Submitted:
17 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
- -
- moderate malnutrition, defined as BMI z-score < -2;
- -
- obesity, defined as BMI z-score > +2;
- -
- short stature, defined as a height z-score < -2;
- -
- tall stature, defined as a height z-score > +2.
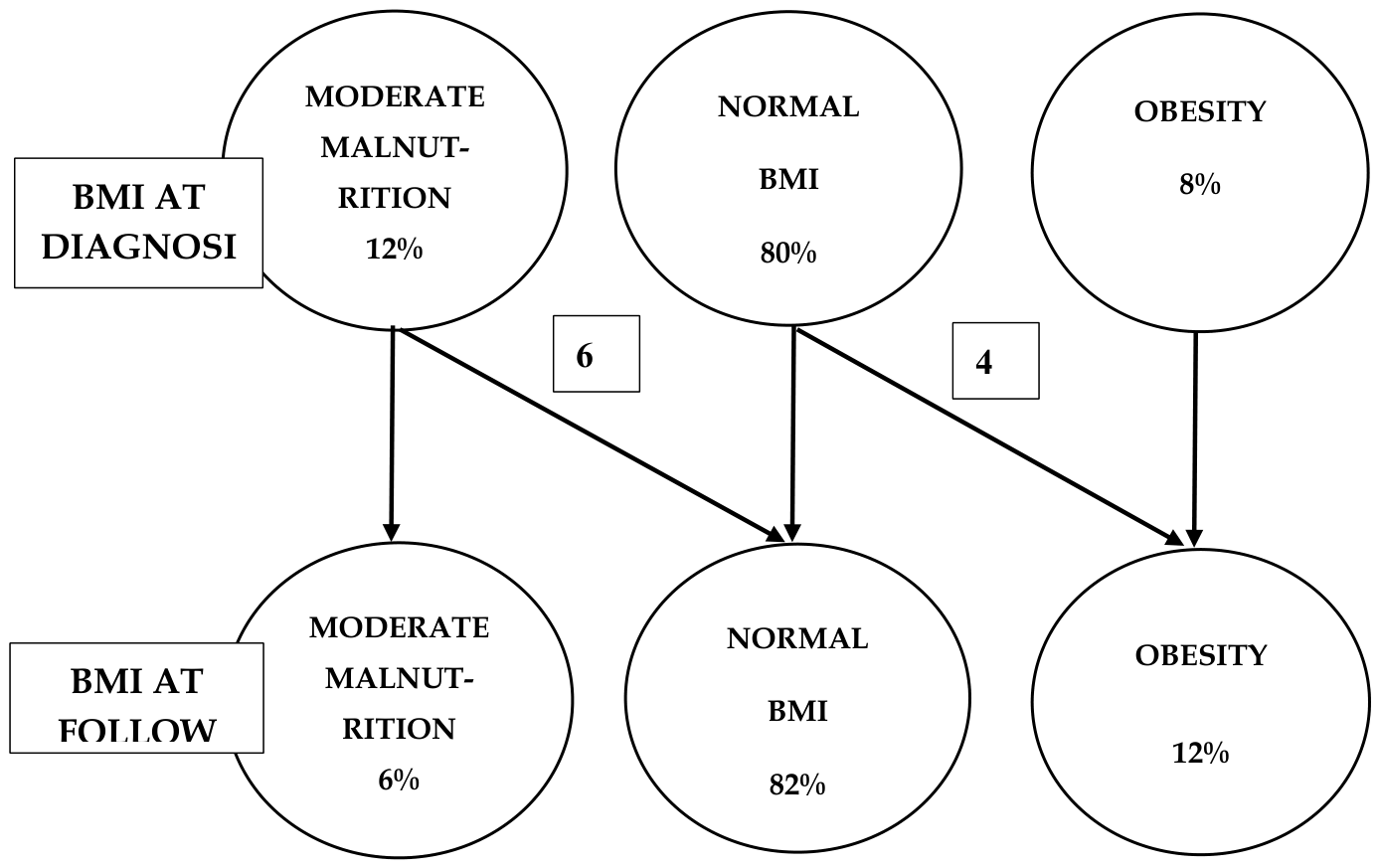
Results
Discussion
Summary of the Results
Comparison with Other Studies
Strengths and Limitations
Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dellon, E.S.; Liacouras, C.A.; Molina-Infante, J.; Furuta, G.T.; Spergel, J.M.; Zevit, N.; Spechler, S.J.; Attwood, S.E.; Straumann, A.; Aceves, S.S.; et al. Updated International Consensus Diagnostic Criteria for Eosinophilic Esophagitis: Proceedings of the AGREE Conference. Gastroenterology 2018, 155, 1022–1033.e10. [CrossRef]
- Dellon, E.S.; Hirano, I. Epidemiology and Natural History of Eosinophilic Esophagitis. Gastroenterology 2018, 154, 319–332.e3. [CrossRef]
- Hoofien A, Dias JA, Malamisura M et al. Pediatric Eosinophilic Esophagitis: Results of the European Retrospective Pediatric Eosinophilic Esophagitis Registry (RetroPEER). J. Pediatr. Gastroenterol. Nutr. 2019,68,552–558.
- Paquet, B.; Bégin, P.; Paradis, L.; Drouin, E.; Roches, A.D. High rate of failure to thrive in a pediatric cohort with eosinophilic esophagitis. Ann. Allergy, Asthma Immunol. 2016, 116, 73–74.e1. [CrossRef]
- Jensen, E.T.; Huang, K.Z.; Chen, H.X.; Landes, L.E.; McConnell, K.A.; Almond, M.A.; Safta, A.M.; Johnston, D.T.; Durban, R.; Jobe, L.; et al. Longitudinal Growth Outcomes Following First-line Treatment for Pediatric Patients With Eosinophilic Esophagitis. J. Pediatr. Gastroenterol. Nutr. 2019, 68, 50–55. [CrossRef]
- Mehta, P.; Furuta, G.T.; Brennan, T.; Henry, M.L.; Maune, N.C.; Sundaram, S.S.; Menard-Katcher, C.; Atkins, D.; Takurukura, F.; Giffen, S.; et al. Nutritional State and Feeding Behaviors of Children With Eosinophilic Esophagitis and Gastroesophageal Reflux Disease. J. Pediatr. Gastroenterol. Nutr. 2018, 66, 603–608. [CrossRef]
- Kovačić, M.; Unić, J.; Mišak, Z.; Jadrešin, O.; Konjik, V.; Kolaček, S.; Hojsak, I. One-year outcomes in children with eosinophilic esophagitis. Esophagus 2018, 16, 162–167. [CrossRef]
- Zdanowicz, K.; Kucharska, M.; Sobaniec-Lotowska, M.E.; Lebensztejn, D.M.; Daniluk, U. Eosinophilic Esophagitis in Children in North-Eastern Poland. J. Clin. Med. 2020, 9, 3869. [CrossRef]
- Mukkada, V.A.; Haas, A.; Maune, N.C.; Capocelli, K.E.; Henry, M.; Gilman, N.; Petersburg, S.; Moore, W.; Lovell, M.A.; Fleischer, D.M.; et al. Feeding Dysfunction in Children With Eosinophilic Gastrointestinal Diseases. Pediatrics 2010, 126, e672–e677. [CrossRef]
- Votto, M.; De Filippo, M.; Olivero, F.; Raffaele, A.; Cereda, E.; De Amici, M.; Testa, G.; Marseglia, G.L.; Licari, A. Malnutrition in Eosinophilic Gastrointestinal Disorders. Nutrients 2020, 13, 128. [CrossRef]
- Hirano, I.; Chan, E.S.; Rank, M.A.; Sharaf, R.N.; Stollman, N.H.; Stukus, D.R.; Wang, K.; Greenhawt, M.; Falck-Ytter, Y.T.; Chachu, K.A.; et al. AGA Institute and the Joint Task Force on Allergy-Immunology Practice Parameters Clinical Guidelines for the Management of Eosinophilic Esophagitis. Gastroenterology 2020, 158, 1776–1786. [CrossRef]
- https://www.who.int/tools/child-growth-standards/software.
- https://www.who.int/tools/growth-reference-data-for-5to19-years/application-tools.
- WHO Expert Committee Physical status: the use and interpretation of anthropometry Report of a WHO Expert Committee Technical Report Series No. 854 World Health Organization, Geneva 1995.
- Becker, P.J.; Nieman Carney, L.; Corkins, M.R.; Monczka, J.; Smith, E.; Smith, S.E.; Spear, B.A.; White, J.V. Consensus Statement of the Academy of Nutrition and Dietetics/American Society for Parenteral and Enteral Nutrition: Indicators Recommended for the Identification and Documentation of Pediatric Malnutrition (Undernutrition). J. Acad. Nutr. Diet. 2014, 114, 1988–2000. [CrossRef]
- Quigley CA, Ranke MB. International classification of pediatric endocrine diagnoses. 2016. www.icped.org.
- Inchley J, Currie D, Budisavljevic S et al., editors. Spotlight on adolescent health and well-being. Findings from the 2017/2018 Health Behaviour in School-aged Children (HBSC) survey in Europe and Canada. International report. Volume 2. Key data. Copenhagen: WHO Regional Office for Europe; 2020. Licence: CC BY-NC-SA 3.0 IGO.
| Characteristic | Value |
Gender
|
42(84) 8 (16%) |
Age at diagnosis
|
12.4 years 1-16 years |
Concomitant diseases:
|
6 4 1 2 7 1 |
| Severity level | At diagnosis n (%) | At the last visit n (%) |
| inactive | 0 (0%) | 2 (4%) |
| mild active | 32 (64%) | 28 (56%) |
| moderate active | 8 (16%) | 4 (8%) |
| Severe | 10 (20%) | 16 (32%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




