Submitted:
17 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Helicobacter pylori infection is a common gastrointestinal condition, more often seen in people living in crowded or less sanitary environments. In many cases, it is not detected until symptoms develop, rather than through routine screening. In this retrospective study, we reviewed patient records from a private laboratory to examine the relationship between gastrointestinal symptoms and H. pylori stool antigen (HpSAg) test results. Patients were also grouped by age to better understand how symptoms present across different life stages. The main aim of this study was to examine whether specific symptoms—particularly burning upper abdominal pain that worsens on an empty stomach—could help clinicians better identify patients who are more likely to benefit from testing. The idea was to support more targeted testing strategies, reduce unnecessary investigations, and ultimately help lower healthcare costs without compromising diagnostic accuracy. We assessed a range of gastrointestinal symptoms, including bloating, belching, nausea, vomiting, difficulty swallowing, early satiety, loss of appetite, weight loss, diarrhea, constipation, and melena, and compared them between infected and non-infected patients and across age groups. Burning epigastric pain was seen more often in patients with H. pylori, and overall, infected patients tended to report more symptoms, although this varies depending on age. It is important to emphasize that this study does not propose using symptoms as a replacement for diagnostic testing. Since the study was retrospective and did not include comparison with gold-standard methods such as the urea breath test or endoscopic biopsy, the findings should be interpreted as associations rather than definitive diagnostic conclusions. Still, the results suggest that certain symptom patterns—particularly burning epigastric pain on an empty stomach—may be useful for identifying patients more likely to benefit from testing. Further prospective studies are needed to confirm these observations. Materials and Methods: This retrospective observational study was conducted at Wilson Laboratory (Shush Square) in Tehran, Iran, using existing patient records. A total of 589 patients were included and classified as Helicobacter pylori–positive or Helicobacter pylori–negative based on stool antigen test (SAT) results. To better explore how symptoms differed across age groups, patients were divided into predefined age categories. These groups were selected to ensure a balanced distribution of cases and to make the comparisons clearer and easier to interpret. All stool antigen tests were performed using a commercially available enzyme immunoassay kit. Because the study was retrospective, detailed information about the assay characteristics—such as whether monoclonal or polyclonal antibodies were used, or the specific antigen targets (e.g., CagA or UreC) was not available. It is also recognized that test performance may vary depending on these factors, with monoclonal-based assays generally showing higher sensitivity and specificity. Gastrointestinal symptoms were extracted from patient records and compared between the H. pylori–positive and H. pylori–negative groups. The relationship between symptom patterns and infection status was then explored across different age groups using descriptive statistical analysis. Results: Of the 589 participants included in the study, 353 (59.9%) were Helicobacter pylori–positive and 236 (40.1%) were H. pylori–negative based on stool antigen test (SAT) results. H. pylori infection was significantly associated with burning epigastric pain that worsens on an empty stomach (p < 0.00001). Other gastrointestinal symptoms also showed significant associations with positive SAT results, including bloating (p = 0.012), persistent vomiting (p = 0.029), dysphagia (p < 0.00001), diarrhea (p = 0.0003), constipation (p = 0.0001), and melena (black, tarry stools) (p < 0.00001). The magnitude of these associations varied across age groups. Burning epigastric pain that worsens on an empty stomach demonstrated a sensitivity of 76.2% and a specificity of 95.8% for identifying H. pylori infection. The positive predictive value (PPV) was 96.4%, and the negative predictive value (NPV) was 72.9%. Conclusions: Overall, having at least one gastrointestinal symptom was strongly associated with Helicobacter pylori infection across all age groups. Among the symptoms studied, burning epigastric pain that worsens when the stomach is empty showed the strongest association with infection. Its relatively high specificity and positive predictive value suggests that it may be helpful in guiding clinicians when deciding which patients should be prioritized for testing. At the same time, because this was a retrospective study, these findings should be interpreted with caution. Further prospective studies are needed to better understand and confirm how useful symptom-based approaches can be in routine clinical practice.
Keywords:
1. Introduction
2. Literature Review
3. Materials and Methods
3.1. Study Design and Setting
3.2. Data Collection and Study Population
3.3. Case Definition and Grouping
3.4. Variables and Outcomes
3.5. Statistical Analysis
4. Results
4.1. Demographic and Clinical Characteristics
- Positive: 269/353 → a = 269, b = 353 - 269 = 84
- Negative: 10/236 → c = 10, d = 236 - 10 = 226
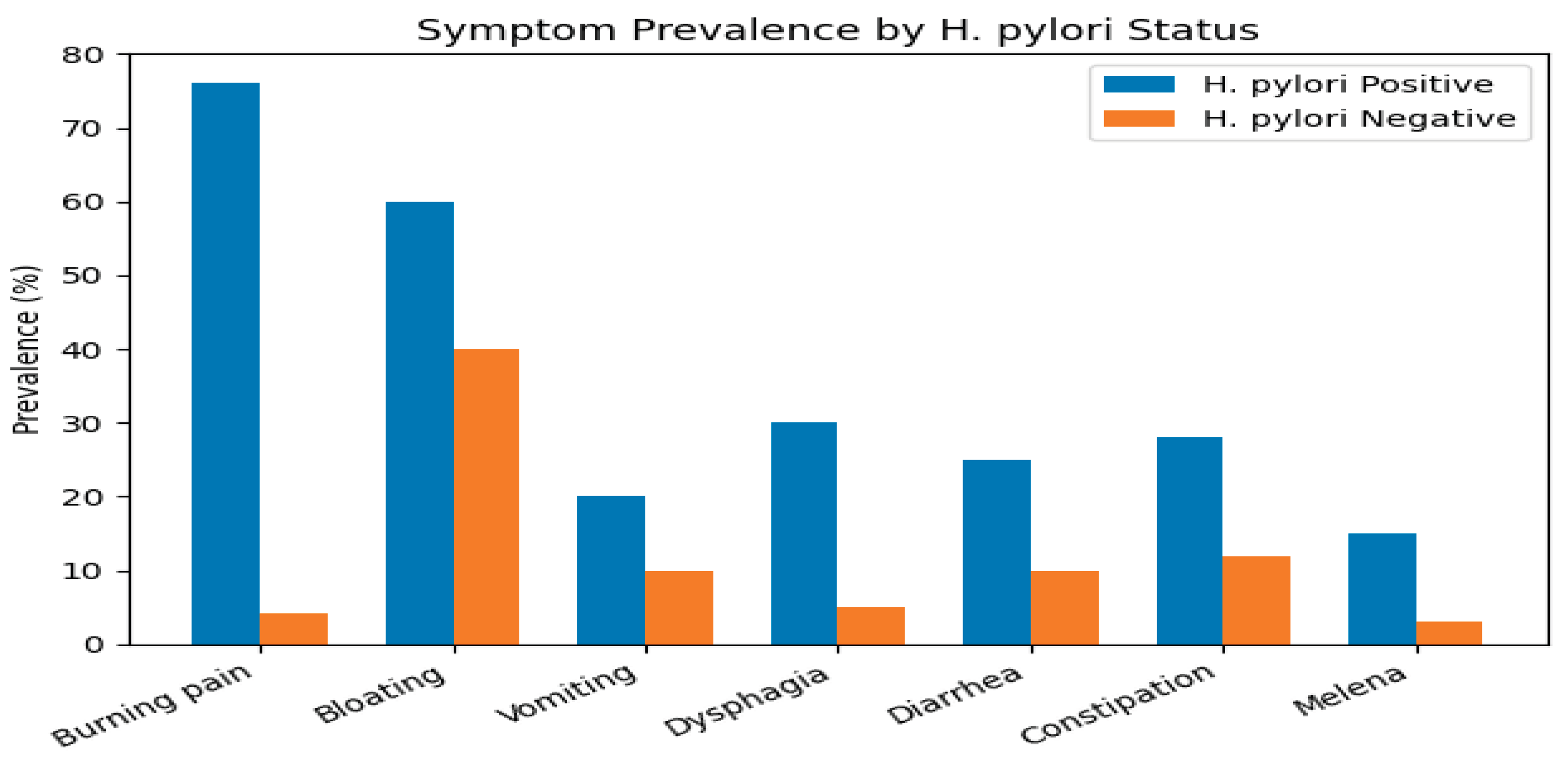
4.2. Prevalence of Symptoms in H. Pylori-Positive and -Negative Patients (Table 10, Figure 2)
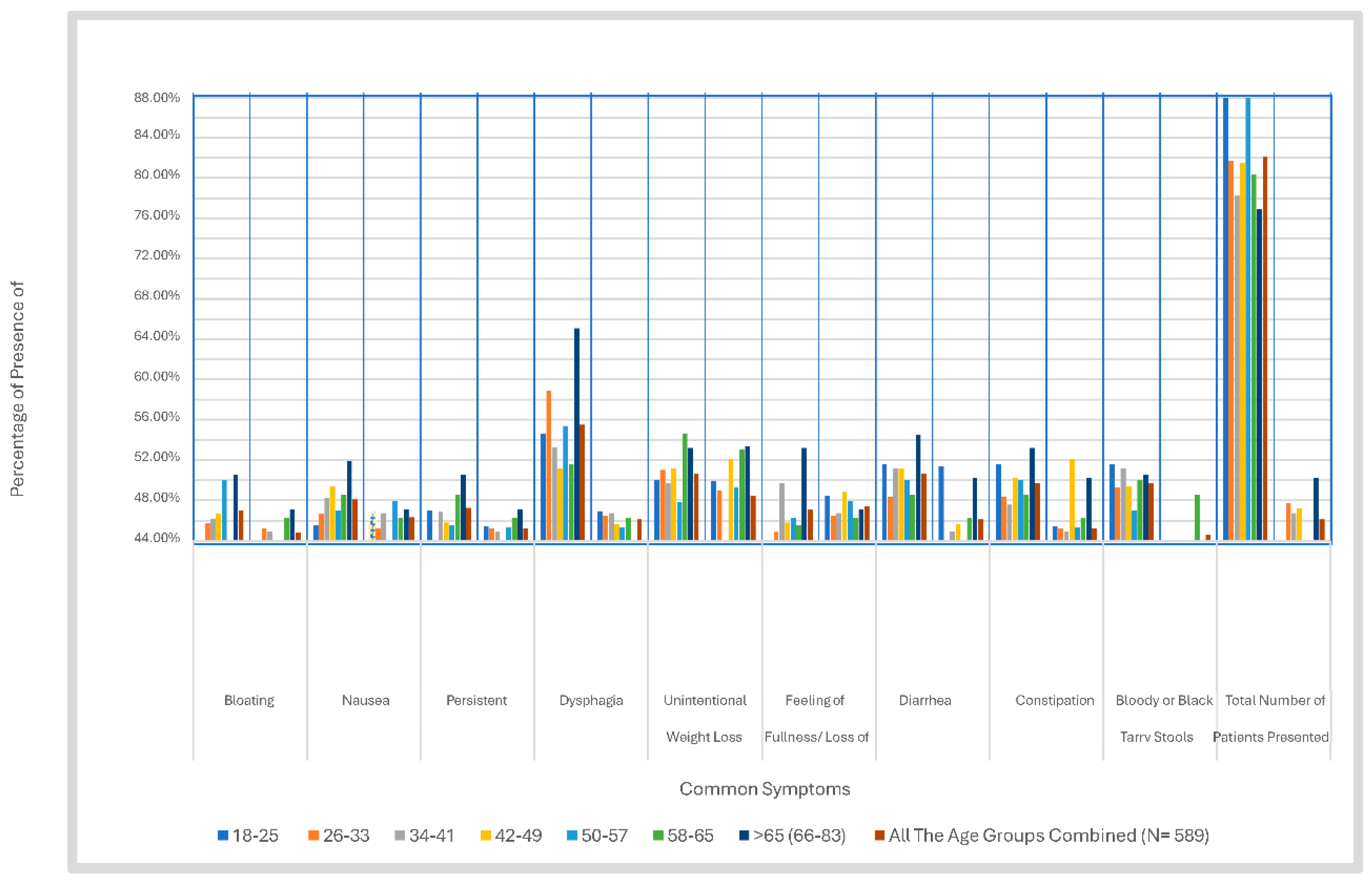
4.3. Prevalence of H. pylori–Associated Symptoms Across Age Groups (Table 4, Table 5, Table 6, Table 7, Table 8, Table 9 and Table 10)
4.3.1. Age Group 18–25 Years (Table 4)
4.3.2. Age Group 26–33 Years (Table 5)
4.3.3. Age Group 34–41 (Table 6)
4.3.4. Age Group 42–49 (Table 7)
4.3.5. Age Group 50–57 (Table 8)
4.3.6. Age Group 58–65 (Table 9)
| Age Groups/ Presented Symptoms | 18-25 | 26-33 | 34-41 | 42-49 | 50-57 | 58-65 | >65 (66-83) | Combined (N= 589) | ||||||||
| Positive For H. pylori |
Negative For H. pylori |
Positive For H. pylori |
Negative For H. pylori |
Positive For H. pylori |
Negative For H. pylori |
Positive For H. pylori |
Negative For H. pylori |
Positive For H. pylori |
Negative For H. pylori |
Positive For H. pylori |
Negative For H. pylori |
Positive For H. pylori |
Negative For H. pylori |
Positive For H. pylori |
Negative For H. pylori |
|
| Bloating | 0.00% | 0.00% | 3.51% | 2.50% | 4.29% | 1.82% | 5.36% | 0.00% | 12.12%* | 0.00% | 0.00% | 4.55% | 13.16% | 6.25% | 5.95%* | 1.69% |
| Nausea | 3.03% | 5.88% | 5.26% | 2.50% | 8.57% | 5.45% | 10.71% | 0.00% | 6.06% | 7.89% | 9.09% | 4.55% | 15.79% | 6.25% | 8.22% | 4.66% |
| Persistent Vomiting | 6.06% | 2.94% | 0.00% | 2.50% | 5.71% | 1.82% | 3.57% | 0.00% | 3.03% | 2.63% | 9.09% | 4.55% | 13.16% | 6.25% | 6.52%* | 2.54% |
| Burning Pain That Worsens When the Stomach Is Empty | 87.88%* | 0.00% | 75.44%* | 7.50% | 68.57%* | 5.45% | 75.00%* | 6.45% | 87.88%* | 0.00% | 72.73%* | 0.00% | 65.79%* | 12.50% | 76.20%* | 4.24% |
| Dysphagia | 21.21% | 5.88% | 29.82%* | 5.00% | 18.57%* | 5.45% | 14.29% | 3.23% | 22.73%* | 2.63% | 15.15% | 4.55% | 42.11%* | 0.00% | 22.95%* | 4.24% |
| Unintentional Weight Loss (Over 3 kg in Past Month) | 12.12% | 11.76% | 14.04% | 10.00% | 11.43%* | 0.00% | 14.29% | 16.13% | 7.58% | 10.53% | 21.21% | 18.18% | 18.42% | 18.75% | 13.31% | 8.90% |
| Feeling of Fullness/ Loss of Appetite | 0.00% | 8.82% | 1.75% | 5.00% | 11.43% | 5.45% | 3.57% | 9.68% | 4.55% | 7.89% | 3.03% | 4.55% | 18.42% | 6.25% | 6.23% | 6.78% |
| Diarrhea | 15.15% | 14.71% | 8.77%* | 0.00% | 14.29%* | 1.82% | 14.29% | 3.23% | 12.12%* | 0.00% | 9.09% | 4.55% | 21.05% | 12.50% | 13.31%* | 4.24% |
| Constipation | 15.15% | 2.94% | 8.77% | 2.50% | 7.14% | 1.82% | 12.50% | 16.13% | 12.12% | 2.63% | 9.09% | 4.55% | 18.42% | 12.50% | 11.33%* | 2.54% |
| Bloody or Black Tarry Stools | 15.15%* | 0.00% | 10.53%* | 0.00% | 14.29%* | 0.00% | 10.71% | 0.00% | 6.06% | 0.00% | 12.12% | 9.09% | 13.16% | 0.00% | 11.33%* | 1.27% |
| Total Number of Patients Presented with Symptoms | 96.97%* | 47.06% | 100.00%* | 32.50% | 100.00%* | 27.27% | 100.00%* | 35.48% | 100.00%* | 34.21% | 100.00%* | 40.91% | 100.00%* | 37.50% | 99.72%* | 35.17% |
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Gastroenterology Organisation. World Gastroenterology Organisation Global Guideline: Helicobacter pylori in developing countries. J Clin Gastroenterol. 2011, 45(5), 383–8. [Google Scholar] [CrossRef] [PubMed]
- Epidemiology of, and risk factors for, Helicobacter pylori infection among 3194 asymptomatic subjects in 17 populations. The EUROGAST Study Group. Gut 1993, 34(12), 1672–6. [CrossRef] [PubMed] [PubMed Central]
- Eusebi, L.H.; Zagari, R.M.; Bazzoli, F. Epidemiology of Helicobacter pylori infection. Helicobacter 2014, 19 Suppl 1, 1–5. [Google Scholar] [CrossRef] [PubMed]
- De Brito, B.B.; da Silva, F.A.F.; Soares, A.S.; Pereira, V.A.; Santos, M.L.C.; Sampaio, M.M.; Neves, P.H.M.; de Melo, F.F. Pathogenesis and clinical management of Helicobacter pylori gastric infection. World J Gastroenterol 2019, 25(37), 5578–5589. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Franceschi, F.; Annalisa, T.; Teresa, D.R.; Giovanna, D.; Ianiro, G.; Franco, S.; Viviana, G.; Valentina, T.; Riccardo, L.L.; Antonio, G. Role of Helicobacter pylori infection on nutrition and metabolism. World J Gastroenterol 2014, 20(36), 12809–17. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Holmes, L., Jr.; Rios, J.; Berice, B.; Benson, J.; Bafford, N.; Parson, K.; Halloran, D. Predictive Effect of Helicobacter pylori in Gastric Carcinoma Development: Systematic Review and Quantitative Evidence Synthesis. Medicines (Basel) 2021, 8(1), 1. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Choi, I.J.; Kook, M.C.; Kim, Y.I.; Cho, S.J.; Lee, J.Y.; Kim, C.G.; Park, B.; Nam, B.H. Helicobacter pylori Therapy for the Prevention of Metachronous Gastric Cancer. N Engl J Med 2018, 378(12), 1085–1095. [Google Scholar] [CrossRef] [PubMed]
- Mestrovic, A.; Bozic, J.; Vukojevic, K.; Tonkic, A. Impact of Different Helicobacter pylori Eradication Therapies on Gastrointestinal Symptoms. Medicina (Kaunas) 2021, 8 57(8), 803. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Suerbaum, S.; Michetti, P. Helicobacter pylori infection. N Engl J Med 2002, 347(15), 1175–1186. [Google Scholar] [CrossRef] [PubMed]
- Hunt, R.H.; Xiao, S.D.; Megraud, F.; Leon-Barua, R.; Bazzoli, F.; van der Merwe, S.; Vaz Coelho, L.G.; Fock, M.; Fedail, S.; Cohen, H.; Malfertheiner, P.; Vakil, N.; Hamid, S.; Goh, K.L.; Wong, B.C.; Krabshuis, J.; Le Mair, A.; World Gastroenterology Organization. Helicobacter pylori in developing countries. World Gastroenterology Organisation Global Guideline. J Gastrointestin Liver Dis. 2011, 20(3), 299–304. [Google Scholar] [PubMed]
- Kabir, S. Detection of Helicobacter pylori in faeces by culture, PCR and enzyme immunoassay. J Med Microbiol. 2001, 50(12), 1021–1029. [Google Scholar] [CrossRef] [PubMed]
- Khalifehgholi, M.; Shamsipour, F.; Ajhdarkosh, H.; Ebrahimi Daryani, N.; Pourmand, M.R.; Hosseini, M.; Ghasemi, A.; Shirazi, M.H. Comparison of five diagnostic methods for Helicobacter pylori. Iran J Microbiol 2013, 5(4), 396–401. [Google Scholar] [PubMed] [PubMed Central]
- Sabbagh, P.; Mohammadnia-Afrouzi, M.; Javanian, M.; Babazadeh, A.; Koppolu, V.; Vasigala, V.R.; Nouri, H.R.; Ebrahimpour, S. Diagnostic methods for Helicobacter pylori infection: ideals, options, and limitations. Eur J Clin Microbiol Infect Dis 2019, 38(1), 55–66. [Google Scholar] [CrossRef] [PubMed]
- Vaira, D.; Vakil, N. Blood, urine, stool, breath, money, and Helicobacter pylori. Gut 2001, 48(3), 287–9. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gisbert, J.P.; Pajares, J.M. Review article: 13C-urea breath test in the diagnosis of Helicobacter pylori infection -- a critical review. Aliment Pharmacol Ther 2004, 20(10), 1001–17. [Google Scholar] [CrossRef] [PubMed]
- Van Doorn, O.J.; Bosman, D.K.; van't Hoff, B.W.; Taminiau, J.A.; ten Kate, F.J.; van der Ende, A. Helicobacter pylori Stool Antigen test: a reliable non-invasive test for the diagnosis of Helicobacter pylori infection in children. Eur J Gastroenterol Hepatol 2001, 13(9), 1061–5. [Google Scholar] [CrossRef] [PubMed]
- Gisbert, J.P.; Pajares, J.M. Stool antigen test for the diagnosis of Helicobacter pylori infection: a systematic review. Helicobacter 2004, 9(4), 347–68. [Google Scholar] [CrossRef] [PubMed]
- Vaira, D.; Vakil, N.; Menegatti, M.; van't Hoff, B.; Ricci, C.; Gatta, L.; Gasbarrini, G.; Quina, M.; Pajares Garcia, J.M.; van Der Ende, A.; van Der Hulst, R.; Anti, M.; Duarte, C.; Gisbert, J.P.; Miglioli, M.; Tytgat, G. The stool antigen test for detection of Helicobacter pylori after eradication therapy. Ann Intern Med. 2002, 136(4), 280–7. [Google Scholar] [CrossRef] [PubMed]
- Demiray, E.; Yilmaz, O.; Sarkis, C.; Soyturk, M.; Simsek, I. Comparison of invasive methods and two different stool antigen tests for diagnosis of H pylori infection in patients with gastric bleeding. World J Gastroenterol 2006, 12(26), 4206–10. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hunt, R.H.; Xiao, S.D.; Megraud, F.; Leon-Barua, R.; Bazzoli, F.; van der Merwe, S.; Vaz Coelho, L.G.; Fock, M.; Fedail, S.; Cohen, H.; Malfertheiner, P.; Vakil, N.; Hamid, S.; Goh, K.L.; Wong, B.C.; Krabshuis, J.; Le Mair, A.; World Gastroenterology Organization. Helicobacter pylori in developing countries. World Gastroenterology Organisation Global Guideline. J Gastrointestin Liver Dis. 2011, 20(3), 299–304. [Google Scholar] [PubMed]
- Santos, I.S.; Boccio, J.; Santos, A.S.; Valle, N.C.; Halal, C.S.; Bachilli, M.C.; Lopes, R.D. Prevalence of Helicobacter pylori infection and associated factors among adults in Southern Brazil: a population-based cross-sectional study. BMC Public Health 2005, 5, 118. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Werdmuller, B.F.; van der Putten, T.B.; Balk, T.G.; Lamers, C.B.; Loffeld, R.J. Clinical presentation of Helicobacter pylori-positive and -negative functional dyspepsia. J Gastroenterol Hepatol. 2000, 15(5), 498–502. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.; Sharif, F.A.; Osman, S.M.; Osman, A.M.; El Sanousi, S.M.; Magzoub, M.; Ibrahim, M.E. Prevalence and Associated Symptoms of Helicobacter pylori Infection among Schoolchildren in Kassala State, East of Sudan. Interdiscip Perspect Infect Dis 2018, 2018, 4325752. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Stenström, B.; Mendis, A.; Marshall, B. Helicobacter pylori--the latest in diagnosis and treatment. Aust Fam Physician 2008, 37(8), 608–12. [Google Scholar] [PubMed]
| Tested Negative for H. pylori | Tested Positive for H. pylori | ||||||
|---|---|---|---|---|---|---|---|
| Symptoms (N=589) | Number of Patients Evaluated | Number of Patients with Symptom |
% of patients with Symptom |
Number of Patients Evaluated | Number of Patients with Symptom |
% of patients with Symptom |
p value |
| Gender |
236 F (145) M (91) |
353 F (206) M (147) |
0.45484 |
||||
| Bloating | 236 | 4 | 1.69% | 353 | 21 | 5.95% | 0.01209 |
| Nausea | 236 | 11 | 4.66% | 353 | 29 | 8.22% | 0.09294 |
| Persistent Vomiting | 236 | 6 | 2.54% | 353 | 23 | 6.52% | 0.02896 |
| Burning Pain That Worsens When the Stomach Is Empty | 236 | 10 | 4.24% | 353 | 269 | 76.20% | 0.00001* |
| Dysphagia | 236 | 10 | 4.24% | 353 | 81 | 22.95% | 0.00001* |
| Unintentional Weight Loss (Over 3 kg in Past Month) | 236 | 21 | 8.90% | 353 | 47 | 13.31% | 0.10028 |
| Feeling of Fullness/ Loss of Appetite | 236 | 16 | 6.78% | 353 | 22 | 6.23% | 0.79103 |
| Diarrhea | 236 | 10 | 4.24% | 353 | 47 | 13.31% | 0.00026 |
| Constipation | 236 | 6 | 2.54% | 353 | 40 | 11.33% | 0.00010 |
| Bloody or Black Tarry Stools | 236 | 3 | 1.27% | 353 | 40 | 11.33% | 0.00001* |
| Total Number of Patients Presented with Symptoms | 236 | 83 | 35.17% | 353 | 352 | 99.72% | 0.00001* |
| Metric | Value |
|---|---|
| Sensitivity | 76.2% |
| Specificity | 95.8% |
| Positive Predictive Value (PPV) | 96.4% |
| Negative Predictive Value (NPV) | 72.9% |
| Symptom | a | b | c | d | OR |
| Dysphagia | 81 | 272 | 10 | 226 | 6.7 |
| Diarrhea | 47 | 306 | 10 | 226 | 3.5 |
| Constipation | 40 | 313 | 6 | 230 | 4.9 |
| Bloody/Black Stools | 40 | 313 | 3 | 233 | 9.97 |
| Tested Positive for H. pylori | Tested Negative for H. pylori | ||||||
|---|---|---|---|---|---|---|---|
| Age Group 18-25 | Number of Patients Evaluated |
Number of Patients with Symptom |
% Of Patients with Symptom |
Number of Patients Evaluated | Number of Patients with Symptom |
% Of Patients with Symptom | p value |
| Gender | 14 (F) 19 (M) |
22 (F) 12 (M) |
0.06744 | ||||
| Bloating | 33 | 0 | 0.00% | 34 | 0 | 0.00% | 1.00000 |
| Nausea | 33 | 1 | 3.03% | 34 | 2 | 5.88% | 0.57252 |
| Persistent Vomiting | 33 | 2 | 6.06% | 34 | 1 | 2.94% | 0.53707 |
| Burning Pain That Worsens When the Stomach Is Empty | 33 | 29 | 87.88% | 34 | 0 | 0.00% | 0.00001* |
| Dysphagia | 33 | 7 | 21.21% | 34 | 2 | 5.88% | 0.06582 |
| Unintentional Weight Loss (Over 3 kg in Past Month) | 33 | 4 | 12.12% | 34 | 4 | 11.76% | 0.96411 |
| Feeling of Fullness/ Loss of Appetite | 33 | 0 | 0.00% | 34 | 3 | 8.82% | 0.08082 |
| Diarrhea | 33 | 5 | 15.15% | 34 | 5 | 14.71% | 0.95918 |
| Constipation | 33 | 5 | 15.15% | 34 | 1 | 2.94% | 0.08013 |
| Bloody or Black Tarry Stools | 33 | 5 | 15.15% | 34 | 0 | 0.00% | 0.01830 |
| Total Number of Patients Presented with Symptoms | 33 | 32 | 96.97% | 34 | 16 | 47.06% | 0.00001* |
| Tested Positive for H. pylori | Tested Negative for H. pylori | ||||||
|---|---|---|---|---|---|---|---|
| Age Group 26-33 | Number of Patients Evaluated | Number of Patients with Symptom | % of Patients with Symptom | Number of Patients Evaluated | Number of Patients with Symptom | % of Patients with Symptom | p value |
| Gender | 31 (F) 26 (M) |
20 (F) 20 (M) |
0.67022 | ||||
| Bloating | 57 | 2 | 3.51% | 40 | 1 | 2.50% | 0.77756 |
| Nausea | 57 | 3 | 5.26% | 40 | 1 | 2.50% | 0.50048 |
| Persistent Vomiting | 57 | 0 | 0.00% | 40 | 1 | 2.50% | 0.23016 |
| Burning Pain That Worsens When the Stomach Is Empty | 57 | 43 | 75.44% | 40 | 3 | 7.50% | 0.00001* |
| Dysphagia | 57 | 17 | 29.82% | 40 | 2 | 5.00% | 0.00242 |
| Unintentional Weight Loss (Over 3 kg in Past Month) | 57 | 8 | 14.04% | 40 | 4 | 10.00% | 0.55240 |
| Feeling of Fullness/ Loss of Appetite | 57 | 1 | 1.75% | 40 | 2 | 5.00% | 0.36339 |
| Diarrhea | 57 | 5 | 8.77% | 40 | 0 | 0.00% | 0.05443 |
| Constipation | 57 | 5 | 8.77% | 40 | 1 | 2.50% | 0.20685 |
| Bloody or Black Tarry Stools | 57 | 6 | 10.53% | 40 | 0 | 0.00% | 0.03413 |
| Total Number of Patients Presented with Symptoms | 57 | 57 | 100.00% | 40 | 13 | 32.50% | 0.00001* |
| Tested Positive for H. pylori | Tested Negative for H. pylori | ||||||
|---|---|---|---|---|---|---|---|
| Age Group 34-41 | Number of Patients Evaluated | Number of Patients with Symptom | % of Patients with Symptom | Number of Patients Evaluated | Number of Patients with Symptom | % of Patients with Symptom | p value |
| Gender | 43 (F) 27 (M) |
27 (F) 28 (M) |
0.16777 | ||||
| Bloating | 70 | 3 | 4.29% | 55 | 1 | 1.82% | 0.43652 |
| Nausea | 70 | 6 | 8.57% | 55 | 3 | 5.45% | 0.50337 |
| Persistent Vomiting | 70 | 4 | 5.71% | 55 | 1 | 1.82% | 0.26985 |
| Burning Pain That Worsens When the Stomach Is Empty | 70 | 48 | 68.57% | 55 | 3 | 5.45% | 0.00001* |
| Dysphagia | 70 | 13 | 18.57% | 55 | 3 | 5.45% | 0.02934 |
| Unintentional Weight Loss (Over 3 kg in Past Month) | 70 | 8 | 11.43% | 55 | 0 | 0.00% | 0.00956 |
| Feeling of Fullness/ Loss of Appetite | 70 | 8 | 11.43% | 55 | 3 | 5.45% | 0.24187 |
| Diarrhea | 70 | 10 | 14.29% | 55 | 1 | 1.82% | 0.01459 |
| Constipation | 70 | 5 | 7.14% | 55 | 1 | 1.82% | 0.16685 |
| Bloody or Black Tarry Stools | 70 | 10 | 14.29% | 55 | 0 | 0.00% | 0.00347 |
| Total Number of Patients Presented with Symptoms | 70 | 70 | 100.00% | 55 | 15 | 27.27% | 0.00001* |
| Tested Positive for H. pylori | Tested Negative for H. pylori | ||||||
|---|---|---|---|---|---|---|---|
| Age Group 42-49 | Number of Patients Evaluated | Number of Patients with Symptom | % of Patients with Symptom | Number of Patients Evaluated | Number of Patients with Symptom | % of Patients with Symptom | p value |
| Gender | 27 (F) 29 (M) |
23 (F) 8 (M) |
0.01891 | ||||
| Bloating, | 56 | 3 | 5.36% | 31 | 0 | 0.00% | 0.18969 |
| Nausea | 56 | 6 | 10.71% | 31 | 0 | 0.00% | 0.05892 |
| Persistent Vomiting | 56 | 2 | 3.57% | 31 | 0 | 0.00% | 0.28710 |
|
Burning Pain That Worsen When Stomach Is Empty |
56 | 42 | 75.00% | 31 | 2 | 6.45% | 0.00001* |
| Dysphagia | 56 | 8 | 14.29% | 31 | 1 | 3.23% | 0.10475 |
|
Unintentional Weight Loss (Over 3 kg in the Past Month) |
56 | 8 | 14.29% | 31 | 5 | 16.13% | 0.81734 |
| Feeling of Fullness/ Loss of Appetite | 56 | 2 | 3.57% | 31 | 3 | 9.68% | 0.24123 |
| Diarrhea | 56 | 8 | 14.29% | 31 | 1 | 3.23% | 0.10475 |
| Constipation | 56 | 7 | 12.50% | 31 | 5 | 16.13% | 0.63827 |
| Bloody or Black Tarry Stools | 56 | 6 | 10.71% | 31 | 0 | 0.00% | 0.05892 |
| Total Number of Patients Presented with Symptoms | 56 | 56 | 100.00% | 31 | 11 | 35.48% | 0.00001* |
| Tested Positive for H. pylori | Tested Negative for H. pylori | ||||||
|---|---|---|---|---|---|---|---|
| Age Group 50-57 | Number of Patients Evaluated | Number of Patients with Symptom | % of Patients with Symptom | Number of Patients Evaluated | Number of Patients with Symptom | % of Patients with Symptom | p value |
| Gender | 48 (F) 18 (M) |
25 (F) 13 (M) |
0.45637 | ||||
| Bloating | 66 | 8 | 12.12% | 38 | 0 | 0.00% | 0.02550 |
| Nausea | 66 | 4 | 6.06% | 38 | 3 | 7.89% | 0.71924 |
| Persistent Vomiting | 66 | 2 | 3.03% | 38 | 1 | 2.63% | 0.90687 |
| Burning Pain That Worsens When the Stomach Is Empty | 66 | 58 | 87.88% | 38 | 0 | 0.00% | 0.00001* |
| Dysphagia | 66 | 15 | 22.73% | 38 | 1 | 2.63% | 0.00624 |
| Unintentional Weight Loss (Over 3 kg in Past Month) | 66 | 5 | 7.58% | 38 | 4 | 10.53% | 0.60631 |
| Feeling of Fullness/ Loss of Appetite | 66 | 3 | 4.55% | 38 | 3 | 7.89% | 0.48055 |
| Diarrhea | 66 | 8 | 12.12% | 38 | 0 | 0.00% | 0.02550 |
| Constipation | 66 | 8 | 12.12% | 38 | 1 | 2.63% | 0.09742 |
| Bloody or Black Tarry Stools | 66 | 4 | 6.06% | 38 | 0 | 0.00% | 0.12171 |
| Total Number of Patients Presented with Symptoms | 66 | 66 | 100.00% | 38 | 13 | 34.21% | 0.00001* |
| Tested Positive for H. pylori | Tested Negative for H. pylori | ||||||
|---|---|---|---|---|---|---|---|
| Age Group 58-65 | Number of Patients Evaluated | Number of Patients with Symptom | % Of Patients with Symptom | Number of Patients Evaluated | Number of Patients with Symptom | % Of Patients with Symptom | p value |
| Gender | 25 (F) 8 (M) |
15 (F) 7 (M) |
0.53656 | ||||
| Bloating, | 33 | 0 | 0.00% | 22 | 1 | 4.55% | 0.21645 |
| Nausea | 33 | 3 | 9.09% | 22 | 1 | 4.55% | 0.52482 |
| Persistent Vomiting | 33 | 3 | 9.09% | 22 | 1 | 4.55% | 0.52482 |
| Burning Pain That Worsen When Stomach Is Empty | 33 | 24 | 72.73% | 22 | 0 | 0.00% | 0.00001* |
| Dysphagia | 33 | 5 | 15.15% | 22 | 1 | 4.55% | 0.21645 |
| Unintentional Weight Loss (Over 3 kg in Past Month) | 33 | 7 | 21.21% | 22 | 4 | 18.18% | 0.78313 |
| Feeling of Fullness/ Loss of Appetite | 33 | 1 | 3.03% | 22 | 1 | 4.55% | 0.76870 |
| Diarrhea | 33 | 3 | 9.09% | 22 | 1 | 4.55% | 0.52482 |
| Constipation | 33 | 3 | 9.09% | 22 | 1 | 4.55% | 0.52482 |
| Bloody or Black Tarry Stools | 33 | 4 | 12.12% | 22 | 2 | 9.09% | 0.72397 |
| Total Number of Patients Presented with Symptoms | 33 | 33 | 100.00% | 22 | 9 | 40.91% | 0.00001* |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




