Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
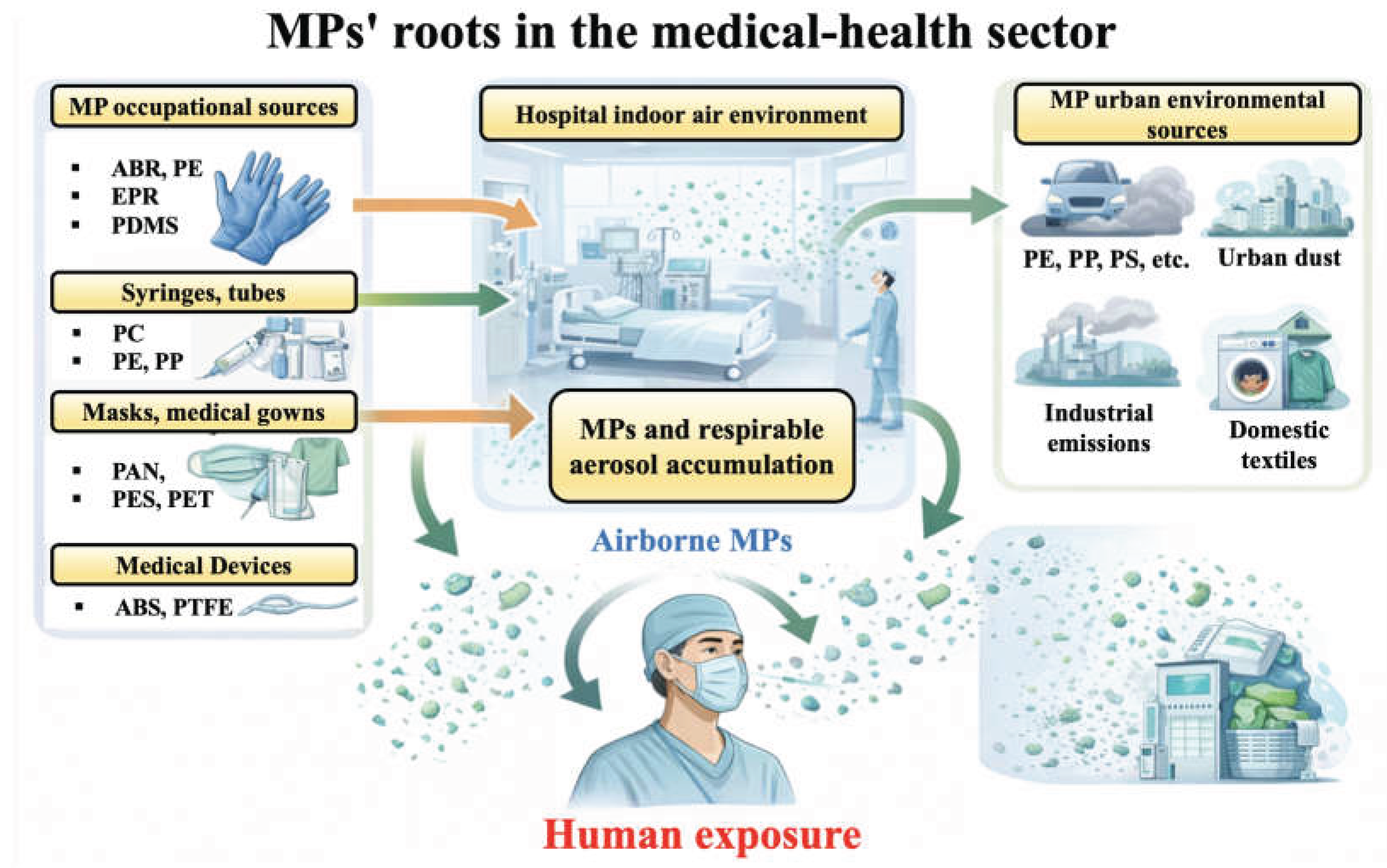
1. Introduction
2. Materials and Methods
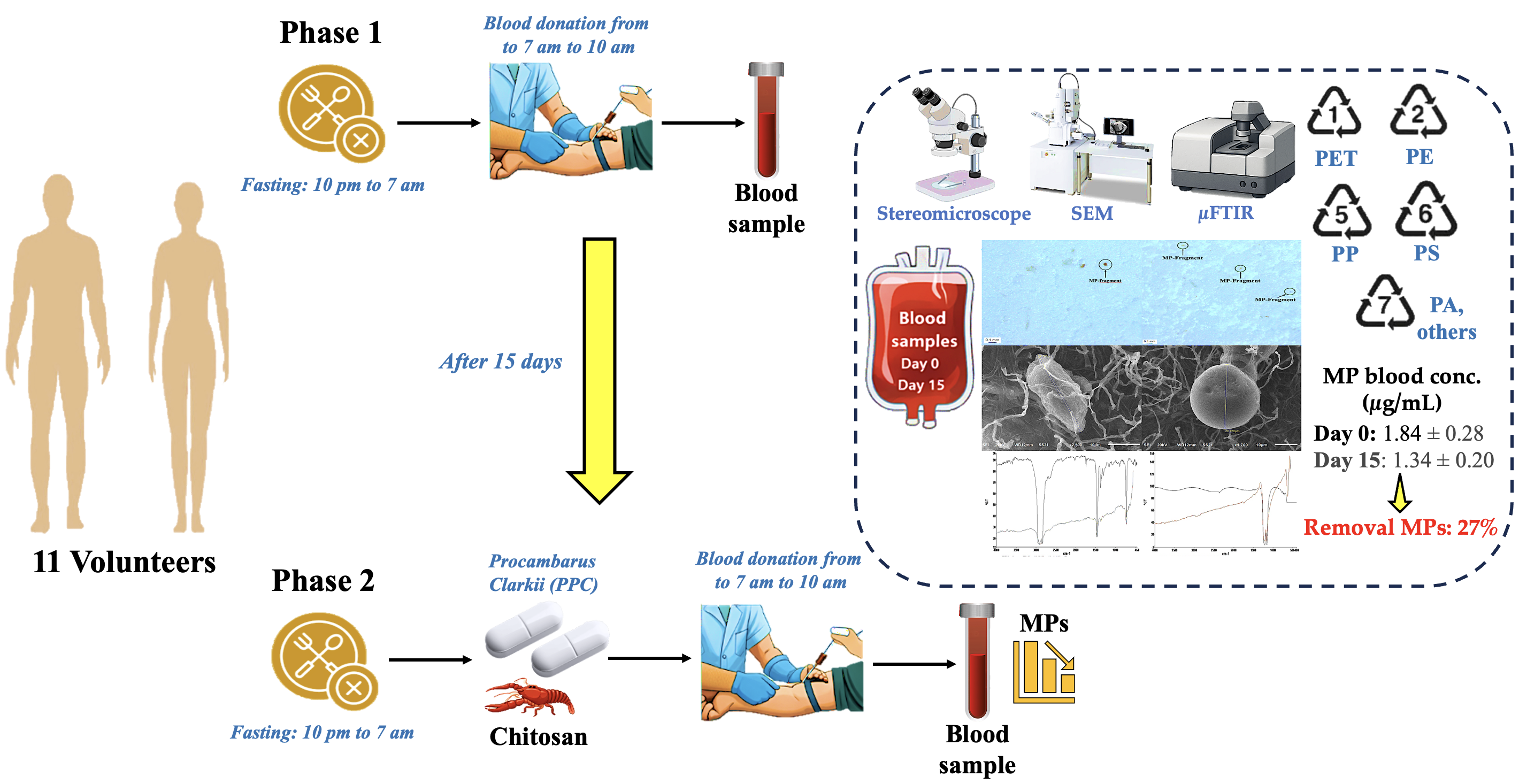
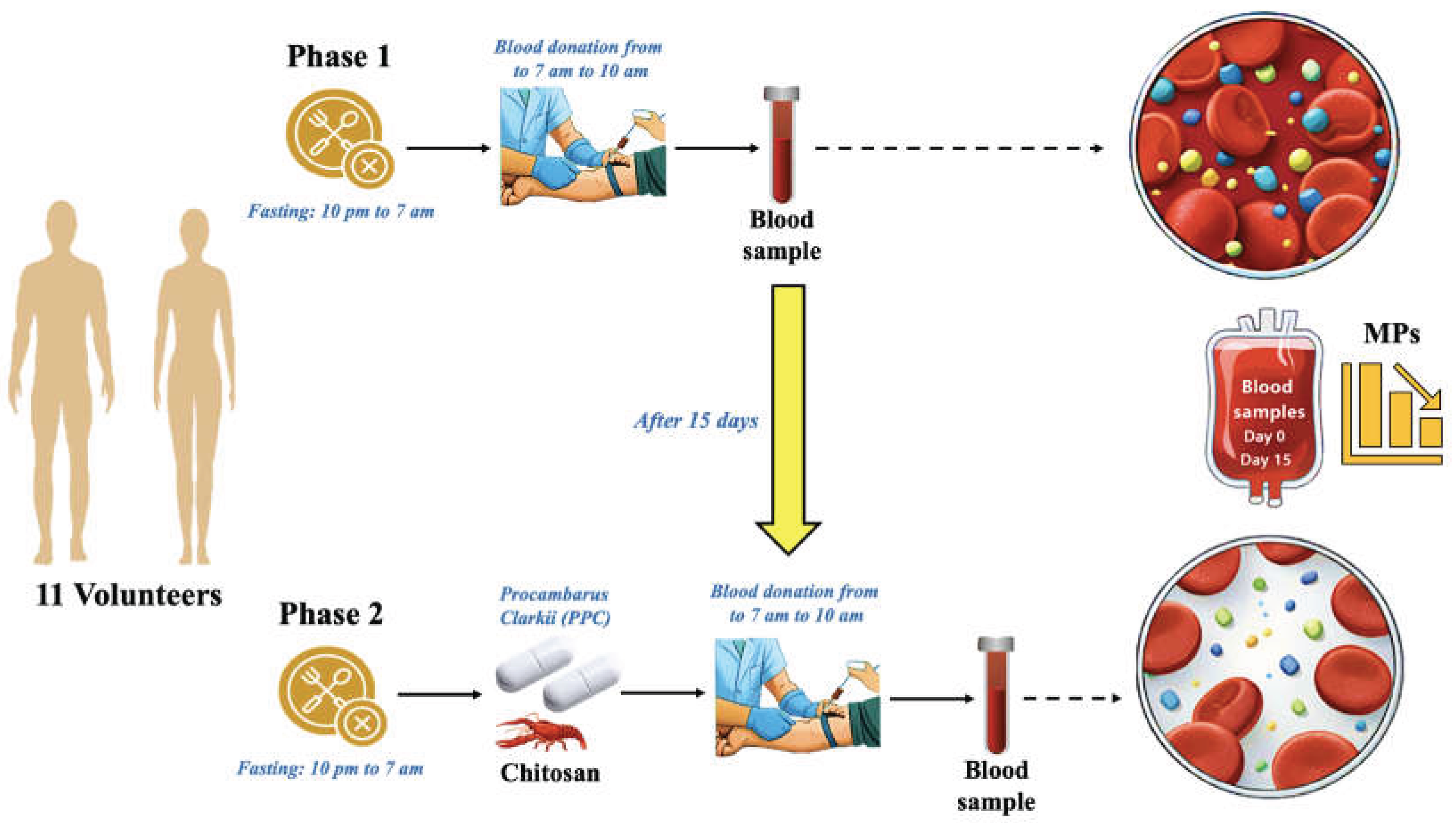
2.1. Experiment Blueprint
2.2. Gathering Samples
2.3. Blood Samples Pretreatment
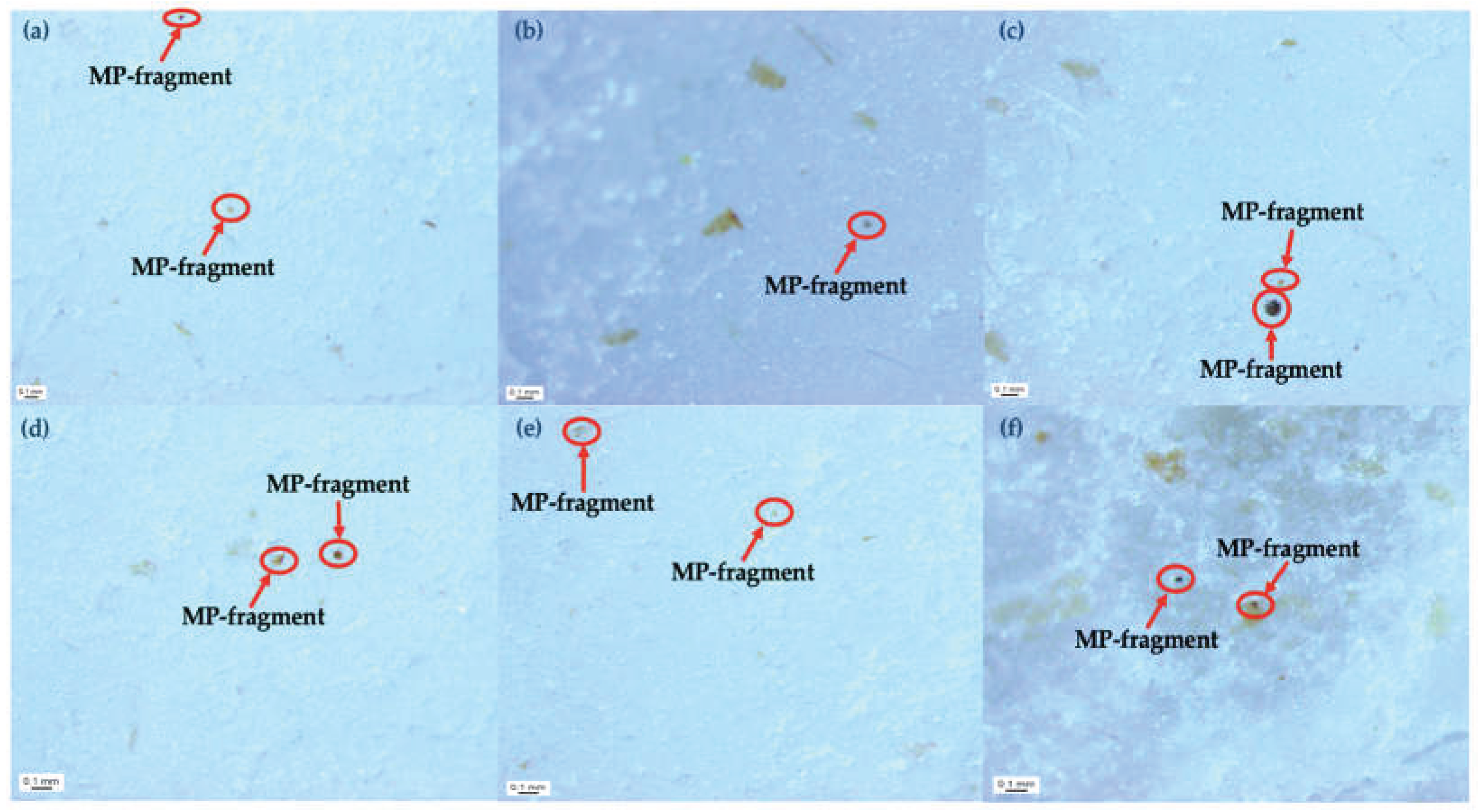
2.4. Microplastic Analysis
2.5. Qyality Assurance and Quality Control (QA/QC)
- Wearing fibre-free lab coats, face masks, and nitrile gloves that have been previously cleaned will stop the operator or clothing from producing particles. Before every use, it was advised that all lab tools, such as forceps, funnels, and filters, be thoroughly cleaned with Type I ultrapure water. Laminar flow should then be used to dry any remaining residue.
- Blank controls: To assess any potential unintentional contamination throughout the process, procedural blanks, or control filters devoid of samples, were processed concurrently with the experimental samples.
- To reduce the filters' exposure to the environment, keep them in closed or shielded systems at all times. These methods provide a high level of assurance that the MPs seen in the samples are solely from the blood samples under investigation and are not the result of contamination from outside sources or experimental process artifacts
2.6. Statistics
2.7. Ethics
3. Results
3.1. Comparative MP Analysis between Phase 1 (baseline) and Phase 2 (PCC) in Cohort Study
3.2. MP Concentrations in Blood and MRT
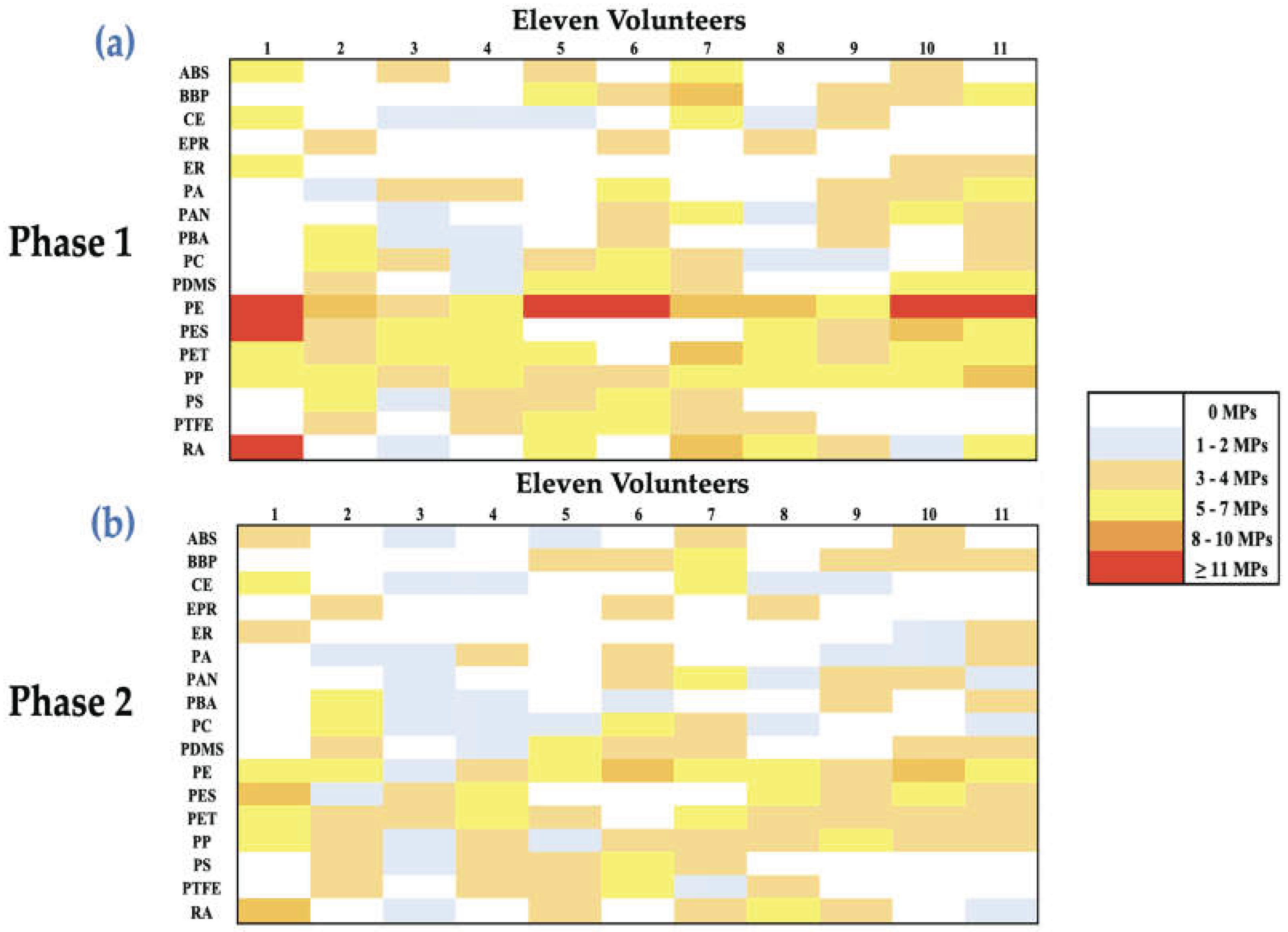
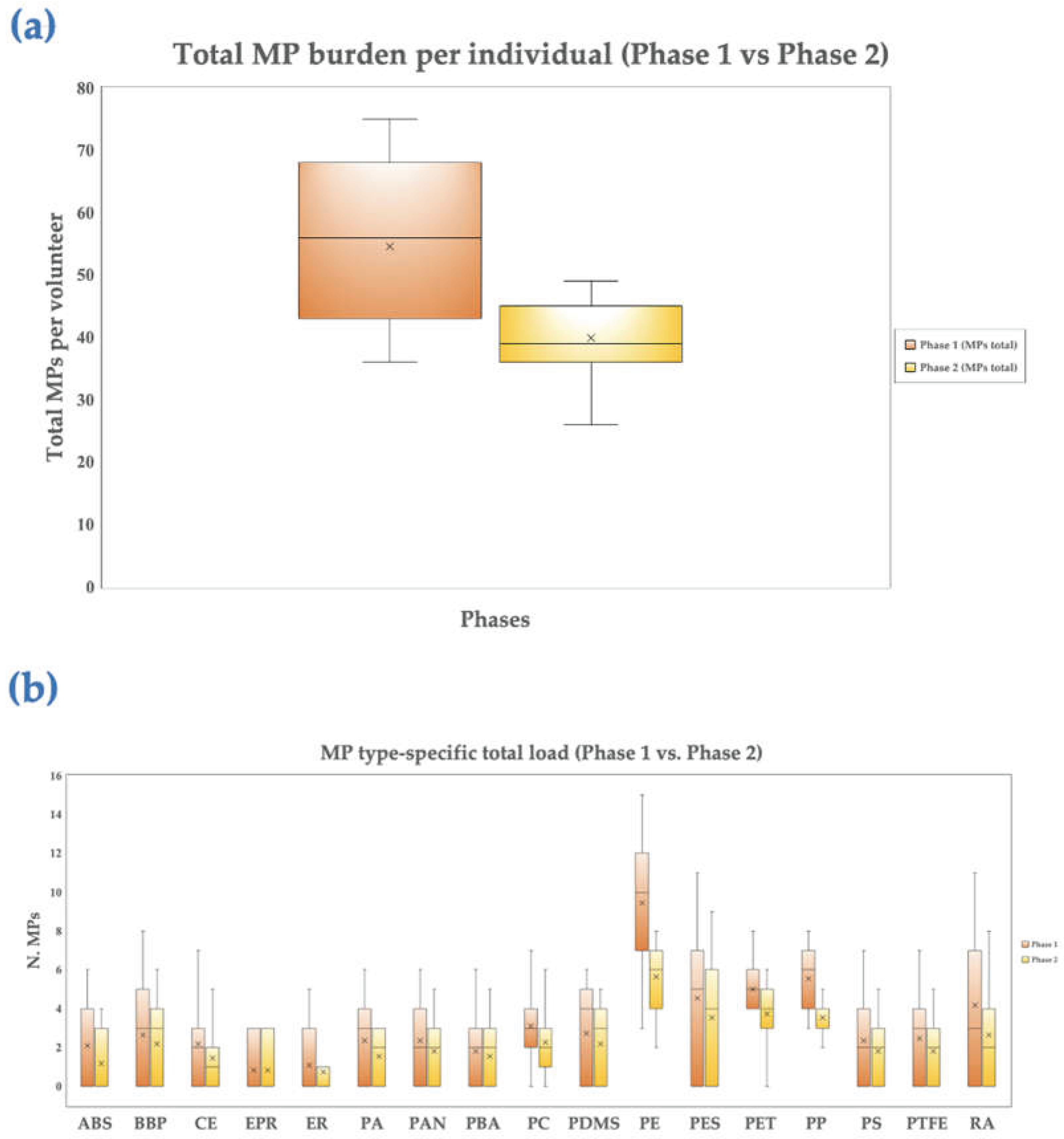
3.3. Heatmap and Boxplots Analysis
4. Discussion
4.1. Size-dependent Effects of PCC on Circulating MPs
4.2. Polymer-specific Effects of PCC on Circulating MPs
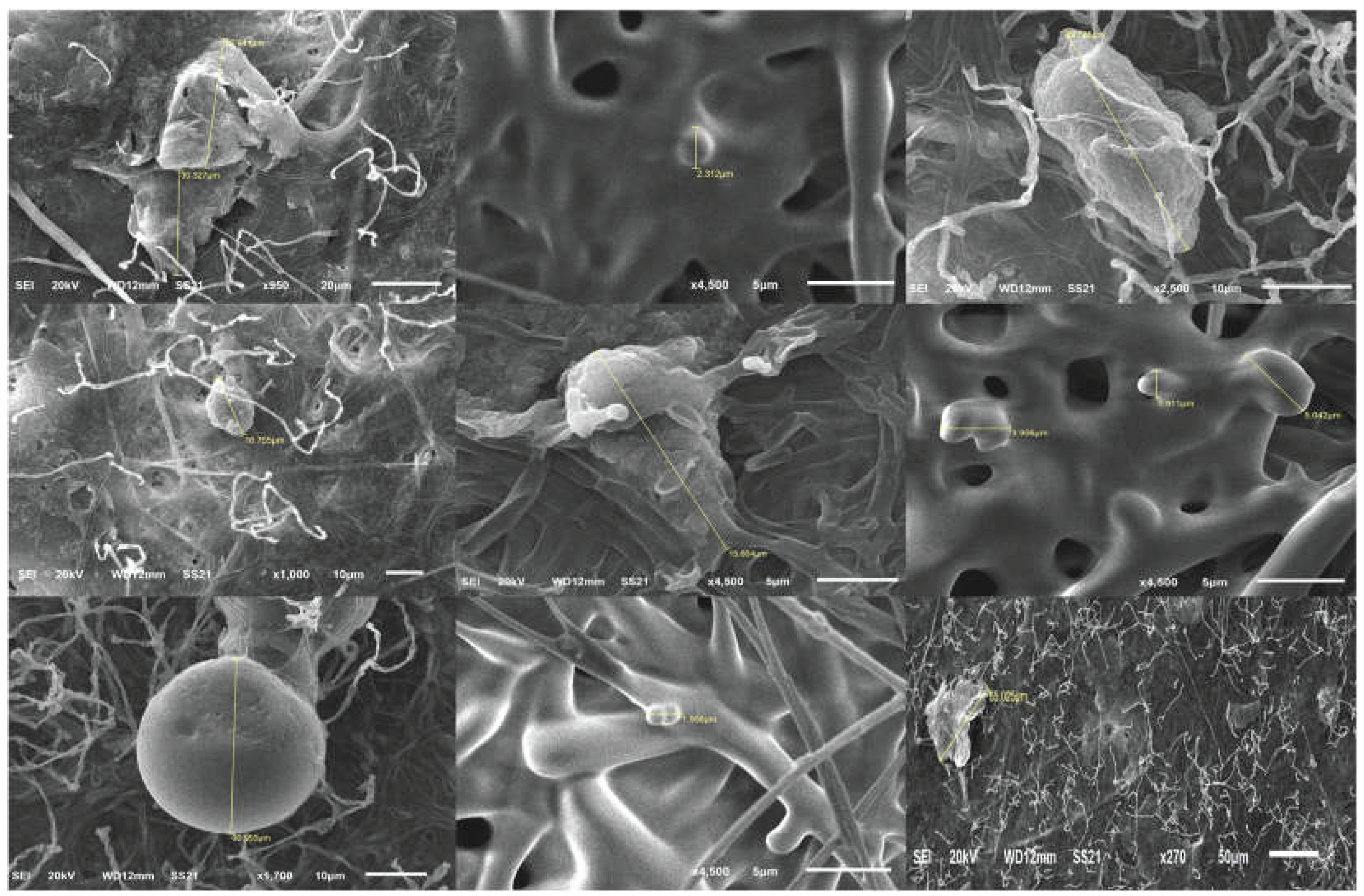
4.3. SEM-based Characterization of the Morphology and Toxicological Relevance of Circulating MPs
4.4. Heatmap and Boxplot Analysis of Polymer-Specific Changes in Circulating MPs following PCC Intervention
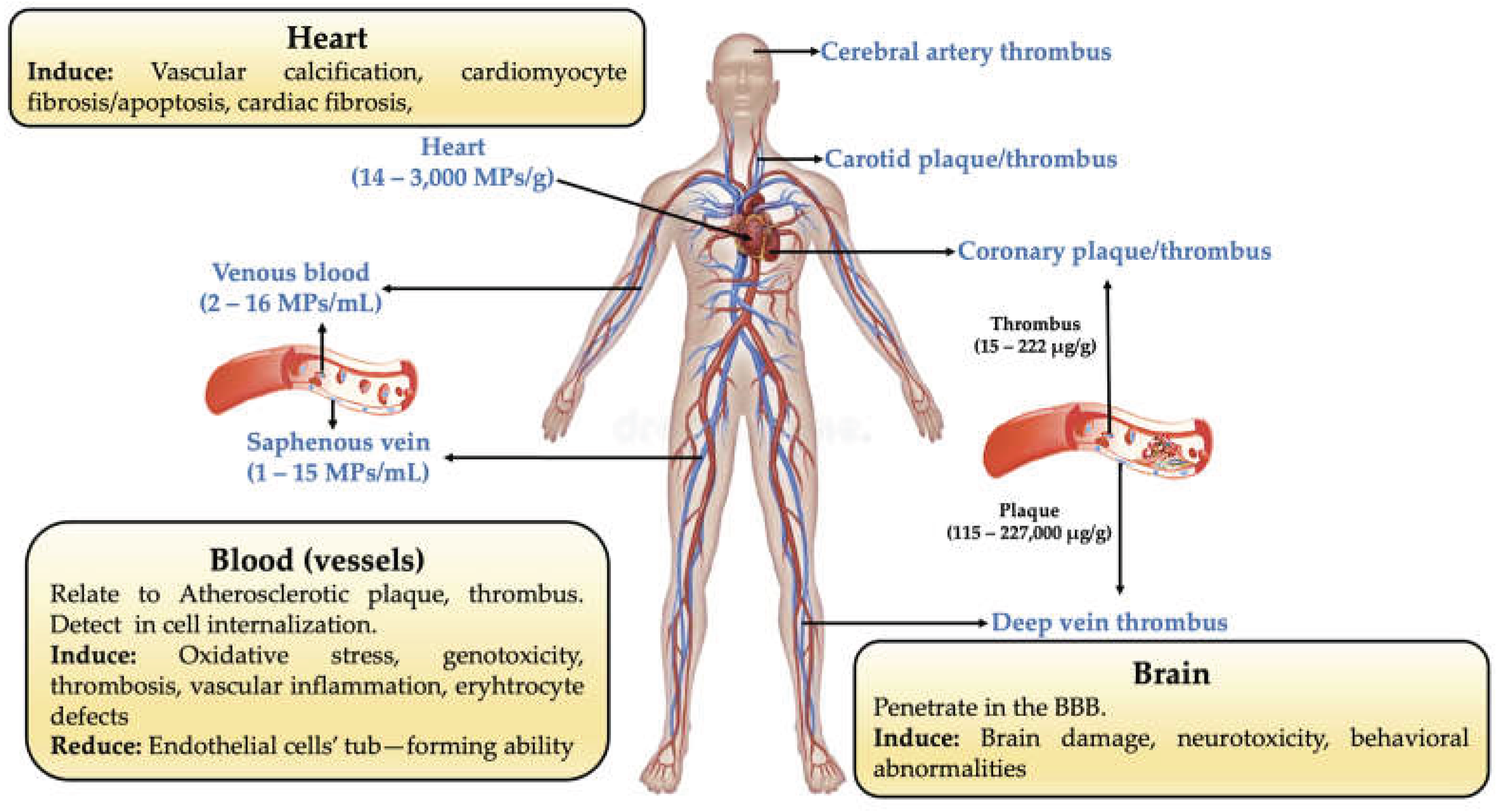
4.5. Systemic Distribution of MPs and their Potential Human Health Risk (i.e., Cardiovascular and Neurovascular Pathology)
4.6. Comparative Analysis of Internalised MPs: Our Findings vs Global Literature Benchmarks
5. Putative Limitations of the Study
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABS | Acrylonitrile Butadiene Styrene |
| AFM-Raman | Atomic Force Microscopy-Raman |
| BBB | Blood-Brain Barrier |
| BBP | Butyl Benzyl Phthalate |
| BMI | Body Mass Index |
| CE | Cellulose (cellulose acetate) |
| CNS | Central Nervous System |
| cP | Centipoise |
| CSF | Cerebrospinal Fluid |
| DDA | Degree of Deacetylation |
| EPR | Ethylene Propylene Rubber |
| ER | Epoxy Resin |
| FDR | Benjamini-Hochberg False Discovery Rate |
| GDPR | General Data Protection Regulations |
| GLPs | Good Field and Laboratory Procedures |
| GUT | Gastrointestinal Tract |
| 1H-NMR | Proton Nuclear Magnetic Resonance |
| IAPS | International Agency for Pharma Standard Supplements |
| LC-MS/MS | Liquid Chromatography-tandem Mass Spectrometry |
| LDIR | Laser Direct Infrared |
| MPs | Microplastics |
| MRT | Mean Residence Time |
| NPs | Nanoplastics |
| PA | Polyamide |
| PAHs | Polycyclic Aromatic Hydrocarbons |
| PAN | Polyacrlonitrile |
| PBA | Polu(butyl acrylate) |
| PC | Polycarbonate |
| PCC | Procambarus clarkii Chitosan |
| PDMS | Polydimethylsiloxane |
| PE | Polyethylene |
| PES | Polyester |
| PET | Polyethylene Terephthalate |
| PLA | Polylactic Acid |
| PP | Polypropylene |
| PPE | Personal Protective Equipment |
| PS | Polystyrene |
| PTFE | Polytetrafluoroethylene |
| Py-GC-MS | Pyrolisis-Gas Chromatography-Mass Spectrometry |
| RA | Rayon |
| ROS | Reactive Oxygen Species |
| SEM | Scanning Electron Microscopy |
| Vd | Distribution Volume |
| µFTIR | Micro-Fourier Transform Infrared Spectroscopy |
| µRaman | Micro-Raman Spectroscopy |
References
- Europe, Plastics. Plastics –The Fast Facts 2025 Plastics Europe, Brussels. 2025. Available online: https://plasticseurope.org/knowledge-hub/plastics-the-fast-facts-2025/ (accessed on 11 February 2026).
- Lee, D. W.; Jung, J.; Park, S. A.; Lee, Y.; Kim, J.; Han, C.; Hong, Y. C. Microplastic particles in human blood and their association with coagulation markers. Sci. Rep. 2024, 14(1), 30419. [Google Scholar] [CrossRef]
- Leslie, H. A.; Van Velzen, M. J.; Brandsma, S. H.; Vethaak, A. D.; Garcia-Vallejo, J. J.; Lamoree, M. H. Discovery and quantification of plastic particle pollution in human blood. Environ. Int. 2022, 163, 107199. [Google Scholar] [CrossRef]
- Tu, P.; Xie, J.; Li, X.; Ma, X.; Xing, M.; Niu, H.; Xu, J. L. What Drives Microplastic Exposure in Human Blood and Feces? Machine Learning Reveals Potential Key Influencing Factors. Environ. Sci. Technol. 2025. [Google Scholar] [CrossRef]
- Roslan, N. S.; Lee, Y. Y.; Ibrahim, Y. S.; Anuar, S. T.; Yusof, K. M. K. K.; Lai, L. A.; Brentnall, T. Detection of microplastics in human tissues and organs: A scoping review. J. Glob. Health 2024, 14, 04179. [Google Scholar] [CrossRef]
- Xu, Z.; Huang, X.; Zhu, Z.; Liang, F.; Hu, H.; Cai, T.; Zhang, W. Elevated blood microplastics and their potential association with Parkinson’s disease. J. Hazard. Mater. 2025, 500, 140431. [Google Scholar] [CrossRef]
- Casella, C.; Cornelli, U.; Ballaz, S.; Zanoni, G.; Merlo, G.; Ramos-Guerrero, L. Plastic smell: a review of the hidden threat of airborne micro and nanoplastics to human health and the environment. Toxics 2025, 13(5), 387. [Google Scholar] [CrossRef] [PubMed]
- Prata, J. C. Airborne microplastics: consequences to human health? Environ. Pollut. 2018, 234, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Stock, V.; Laurisch, C.; Franke, J.; Dönmez, M. H.; Voss, L.; Böhmert, L.; Sieg, H. Uptake and cellular effects of PE, PP, PET and PVC microplastic particles. Toxicol. In Vitro 2021, 70, 105021. [Google Scholar] [CrossRef] [PubMed]
- Jenner, L. C.; Rotchell, J. M.; Bennett, R. T.; Cowen, M.; Tentzeris, V.; Sadofsky, L. R. Detection of microplastics in human lung tissue using μFTIR spectroscopy. Sci. Total Environ. 2022, 831, 154907. [Google Scholar] [CrossRef]
- Leonard, S. V.; Liddle, C. R.; Atherall, C. A.; Chapman, E.; Watkins, M.; Calaminus, S. D.; Rotchell, J. M. Microplastics in human blood: Polymer types, concentrations and characterisation using μFTIR. Environ. Int. 2024, 188, 108751. [Google Scholar] [CrossRef]
- Casella, C.; Ballaz, S. J. Genotoxic and neurotoxic potential of intracellular nanoplastics: A review. J. Appl. Toxicol. 2024, 44(11), 1657–1678. [Google Scholar] [CrossRef]
- Casella, C.; Cornelli, U. Pancreatic Cancer in Relation to Food Expenditure: Difference Between Northern and Southern Italian Regions. Green Health 2026, 2(1), 4. [Google Scholar] [CrossRef]
- Cornelli, U.; Casella, C.; Belcaro, G.; Cesarone, M. R.; Marucci, S.; Rondanelli, M.; Zanoni, G. Definition of Emerging Microplastic Syndrome Based on Clinical and Epidemiological Evidence: A Narrative Review. Microplastics 2025, 4(4), 93. [Google Scholar] [CrossRef]
- Casella, C.; Dondi, D.; Vadivel, D. Do microplastics (MPs) and nanoplastics (NPs) directly contribute to human carcinogenesis? Environ. Pollut. 2025, 127343. [Google Scholar] [CrossRef]
- Brits, M.; Van Velzen, M. J.; Sefiloglu, F. Ö; Scibetta, L.; Groenewoud, Q.; Garcia-Vallejo, J. J.; Lamoree, M. H. Quantitation of micro and nanoplastics in human blood by pyrolysis-gas chromatography–mass spectrometry. Microplast. Nanoplast 2024, 4(1), 12. [Google Scholar] [CrossRef]
- Casella, C.; Cornelli, U.; Ballaz, S.; Recchia, M.; Zanoni, G.; Ramos-Guerrero, L. Preliminary study on PCC-chitosan’s ability to enhance microplastic excretion in human stools from healthy volunteers. Foods 2025, 14(13), 2190. [Google Scholar] [CrossRef] [PubMed]
- Gallaher, C. M.; Munion, J.; Hesslink, R., Jr.; Wise, J.; Gallaher, D. D. Cholesterol reduction by glucomannan and chitosan is mediated by changes in cholesterol absorption and bile acid and fat excretion in rats. J. Nutr. 2000, 130(11), 2753–2759. [Google Scholar] [CrossRef] [PubMed]
- Saito, Y.; Hinoi, T.; Adachi, T.; Miguchi, M.; Niitsu, H.; Kochi, M.; Ohdan, H. Synbiotics suppress colitis-induced tumorigenesis in a colon-specific cancer mouse model. PLoS One 2019, 14(6), e0216393. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X. Y.; Dai, A. L.; Zhang, X. K.; Kuroiwa, K.; Kodaira, R.; Shimosaka, M.; Okazaki, M. Purification and characterization of chitosanase and exo-β-D-glucosaminidase from a koji mold, Aspergillus oryzae IAM2660. Biosci. Biotechnol. Biochem. 2000, 64(9), 1896–1902. [Google Scholar] [CrossRef]
- Belcaro, G.; Cornelli, U.; Cox, D.; Dugall, M.; Cesarone, M. R.; Ledda, A.; Cotellese, R. Intestinal fat absorption shifting by polyglucosamine biopolymer: control of lipids and reduction of progression of early subclinical atherosclerosis. Minerva Gastroenterol. 2023. [Google Scholar] [CrossRef]
- Choi, Y. J.; Kim, E. J.; Piao, Z.; Yun, Y. C.; Shin, Y. C. Purification and characterization of chitosanase from Bacillus sp. strain KCTC 0377BP and its application for the production of chitosan oligosaccharides. Appl. Environ. Microbiol. 2004, 70(8), 4522–4531. [Google Scholar] [CrossRef] [PubMed]
- Konagaya, Y.; Tsuchiya, C.; Sugita, H. Purification and characterization of chitinases from Clostridium sp. E-16 isolated from the intestinal tract of the South American sea lion (Otaria flavescens). Lett. Appl. Microbiol. 2006, 43(2), 187–193. [Google Scholar] [CrossRef] [PubMed]
- Omar, B.A.; Elmasry, R.; Eita, A.; Soliman, M.M.; El-Tahan, A.M.; Sitohy, M. Upgrading the preparation of high-quality chitosan from Procambarus clarkii wastes over the traditional isolation of shrimp chitosan. Saudi J. Biol. Sci. 2022, 29, 911–919. [Google Scholar] [CrossRef]
- Pavlov, G.M.; Gosteva, A.A. Current analysis of Huggins and Kraemer plots for determining the intrinsic viscosity of macro-molecules and corresponding dimensionless parameters. Polym. Sci. 2023, 64, 586–590. [Google Scholar] [CrossRef]
- Casella, C.; Sol, D.; Laca, A.; Díaz, M. Microplastic retention in secondary sewage sludge: characterization and influence of solid concentration. Appl. Sci. 2025, 15(7), 3557. [Google Scholar] [CrossRef]
- Field, D. T.; Green, J. L.; Bennett, R.; Jenner, L. C.; Sadofsky, L. R.; Chapman, E.; Rotchell, J. M. Microplastics in the surgical environment. Environ. Int. 2022, 170, 107630. [Google Scholar] [CrossRef]
- Liu, D.; Shimizu, M. Ingesting chitosan can promote excretion of microplastics. Sci. Rep. 2025, 15(1), 14041. [Google Scholar] [CrossRef]
- Sun, H.; Su, X.; Mao, J.; Liu, Y.; Li, G.; Du, Q. Microplastics in maternal blood, fetal appendages, and umbilical vein blood. Ecotoxicol. Environ. Saf. 2024, 287, 117300. [Google Scholar] [CrossRef]
- Rangrazi, A.; Bonyadi, Z.; Sarkhosh, M. Investigation of airborne microplastics emission and characteristics in hospital laundry environments. Sci. Rep. 2026. [Google Scholar] [CrossRef]
- Shruti, V. C.; Pérez-Guevara, F.; Elizalde-Martínez, I.; Kutralam-Muniasamy, G. Reusable masks for COVID-19: a missing piece of the microplastic problem during the global health crisis. Mar. Pollut. Bull. 2020, 161, 111777. [Google Scholar] [CrossRef]
- Liu, M.; Xu, H.; Feng, R.; Gu, Y.; Bai, Y.; Zhang, N.; Cao, J. Chemical composition and potential health risks of tire and road wear microplastics from light-duty vehicles in an urban tunnel in China. Environ. Pollut. 2023, 330, 121835. [Google Scholar] [CrossRef]
- Sun, X.; Song, R.; Liu, J.; Yan, S.; Li, Y.; Jin, X.; Su, H. Characterization of airborne microplastics at different workplaces of the poly (ethylene: propylene: diene)(EPDM) rubber industry. Environ. Sci. Pollut. Res. 2023, 30(32), 78839–78848. [Google Scholar] [CrossRef]
- Abdulrahman, A. Investigating the link between microplastic exposure (benzyl butyl phthalate) and neurodegenerative diseases using high-performance computational toxicology. Toxicol. Res. 2025, 14(1), tfae211. [Google Scholar] [CrossRef]
- Li, Z.; Xu, T.; Peng, L.; Tang, X.; Chi, Q.; Li, M.; Li, S. Polystyrene nanoplastics aggravates lipopolysaccharide-induced apoptosis in mouse kidney cells by regulating IRE1/XBP1 endoplasmic reticulum stress pathway via oxidative stress. J. Cell. Physiol. 2023, 238(1), 151–164. [Google Scholar] [CrossRef]
- Liu, K.; Wang, X.; Fang, T.; Xu, P.; Zhu, L.; Li, D. Source and potential risk assessment of suspended atmospheric microplastics in Shanghai. Sci. Total Environ. 2019, 675, 462–471. [Google Scholar] [CrossRef]
- Mehta, N.; Kozielska, B.; Lunagaria, J.; Ladva, K. D. Characterization and exposure assessment of indoor microplastics in hospital indoor settled dust: First insides from India. Build. Environ. 2025, 279, 113065. [Google Scholar] [CrossRef]
- Clarke, M. W.; Burnett, J. R.; Croft, K. D. Vitamin E in human health and disease. Crit. Rev. Clin. Lab. Sci. 2008, 45(5), 417–450. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Hu, X.; Qin, L.; Yin, D. Evaluating the effect of different modified microplastics on the availability of polycyclic aromatic hydrocarbons. Water Res. 2020, 170, 115290. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M. D.; Elanjickal, A. I.; Mankar, J. S.; Krupadam, R. J. Assessment of cancer risk of microplastics enriched with polycyclic aromatic hydrocarbons. J. Hazard. Mater. 2020, 398, 122994. [Google Scholar] [CrossRef]
- Picone, D.; Carista, A.; Piano, G. M. G. L.; Manna, O. M.; Burgio, S.; Bucchieri, F.; Fucarino, A. Airborne microplastics: a Trojan horse for respiratory dysfunction and multiorgan damage. J. Biol. Res. 2025, 98. [Google Scholar] [CrossRef]
- Wen, J.; Lin, Y. Invisible invaders: unveiling the carcinogenic threat of microplastics and nanoplastics in colorectal cancer-a systematic review. Front. Public Health 2025, 13, 1653245. [Google Scholar] [CrossRef]
- Marfella, R.; Prattichizzo, F.; Sardu, C.; Fulgenzi, G.; Graciotti, L.; Spadoni, T.; Paolisso, G. Microplastics and nanoplastics in atheromas and cardiovascular events. New Engl. J. Med. 2024, 390(10), 900–910. [Google Scholar] [CrossRef] [PubMed]
- Nihart, A. J.; Garcia, M. A.; El Hayek, E.; Liu, R.; Olewine, M.; Kingston, J. D.; Campen, M. J. Bioaccumulation of microplastics in decedent human brains. Nat. Med. 2025, 31(4), 1114–1119. [Google Scholar] [CrossRef]
- Rondanelli, M.; Perna, S.; Della Porta, M.; Lombardoni, F.; Patelli, Z.; Nichetti, M.; Cazzola, R. A randomized Double-Blind Placebo-Controlled clinical study to evaluate the effect on the weight of a medical device with polyglucosamine L112 in a group of overweight and obese subjects. Nutrients 2023, 15(16), 3516. [Google Scholar] [CrossRef]
- Stock, V.; Böhmert, L.; Lisicki, E.; Block, R.; Cara-Carmona, J.; Pack, L. K.; Lampen, A. Uptake and effects of orally ingested polystyrene microplastic particles in vitro and in vivo. Arch. Toxicol. 2019, 93(7), 1817–1833. [Google Scholar] [CrossRef] [PubMed]
- Wright, S. L.; Kelly, F. J. Plastic and human health: a micro issue? Environ. Sci. Technol. 2017, 51(12), 6634–6647. [Google Scholar] [CrossRef]
- Koelmans, A. A.; Bakir, A.; Burton, G. A.; Janssen, C. R. Microplastic as a vector for chemicals in the aquatic environment: critical review and model-supported reinterpretation of empirical studies. Environ. Sci. Technol. 2016, 50(7), 3315–3326. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Joshi, A. S.; Singh, H.; Mijakovic, I. Medical importance and pharmacokinetics of gold nanoparticles in the human body. Mol. Cancer 2025, 24(1), 252. [Google Scholar] [CrossRef]
- Casella, C.; Cornelli, U.; Zanoni, G.; Moncayo, P.; Ramos-Guerrero, L. Health risks from microplastics in intravenous infusions: evidence from Italy, Spain, and Ecuador. Toxics 2025, 13(7), 597. [Google Scholar] [CrossRef]
- Luo, Y.; Xu, X.; Yin, Q.; Liu, S.; Xing, M.; Jin, X.; Zhang, H. Mapping micro (nano) plastics in various organ systems: Their emerging links to human diseases? TrAC Trends Anal. Chem. 2025, 183, 118114. [Google Scholar] [CrossRef]
- Marfella, R.; Prattichizzo, F.; Barbieri, M.; Paolisso, P.; Scisciola, L.; Basilicata, M. G.; Paolisso, G. Microplastics and atherosclerosis: Mechanisms. Ann. Rev. Pharmacol. Toxicol. 2026, 66(1), 369–390. [Google Scholar] [CrossRef]
- Gural, A.; Barshtein, G. Are Polymeric Microparticles Dangerous for Red Blood Cells? Appl. Sci. 2026, 16(5), 2302. [Google Scholar] [CrossRef]
- Abdel-Zaher, S.; Mohamed, M. S.; Sayed, A. E. D. H. Hemotoxic effects of polyethylene microplastics on mice. Front. Physiol. 2023, 14, 1072797. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. Q.; Lykaki, M.; Alrajoula, M. T.; Markiewicz, M.; Kraas, C.; Kolbe, S.; Stolte, S. Microplastics from textile origin–emission and reduction measures. Green Chem. 2021, 23(15), 5247–5271. [Google Scholar] [CrossRef]
- Iovino, P.; Fenti, A.; Belmonte, M.; Marfella, L. V.; Tortorella, G.; Paolisso, G. Analytical techniques for detecting micro-and nanoplastics in blood and vascular tissues: Strengths and limitations. Polish Heart J. Kardiol. Pol. 2026. [Google Scholar] [CrossRef]
- Akhbarizadeh, R.; Dobaradaran, S.; Nabipour, I.; Tangestani, M.; Abedi, D.; Javanfekr, F.; Zendehboodi, A. Abandoned Covid-19 personal protective equipment along the Bushehr shores, the Persian Gulf: an emerging source of secondary microplastics in coastlines. Mar. Pollut. Bull. 2021, 168, 112386. [Google Scholar] [CrossRef]
- Khan, M. T.; Shah, I. A.; Hossain, M. F.; Akther, N.; Zhou, Y.; Khan, M. S.; Ihsanullah, I. Personal protective equipment (PPE) disposal during COVID-19: An emerging source of microplastic and microfiber pollution in the environment. Sci. Total Environ. 2023, 860, 160322. [Google Scholar] [CrossRef] [PubMed]
- Saqib, M.; Fan, Y.; Hao, R.; Zhang, B. Optical imaging of nanoscale electrochemical interfaces in energy applications. Nano Energy 2021, 90, 106539. [Google Scholar] [CrossRef]
- Hamimed, S.; Kamah, A.; Kamah, F. Z.; Aggoun, K. Polyethylene terephthalate and polypropylene microplastic bioaccumulation in human blood and cancerous tissues in Algerian cases. Naunyn-Schmiedeberg Arch. Pharmacol. 2026, 1–17. [Google Scholar] [CrossRef]
- Wang, T.; Yi, Z.; Tan, Y.; Huang, Y.; Li, T.; Lu, W.; Guo, S. Detection of micro-and nanoplastics in cerebrospinal fluid and blood: Implications for brain diseases. Ecotoxicol. Environ. Saf. 2026, 309, 119614. [Google Scholar] [CrossRef]
- Nardella, F.; Brits, M.; van Velzen, M. J.; Scibetta, L.; Durkin, A.; Vermeulen, R.; Lamoree, M. H. Advancing pyrolysis-gas chromatography-mass spectrometry for the accurate quantification of micro-and nanoplastics in human blood. Microplast. Nanoplast. 2025, 5(1), 48. [Google Scholar] [CrossRef]
- Guan, Q.; Jiang, J.; Huang, Y.; Wang, Q.; Liu, Z.; Ma, X.; Xia, Y. The landscape of micron-scale particles including microplastics in human enclosed body fluids. J. Hazard. Mater. 2023, 442, 130138. [Google Scholar] [CrossRef] [PubMed]
- Salvia, R.; Rico, L. G.; Bradford, J. A.; Ward, M. D.; Olszowy, M. W.; Martínez, C.; Petriz, J. Fast-screening flow cytometry method for detecting nanoplastics in human peripheral blood. MethodsX 2023, 10, 102057. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Xie, E.; Du, Z.; Peng, Z.; Han, Z.; Li, L.; Yang, X. Detection of various microplastics in patients undergoing cardiac surgery. Environ. Sci. Technol. 2023, 57(30), 10911–10918. [Google Scholar] [CrossRef] [PubMed]
- Çobanoğlu, H.; Belivermiş, M.; Sıkdokur, E.; Kılıç, Ö; Çayır, A. Genotoxic and cytotoxic effects of polyethylene microplastics on human peripheral blood lymphocytes. Chemosphere 2021, 272, 129805. [Google Scholar] [CrossRef]
| Volunteers | Sex | Age (Year) | Height (m) | Weigth (Kg) | BMI (Kg/m2) | Area of residence | Professional sector |
|---|---|---|---|---|---|---|---|
| 001 | M | 42 | 1.82 | 92 | 27.8 | Metropolitan area | Healthcare sector |
| 002 | F | 43 | 1.65 | 52 | 19.1 | ||
| 003 | F | 19 | 1.60 | 45 | 17.6 | ||
| 004 | F | 18 | 1.70 | 60 | 20.8 | ||
| 005 | M | 46 | 1.80 | 110 | 34.0 | ||
| 006 | M | 49 | 1.70 | 100 | 34.6 | ||
| 007 | M | 52 | 1.75 | 85 | 27.8 | ||
| 008 | F | 48 | 1.68 | 76 | 26.9 | ||
| 009 | F | 53 | 1.60 | 54 | 21.1 | ||
| 010 | M | 35 | 1.62 | 50 | 19.1 | ||
| 011 | M | 25 | 1.75 | 63 | 20.6 | ||
| MEAN | 39 | 1.70 | 72 | 24.5 | - | ||
| SD | 13 | 0.08 | 22 | 6.1 | - | ||
| Volunteers | Sex | Age (Year) | Height (m) | Weigth (Kg) | BMI (Kg/m2) | Area of residence | Professional sector |
|---|---|---|---|---|---|---|---|
| 012 | F | 20 | 1.64 | 55 | 20.4 | Metropolitan area | Healthcare sector |
| 013 | M | 43 | 1.75 | 81 | 26.4 | ||
| 014 | M | 26 | 1.81 | 78 | 23.8 | ||
| 015 | M | 51 | 1.74 | 83 | 27.4 | ||
| 016 | F | 35 | 1.60 | 62 | 24.2 | ||
| 017 | F | 29 | 1.71 | 63 | 21.5 | ||
| 018 | F | 47 | 1.62 | 58 | 22.1 | ||
| 019 | M | 32 | 1.88 | 80 | 23.2 | ||
| 020 | F | 46 | 1.67 | 66 | 23.7 | ||
| 021 | M | 24 | 1.84 | 84 | 24.8 | ||
| MEAN | 35 | 1.73 | 74 | 24.5 | - | ||
| SD | 11 | 0.10 | 12 | 6.1 | - | ||
| Volunteer | Phase 1 N. MPs/10 mL |
Phase 2 N. MPs/10 mL |
Phase 1 MP Conc. (µg/mL) |
Phase 2 MP Conc. (µg/mL) |
MP Removal (%) | MP Type* |
|---|---|---|---|---|---|---|
| 001 | 69 | 49 | 2.05 | 1.31 | 35.6 | ABS, CE, ER, PA, PAN, PE, PES, PET, PP, RA |
| 002 | 51 | 40 | 1.67 | 1.30 | 22.2 | EPR, PA, PBA, PC, PDMS, PE, PES, PET, PP, PS, PTFE |
| 003 | 36 | 26 | 1.54 | 1.14 | 26.0 | ABS, CE, PA, PAN, PBA, PC, PE, PES, PET, PP, PS, RA |
| 004 | 40 | 36 | 1.74 | 1.51 | 13.2 | CE, PA, PBA, PC, PDMS, PE, PES, PET, PP, PS, PTFE |
| 005 | 57 | 36 | 2.23 | 1.41 | 36.8 | ABS, BBP, CE, PC, PDMS, PE, PET, PP, PS, PTFE, RA |
| 006 | 61 | 45 | 1.51 | 1.12 | 25.8 | BBP, EPR, PA, PAN, PBA, PC, PDMS, PE, PP, PS, PTFE |
| 007 | 75 | 49 | 1.89 | 1.23 | 34.9 | ABS, BBP, CE, PAN, PC, PDMS, PE, PET, PP, PS, PTFE, RA |
| 008 | 44 | 37 | 1.94 | 1.68 | 13.4 | CE, EPR, PAN, PC, PE, PES, PET, PP, PTFE, RA |
| 009 | 43 | 38 | 1.40 | 1.16 | 17.1 | BBP, CE, PA, PAN, PBA, PC, PE, PES, PET, PP, RA |
| 010 | 56 | 44 | 2.12 | 1.67 | 21.2 | ABS, BBP, ER, PA, PAN, PDMS, PE, PES, PET, PP, RA |
| 011 | 68 | 39 | 2.10 | 1.19 | 43.3 | BBP, ER, PA, PAN, PBA, PC, PDMS, PE, PES, PET, PP, RA |
| MEAN | 55 | 40 | 1.84 | 1.34 | 26.3 | - |
| SD | 13 | 7 | 0.28 | 0.20 | 10.1 | - |
| *ABS: Acrylonitrile Butadiene Styrene; BBP: Butyl Benzyl Phthalate; CE: Cellulose (acetate cellulose); EPR: Ethylene propylene rubber; ER: Epoxy resin; PA: Polyamide; PAN: Polyacrylonitrile; PBA: Poly(butyl acrylate); PC: Polycarbonate; PDMS: Polydimethylsiloxane; PE: Polyethylene; PES: Polyester; PET: Polyethylene terephthalate; PP: Polypropylene; PS: Polystyrene; PTFE: Polytetrafluoroethylene (Teflon); RA: Rayon | ||||||
| Volunteer | Phase 1 N. MPs/10 mL |
Phase 2 N. MPs/10 mL |
Phase 1 MP Conc. (µg/mL) |
Phase 2 MP Conc. (µg/mL) |
MP Removal (%) | MP Type* |
|---|---|---|---|---|---|---|
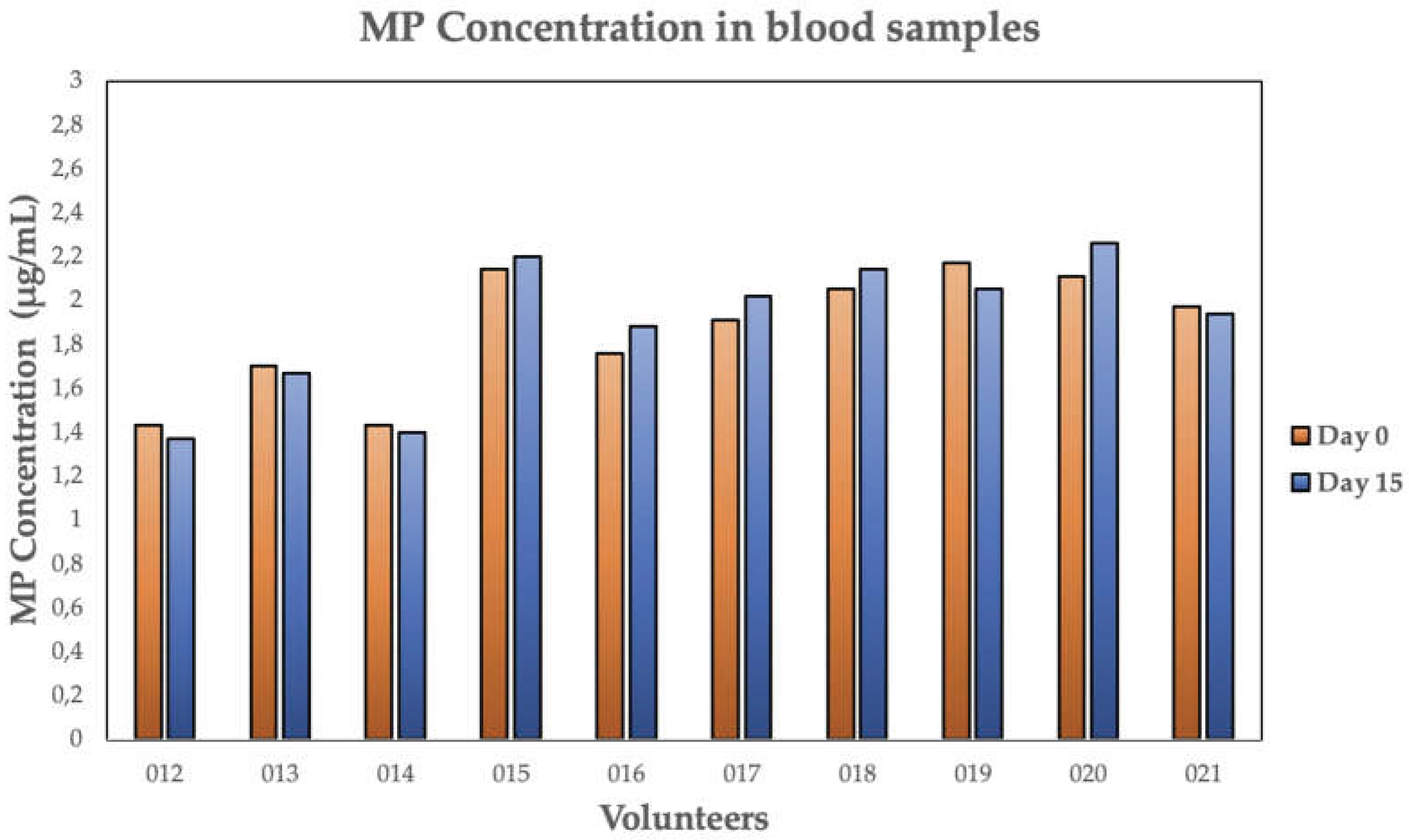
| 012 | 48 | 46 | 1.43 | 1.37 | 4.2 | ABS, CE, EPR, ER, PA, PE, PES, PET, RA |
| 013 | 57 | 56 | 1.70 | 1.67 | 1.8 | BBP, CE, PA, PBA, PC, PE, PES, PET, PP, PS |
| 014 | 48 | 47 | 1.43 | 1.40 | 2.1 | CE, PA, PAN, PBA, PC, PE, PES, PET, PP, PS |
| 015 | 72 | 74 | 2.14 | 2.20 | - | ABS, EPR, PA, PBA, PC, PDMS, PE, PET, PP, PS, PTFE, RA |
| 016 | 59 | 63 | 1.76 | 1.88 | - | ABS, BBP, CE, EPR, PDMS, PE, PET, PP, PTFE, RA |
| 017 | 64 | 68 | 1.91 | 2.02 | - | BBP, EPR, PA, PAN, PBA, PC, PDMS, PE, PES, PP, PS, PTFE |
| 018 | 69 | 72 | 2.05 | 2.14 | - | ABS, BBP, CE, PC, PDMS, PE, PET, PP, PS, PTFE, RA |
| 019 | 73 | 69 | 2.17 | 2.05 | 5,5 | ABS, BBP, CE, EPR, PAN, PC, PE, PES, PET, PP, PTFE, RA |
| 020 | 71 | 76 | 2.11 | 2.26 | - | PA, PAN, PBA, PC, PDMS PE, PES, PET, PP, PS, RA |
| 021 | 66 | 65 | 1.97 | 1.94 | 1.5 | ABS, EPR, PA, PBA, PC, PDMS, PE, PET, PP, RA |
| MEAN | 63 | 64 | 1.87 | 1.85 | 3.0 | - |
| SD | 9 | 11 | 0.28 | 0.30 | 1.8 | - |
| *ABS: Acrylonitrile Butadiene Styrene; BBP: Butyl Benzyl Phthalate; CE: Cellulose (acetate cellulose); EPR: Ethylene propylene rubber; ER: Epoxy resin; PA: Polyamide; PAN: Polyacrylonitrile; PBA: Poly(butyl acrylate); PC: Polycarbonate; PDMS: Polydimethylsiloxane; PE: Polyethylene; PES: Polyester; PET: Polyethylene terephthalate; PP: Polypropylene; PS: Polystyrene; PTFE: Polytetrafluoroethylene (Teflon); RA: Rayon | ||||||
| Healthy Volunteer |
MP Concentration (µg/mL) | % Decrease in MP concentrations | MRT (days) | |
|---|---|---|---|---|
| Phase 1 | Phase 2 | |||
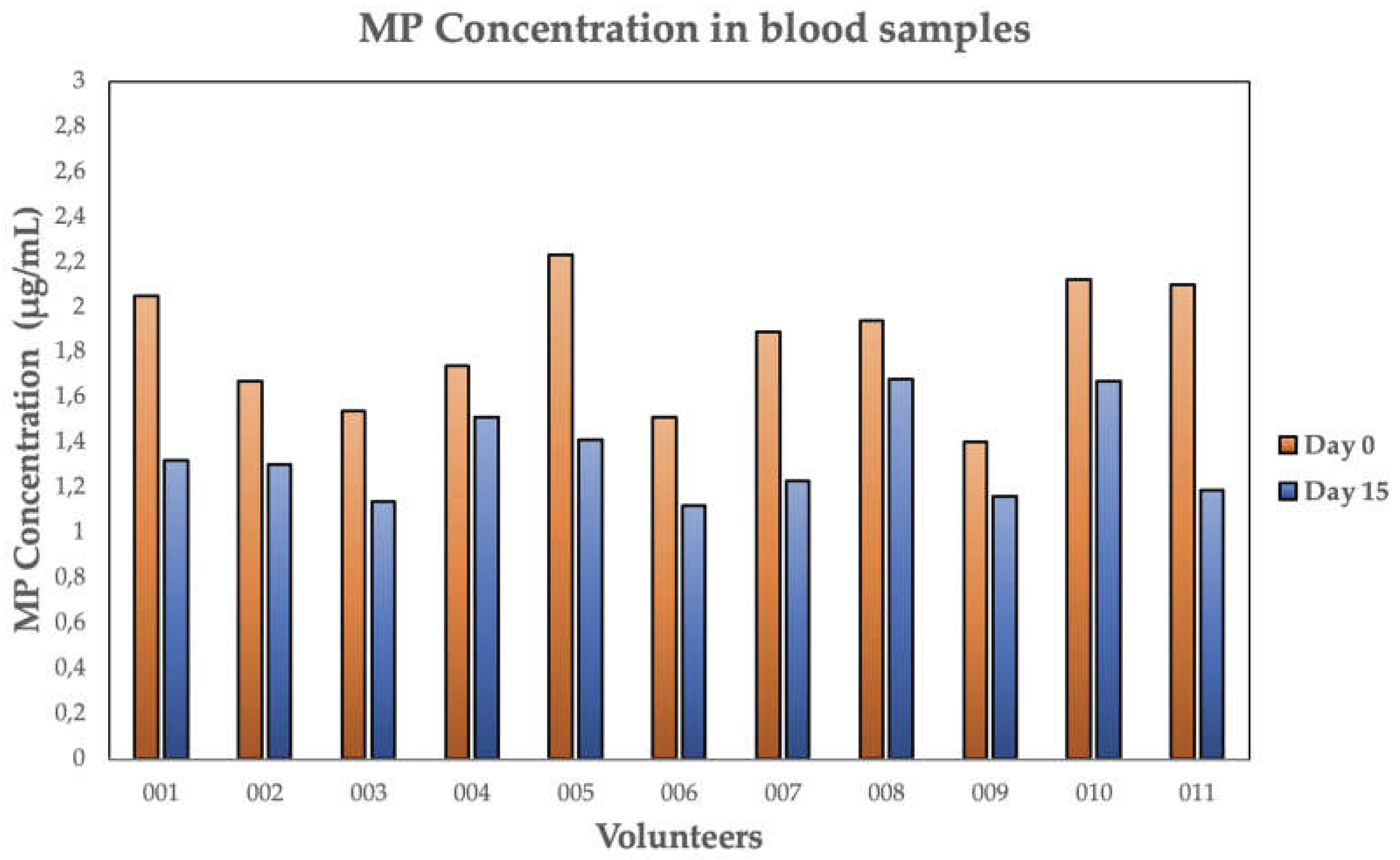
| 001 | 2.05 | 1.32 | 35.6 | 34.1 |
| 002 | 1.67 | 1.30 | 22.2 | 59.6 |
| 003 | 1.54 | 1.14 | 26.0 | 49.7 |
| 004 | 1.74 | 1.51 | 13.2 | 106.4 |
| 005 | 2.23 | 1.41 | 36.8 | 32.8 |
| 006 | 1.51 | 1.12 | 25.8 | 50.3 |
| 007 | 1.89 | 1.23 | 34.9 | 35.0 |
| 008 | 1.94 | 1.68 | 13.4 | 105.0 |
| 009 | 1.40 | 1.16 | 17.1 | 79.4 |
| 010 | 2.21 | 1.67 | 21.2 | 62.8 |
| 011 | 2.10 | 1.19 | 43.3 | 26.5 |
| MEAN | 1.84 | 1.34* | 26.3 | 58.3 |
| SD | 0.28 | 0.20 | 10.1 | 28.0 |
| *Statistical significance was determined using Tukey’s post-hoc test; p < 0.01 indicates a significant difference between Phase 1 and Phase 2. | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




