Submitted:
19 April 2026
Posted:
23 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Participants
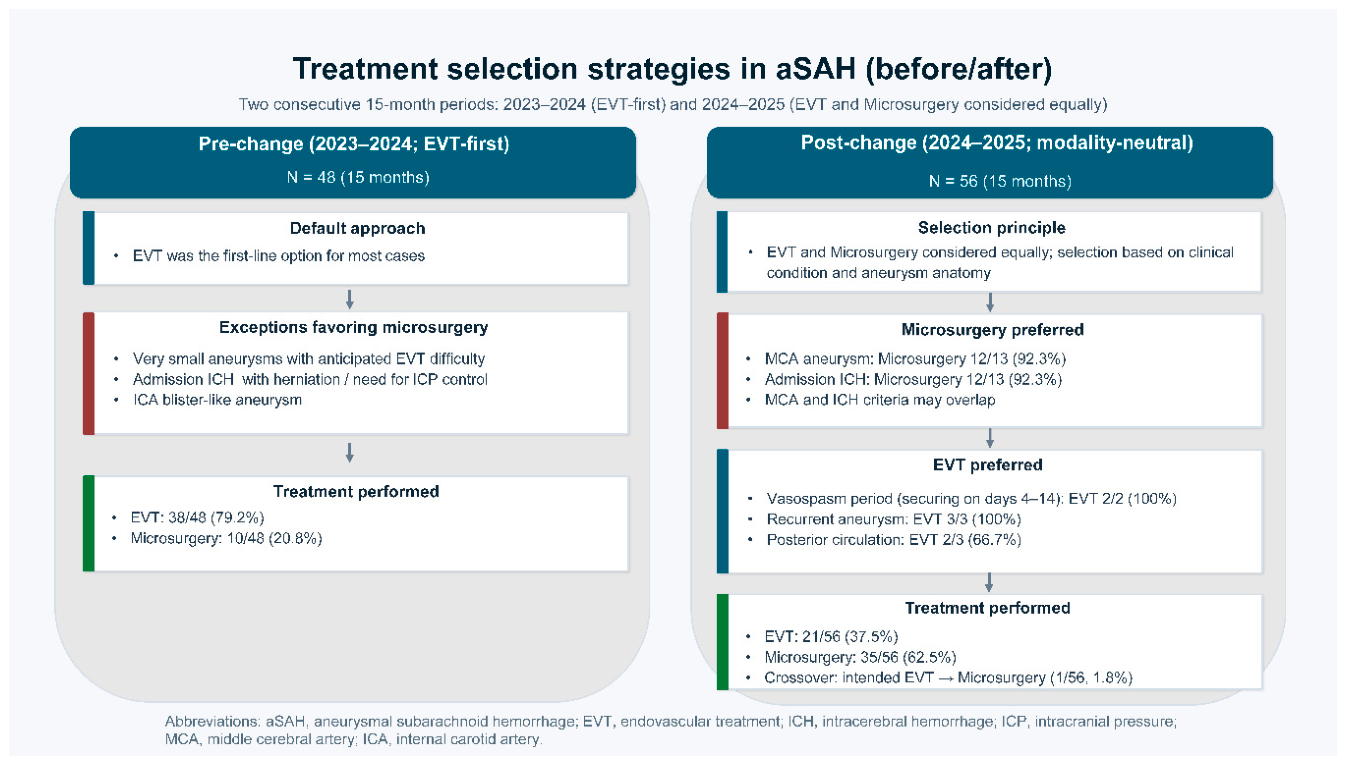
2.3. Treatment Strategy
2.4. Outcomes
2.5. Statistical Analysis
3. Results
3.1. Study Population and Treatment Selection
3.2. Baseline Characteristics
3.3. Primary Outcome (90-Day mRS 0–2)
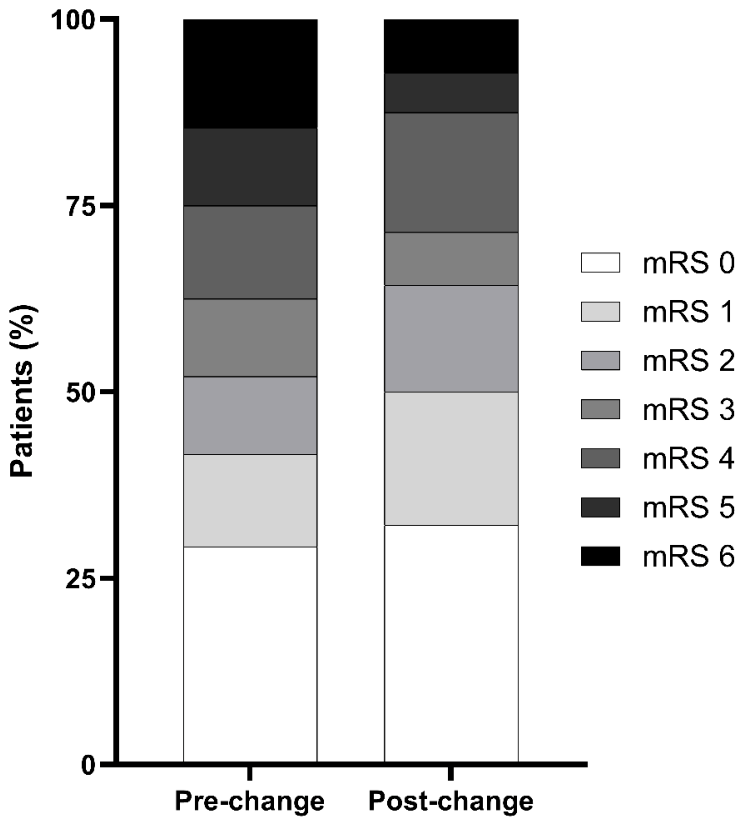
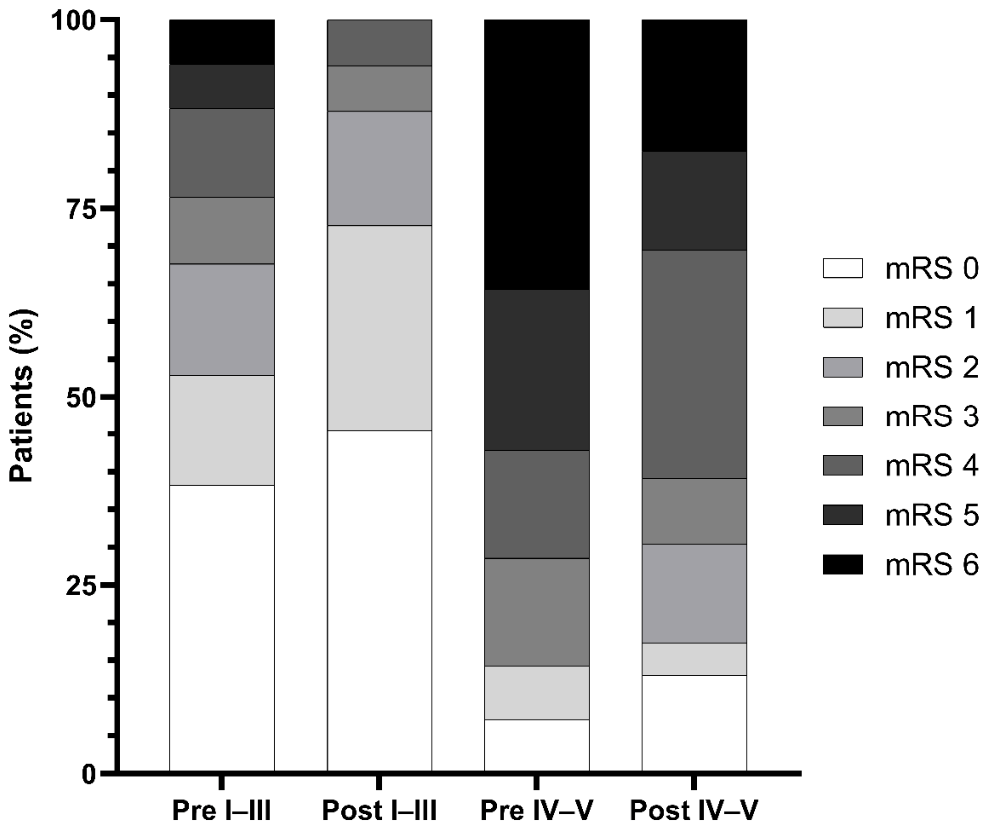
3.4. Shift Analysis of 90-Day mRS (0–6)
3.5. Secondary Outcomes and Perioperative Events
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Molyneux, A.; Kerr, R.; Stratton, I.; Sandercock, P.; Clarke, M.; Shrimpton, J.; Holman, R.; International Subarachnoid Aneurysm Trial (ISAT) Collaborative Group. International Subarachnoid Aneurysm Trial (ISAT) of Neurosurgical Clipping versus Endovascular Coiling in 2143 Patients with Ruptured Intracranial Aneurysms: A Randomised Trial. Lancet 2002, 360, 1267–1274. [Google Scholar] [CrossRef]
- Molyneux, A.J.; Kerr, R.S.C.; Yu, L.-M.; Clarke, M.; Sneade, M.; Yarnold, J.A.; Sandercock, P.; International Subarachnoid Aneurysm Trial (ISAT) Collaborative Group. International Subarachnoid Aneurysm Trial (ISAT) of Neurosurgical Clipping versus Endovascular Coiling in 2143 Patients with Ruptured Intracranial Aneurysms: A Randomised Comparison of Effects on Survival, Dependency, Seizures, Rebleeding, Subgroups, and Aneurysm Occlusion. Lancet 2005, 366, 809–817. [Google Scholar] [CrossRef]
- Hoh, B.L.; Ko, N.U.; Amin-Hanjani, S.; Chou, S.H.-Y.; Cruz-Flores, S.; Dangayach, N.S.; Derdeyn, C.P.; Du, R.; Hänggi, D.; Hetts, S.W.; et al. 2023 Guideline for the Management of Patients With Aneurysmal Subarachnoid Hemorrhage: A Guideline From the American Heart Association/American Stroke Association. Stroke 2023, 54, e314–e370. [Google Scholar] [CrossRef]
- Robba, C.; Busl, K.M.; Claassen, J.; Diringer, M.N.; Helbok, R.; Park, S.; Rabinstein, A.; Treggiari, M.; Vergouwen, M.D.I.; Citerio, G. Contemporary Management of Aneurysmal Subarachnoid Haemorrhage. An Update for the Intensivist. Intensive Care Med. 2024, 50, 646–664. [Google Scholar] [CrossRef] [PubMed]
- Mayer, S.A.; Bruder, N.; Citerio, G.; Defreyne, L.; Dubois, C.; Gupta, R.; Higashida, R.; Marr, A.; Nguyen, T.N.; Roux, S.; et al. REACT: A Randomized Trial to Assess the Efficacy and Safety of Clazosentan for Preventing Clinical Deterioration Due to Delayed Cerebral Ischemia after Aneurysmal Subarachnoid Hemorrhage. J. Neurosurg. 2025, 142, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Darsaut, T.E.; Jack, A.S.; Kerr, R.S.; Raymond, J. International Subarachnoid Aneurysm Trial—ISAT Part II: Study Protocol for a Randomized Controlled Trial. Trials 2013, 14, 156. [Google Scholar] [CrossRef]
- De Winkel, J.; Roozenbeek, B.; Dijkland, S.A.; Dammers, R.; Van Doormaal, P.-J.; Van Der Jagt, M.; Van Klaveren, D.; Dippel, D.W.J.; Lingsma, H.F. Endovascular versus Neurosurgical Aneurysm Treatment: Study Protocol for the Development and Validation of a Clinical Prediction Tool for Individualised Decision Making. BMJ Open 2022, 12, e065903. [Google Scholar] [CrossRef] [PubMed]
- Schatlo, B.; Fathi, A.-R.; Fandino, J. Management of Aneurysmal Subarachnoid Haemorrhage. Swiss Med. Wkly. 2014, 144, w13934. [Google Scholar] [CrossRef]
- Salaud, C.; Hamel, O.; Riem, T.; Desal, H.; Buffenoir, K. Management of Aneurysmal Subarachnoid Haemorrhage with Intracerebral Hematoma: Is There an Indication for Coiling First? Study of 44 Cases. Interv. Neuroradiol. 2016, 22, 5–11. [Google Scholar] [CrossRef]
- Bohnstedt, B.N.; Nguyen, H.S.; Kulwin, C.G.; Shoja, M.M.; Helbig, G.M.; Leipzig, T.J.; Payner, T.D.; Cohen-Gadol, A.A. Outcomes for Clip Ligation and Hematoma Evacuation Associated with 102 Patients with Ruptured Middle Cerebral Artery Aneurysms. World Neurosurg. 2013, 80, 335–341. [Google Scholar] [CrossRef]
- Koiso, T.; Satow, T.; Hamano, E.; Nishimura, M.; Takahashi, J.C.; Kataoka, H. Coiling for Ruptured Aneurysms in the Vasospasm Period: Safety and Efficacy Based on a Propensity Score Analysis. J. Neuroendovascular Ther. 2022, 16, 141–146. [Google Scholar] [CrossRef]
- Li, W.; Zhu, W.; Sun, X.; Liu, J.; Wang, Y.; Wang, K.; Zhang, Y.; Yang, X.; Zhang, Y. Retreatment With Flow Diverters and Coiling for Recurrent Aneurysms After Initial Endovascular Treatment: A Propensity Score-Matched Comparative Analysis. Front. Neurol. 2021, 12, 625652. [Google Scholar] [CrossRef]
- Ohkuma, H.; Shimamura, N.; Naraoka, M.; Katagai, T. Aneurysmal Subarachnoid Hemorrhage in the Elderly over Age 75: A Systematic Review. Neurol. Med.-Chir. (Tokyo) 2017, 57, 575–583. [Google Scholar] [CrossRef]
- Goldberg, J.; Schoeni, D.; Mordasini, P.; Z’Graggen, W.; Gralla, J.; Raabe, A.; Beck, J.; Fung, C. Survival and Outcome After Poor-Grade Aneurysmal Subarachnoid Hemorrhage in Elderly Patients. Stroke 2018, 49, 2883–2889. [Google Scholar] [CrossRef] [PubMed]
- da Silva Júnior, N.R.; Trivelato, F.P.; Nakiri, G.S.; Rezende, M.T.S.; de Castro-Afonso, L.H.; Abud, T.G.; Vanzin, J.R.; Manzato, L.B.; Ulhôa, A.C.; Abud, D.G.; et al. Endovascular Treatment of Residual or Recurrent Intracranial Aneurysms after Surgical Clipping. J. Cerebrovasc. Endovasc. Neurosurg. 2021, 23, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Fraser, J.F.; Heit, J.J.; Mascitelli, J.R.; Tsai, J.P. Decoding the Data: A Comment on the American Heart Association/American Stroke Association (AHA/ASA) 2023 Guideline for the Management of Patients with Aneurysmal Subarachnoid Hemorrhage. J. NeuroInterv. Surg. 2023, 15, 835–837. [Google Scholar] [CrossRef] [PubMed]
- Vergouwen, M.D.I.; Vermeulen, M.; Van Gijn, J.; Rinkel, G.J.E.; Wijdicks, E.F.; Muizelaar, J.P.; Mendelow, A.D.; Juvela, S.; Yonas, H.; Terbrugge, K.G.; et al. Definition of Delayed Cerebral Ischemia After Aneurysmal Subarachnoid Hemorrhage as an Outcome Event in Clinical Trials and Observational Studies: Proposal of a Multidisciplinary Research Group. Stroke 2010, 41, 2391–2395. [Google Scholar] [CrossRef]
- Della Pepa, G.M.; Scerrati, A.; Albanese, A.; Marchese, E.; Maira, G.; Sabatino, G. Protective Effect of External Ventricular Drainage on Cerebral Vasospasm. A Retrospective Study on Aneurysmal SAH Treated Endovascularly. Clin. Neurol. Neurosurg. 2014, 124, 97–101. [Google Scholar] [CrossRef]
- Wolf, S.; Mielke, D.; Barner, C.; Malinova, V.; Kerz, T.; Wostrack, M.; Czorlich, P.; Salih, F.; Engel, D.C.; Ehlert, A.; et al. Effectiveness of Lumbar Cerebrospinal Fluid Drain Among Patients With Aneurysmal Subarachnoid Hemorrhage: A Randomized Clinical Trial. JAMA Neurol. 2023, 80, 833. [Google Scholar] [CrossRef]
- Endo, H.; Hagihara, Y.; Kimura, N.; Takizawa, K.; Niizuma, K.; Togo, O.; Tominaga, T. Effects of Clazosentan on Cerebral Vasospasm-Related Morbidity and All-Cause Mortality after Aneurysmal Subarachnoid Hemorrhage: Two Randomized Phase 3 Trials in Japanese Patients. J. Neurosurg. 2022, 137, 1707–1717. [Google Scholar] [CrossRef]
- Firth, D. Bias Reduction of Maximum Likelihood Estimates. Biometrika 1993, 80, 27–38. [Google Scholar] [CrossRef]
- Heinze, G.; Schemper, M. A Solution to the Problem of Separation in Logistic Regression. Stat. Med. 2002, 21, 2409–2419. [Google Scholar] [CrossRef] [PubMed]
- Mehta, T.; Datta, N.; Patel, S.; Mehta, K.; Hussain, M.; Kureshi, I.; Ollenschleger, M.; Nouh, A. Trends in Endovascular Treatment of Aneurysmal Subarachnoid Hemorrhages. Interv. Neurol. 2017, 6, 236–241. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Pre-Change (2023–2024) N = 48 |
Post-Change (2024–2025) N = 56 |
p Value |
|---|---|---|---|
| Age, mean (SD), years | 65.5 (15.0) | 65.4 (14.1) | 0.960 |
| Female sex | 36 (75.0%) | 42 (75.0%) | 1.000 |
| Current smoker | 16 (33.3%) | 20 (35.7%) | 0.839 |
| Hypertension | 20 (41.7%) | 22 (39.3%) | 0.843 |
| Dyslipidemia | 12 (25.0%) | 9 (16.1%) | 0.329 |
| Diabetes mellitus | 2 (4.2%) | 7 (12.5%) | 0.172 |
| WFNS grades IV–V | 14 (29.2%) | 23 (41.1%) | 0.225 |
| Modified Fisher grades 3–4 | 30 (62.5%) | 30 (53.6%) | 0.427 |
| ICH on admission | 15 (31.2%) | 13 (23.2%) | 0.383 |
| Aneurysm location | 0.649 | ||
| ACA/ACom | 15 (31.2%) | 24 (42.9%) | |
| MCA | 15 (31.2%) | 13 (23.2%) | |
| ICA | 15 (31.2%) | 16 (28.6%) | |
| Posterior circulation | 3 (6.2%) | 3 (5.4%) | |
| Treatment modality | < 0.001 | ||
| Endovascular treatment | 38 (79.2%) | 21 (37.5%) | |
| Microsurgery | 10 (20.8%) | 35 (62.5%) |
| Variable | Contrast | Primary Analysis (Firth) aOR (95% CI) |
p Value | Sensitivity Analysis (Conventional) aOR (95% CI) |
p Value |
|---|---|---|---|---|---|
| Study period | Post-change vs. pre-change |
3.82 (1.31–12.79) | 0.009 | 4.41 (1.43–15.95) | 0.009 |
| Age | Per 10 years | 0.43 (0.25–0.66) | <0.001 | 0.39 (0.22–0.63) | <0.001 |
| Pre-morbid mRS | ≥2 vs. 0–1 | 1.12 (0.14–7.79) | 1.000 | 1.06 (0.11–8.31) | 0.955 |
| WFNS grade | IV–V vs. I–III | 0.05 (0.01–0.15) | <0.001 | 0.03 (0.01–0.12) | <0.001 |
| Outcome | Time Window | Pre-Change (2023–2024) N = 48 |
Post-Change (2024–2025) N = 56 |
p Value |
|---|---|---|---|---|
| Perioperative complications (any) | Intraoperative or within 30 days |
7 (14.6%) | 6 (10.7%) | 0.569 |
| Delayed cerebral ischemia | During hospitalization | 8 (16.7%) | 7 (12.5%) | 0.586 |
| CSF drainage (EVD or ELD) |
Used within 0–14 days from onset | 30 (62.5%) | 39 (69.6%) | 0.533 |
| Clazosentan use | Administered within 0–14 days from onset | 34 (70.8%) | 41 (73.2%) | 0.829 |
| Shunt-dependent hydrocephalus | Requiring permanent CSF diversion | 13 (27.1%) | 16 (28.6%) | 1.000 |
| Aneurysm recurrence | Imaging-confirmed within 3 months after aneurysm securing | 7 (14.6%) | 4 (7.1%) | 0.338 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




