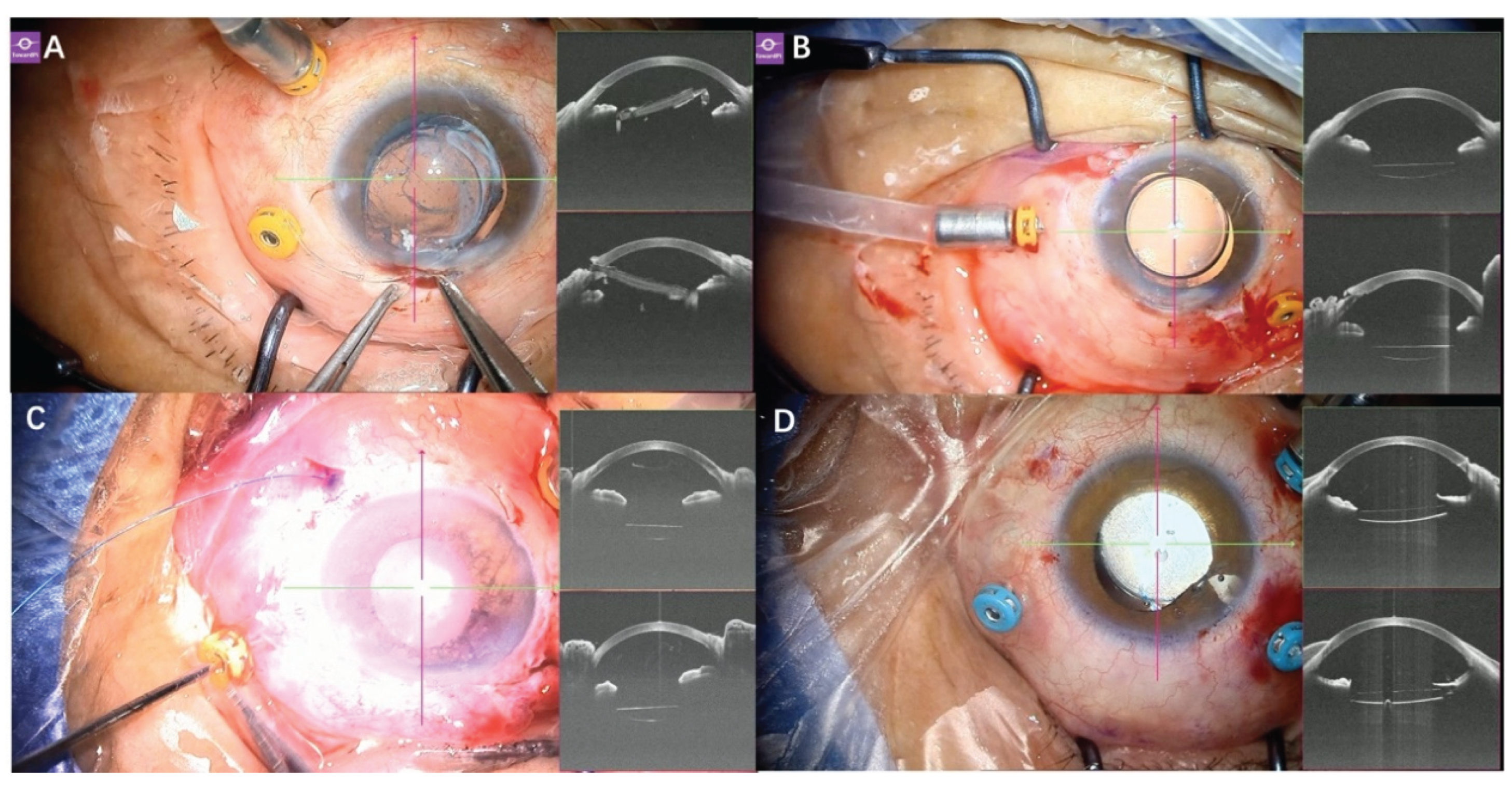
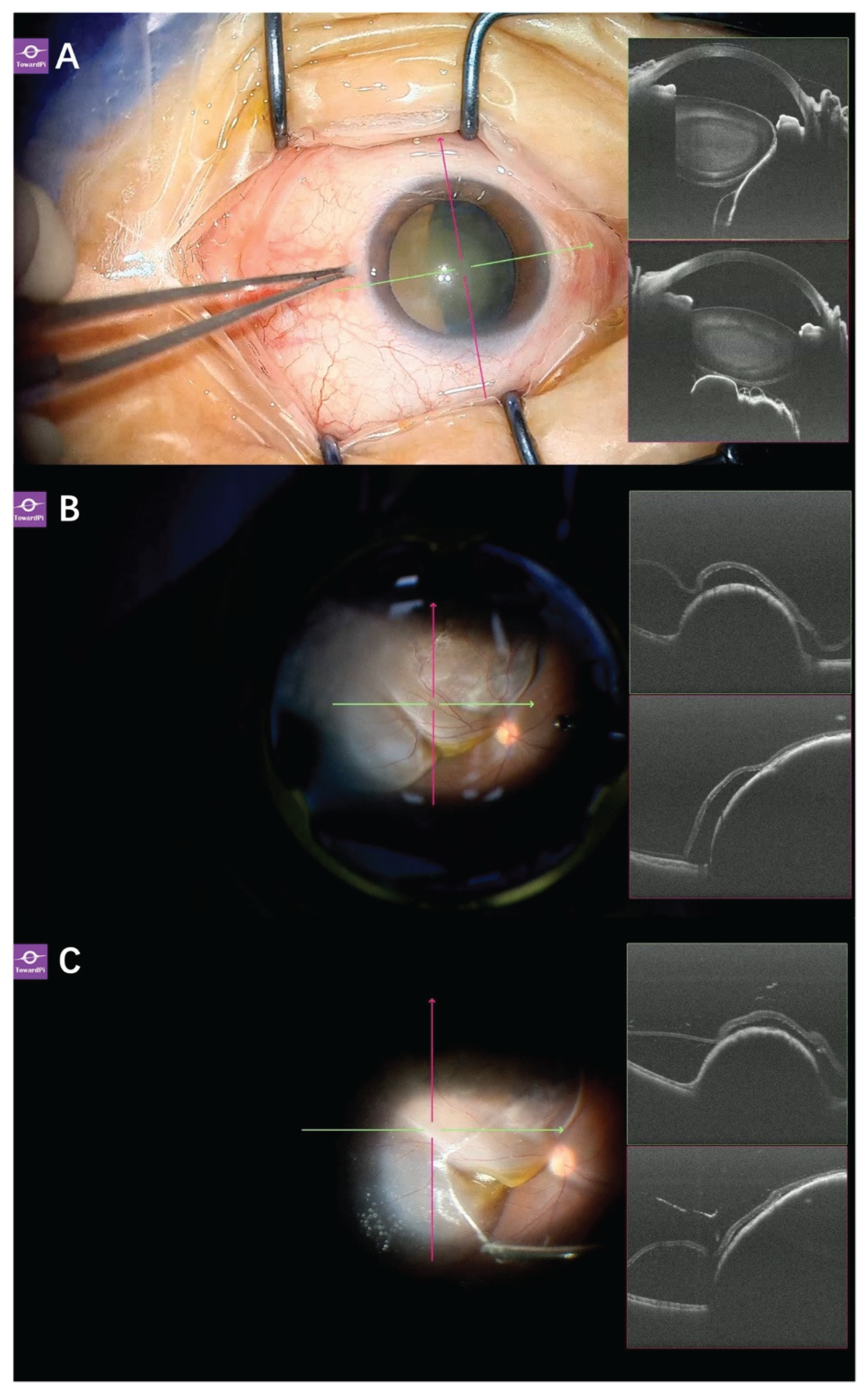
Figure 1.
Appl. ation of real-time SS-iOCT in anterior segment surgery. (A) The microscope is equipped with a “Mendez Ring” for astigmatism correction and IOL ciliary sulcus fixation; (B) In addition to the standard line-scan mode, it can display the angle structures at eight clock hours during anterior segment surgery; (C) iOCT demonstrates the layered structure of the lesion in a 33-year-old male MHRD patient with adherent leucoma, facilitating teaching and illustrating the iris-cornea relationship; (D) iOCT reveals posterior synechiae during phacovitrectomy in a 66-year-old female with RRD; (E) A 47-year-old female patient with retinal detachment secondary to coloboma of the choroid, microcornea (diameter 7×6 mm), and nuclear cataract graded N4. Anterior segment OCT was used to observe the anterior chamber structures and assist in determining the location of the corneal incision. Based on the assessment of anterior segment OCT, the angle structure was like that of a normal cornea, and a clear corneal incision was performed; (F) SS-iOCT confirmed that the corneal incision was appropriately positioned; (G) In a 33-year-old male with recurrent retinal detachment, SS-iOCT revealed angle adhesions and silicone oil droplets in the anterior chamber; (H) Goniosynechialysis was performed with a viscoelastic agent, and OCT confirmed the angle had been re-opened after its removal.
Figure 1.
Appl. ation of real-time SS-iOCT in anterior segment surgery. (A) The microscope is equipped with a “Mendez Ring” for astigmatism correction and IOL ciliary sulcus fixation; (B) In addition to the standard line-scan mode, it can display the angle structures at eight clock hours during anterior segment surgery; (C) iOCT demonstrates the layered structure of the lesion in a 33-year-old male MHRD patient with adherent leucoma, facilitating teaching and illustrating the iris-cornea relationship; (D) iOCT reveals posterior synechiae during phacovitrectomy in a 66-year-old female with RRD; (E) A 47-year-old female patient with retinal detachment secondary to coloboma of the choroid, microcornea (diameter 7×6 mm), and nuclear cataract graded N4. Anterior segment OCT was used to observe the anterior chamber structures and assist in determining the location of the corneal incision. Based on the assessment of anterior segment OCT, the angle structure was like that of a normal cornea, and a clear corneal incision was performed; (F) SS-iOCT confirmed that the corneal incision was appropriately positioned; (G) In a 33-year-old male with recurrent retinal detachment, SS-iOCT revealed angle adhesions and silicone oil droplets in the anterior chamber; (H) Goniosynechialysis was performed with a viscoelastic agent, and OCT confirmed the angle had been re-opened after its removal.
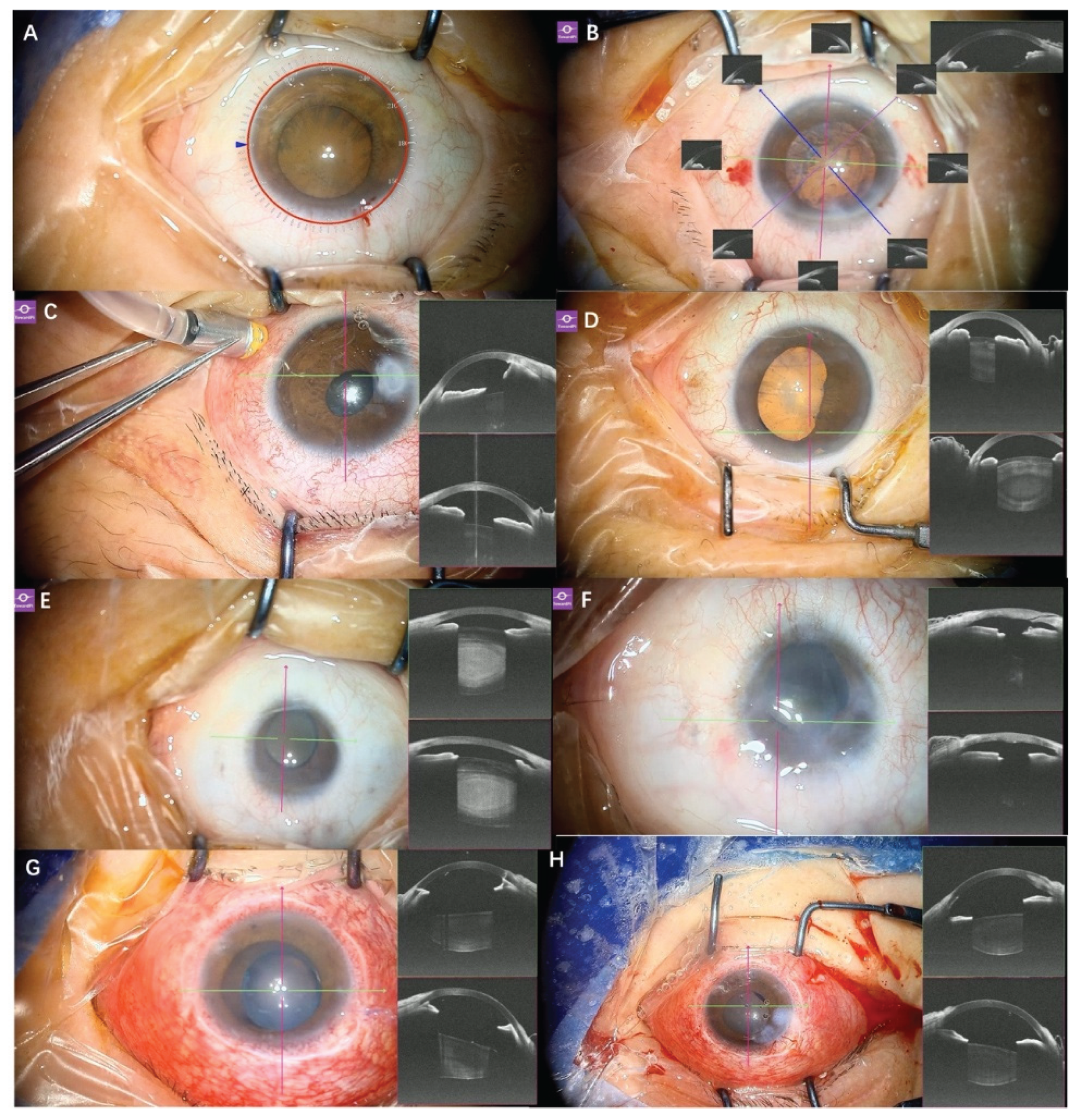
Figure 2.
Application of real-time SS-iOCT in cataract surgery. (A) In a 79-year-old male patient with vitreomacular Traction (VMT) undergoing phacovitrectomy, SS-iOCT clearly visualized the corneal incision; (B) For this 72-year-old PCV patient undergoing phacovitrectomy, SS-iOCT showed clear images following hydrodelineation (golden ring under the microscope) and hydrodissection; (C) In the 79-year-old male patient with VMT, the anterior surge of the cortex, posterior capsule, and anterior vitreous following phacoemulsification indicated intraoperative disturbance of the vitreous during cataract surgery; (D) In a 65-year-old female patient with a macular hole (MH) undergoing phacovitrectomy, following cortical aspiration, OCT revealed anterior vitreous prolapse and apposition of the anterior and posterior capsules, despite a watertight incision and normal anterior chamber depth. This indicated that the vitreous had been disturbed during the cataract surgery; (E) In this 77-year-old female patient with LMH undergoing phacovitrectomy, prior to IOL insertion, OCT clearly demonstrated the capsular bag being expanded with a viscoelastic agent, facilitating IOL implantation; (F) In this LMH patient, after IOL implantation and aspiration of the viscoelastic agent from the capsular bag, posterior capsule folds were observed under both the microscope and OCT; (G) In this VMT patient, after the IOL was implanted in the capsular bag, OCT confirmed the IOL was well-positioned; (H) Following fluid-air exchange, OCT was still able to visualize the IOL position despite the presence of gas in the anterior chamber.
Figure 2.
Application of real-time SS-iOCT in cataract surgery. (A) In a 79-year-old male patient with vitreomacular Traction (VMT) undergoing phacovitrectomy, SS-iOCT clearly visualized the corneal incision; (B) For this 72-year-old PCV patient undergoing phacovitrectomy, SS-iOCT showed clear images following hydrodelineation (golden ring under the microscope) and hydrodissection; (C) In the 79-year-old male patient with VMT, the anterior surge of the cortex, posterior capsule, and anterior vitreous following phacoemulsification indicated intraoperative disturbance of the vitreous during cataract surgery; (D) In a 65-year-old female patient with a macular hole (MH) undergoing phacovitrectomy, following cortical aspiration, OCT revealed anterior vitreous prolapse and apposition of the anterior and posterior capsules, despite a watertight incision and normal anterior chamber depth. This indicated that the vitreous had been disturbed during the cataract surgery; (E) In this 77-year-old female patient with LMH undergoing phacovitrectomy, prior to IOL insertion, OCT clearly demonstrated the capsular bag being expanded with a viscoelastic agent, facilitating IOL implantation; (F) In this LMH patient, after IOL implantation and aspiration of the viscoelastic agent from the capsular bag, posterior capsule folds were observed under both the microscope and OCT; (G) In this VMT patient, after the IOL was implanted in the capsular bag, OCT confirmed the IOL was well-positioned; (H) Following fluid-air exchange, OCT was still able to visualize the IOL position despite the presence of gas in the anterior chamber.
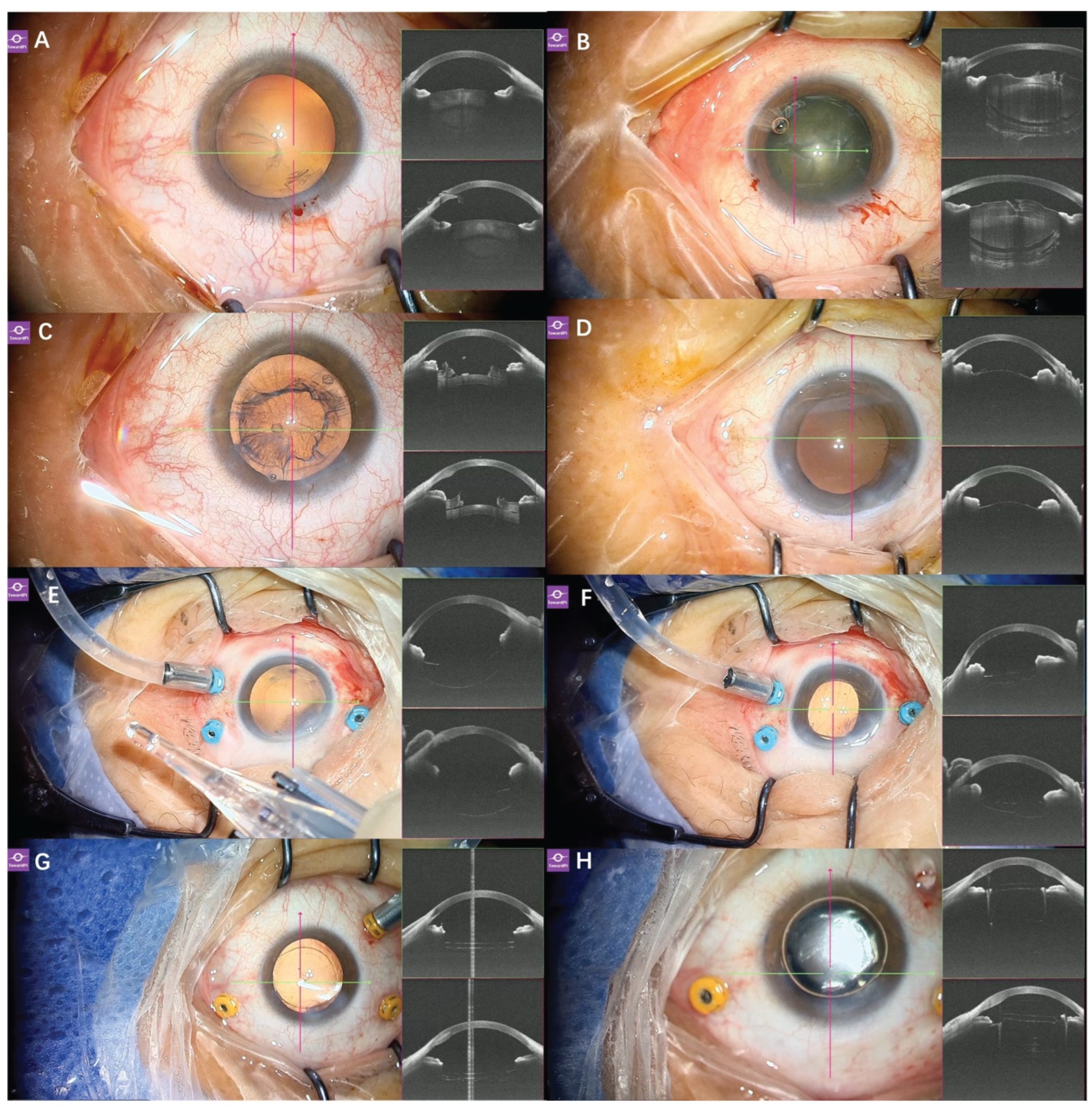
Figure 3.
Application of real-time SS-iOCT in IOL ciliary sulcus fixation surgery. (A) In a 57-year-old patient with a dislocated IOL in the vitreous cavity, SS-iOCT confirmed that the IOL and capsular tension ring were removed through a clear corneal incision without contacting the corneal endothelium; (B) Following the Yamane technique, SS-iOCT confirmed that the IOL was well-positioned; (C) In a 62-year-old female patient undergoing IOL ciliary sulcus fixation with prolene sutures, intraoperative adjustments were made based on the IOL position along two perpendicular meridians as visualized by SS-iOCT. This helped ensure optimal IOL positioning and minimize postoperative astigmatism; (D) In a 59-year-old male patient, SS-iOCT demonstrated the immediate postoperative result following combined Yamane technique and iris suture, confirming that the IOL was well-positioned.
Figure 3.
Application of real-time SS-iOCT in IOL ciliary sulcus fixation surgery. (A) In a 57-year-old patient with a dislocated IOL in the vitreous cavity, SS-iOCT confirmed that the IOL and capsular tension ring were removed through a clear corneal incision without contacting the corneal endothelium; (B) Following the Yamane technique, SS-iOCT confirmed that the IOL was well-positioned; (C) In a 62-year-old female patient undergoing IOL ciliary sulcus fixation with prolene sutures, intraoperative adjustments were made based on the IOL position along two perpendicular meridians as visualized by SS-iOCT. This helped ensure optimal IOL positioning and minimize postoperative astigmatism; (D) In a 59-year-old male patient, SS-iOCT demonstrated the immediate postoperative result following combined Yamane technique and iris suture, confirming that the IOL was well-positioned.
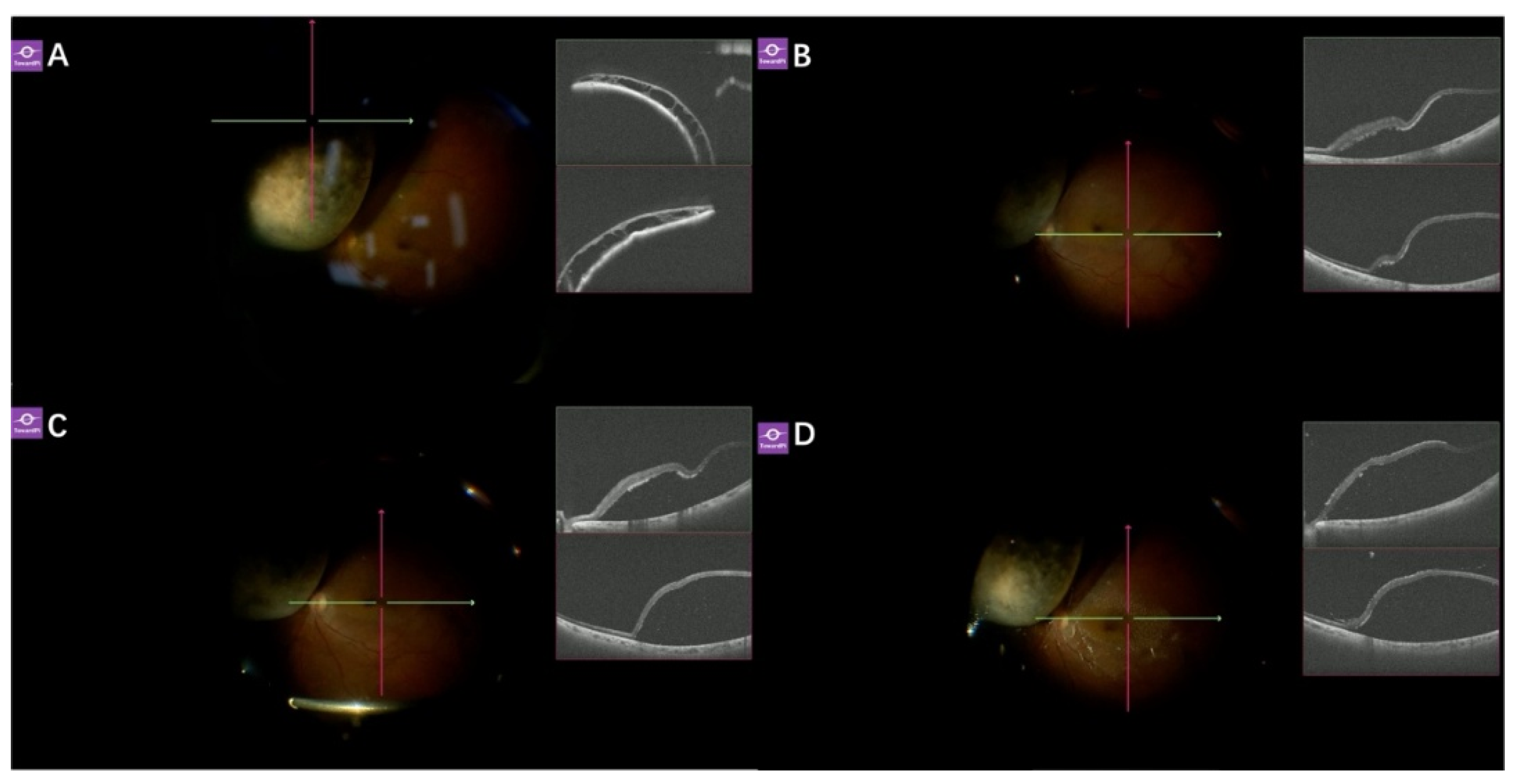
Figure 4.
Application of real-time SS-iOCT in RRD surgery. (A) In an 18-year-old male patient with RRD undergoing 25G endoilluminator-assisted scleral buckling, in addition to a definite superior retinal break and detachment, there was an area of peripheral degeneration inferiorly where the presence of retinal detachment was difficult to determine. SS-iOCT revealed that the inferior degenerative area was associated with shallow retinal detachment; (B) Despite scleral indentation, shallow retinal detachment persisted. Therefore, an external buckle was also applied to the inferior quadrant during surgery; (C) A 48-year-old female patient with RRD presented with extensive preoperative retinal detachment that precluded evaluation of macular structure from imaging. With an axial length of 30.12 mm and diffuse posterior pole atrophy, it was difficult to determine whether a macular hole was present. Performing ILM peeling in the setting of macular thinning would increase the risk of postoperative macular hole formation. SS-iOCT confirmed the absence of a macular hole, and consequently, ILM peeling was not performed during surgery; (D) In a 47-year-old female patient with choroidal coloboma associated retinal detachment, SS-iOCT facilitated the identification of structures within the colobomatous area, including the optic disc and prepapillary glial tissue located in the region of the choroidal coloboma; (E) After perfluoro-n-octane injection, the optic disc appeared clearer under the microscope compared to its detached state, and OCT was able to clearly visualize the optic disc; (F)Due to the absence of a typical highly reflective RPE layer within the choroidal coloboma area, the structures were difficult to identify.
Figure 4.
Application of real-time SS-iOCT in RRD surgery. (A) In an 18-year-old male patient with RRD undergoing 25G endoilluminator-assisted scleral buckling, in addition to a definite superior retinal break and detachment, there was an area of peripheral degeneration inferiorly where the presence of retinal detachment was difficult to determine. SS-iOCT revealed that the inferior degenerative area was associated with shallow retinal detachment; (B) Despite scleral indentation, shallow retinal detachment persisted. Therefore, an external buckle was also applied to the inferior quadrant during surgery; (C) A 48-year-old female patient with RRD presented with extensive preoperative retinal detachment that precluded evaluation of macular structure from imaging. With an axial length of 30.12 mm and diffuse posterior pole atrophy, it was difficult to determine whether a macular hole was present. Performing ILM peeling in the setting of macular thinning would increase the risk of postoperative macular hole formation. SS-iOCT confirmed the absence of a macular hole, and consequently, ILM peeling was not performed during surgery; (D) In a 47-year-old female patient with choroidal coloboma associated retinal detachment, SS-iOCT facilitated the identification of structures within the colobomatous area, including the optic disc and prepapillary glial tissue located in the region of the choroidal coloboma; (E) After perfluoro-n-octane injection, the optic disc appeared clearer under the microscope compared to its detached state, and OCT was able to clearly visualize the optic disc; (F)Due to the absence of a typical highly reflective RPE layer within the choroidal coloboma area, the structures were difficult to identify.
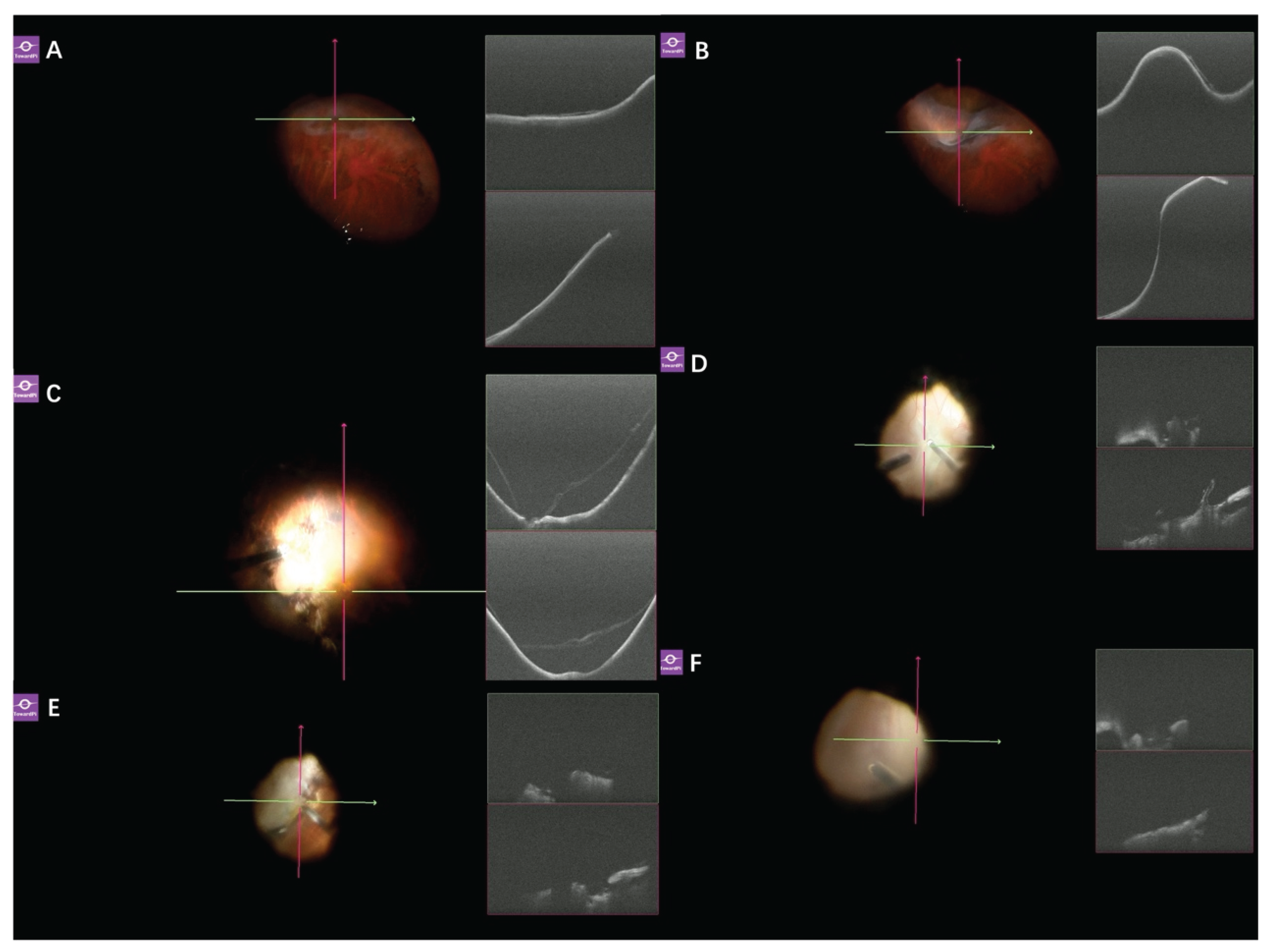
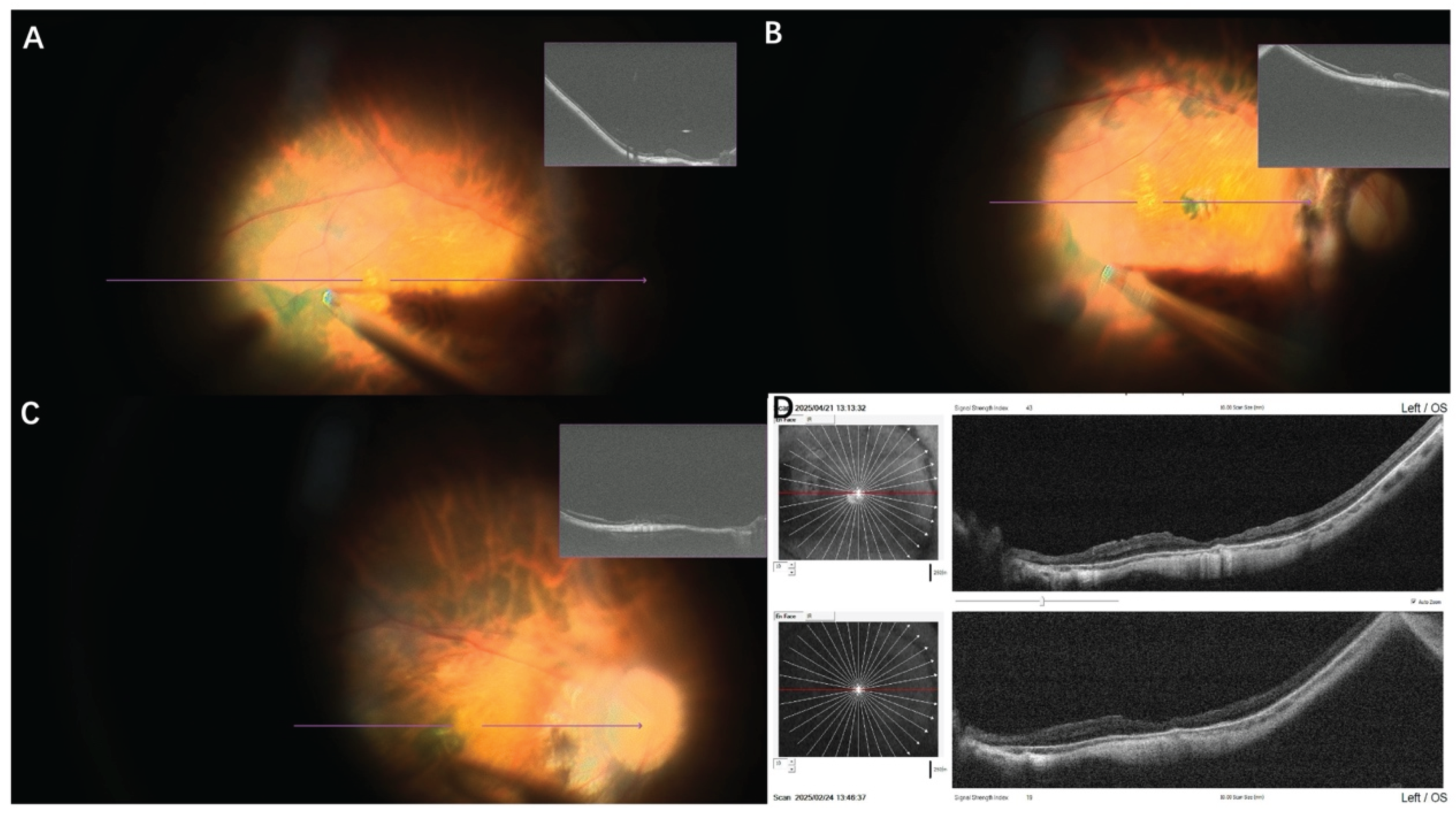
Figure 5.
Application of real-time SS-iOCT in a 58-year-old female with a macular hole measuring 921 μm in diameter and an axial length of 30.12 mm, who underwent PPV with ILM insertion technique. (A) The margins of the hole appeared rigid, without significant elevation; (B) An ILM flap was created and plugged into the macular hole. SS-iOCT revealed the inserted ILM flap positioned on the temporal side of the hole; (C) Another ILM flap was created and inserted into the nasal side of the macular hole to improve the macular hole closure rate; (D) The macular hole closed three months postoperatively (February 2025).
Figure 5.
Application of real-time SS-iOCT in a 58-year-old female with a macular hole measuring 921 μm in diameter and an axial length of 30.12 mm, who underwent PPV with ILM insertion technique. (A) The margins of the hole appeared rigid, without significant elevation; (B) An ILM flap was created and plugged into the macular hole. SS-iOCT revealed the inserted ILM flap positioned on the temporal side of the hole; (C) Another ILM flap was created and inserted into the nasal side of the macular hole to improve the macular hole closure rate; (D) The macular hole closed three months postoperatively (February 2025).
Figure 6.
Application of real-time SS-iOCT in a 66-year-old female with a macular hole measuring 2227 μm in diameter and an axial length of 22.32mm, who underwent PPV with amniotic membrane insertion technique. (A) The margins of the hole appeared rigid; (B) The amniotic membrane was trimmed and then plugged into the macular hole without touching the RPE; (C) The nasal side had already been inserted into the hole; (D) The inserted amniotic membrane remained visible after fluid-air exchange.
Figure 6.
Application of real-time SS-iOCT in a 66-year-old female with a macular hole measuring 2227 μm in diameter and an axial length of 22.32mm, who underwent PPV with amniotic membrane insertion technique. (A) The margins of the hole appeared rigid; (B) The amniotic membrane was trimmed and then plugged into the macular hole without touching the RPE; (C) The nasal side had already been inserted into the hole; (D) The inserted amniotic membrane remained visible after fluid-air exchange.
Figure 7.
Application of real-time SS-iOCT in a 79-year-old male with VMT and an axial length of 34.29mm, who underwent phacovitrectomy. (A) In an eye with an axial length of 34 mm, SS-iOCT was still able to clearly visualize macular schisis and vitreous traction; (B) Even with ICG staining present and not yet aspirated, OCT was still able to produce clear images; (C) Even without endoillumination, OCT was able to produce clear images under the light source of the operating microscope; (D) Even with the microscope light source turned off, OCT was still able to capture images.
Figure 7.
Application of real-time SS-iOCT in a 79-year-old male with VMT and an axial length of 34.29mm, who underwent phacovitrectomy. (A) In an eye with an axial length of 34 mm, SS-iOCT was still able to clearly visualize macular schisis and vitreous traction; (B) Even with ICG staining present and not yet aspirated, OCT was still able to produce clear images; (C) Even without endoillumination, OCT was able to produce clear images under the light source of the operating microscope; (D) Even with the microscope light source turned off, OCT was still able to capture images.
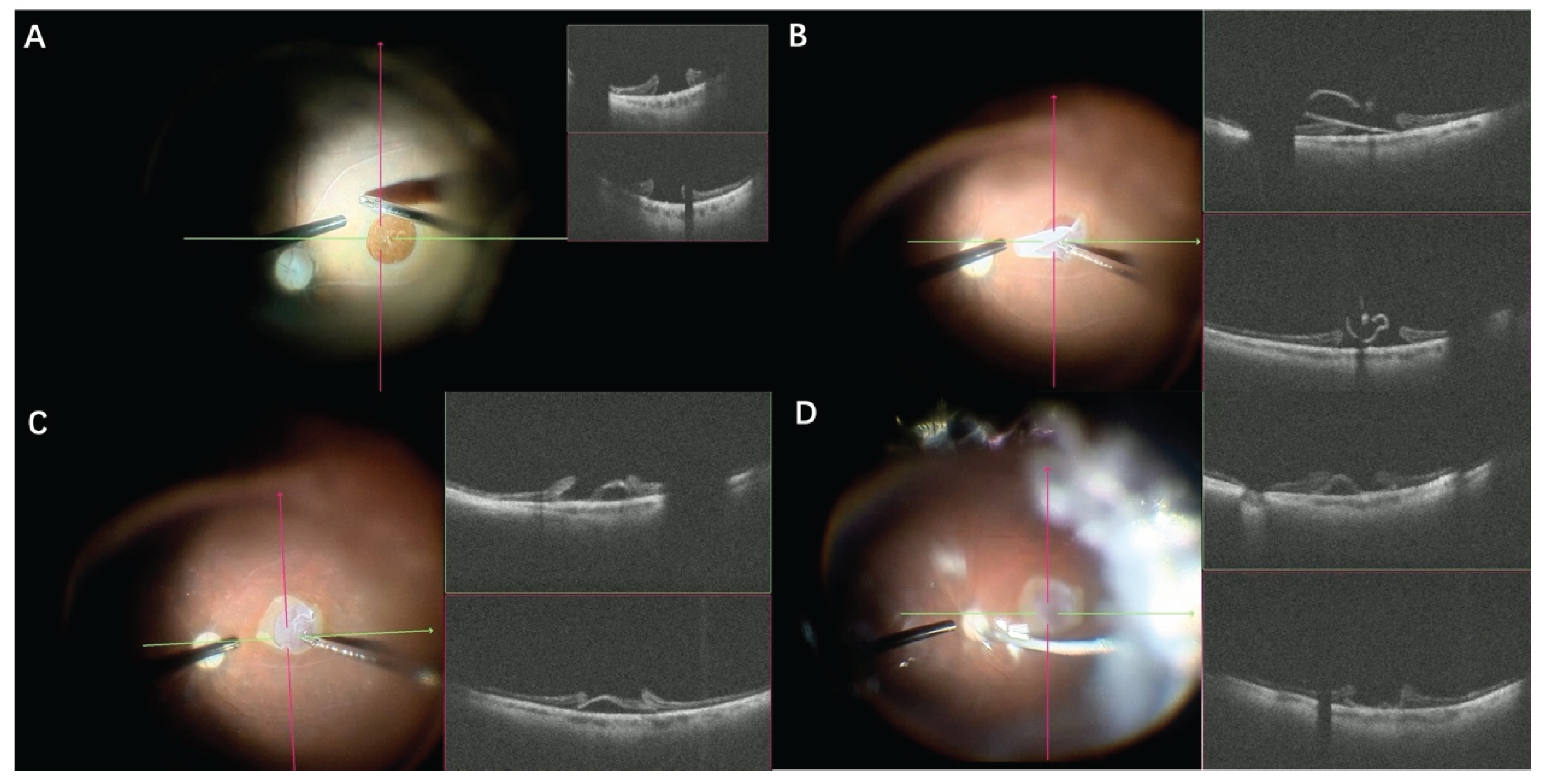
Figure 8.
Application of real-time SS-iOCT in a 72-year-old male with PCV and vitreous hemorrhage, who underwent phacovitrectomy. (A) SS-iOCT clearly demonstrates the cataract as well as the sand-like opacities within the anterior vitreous; (B) SS-iOCT clearly demonstrated the anteriorly displaced posterior capsule adhered to the anterior capsule, as well as the highly reflective anterior hyaloid membrane adherent to the posterior capsule following cataract surgery; (C) When the vitrectomy probe engaged the anterior hyaloid membrane, SS-iOCT showed the separation of the anterior hyaloid membrane from the posterior capsule; (D) After clearing the anterior hyaloid membrane, viscoelastic agent was injected into the capsular bag. SS-iOCT revealed the absence of the anterior hyaloid membrane, with a distinct separation between the anterior and posterior capsules, appearing as two hyperreflective bands. The posterior capsule remained intact; (E) After ILM peeling, SS-iOCT revealed that the 41G needle had touched the retina, causing indentation of the retinal tissue; (F) After successful injection, fluctuation of the subretinal blood could be observed, confirming that t-PA had been delivered into the subretinal space.
Figure 8.
Application of real-time SS-iOCT in a 72-year-old male with PCV and vitreous hemorrhage, who underwent phacovitrectomy. (A) SS-iOCT clearly demonstrates the cataract as well as the sand-like opacities within the anterior vitreous; (B) SS-iOCT clearly demonstrated the anteriorly displaced posterior capsule adhered to the anterior capsule, as well as the highly reflective anterior hyaloid membrane adherent to the posterior capsule following cataract surgery; (C) When the vitrectomy probe engaged the anterior hyaloid membrane, SS-iOCT showed the separation of the anterior hyaloid membrane from the posterior capsule; (D) After clearing the anterior hyaloid membrane, viscoelastic agent was injected into the capsular bag. SS-iOCT revealed the absence of the anterior hyaloid membrane, with a distinct separation between the anterior and posterior capsules, appearing as two hyperreflective bands. The posterior capsule remained intact; (E) After ILM peeling, SS-iOCT revealed that the 41G needle had touched the retina, causing indentation of the retinal tissue; (F) After successful injection, fluctuation of the subretinal blood could be observed, confirming that t-PA had been delivered into the subretinal space.
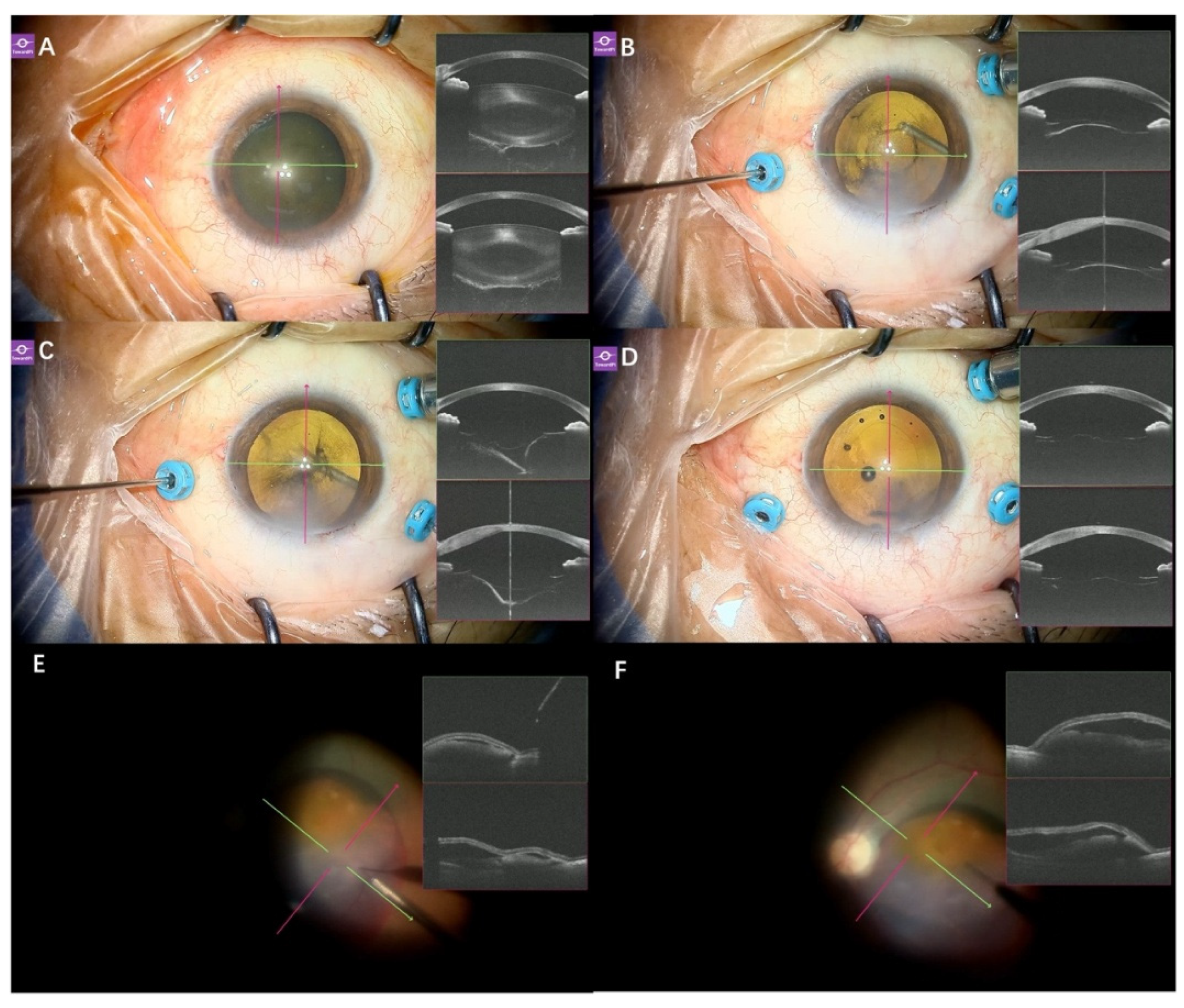
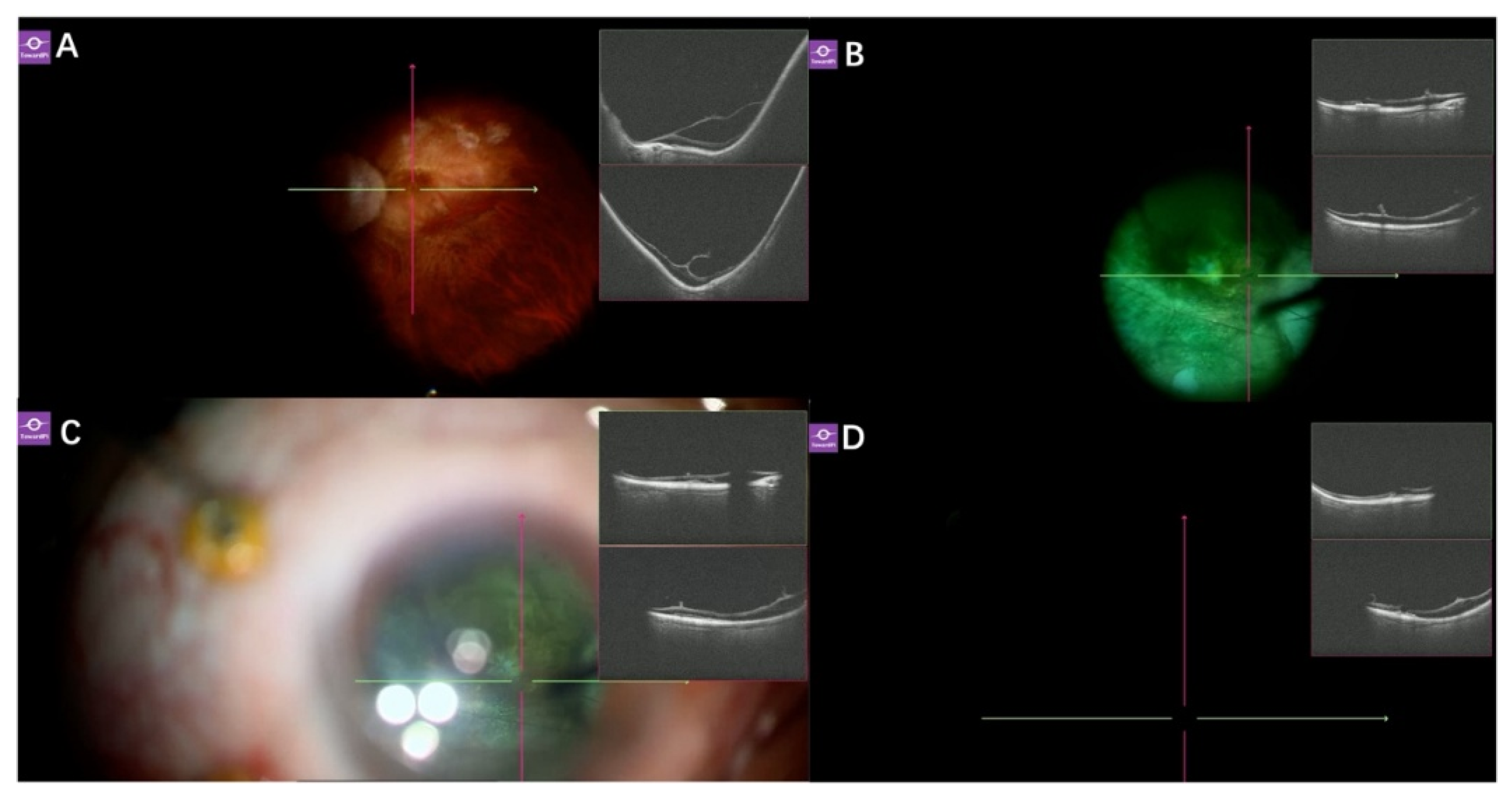
Figure 9.
Surgical imaging and real-time SS-iOCT of a 58-year-old male diagnosed with choroidal melanoma undergoing PPV and endoresection. (A) Prior to vitrectomy, microscope view reveals an elevated lesion with overlying retinal degeneration and cystoid changes; (B) Microscope view shows pigmentary abnormalities in the macular region. SS-iOCT clearly demonstrates subretinal hyper-reflective material, tumor-associated retinal pigmentation (TARP); (C) SS-iOCT reveals retinal detachment with TARP and punctate hyper-reflective foci within the subretinal fluid; (D) Following TA staining, TA particles are visible on the retinal surface, exhibiting sharper borders and higher reflectivity compared to TARP.
Figure 9.
Surgical imaging and real-time SS-iOCT of a 58-year-old male diagnosed with choroidal melanoma undergoing PPV and endoresection. (A) Prior to vitrectomy, microscope view reveals an elevated lesion with overlying retinal degeneration and cystoid changes; (B) Microscope view shows pigmentary abnormalities in the macular region. SS-iOCT clearly demonstrates subretinal hyper-reflective material, tumor-associated retinal pigmentation (TARP); (C) SS-iOCT reveals retinal detachment with TARP and punctate hyper-reflective foci within the subretinal fluid; (D) Following TA staining, TA particles are visible on the retinal surface, exhibiting sharper borders and higher reflectivity compared to TARP.
Figure 10.
Application of real-time SS-iOCT in PPV and endoresection surgery. (A) In a 48-year-old female patient with an intraocular mass, SS-iOCT clearly demonstrated the mass in contact with the lens, as well as the retinal detachment on the surface of the mass; (B) In an 18-year-old female patient with uveal melanoma (UM), SS-iOCT revealed retinal detachment on the surface of the tumor. TARP was also observed on SS-iOCT; (C) After artificial posterior vitreous detachment (PVD) induction, a Weiss ring was observed, and SS-iOCT clearly visualized the posterior vitreous cortex.
Figure 10.
Application of real-time SS-iOCT in PPV and endoresection surgery. (A) In a 48-year-old female patient with an intraocular mass, SS-iOCT clearly demonstrated the mass in contact with the lens, as well as the retinal detachment on the surface of the mass; (B) In an 18-year-old female patient with uveal melanoma (UM), SS-iOCT revealed retinal detachment on the surface of the tumor. TARP was also observed on SS-iOCT; (C) After artificial posterior vitreous detachment (PVD) induction, a Weiss ring was observed, and SS-iOCT clearly visualized the posterior vitreous cortex.