Submitted:
17 April 2026
Posted:
18 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Definitions
2.3. Data Collection
2.4. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Microbiological Findings
3.3. Management
3.4. Complication
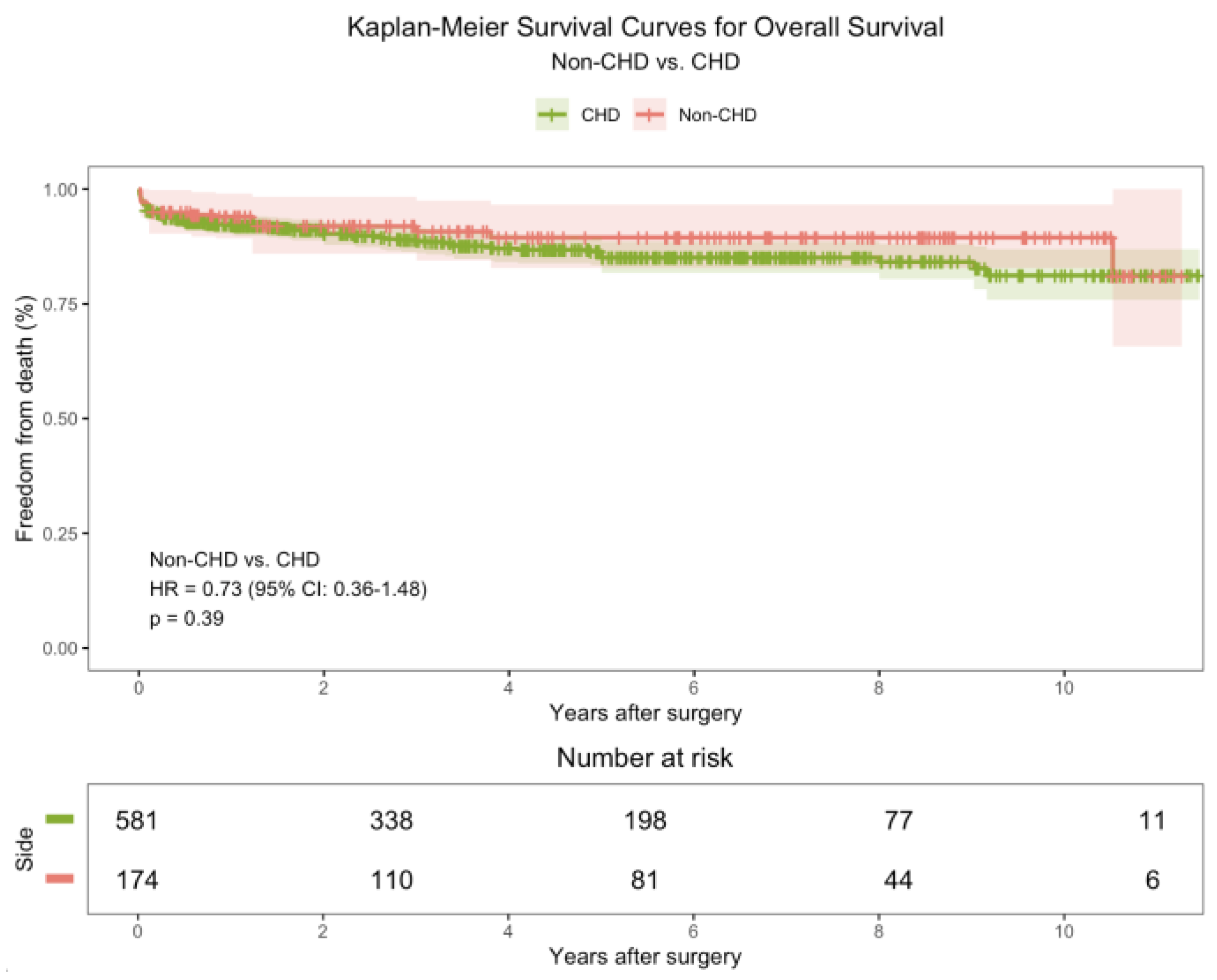
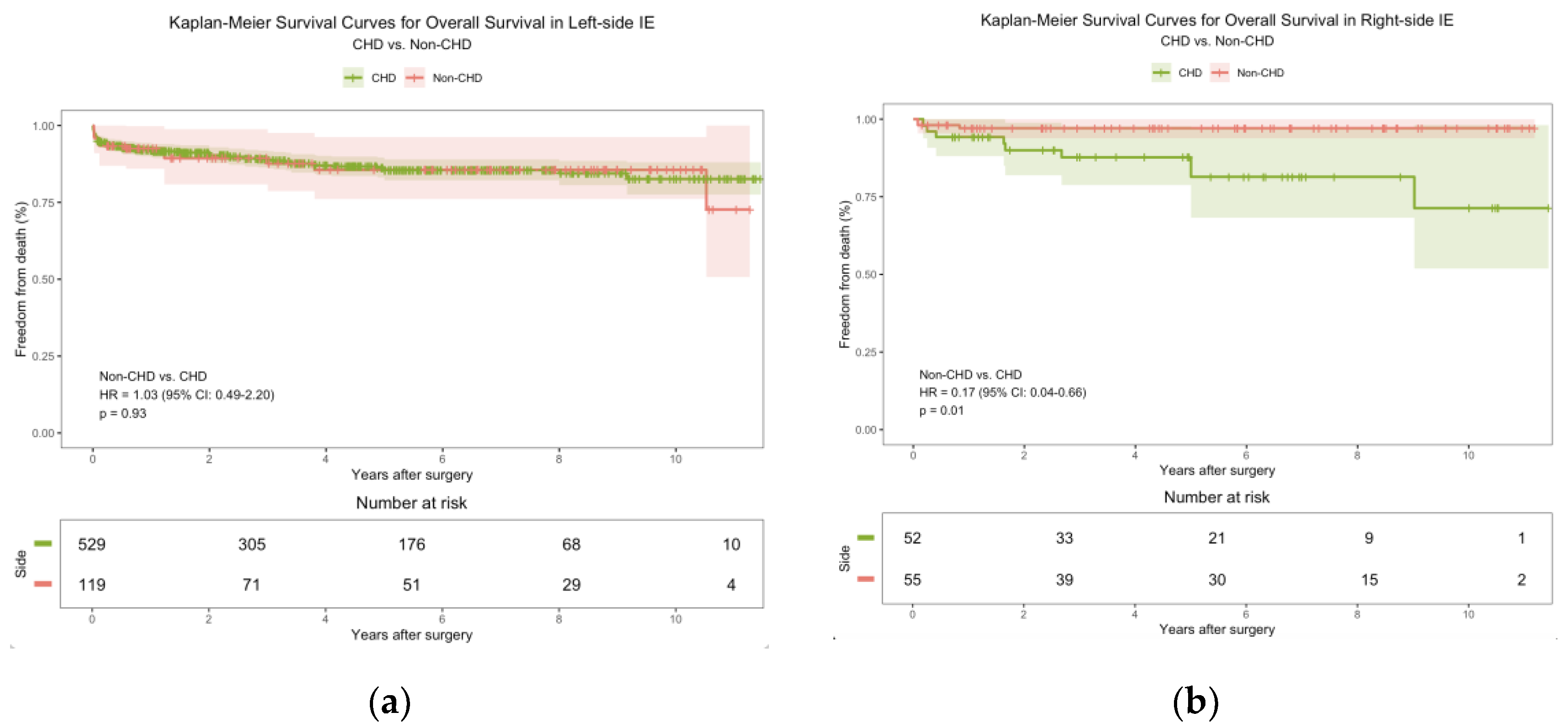
3.5. Outcome
4. Discussion
4.1. Clinical Characteristics
4.2. Microbiological Profile
4.3. Outcomes and Prognosis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CHD | Congenital Heart Disease |
| CPB | Cardiopulmonary Bypass |
| CRRT | Continuous Renal Replacement Therapy |
| MRSA | Methicillin-Resistant Staphylococcus Aureus |
| ECMO | Extracorporeal Membrane Oxygenation |
| ICU | Intensive Care Unit |
| IE | Infective Endocarditis |
| IPTW | Inverse Probability of Treatment Weighting |
References
- Dellborg, M.; et al. Adults With Congenital Heart Disease: Trends in Event-Free Survival Past Middle Age. Circulation 2023, 147(12), 930–938. [Google Scholar] [CrossRef]
- A Mandalenakis, Z.; et al. Survival in Children With Congenital Heart Disease: Have We Reached a Peak at 97%? J Am Heart Assoc 2020, 9(22), p. e017704. [Google Scholar] [CrossRef]
- Bergh, N.; et al. Risk of Heart Failure in Congenital Heart Disease: A Nationwide Register-Based Cohort Study. Circulation 2023, 147(12), 982–984. [Google Scholar] [CrossRef]
- Fedchenko, M.; et al. Risk and predictors of first-time infective endocarditis in adult patients with congenital heart disease-A nationwide, register-based study. Int J Cardiol 2025. 426, p. 133081. [Google Scholar] [CrossRef] [PubMed]
- Østergaard, L.; et al. Temporal changes in infective endocarditis guidelines during the last 12 years: High-level evidence needed. Am Heart J 2017, 193, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Snygg-Martin, U.; et al. Cumulative Incidence of Infective Endocarditis in Patients with Congenital Heart Disease: A Nationwide, Case-Control Study Over Nine Decades. Clin Infect Dis 2021, 73(8), 1469–1475. [Google Scholar] [CrossRef] [PubMed]
- Brida, M.; et al. Infective endocarditis in adults with congenital heart disease: Contemporary management and related outcomes in Central and South-Eastern European region. Int J Cardiol 2023. 377, 45–50. [Google Scholar] [CrossRef]
- Havers-Borgersen, E.; et al. Long-term incidence of infective endocarditis among patients with congenital heart disease. Am Heart J 2023. 259, 9–20. [Google Scholar] [CrossRef]
- van Melle, J.P.; et al. Infective endocarditis in adult patients with congenital heart disease. Int J Cardiol 2023, 370, 178–185. [Google Scholar] [CrossRef]
- Havers-Borgersen, E.; et al. Infective endocarditis with or without congenital heart disease: clinical features and outcomes. Eur Heart J 2024, 45(44), 4704–4715. [Google Scholar] [CrossRef]
- Vicent, L.; et al. Infective endocarditis in children and adolescents: a different profile with clinical implications. Pediatr Res 2022, 92(5), 1400–1406. [Google Scholar] [CrossRef]
- Cahill, T.J.; et al. Contemporary epidemiology of infective endocarditis in patients with congenital heart disease: A UK prospective study. Am Heart J 2019, 215, 70–77. [Google Scholar] [CrossRef]
- Moore, B.; et al. Incidence, predictors and outcomes of infective endocarditis in a contemporary adult congenital heart disease population. Int J Cardiol 2017, 249, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Tutarel, O.; et al. Infective endocarditis in adults with congenital heart disease remains a lethal disease. Heart 2018, 104(2), 161–165. [Google Scholar] [CrossRef] [PubMed]
- Khouchoua, S.; et al. Mycotic Pulmonary Artery Aneurysm a Rare Complication of Ventricular Septal Defect With Infective Endocarditis: A Case Report. Clin Med Insights Case Rep 2024, 17, 11795476241238884. [Google Scholar] [CrossRef] [PubMed]
- Durante-Mangoni, E.; et al. Current features of infective endocarditis in elderly patients: results of the International Collaboration on Endocarditis Prospective Cohort Study. Arch Intern Med 2008, 168(19), 2095–103. [Google Scholar] [CrossRef]
- Remadi, J.P.; et al. Infective endocarditis in elderly patients: clinical characteristics and outcome. Eur J Cardiothorac Surg 2009, 35(1), 123–9. [Google Scholar] [CrossRef]
- Desimone, D.C.; et al. Incidence of infective endocarditis caused by viridans group streptococci before and after publication of the 2007 American Heart Association's endocarditis prevention guidelines. Circulation 2012, 126(1), 60–4. [Google Scholar] [CrossRef]
- Ly, R.; et al. Predictive factors of death associated with infective endocarditis in adult patients with congenital heart disease. Eur Heart J Acute Cardiovasc Care 2021, 10(3), 320–328. [Google Scholar] [CrossRef]
- Mylotte, D.; et al. Incidence, Predictors, and Mortality of Infective Endocarditis in Adults With Congenital Heart Disease Without Prosthetic Valves. Am J Cardiol 2017, 120(12), 2278–2283. [Google Scholar] [CrossRef]
- Verzelloni Sef, A.; et al. Factors associated with long-term outcomes in adult congenital heart disease patients with infective endocarditis: a 16-year tertiary single-centre experience. Eur J Cardiothorac Surg 2023, 63(5). [Google Scholar] [CrossRef] [PubMed]
- Lee, M.R.; et al. Clinical features of right-sided infective endocarditis occurring in non-drug users. J Korean Med Sci 2014, 29(6), 776–81. [Google Scholar] [CrossRef] [PubMed]
- Shmueli, H.; et al. Right-Sided Infective Endocarditis 2020: Challenges and Updates in Diagnosis and Treatment. J Am Heart Assoc 2020, 9(15), p. e017293. [Google Scholar] [CrossRef] [PubMed]
| Overall 773 |
Non-CHD 585 (75.7%) |
CHD 188 (24.3%) |
p-value | |
|---|---|---|---|---|
| Baseline Characteristics | ||||
| Male | 531 (69%) | 409 (70%) | 122 (65%) | 0.2 |
| Age | 46.5 ± 15.1 | 48.6 ± 15.0 | 39.8 ± 13.4 | <0.001 |
| Body mass index, kg/m² | 22.4 ± 3.7 | 22.6 ± 3.7 | 21.7 ± 3.6 | 0.003 |
| Comorbidities | ||||
| Hypertension | 193 (25%) | 172 (29%) | 21 (11%) | <0.001 |
| Coronary artery disease | 73 (9%) | 63 (11%) | 10 (5%) | 0.03 |
| Diabetes mellitus | 87 (11%) | 73 (12%) | 14 (7%) | 0.06 |
| Pneumonia | 68 (9%) | 47 (8%) | 21 (11%) | 0.2 |
| COPD | 17 (2%) | 15 (3%) | 2 (1%) | 0.4 |
| Heart failure | 236 (31%) | 179 (31%) | 57 (30%) | >0.9 |
| pre_dialysis | 31 (4%) | 28 (5%) | 3 (2%) | 0.052 |
| Previous IE | 20 (3%) | 19 (3%) | 1 (1%) | 0.060 |
| Echocardiographic | ||||
| LVEF≤50% | 32 (4%) | 20 (3%) | 12 (6%) | 0.076 |
| Perivalvular Abscess | 107 (14%) | 86 (15%) | 21 (11%) | 0.2 |
| Huge Vegetation | 445 (58%) | 333 (57%) | 112 (60%) | 0.5 |
| Affected Valves | ||||
| Aortic valve | 369 (48%) | 261 (45%) | 108 (57%) | 0.002 |
| Mitral valve | 430 (56%) | 378 (65%) | 52 (28%) | <0.001 |
| Triple valve | 93 (12%) | 41 (7%) | 52 (28%) | <0.001 |
| Pulmonary valve | 44 (6%) | 10 (2%) | 34 (18%) | <0.001 |
| Multiple valves | 156 (20%) | 107 (18%) | 49 (26%) | 0.021 |
| Prosthetic valve endocarditis | 126 (16%) | 105 (18%) | 21 (11%) | 0.029 |
| Left-side | 645 (83%) | 534 (91%) | 111 (59%) | <0.001 |
| Right-side | 97 (13%) | 39 (7%) | 58 (31%) | <0.001 |
| Double-side | 33 (4%) | 12 (2%) | 21 (11%) | <0.001 |
| Embolic events | ||||
| Overall Embolism | 324 (42%) | 257 (44%) | 67 (36%) | 0.045 |
| Cerebrovascular | 182 (24%) | 154 (26%) | 28 (15%) | <0.001 |
| Lung | 47 (6%) | 28 (5%) | 19 (10%) | 0.008 |
| Values are reported as n (%), median (IQR) or mean (± Standard Deviation). Abbreviations: IE = infective endocarditis; COPD = Chronic Obstructive Pulmonary Disease; eGFR = estimated glomerular filtration rate; LVEF = left ventricular ejection fraction. | ||||
| Overall 773 |
Non-CHD 585 (75.7%) |
CHD 188 (24.3%) |
p-value | |
|---|---|---|---|---|
| Pathogen | ||||
| Blood culture positive | 596 (77%) | 454 (78%) | 142 (76%) | 0.6 |
| Streptococcus | 360 (47%) | 264 (45%) | 96 (51%) | 0.2 |
| Staphylococcus | 124 (16%) | 99 (17%) | 25 (13%) | 0.2 |
| S_aureus | 83 (11%) | 66 (11%) | 17 (9%) | 0.4 |
| MRSA | 42 (5%) | 36 (6%) | 6 (3%) | 0.12 |
| Enterococcus | 34 (4%) | 30 (5%) | 4 (2%) | 0.081 |
| HACEK | 19 (2%) | 14 (2%) | 5 (3%) | 0.8 |
| Fungi | 16 (2%) | 16 (3%) | 0 (0%) | 0.017 |
| Values are reported as n (%), median (IQR) or mean (± Standard Deviation). | ||||
| Overall 773 |
Non-CHD 585 (75.7%) |
CHD 188 (24.3%) |
p-value | |
|---|---|---|---|---|
| Re-operation | 101 (13%) | 93 (16%) | 8 (4%) | <0.001 |
| Surgical timing | 0.5 | |||
| Elective | 656 (85%) | 501 (86%) | 155 (82%) | |
| Urgent | 55 (7%) | 41 (7%) | 14 (7%) | |
| Emergency | 62 (8%) | 43 (7%) | 19 (10%) | |
| CPB, min | 148.2 ± 72.9 | 150.7 ± 77.0 | 140.7 ± 57.8 | 0.12 |
| Abbreviations: CPB = cardiopulmonary bypass time | ||||
|
Overall 773 |
Non-CHD 585(75.7%) |
CHD 188(24.3%) |
OR (95CI%) | p-value | |
| CNS | 22 (3%) | 18 (3%) | 4 (2%) | 1.03 (0.25-4.25) | 0.96 |
| Long ventilation | 166 (21%) | 133 (23%) | 33 (18%) | 1.07 (0.64-1.80) | 0.80 |
| Pneumonia | 81 (10%) | 64 (11%) | 17 (9%) | 1.89 (0.46-1.72) | 0.73 |
| Atrial fibrillation | 111 (14%) | 92 (16%) | 19 (10%) | 1.19 (0.66-2.15) | 0.57 |
| Atrioventricular block | 33 (4%) | 25 (4%) | 8 (4%) | 0.77 (0.31-1.90) | 0.59 |
| Re-thoracotomy | 44 (6%) | 38 (6%) | 6 (3%) | 1.85 (0.66-5.18) | 0.29 |
| Low Cardiac Output Syndrome | 61 (8%) | 45 (8%) | 16 (9%) | 0.55 (0.28-1.08) | 0.10 |
| Septic shock | 33 (4%) | 28 (5%) | 5 (3%) | 1.39 (0.49-3.95) | 0.59 |
| ECMO | 15 (2%) | 11 (2%) | 4 (2%) | 0.60 (0.15-2.44) | 0.48 |
| Pacemaker | 20 (3%) | 12 (2%) | 8 (4%) | 0.39 (0.15-1.05) | 0.06 |
| CRRT | 85 (11%) | 69 (12%) | 16 (9%) | 1.13 (0.58-2.19) | 0.72 |
| Treatment Time | |||||
| ICU time, days | 4.0 ± 6.3 | 4.2 ± 7.0 | 3.2 ± 3.5 | 0.57 (0.26-1.27) | 0.17 |
| Hospital time, days | 13.2 ± 11.3 | 13.5 ± 12.3 | 12.3 ± 7.3 | 0.51 (0.10-2.65) | 0.42 |
| Early mortality | 33 (4%) | 26 (4%) | 7 (4%) | 0.59 (0.22-1.62) | 0.31 |
| Values are reported as odds ratios (ORs) with 95% confidence intervals (CIs). Inverse probability of treatment weighting (IPTW) was applied to balance baseline covariates. Non-CHD was specified as the reference group in all weighted logistic regression models. An OR >1 indicates higher odds of the outcome in Non-CHD compared with CHD. | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




