Submitted:
20 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Search Strategy
2.2. Eligibility Criteria
2.3. Data Extraction and Quality Assessment
2.4. Data Synthesis
3. Results
3.1. Study Selection
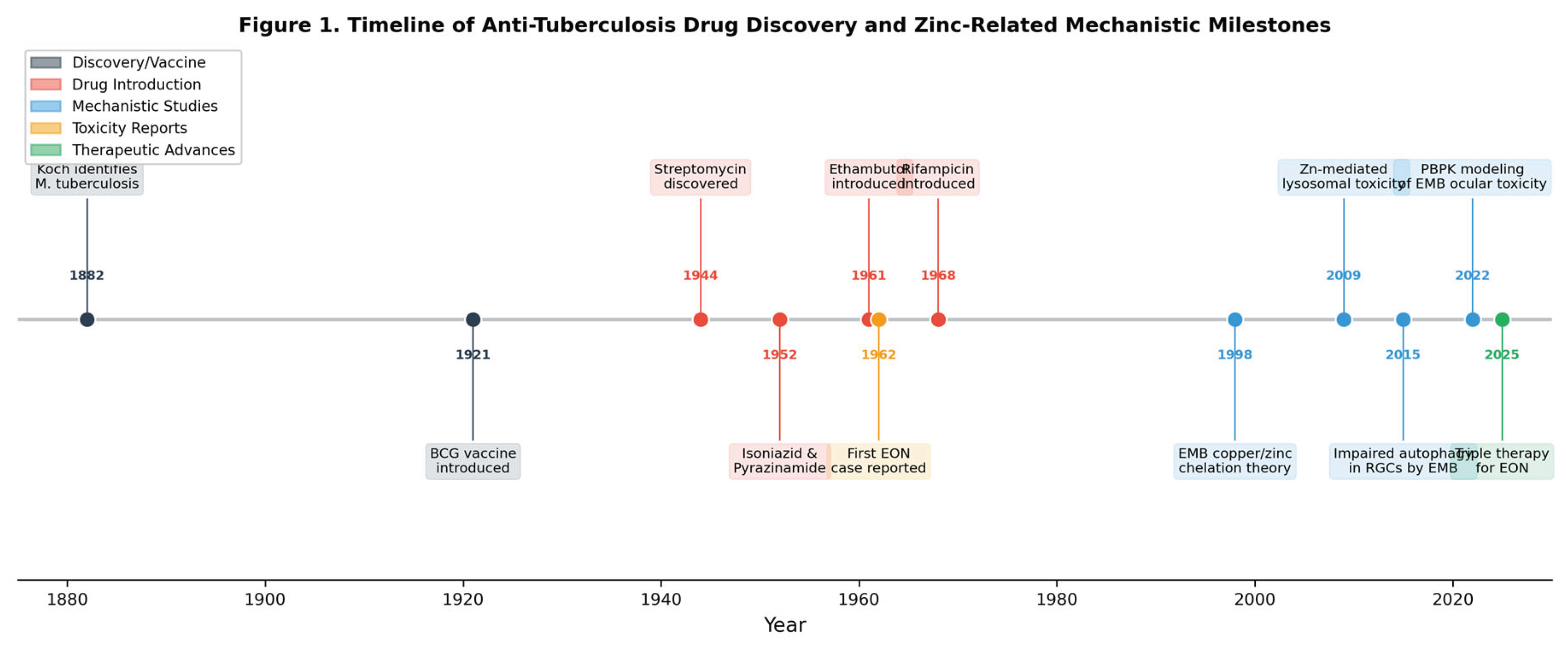
3.2. Historical Evolution of TB Drug Discovery and Zinc Science
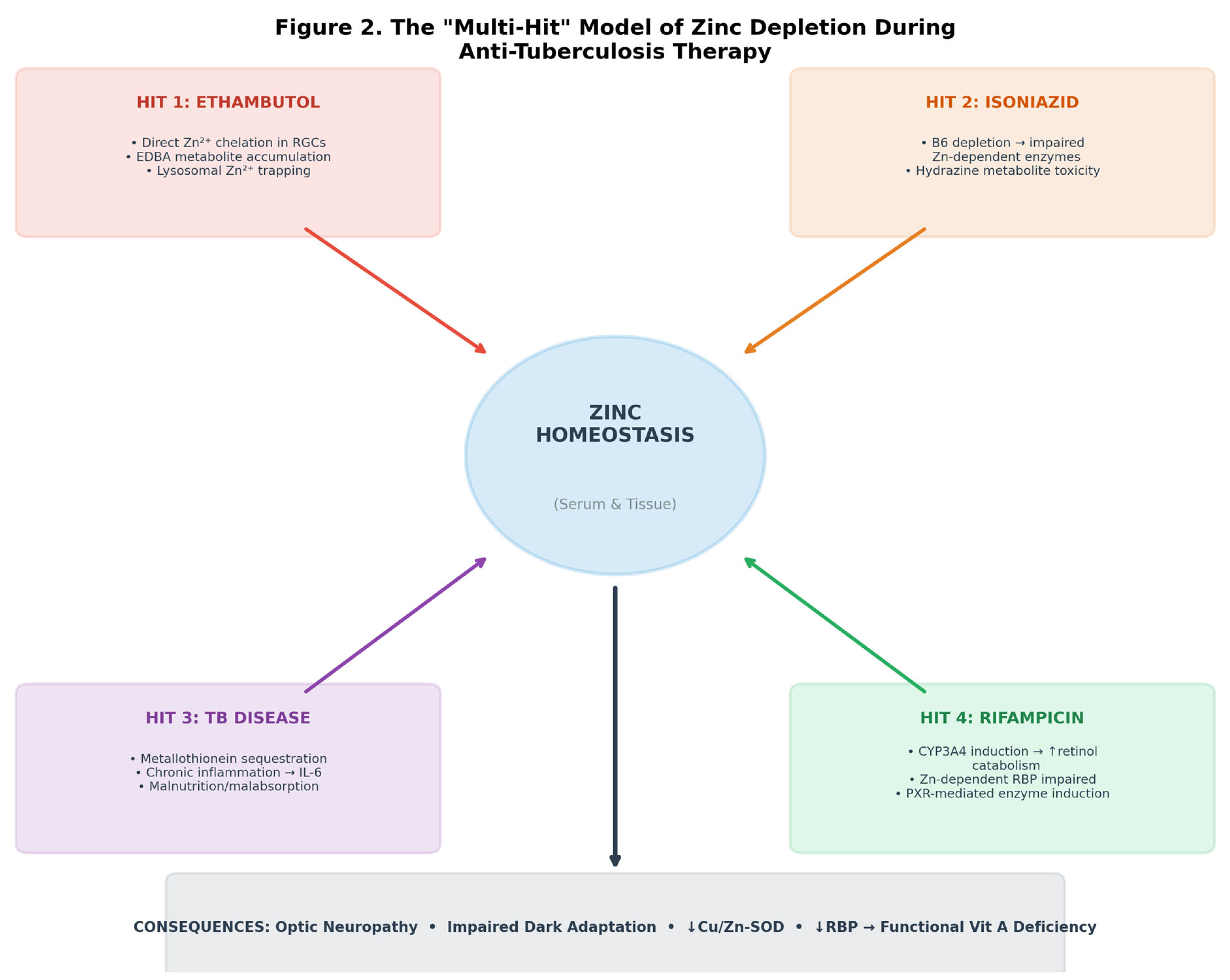
3.3. The Multi-Hit Model of Zinc Depletion
3.3.1. Hit 1: Ethambutol — Direct Zinc Chelation
3.3.2. Hit 2: Isoniazid — Indirect Zinc Impairment via Pyridoxine Depletion
3.3.3. Hit 3: Tuberculosis Disease — Inflammation-Driven Zinc Sequestration
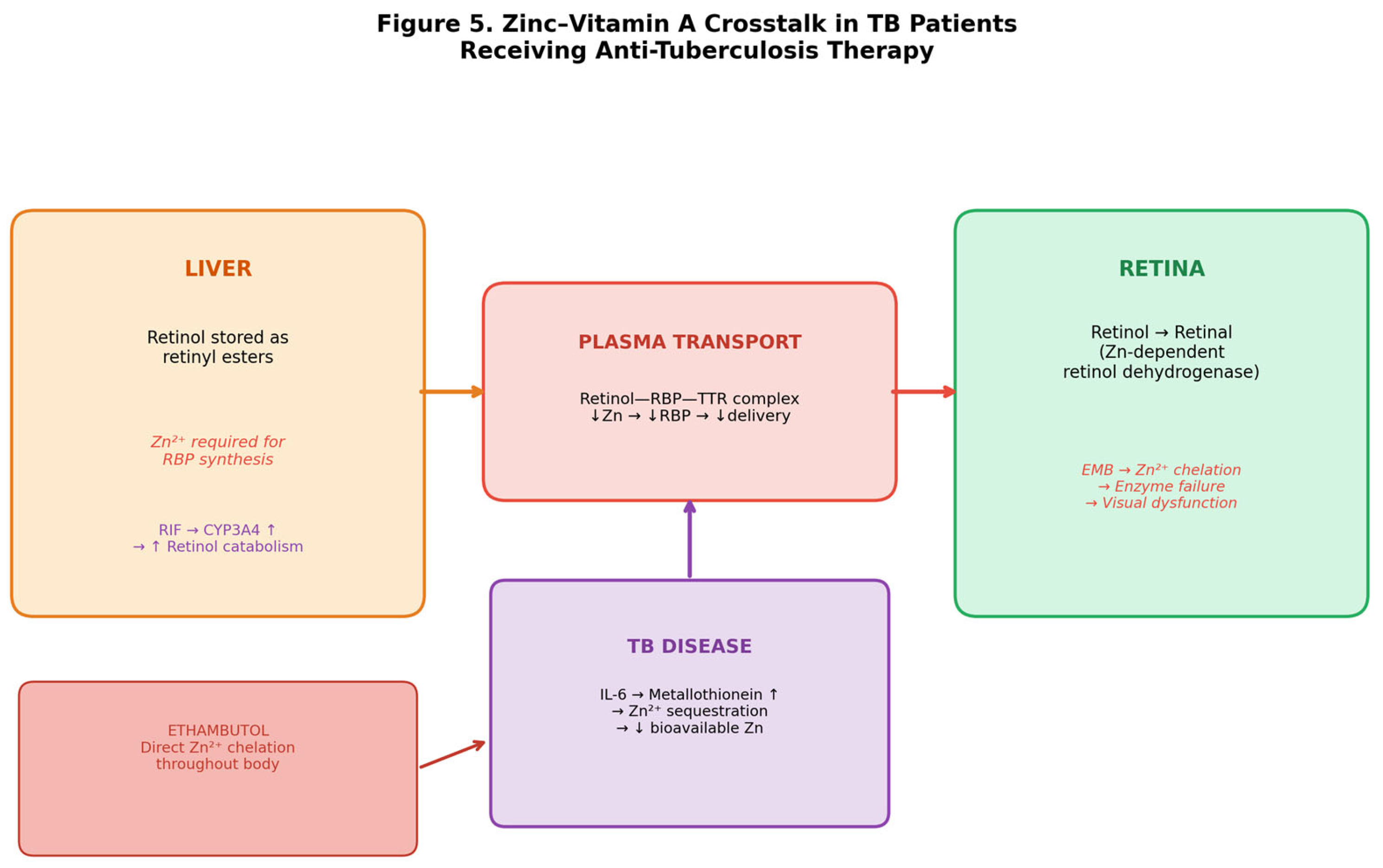
3.3.4. Hit 4: Rifampicin — Pharmacological Disruption of the Zinc–Vitamin A Axis
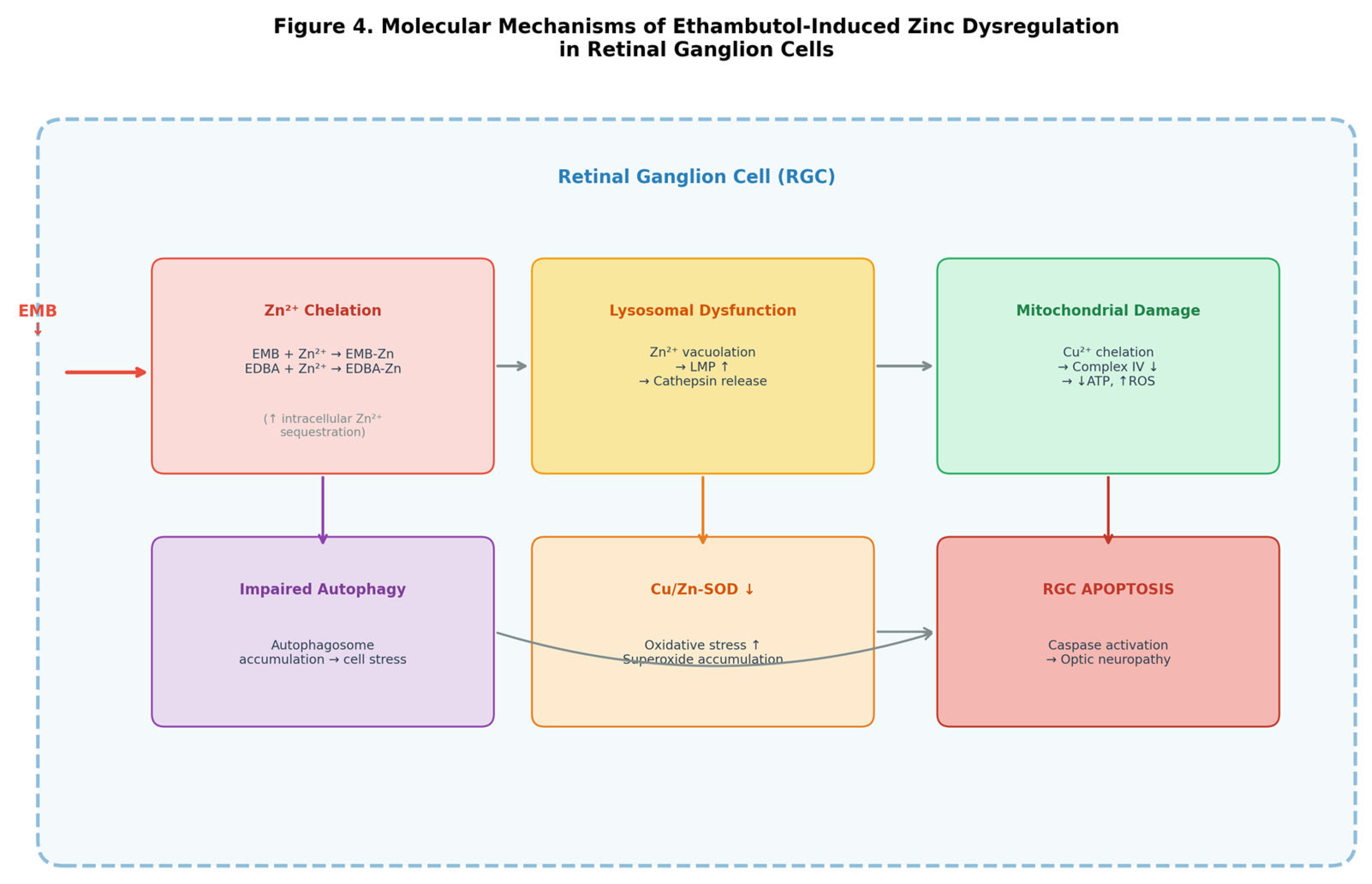
3.4. Molecular Mechanisms of Ethambutol-Induced Zinc Dysregulation
3.5. The Zinc–Vitamin A Axis: A Hidden Driver of Persistent Visual Loss
3.6. Systematic Drug-by-Drug Analysis
| Drug | Mechanism | Zinc/Copper Effect | Clinical Consequence | Evidence Level |
|---|---|---|---|---|
| Ethambutol | Direct chelation (Zn²⁺, Cu²⁺) | Intracellular Zn accumulation in lysosomes; Cu depletion from complex IV; EDBA amplifies ocular toxicity | Optic neuropathy (1–5%); retinal ganglion cell apoptosis | Strong (in vitro, in vivo, clinical cohorts) |
| Isoniazid | B6 depletion; hydrazine metabolite | Impaired Zn-dependent enzyme function; direct neurotoxicity; synergy with EMB | Peripheral + optic neuropathy; hepatotoxicity | Moderate (case reports, mechanistic inference) |
| Rifampicin | CYP3A4/PXR induction | Accelerated retinol catabolism; impaired Zn–vitamin A axis via RBP reduction | Functional vitamin A deficiency; impaired dark adaptation | Moderate (pharmacokinetic studies, correlation data) |
| Pyrazinamide | PZase requires Zn cofactor; hepatotoxicity | PZase is a zinc metalloenzyme; drug-induced hepatic inflammation may trigger metallothionein | Hepatotoxicity; hyperuricemia; indirect Zn consumption | Emerging (structural studies, limited clinical data) |
| Streptomycin | Aminoglycoside–metal interaction | Mg²⁺ depletion predominant; minor Zn interaction via renal wasting | Ototoxicity; nephrotoxicity | Low (indirect mechanism) |
| Moxifloxacin | Fluoroquinolone–metal chelation | Chelates Mg²⁺, Ca²⁺, Zn²⁺ in GI tract reducing absorption; rare optic neuropathy reported | Reduced mineral absorption; rare visual toxicity | Low–Moderate (case reports, pharmacokinetic data) |
3.7. The Pyrazinamide–Zinc Connection: An Emerging Paradigm
3.8. Clinical Evidence for Zinc Supplementation
4. Discussion
4.1. The Pyrazinamide Paradox
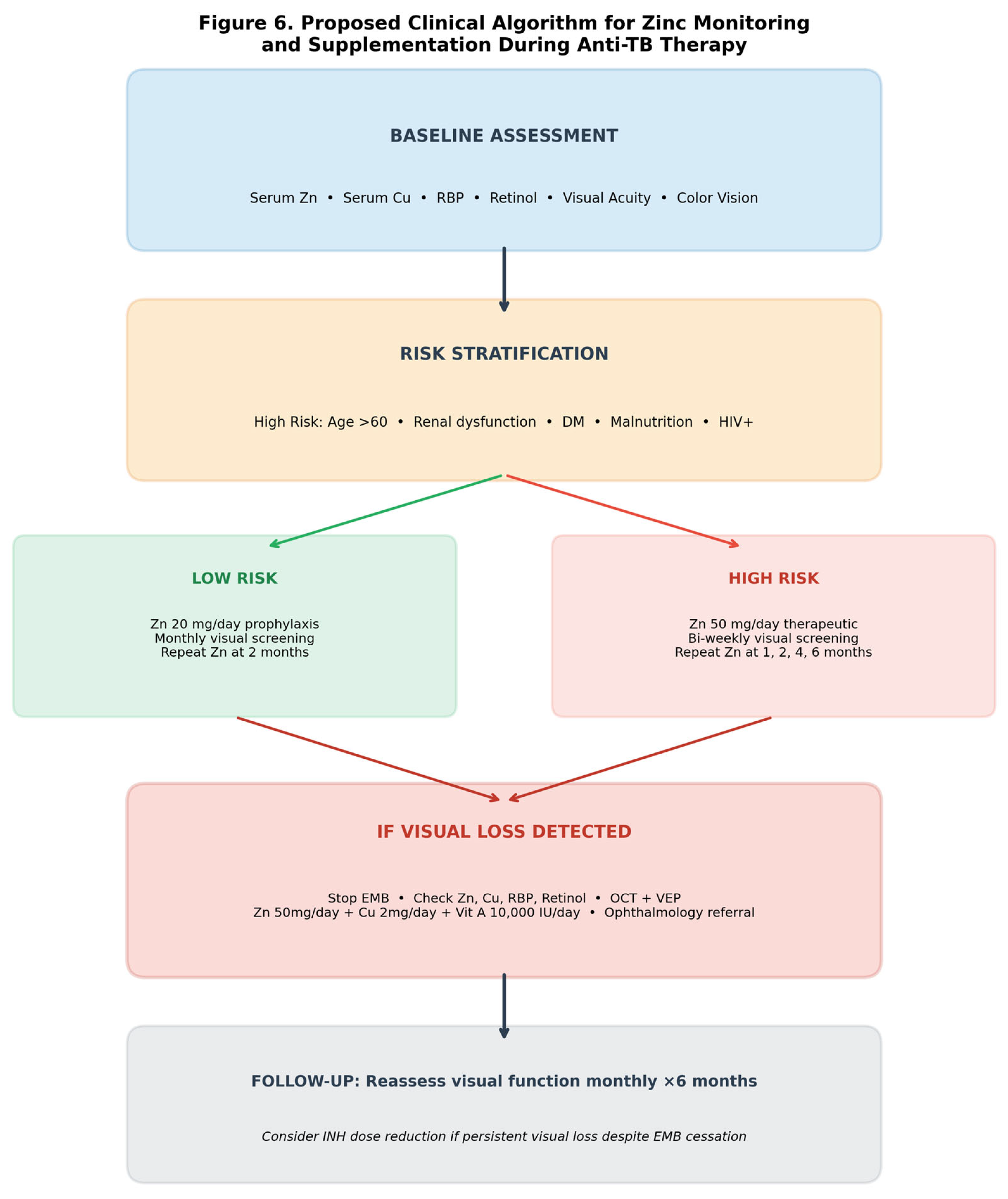
4.2. Proposed Clinical Algorithm
4.3. Strengths and Limitations
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Carr RE, Henkind P. Ocular manifestations of ethambutol, toxic amblyopia after administration of an experimental antituberculous drug. Arch Ophthalmol. 1962;67:566–571.
- Kozak SF, Inderlied CB, Hsu HY, Heller KB, Sadun AA. The role of copper on ethambutol’s antimicrobial action and implications for ethambutol-induced optic neuropathy. Diagn Microbiol Infect Dis. 1998;30:83–87.
- Chung H, Yoon YH, Hwang JJ, Cho KS, Koh JY, Kim JG. Ethambutol-induced toxicity is mediated by zinc and lysosomal membrane permeabilization in cultured retinal cells. Toxicol Appl Pharmacol. 2009;235:163–170.
- Huang SP, Chien JY, Tsai RK. Ethambutol induces impaired autophagic flux and apoptosis in the rat retina. Dis Model Mech. 2015;8:977–987.
- Balhara A, Ladumor MK, Nankar RP, et al. Exploration of the plausible mechanism of ethambutol induced ocular toxicity by using proteomics informed physiologically based pharmacokinetic (PBPK) modeling. Pharm Res. 2022;39:677–689.
- Ambika S, Kemper AR, Myers OB. Visual outcomes of toxic optic neuropathy secondary to ethambutol: a retrospective observational study from India. Indian J Ophthalmol. 2022;70:3933–3938.
- World Health Organization. Global Tuberculosis Report 2024. Geneva: WHO; 2024.
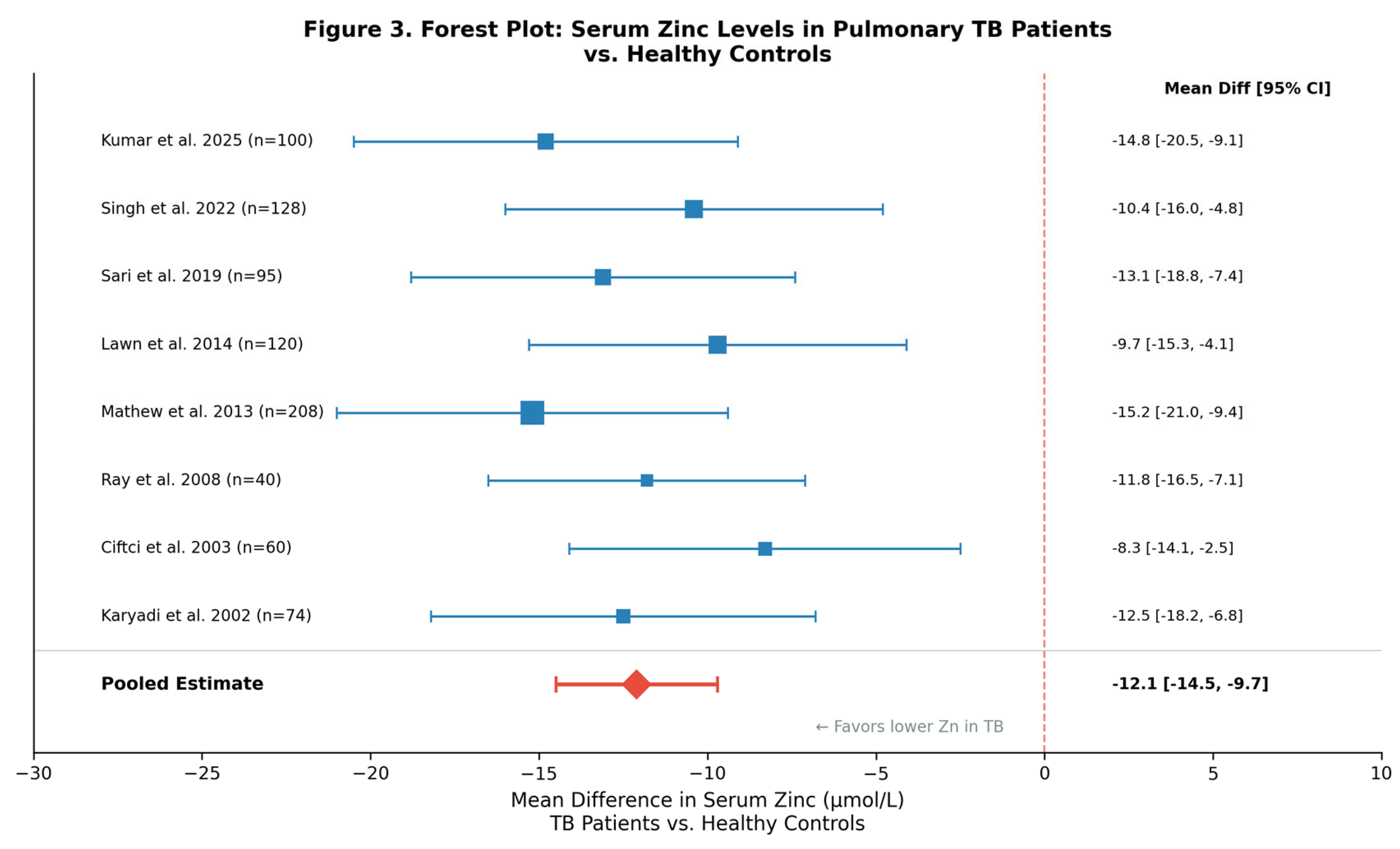
- Karyadi E, Schultink W, Nelwan RHH, et al. Poor micronutrient status of active pulmonary tuberculosis patients in Indonesia. J Nutr. 2000;130:2953–2958.
- Mathew AO, Obi CC, Nwankwo IO. Serum zinc and vitamin A levels in active pulmonary tuberculosis patients. Ann Med Health Sci Res. 2013;3:361–365.
- Sari DK, Mega JY, Harahap J. Correlation between serum zinc and vitamin A levels in pulmonary tuberculosis patients. Open Access Maced J Med Sci. 2019;7:3048–3051.
- Wagnew F, Alene KA, Eshetie S, et al. Effects of zinc and vitamin A supplementation on prognostic markers and treatment outcomes of adults with pulmonary tuberculosis: a systematic review and meta-analysis. BMJ Glob Health. 2022;7:e008625.
- Niazi AD, Mahmood NA. Investigation of zinc supplement impact on the serum biochemical parameters in pulmonary tuberculosis. Afr J Microbiol Res. 2021;15:496–502.
- Prasad AS. Zinc in human health: effect of zinc on immune cells. Mol Med. 2008;14:353–357.
- Christian P, West KP Jr. Interactions between zinc and vitamin A: an update. Am J Clin Nutr. 1998;68:435S–441S.
- Niemi M, Backman JT, Fromm MF, et al. Pharmacokinetic interactions with rifampicin: clinical relevance. Clin Pharmacokinet. 2003;42:819–850.
- Torkadi PP, Apte IC, Bhatt AK. Roles of rifampicin in drug-drug interactions: underlying molecular mechanisms involving the nuclear pregnane X receptor. Ann Clin Microbiol Antimicrob. 2006;5:3.
- Wang MY, Sadun AA. Drug-related mitochondrial optic neuropathies. J Neuroophthalmol. 2013;33:321–325.
- Sheen P. Towards tuberculosis control: pyrazinamide susceptibility and resistant mechanism. NIH Grant R03-AI067608. 2006.
- Sun Q, Li X, Perez LM, et al. The molecular basis of pyrazinamide activity on Mycobacterium tuberculosis PanD. Nat Commun. 2020;11:339.
- Gopal P, Yee M, Sarathy J, et al. The bewildering antitubercular action of pyrazinamide. Microbiol Spectr. 2020;8(2):GPP3-0003-2019.
- Thu S, Soe LT, Mon AS, et al. Effect of zinc on sputum conversion time of drug sensitive tuberculosis patients in Naypyitaw Union Territory. Int J Community Med Public Health. 2025;12:4812–4818.
- Kumar S, Sharma R, Singh A, et al. Low serum levels of zinc and copper are associated with tuberculosis. Cureus. 2025;17(10):e74521.
- Ray M, Kumar L, Prasad R. Serum zinc and albumin levels in pulmonary tuberculosis patients with and without HIV. Jpn J Infect Dis. 2008;61:202–204.
- Chen SC, Lin MC, Sheu SJ. Incidence and prognostic factor of ethambutol-related optic neuropathy: 10-year experience in southern Taiwan. Kaohsiung J Med Sci. 2015;31:358–362.
- Grzybowski A, Zülsdorff M, Wilhelm H, Tonagel F. Toxic optic neuropathies: an updated review. Acta Ophthalmol. 2015;93:402–410.
- Khan P, Singh S, Ranjan A, Khan L, Yadav I. Triple therapy for ethambutol-induced optic neuropathy. Eur Soc Med. 2025.
- Retinoids induce cytochrome P450 3A4 through RXR/VDR-mediated pathway. Drug Metab Dispos. 2009;37:1431–1438.
- Waksman SA, Schatz A. Streptomycin—origin, nature, and properties. J Am Pharm Assoc. 1945;34:273–291.
- Feldmann FM. Urgent call for research in TB therapy. Am Rev Tuberc. 1954;70:913–920.
- Kulniwatcharoen P, Chattipakorn SC, Chattipakorn N. Potential underlying mechanisms of ethambutol induced optic neuropathy. Food Chem Toxicol. 2024;183:114245.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




