping |
| External contractors | Construction / refurbishment contractors | Work on water networks, tanks and spa facilities during renovations (short-term but intense exposure) |
| External / regulatory / safety | Public health inspectors / environmental officers | Inspections and sampling in mechanical rooms, plant rooms, recreational water systems and other high-risk water installations |
| External / regulatory / safety | Occupational / technical safety specialists | Risk assessments and audits in plant rooms, technical areas and high-risk installations |
| Risk factors | Odds ratio | 95% CI | R.R. | 95% CI | Risk difference (%) | 95% CI | p value* |
|---|---|---|---|---|---|---|---|
| Free residual chlorine <0.2 mg/L | 4.59 | 2.48–8.50 | 2.90 | 1.92–4.39 | 30.84 | 17.84–43.84 | <0.0001 (χ2) |
| Cold water >20 °C | 2.18 | 0.26–18.41 | 1.87 | 0.30–11.57 | 12.36 | −14.09–38.80 | 0.6799 (Fisher) |
| Cold water >25 °C | 1.23 | 0.72–2.10 | 1.17 | 0.79–1.73 | 4.01 | −6.27–14.29 | 0.4988 (Fisher) |
| Hot water <45 °C | 1.55 | 0.87–2.77 | 1.31 | 0.93–1.85 | 10.37 | −3.51–24.25 | 0.1739 (Fisher) |
| Hot water <50 °C | 4.50 | 2.18–9.29 | 3.00 | 1.65–5.44 | 28.57 | 17.92–39.23 | <0.0001 (χ2) |
| Hot water <55 °C | 4.50 | 2.18–9.29 | 3.00 | 1.65–5.44 | 28.57 | 17.92–39.23 | <0.0001 (χ2) |
| Boiler outlet temperature <60 °C | 2.25 | 0.38–13.35 | 1.76 | 0.47–6.62 | 16.91 | −16.79–50.61 | 0.4414 (Fisher) |
| Boiler return temperature <50 oC | 2.70 | 0.51–14.37 | 1.77 | 0.67–4.71 | 23.78 | −14.89–62.45 | 0.4081 (Fisher) |
| Star classification <4 | 2.33 | 1.43–3.80 | 1.89 | 1.28–2.80 | 15.64 | 7.92–23.36 | 0.0004 (Fisher) |
| Sample from the TALD case room | 1.85 | 1.17–2.95 | 1.48 | 1.12–1.96 | 14.15 | 3.22–25.09 | 0.0104 (Fisher) |
| Functional area | Scored n | SD | |
|---|---|---|---|
| Kitchens and Food & Beverage Areas | 14 | 2.607 | 1.130 |
| Machinery Rooms and Water Production/Storage Systems | 90 | 2.022 | 1.503 |
| Guest Rooms | 405 | 1.874 | 1.404 |
| Recreational Areas (Pools/Spa) | 63 | 1.825 | 1.097 |
| Gardens and Outdoor Plumbing/Irrigation | 6 | 1.750 | 0.758 |
| Worker Group | Primary Exposure Zone | Estimated Daily Inhaled Dose (d, CFU/day) | |
|---|---|---|---|
| Kitchen staff (chefs/cooks) | Kitchens / F&B | 3 | 6.2 x 10−3 |
| Stewarding / dishwashing | Kitchens / F&B | 3 | 6.2 x 10−3 |
| Spa therapist / attendant | Recreational Areas | 3 | 5.1 x 10−3 |
| Plumbing technician | Machinery Rooms | 2 | 3.1 x 10−3 |
| HVAC technician | Machinery Rooms | 2 | 3.1 x 10−3 |
| Pool/Spa technician | Recreational Areas | 2 | 2.8 x 10−3 |
| Room attendant (housekeeper) | Guest Rooms | 3 | 2.5 x 10−3 |
| General maintenance | All zones (weighted mean exposure scenario) | 3 | 2.2 x 10−3 |
| Public area cleaner | Public and spa shower areas | 3 | 2.1 x 10−3 |
| Public health inspectors | High-exposure sampling points | 1 | 1.5 x 10−3 |
| Lifeguard | Recreational Areas | 3 | 1.1 x 10−3 |
| Occupational safety spec. | Technical service areas | 1 | 0.9 x 10−3 |
| External HVAC contractors | Machinery Rooms | 1 | 0.8 x 10−3 |
| Gardener / landscaping | Gardens / Irrigation | 2 | 0.8 x 10−3 |
| Security / General services | General hotel service areas | 1 | 0.4 x 10−3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




