Submitted:
16 April 2026
Posted:
16 April 2026
You are already at the latest version
Abstract
This work develops and analyzes a mathematical model of SARS-CoV-2 infection within the human host, incorporating susceptible and infected epithelial cells, viral particles, ACE2 receptors, cytotoxic T lymphocytes (CTLs), and antibodies. The basic reproduction number and equilibrium points are derived, with stability analysis showing that the disease-free equilibrium is maintained when \( \mathcal{R}_0 < 1 \), while an endemic equilibrium arises for \( \mathcal{R}_0 > 1 \). To capture therapeutic intervention, an impulsive control framework based on antibody-mediated drug administration is introduced. Within this framework, the existence and stability of a disease-free periodic orbit are established through the impulsive reproduction number, \( \mathcal{R}_0^{imp} \), with stability ensured when \( \mathcal{R}_0^{imp} < 1 \). Numerical simulations confirm the analytical results, demonstrating the effectiveness of impulsive therapy in achieving viral eradication. Additionally, Hopf bifurcating periodic solutions are observed under elevated viral replication and infection rates. The proposed model provides new insights into the interaction between viral dynamics, immune response, and impulsive therapeutic strategies, offering a rigorous foundation for advancing treatment approaches against SARS-CoV-2.
Keywords:
1. Introduction
2. Mathematical Model Derivation
- (i)
- Uninfected susceptible epithelial cells, , located in the respiratory tract (lungs, nasal, tracheal, and bronchial tissues),
- (ii)
- Infected virus-producing epithelial cells, ,
- (iii)
- Free virus particles, ,
- (iv)
- ACE2 receptors on epithelial cells, ,
- (v)
- Cytotoxic T lymphocytes (CTLs) targeting infected cells, , and
- (vi)
- Antibodies produced by B cells in response to infection, .
- The model incorporates the following biological mechanisms:
3. Dynamics Study Using the Model
3.1. Equilibria
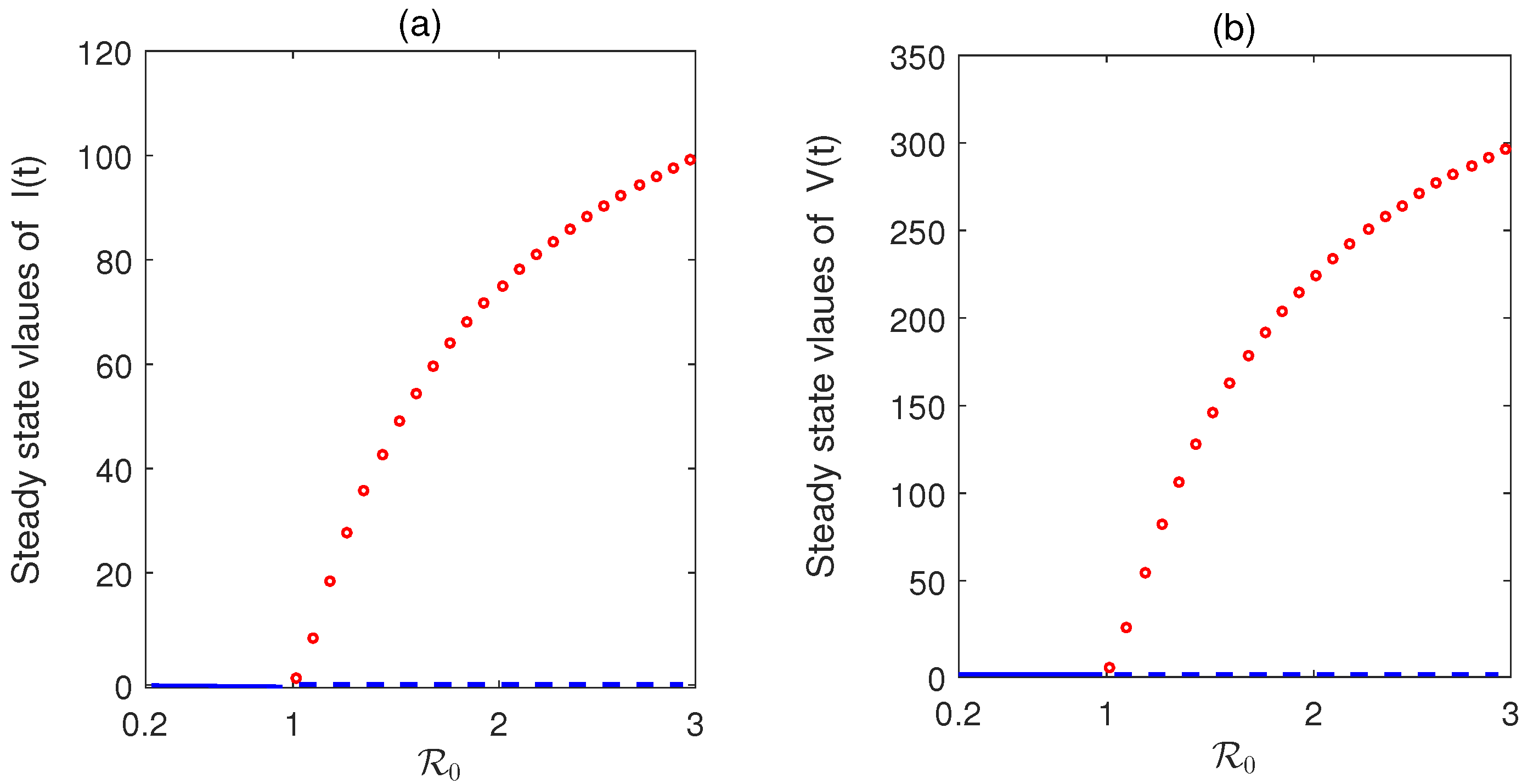
3.2. The Basic Reproduction Number
3.3. Stability Analysis
3.3.1. Stability Analysis of the Disease-Free Equilibrium
- (i)
- If , then , and all eigenvalues have negative real parts. The DFE is locally asymptotically stable.
- (ii)
- If , then , and one eigenvalue becomes positive. The DFE is unstable.
3.3.2. Stability of
- i)
- ii)
- iii)
- iv)
- v)
- vi)
- Transversality condition given below should be satisfied:
4. The System with Impulsive Control
5. Dynamics of the Impulsive System
5.1. The Disease-Free Periodic Orbit
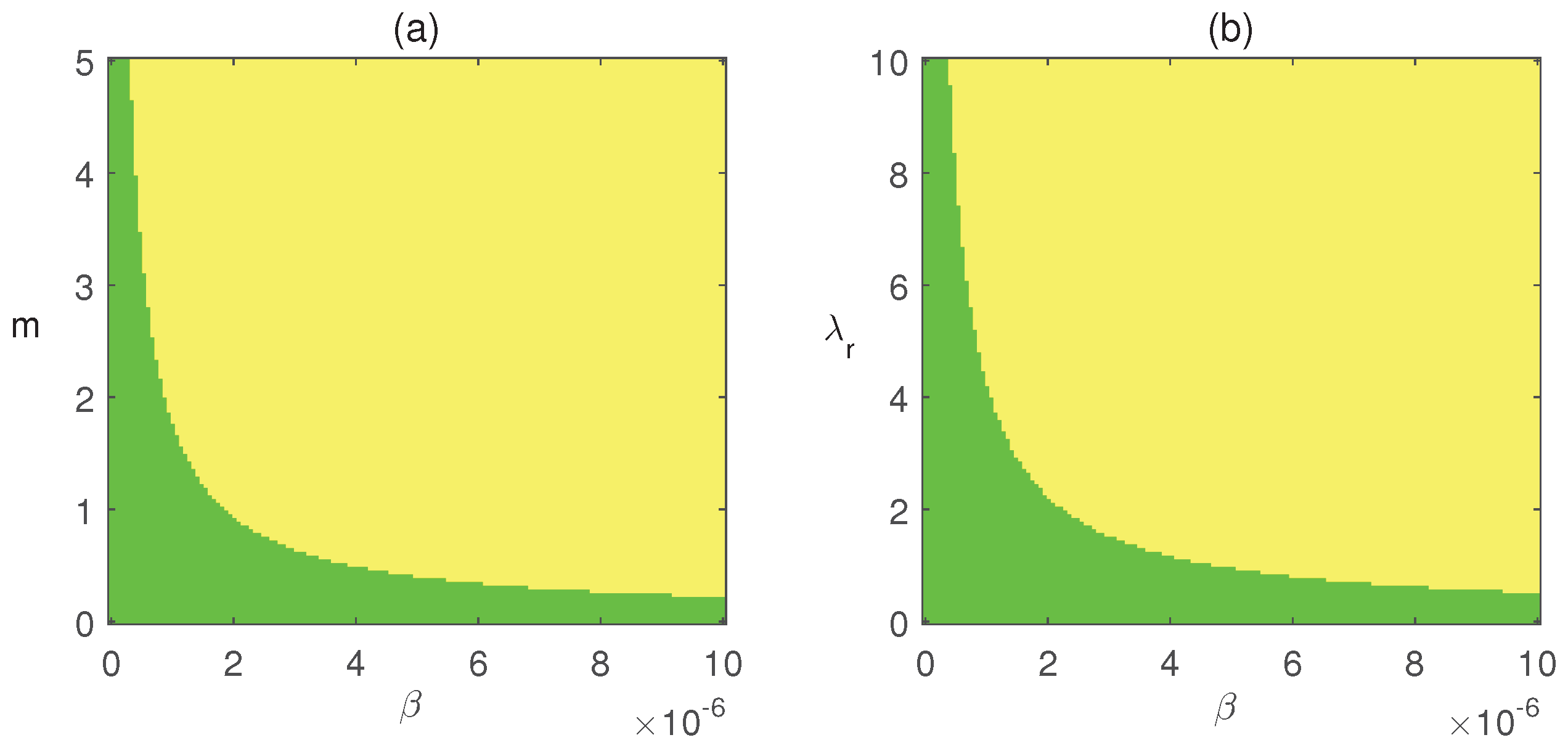
5.1.1. Impulsive Reproduction Number
5.1.2. Floquet Stability Criterion for
5.1.3. Antibody Periodic Level and Stability Analysis
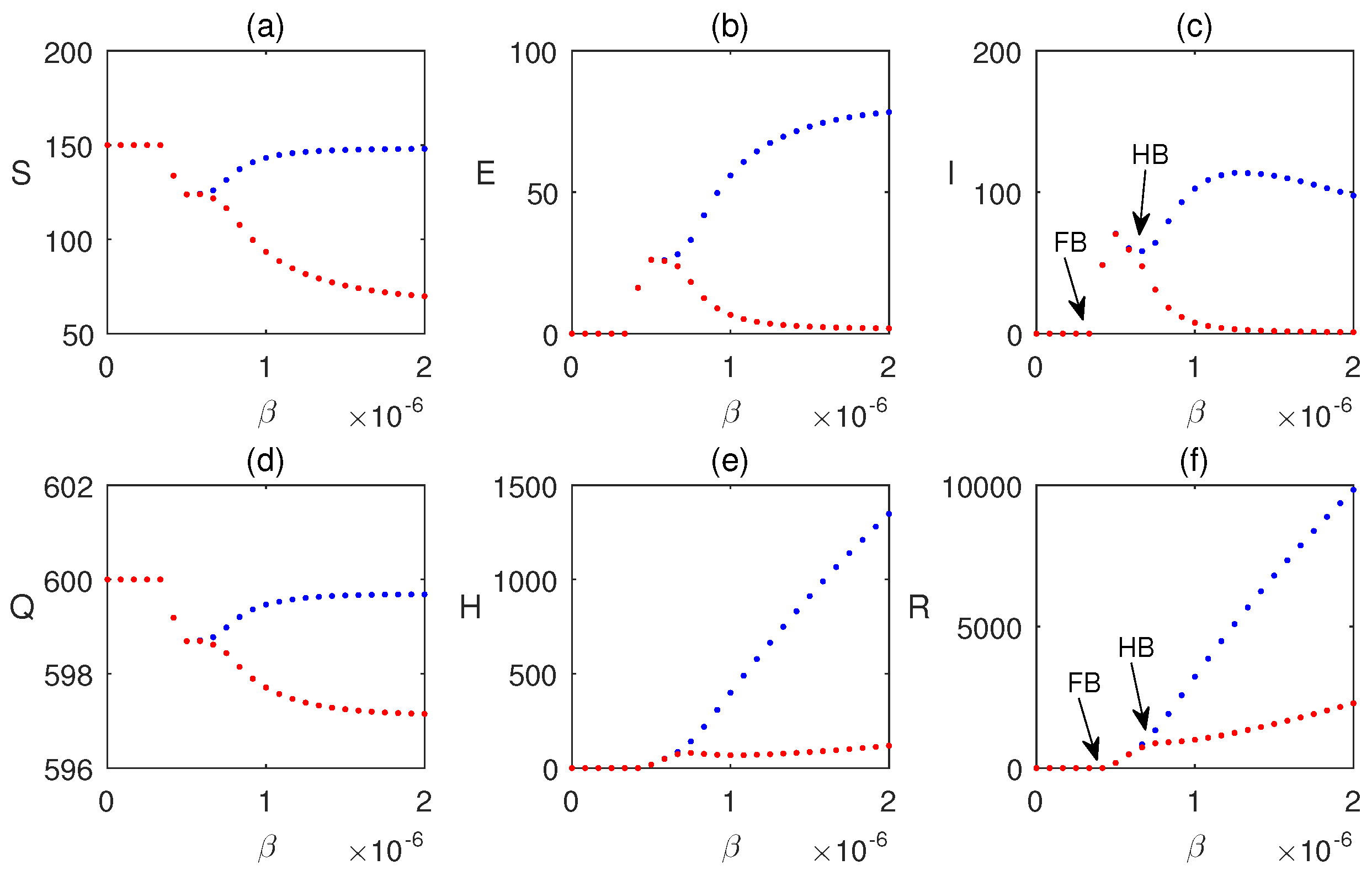
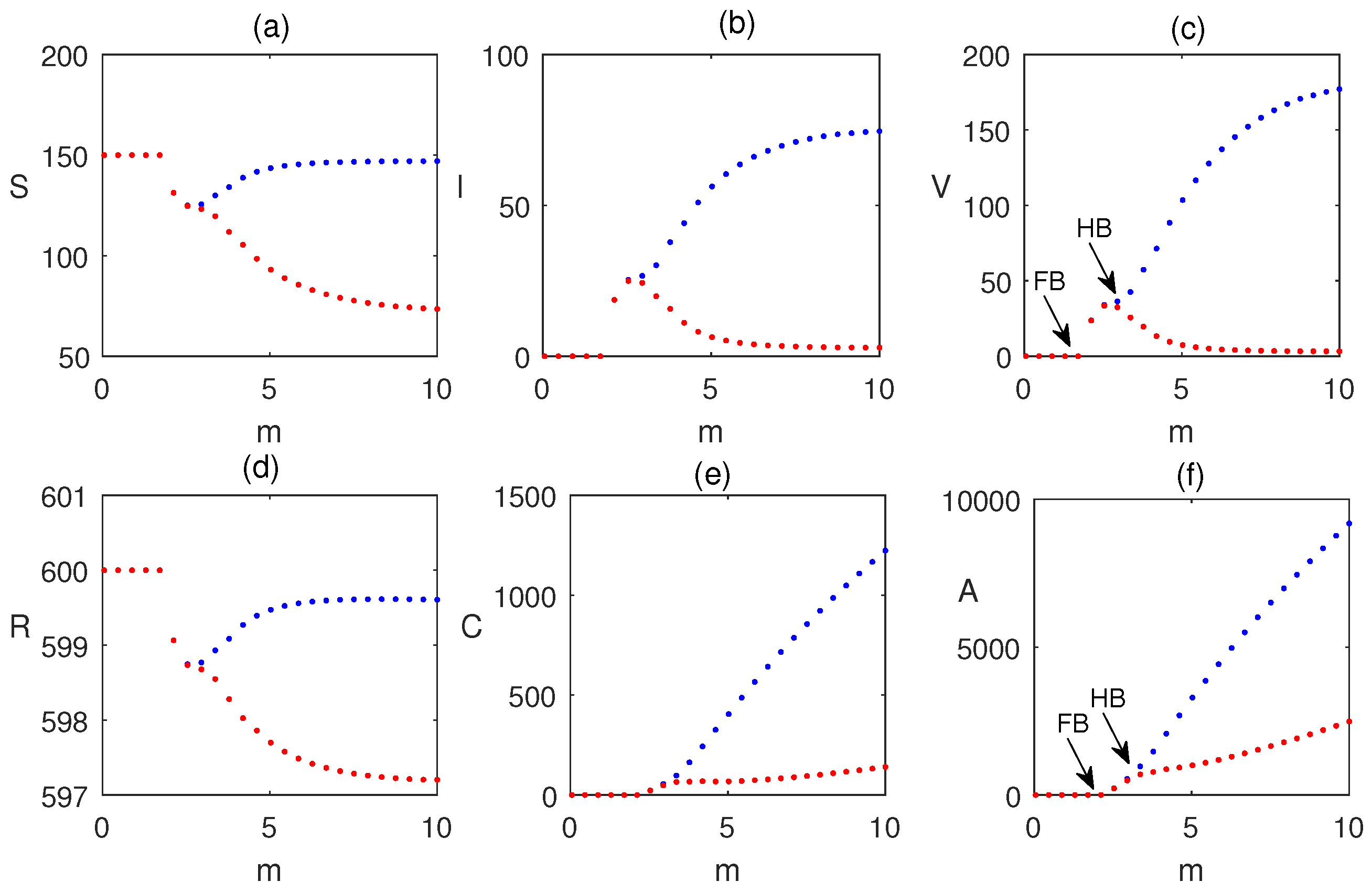
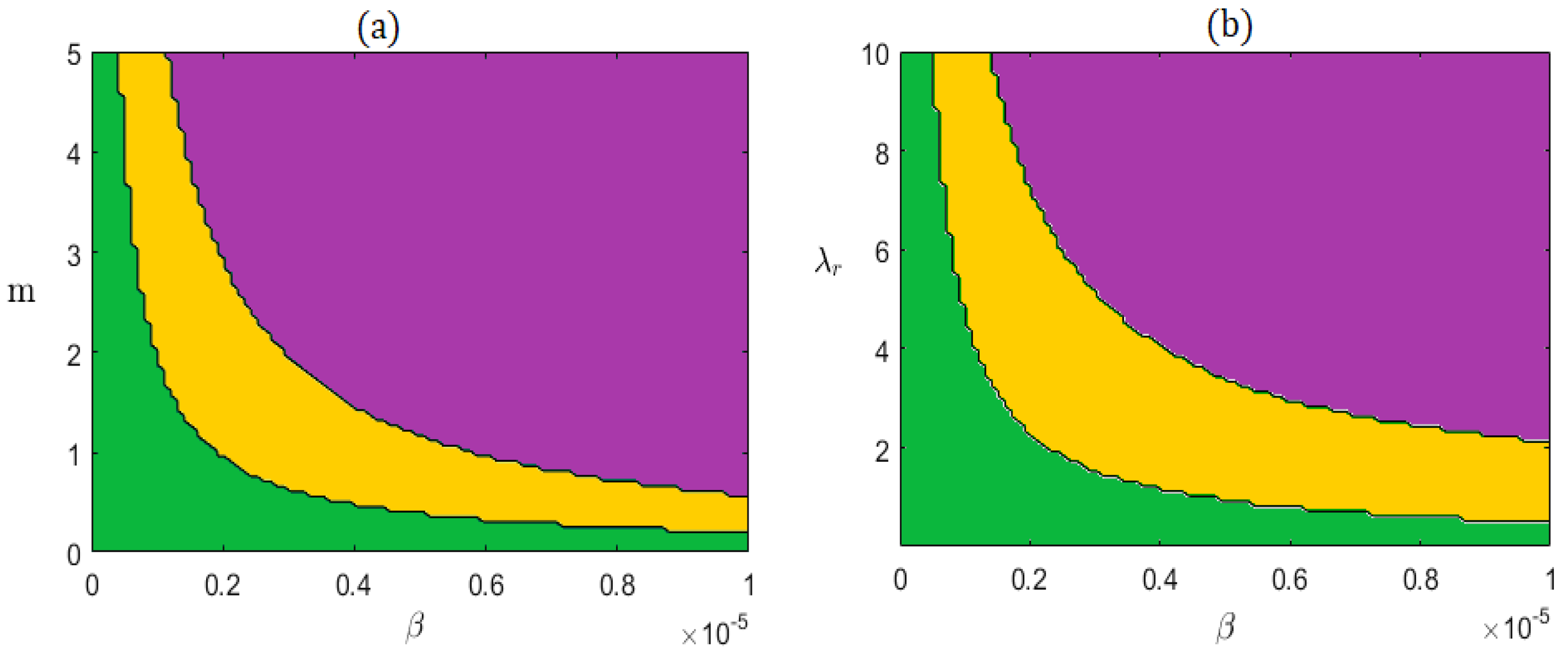
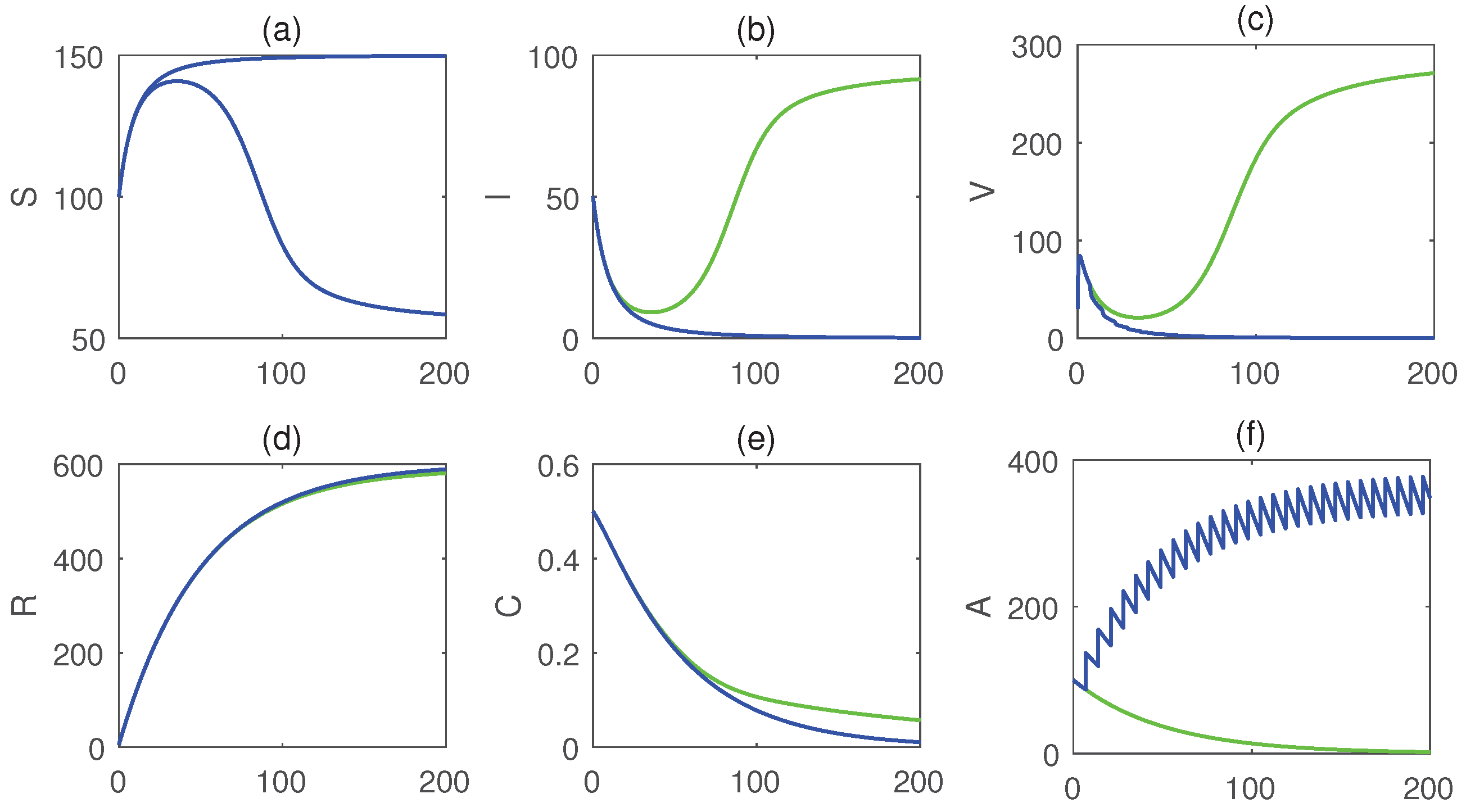
6. Numerical Simulation
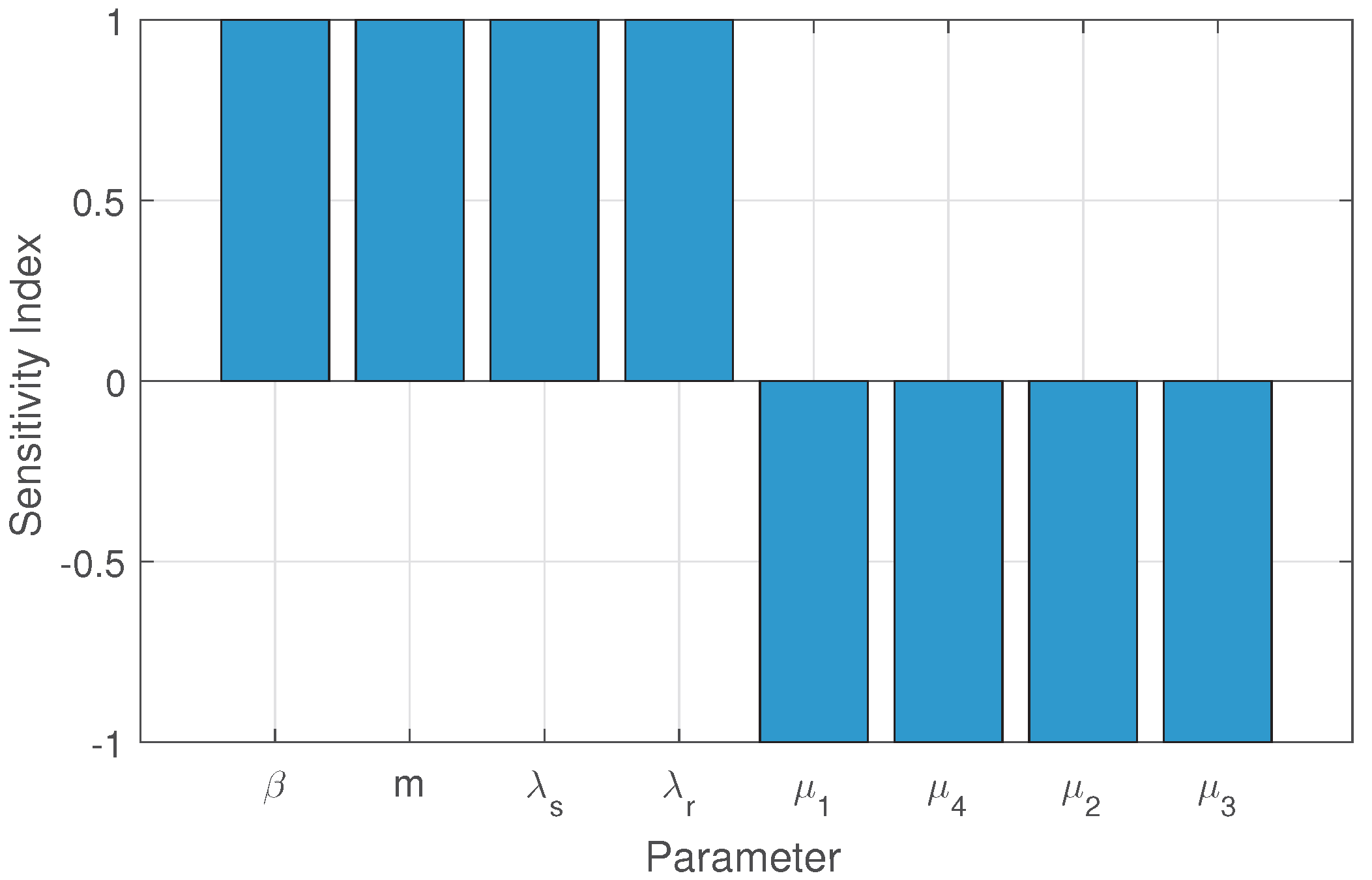
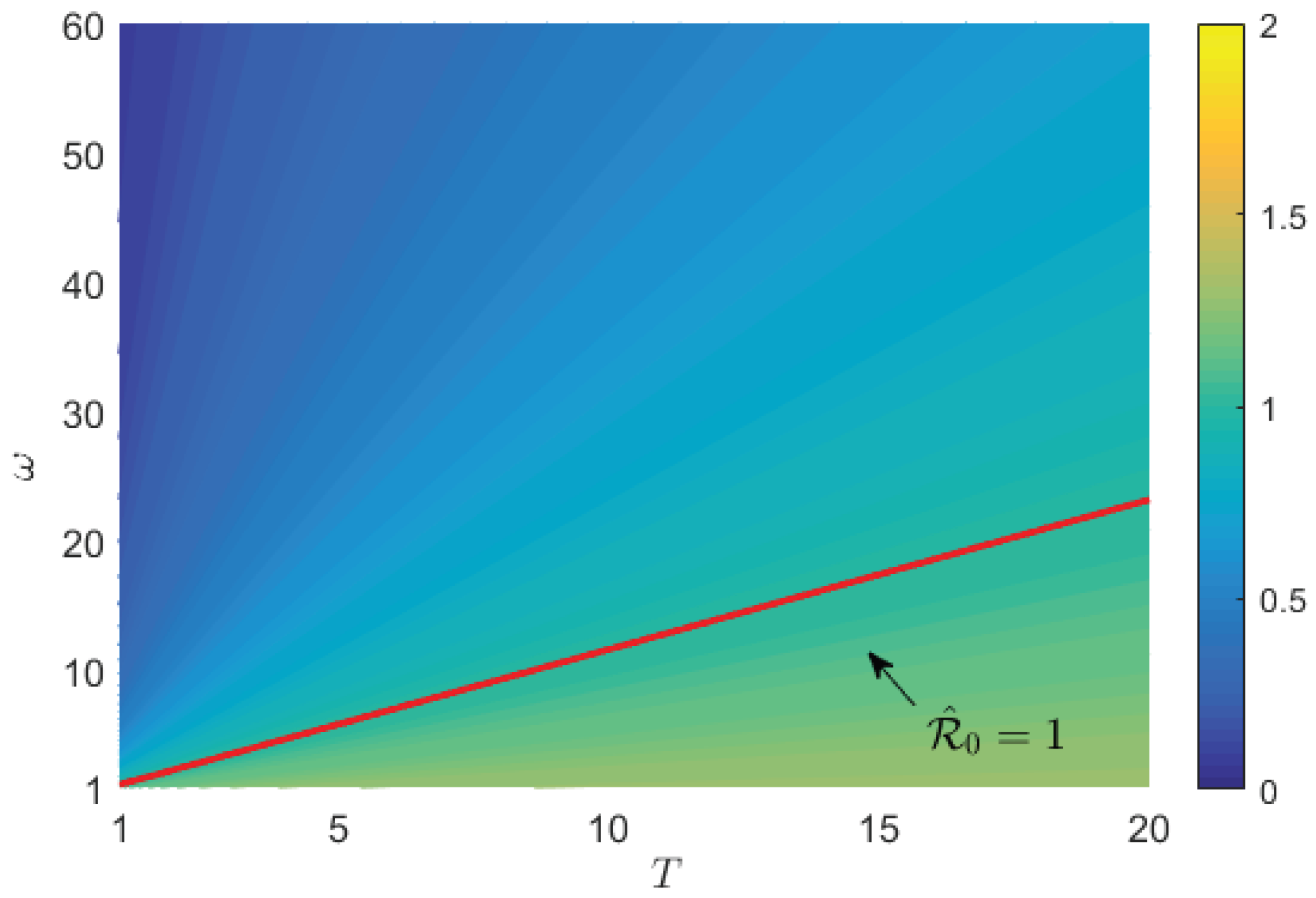
6.1. Sensitivity Analysis Using
| Variables/Parameters | Explanation | Assigned Values |
|---|---|---|
| S | Count of susceptible (uninfected target) cells | — |
| I | Population of SARS-CoV-2 infected (virus-producing) cells | — |
| V | Quantity of free SARS-CoV-2 viral particles | — |
| R | Number of ACE2 receptors present on epithelial cells | — |
| C | Population of cytotoxic T lymphocytes (CTLs) | — |
| Rate of ACE2 receptor production | 12 | |
| Infection transmission rate | ||
| Production rate of epithelial cells | 15 | |
| Natural death rate of uninfected epithelial cells | 0.1 | |
| Death rate of infected epithelial cells | 0.1 | |
| Clearance rate of viral particles | 1.67 | |
| Clearance rate of ACE2 receptors | 0.02 | |
| Elimination rate of immune cells | 0.02 | |
| p | Intracellular replication rate of virus | |
| q | Rate of virus neutralization by antibodies | |
| m | Number of virions generated per cell | |
| Antibody response rate from immune cells | 0.82 | |
| Clearance rate of antibodies | 0.02 |
6.2. Dynamics Without Impulses
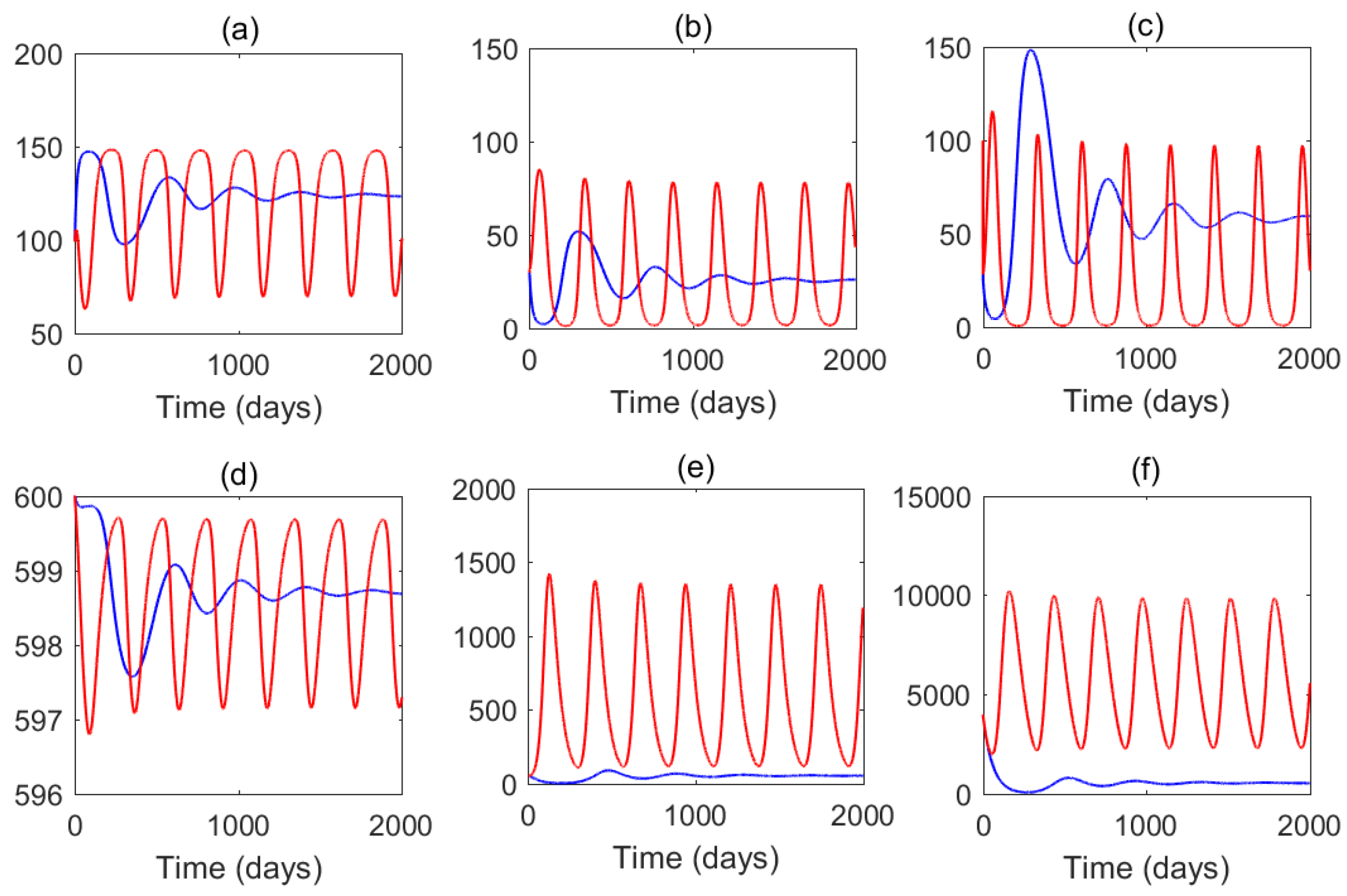
6.3. Dynamics with Impulses
7. Discussion and Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Giovanetti, M.; Branda, F.; Cella, E.; Scarpa, F.; Bazzani, L.; Ciccozzi, A.; Slavov, S.N.; Benvenuto, D.; Sanna, D.; Casu, M.; et al. Epidemic history and evolution of an emerging threat of international concern, the severe acute respiratory syndrome coronavirus 2. Journal of Medical Virology 2023, 95, e29012. [Google Scholar] [CrossRef] [PubMed]
- Jackson, C.B.; Farzan, M.; Chen, B.; Choe, H. Mechanisms of SARS-CoV-2 entry into cells. Nature reviews Molecular cell biology 2022, 23, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Sadria, M.; Layton, A.T. Modeling within-Host SARS-CoV-2 Infection Dynamics and Potential Treatments. Viruses 2021, 13, 1141. [Google Scholar] [CrossRef] [PubMed]
- Song, T.; Wang, Y.; Gu, X.; Qiao, S. Modeling the Within-Host Dynamics of SARS-CoV-2 Infection Based on Antiviral Treatment. Mathematics 2023, 11, 3485. [Google Scholar] [CrossRef]
- Lythgoe, K.A.; Hall, M.; Ferretti, L.; de Cesare, M.; et al. SARS-CoV-2 within-host diversity and transmission. Science 2021, 372, eabg0821. [Google Scholar] [CrossRef]
- Sadria, M.; Layton, A.T. Modeling within-Host SARS-CoV-2 Infection Dynamics and Potential Treatments. Viruses 2021, 13, 1141. [Google Scholar] [CrossRef]
- Gruell, H.; Vanshylla, K.; Weber, T.; Barnes, C.O.; Kreer, C.; Klein, F. Antibody-mediated neutralization of SARS-CoV-2. Immunity 2022, 55, 925–944. [Google Scholar] [CrossRef]
- Post, N.; Eddy, D.; Huntley, C.; Van Schalkwyk, M.C.; Shrotri, M.; Leeman, D.; Rigby, S.; Williams, S.V.; Bermingham, W.H.; Kellam, P.; et al. Antibody response to SARS-CoV-2 infection in humans: A systematic review. PloS one 2020, 15, e0244126. [Google Scholar] [CrossRef]
- Chatterjee, A.N.; Al Basir, F. A model for SARS-CoV-2 infection with treatment. Computational and Mathematical Methods in Medicine 2020, 2020, 1352982. [Google Scholar] [CrossRef]
- Nath, B.J.; Dehingia, K.; Mishra, V.N.; Chu, Y.M.; Sarmah, H.K. Mathematical analysis of a within-host model of SARS-CoV-2. Advances in Continuous and Discrete Models 2021, 2021. [Google Scholar] [CrossRef]
- Song, T.; Wang, Y.; Gu, X.; Qiao, S. Modeling the Within-Host Dynamics of SARS-CoV-2 Infection Based on Antiviral Treatment. Mathematics 2023, 11, 3485. [Google Scholar] [CrossRef]
- Kumar, A.; et al. Within Host Dynamics of SARS-CoV-2 in Humans: Modeling Immune Response and Cytokine Storm. SN Computer Science 2021, 2, 1–10. [Google Scholar]
- Hemmatzadeh, Z.; Roomi, V.; Gharahasanlou, T.K. Stability, Hopf bifurcation and numerical simulation of an HIV model with two modes of transmission and with cellular and humoral immunity. International Journal of Bifurcation and Chaos 2023, 33, 2350162. [Google Scholar] [CrossRef]
- Chatterjee, A.N.; Sharma, S.K.; Al Basir, F. A compartmental approach to modeling the measles disease: a fractional order optimal control model. Fractal and Fractional 2024, 8, 446. [Google Scholar] [CrossRef]
- Wang, M.; Zhao, Y.; Zhang, C.; Lou, J. THE WITHIN-HOST VIRAL KINETICS OF SARS-COV-2. Journal of Applied Analysis & Computation 2023, 13, 2121–2152. [Google Scholar] [CrossRef]
- Homburg, A.J.; et al. Bifurcation Theory; American Mathematical Society, 2024; Vol. 246. [Google Scholar]
- Al Basir, F.; Rajak, B.; Rahman, B.; Hattaf, K. Hopf bifurcation analysis and optimal control of an infectious disease with awareness campaign and treatment. Axioms 2023, 12, 608. [Google Scholar] [CrossRef]
- Kuniya, T. Hopf bifurcation in an age-structured SIR epidemic model. Applied Mathematics Letters 2019, 92, 22–28. [Google Scholar] [CrossRef]
- Wang, J.; Chen, L. Hopf bifurcation and pattern formation in a modified SIR model. Frontiers in Physics 2023, 11, 1294451. [Google Scholar]
- Kumar, A.; Singh, R. Hopf bifurcation and optimal control in infectious disease models. Axioms 2022, 12, 608. [Google Scholar]
- Co., S. Late-Breaking at CROI 2025: SCORPIO-PEP Phase 3 Trial: Ensitrelvir. 2025. Available online: https://www.shionogi.com/global/en/news/2025/03/20250313.html.
- Hayden, F.G.; Shinkai, M.; Clark, T.W.e.a. Ensitrelvir to Prevent COVID-19 in Households: SCORPIO-PEP Phase III Trial. In Proceedings of the Conference on Retroviruses and Opportunistic Infections (CROI); University of Virginia School of Medicine, 2025. [Google Scholar]
- Luetkemeyer, A.F.; Chew, K.W.; Lacey, S.e.a. Ensitrelvir for the Treatment of Nonhospitalized Adults with COVID-19: Results from the SCORPIO-HR Trial. Clinical Infectious Diseases 2025, 80, 1235–1244. [Google Scholar] [CrossRef]
- Food, U.; Administration, D. COVID-19 Treatment Guidelines: Paxlovid, Remdesivir, and Molnupiravir. 2025. Available online: https://www.fda.gov.
- Organization, W.H. Therapeutics and COVID-19: Living Guideline. 2025. Available online: https://www.who.int.
- Food, U.; Administration, D.; et al. FDA authorizes revisions to fact sheets to address SARS-CoV-2 variants for monoclonal antibody products under emergency use authorization. 2021. [Google Scholar]
- of America, I.D.S. IDSA Guidelines on the Treatment and Management of COVID-19. 2023. (accessed on 2025-11-12).
- Taylor, P.; et al. Monoclonal antibody therapies against SARS-CoV-2. The Lancet Infectious Diseases 2022, 22, e311–e319. [Google Scholar] [CrossRef]
- Deeks, E.D. Casirivimab/imdevimab: first approval. Drugs 2021, 81, 2047–2055. [Google Scholar] [CrossRef] [PubMed]
- Chavda, V.P.; Prajapati, R.; Lathigara, D.; Nagar, B.; Kukadiya, J.; Redwan, E.M.; Uversky, V.N.; Kher, M.N.; Patel, R. Therapeutic monoclonal antibodies for COVID-19 management: an update. Expert opinion on biological therapy 2022, 22, 763–780. [Google Scholar] [CrossRef] [PubMed]
- Moya, J.; Temech, M.; Parra, S.; Juarez, E.; Hernandez-Loy, R.; Gutierrez, J.C.M.; Diaz, J.; Hussain, R.; Segal, S.; Xu, C.; et al. Safety, virology, pharmacokinetics, and clinical experience of high-dose intravenous sotrovimab for the treatment of mild to moderate COVID-19: an open-label clinical trial. In Proceedings of the Open Forum Infectious Diseases; Oxford University Press US, 2023; Vol. 10, p. ofad344. [Google Scholar]
- Levin, M.J.; Ustianowski, A.; De Wit, S.; Launay, O.; Avila, M.; Templeton, A.; Yuan, Y.; Seegobin, S.; Ellery, A.; Levinson, D.J.; et al. Intramuscular AZD7442 (tixagevimab–cilgavimab) for prevention of Covid-19. New England Journal of Medicine 2022, 386, 2188–2200. [Google Scholar] [CrossRef] [PubMed]
- Du, S.; Yuan, W. Mathematical modeling of interaction between innate and adaptive immune responses in COVID-19 and implications for viral pathogenesis. Journal of Medical Virology 2020, 92, 1615–1628. [Google Scholar] [CrossRef]
- Nicholls, J.M.; Chan, M.C.W.; Chan, W.Y.; Wong, H.K.; Cheung, C.Y.; Kwong, D.L.W.; et al. Tropism of avian influenza A (H5N1) in the upper and lower respiratory tract. Nature Medicine 2007, 13, 147–149. [Google Scholar] [CrossRef]
- Zhou, Q.; Zhang, L.; Dong, Y.; Wang, Y.; Zhang, B.; Zhou, S.; et al. The role of SARS-CoV-2-mediated NF-κB activation in COVID-19 patients. Hypertension Research 2024, 47, 375–384. [Google Scholar] [CrossRef]
- van den Driessche, P.; Watmough, J. Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Mathematical Biosciences 2002, 180, 29–48. [Google Scholar] [CrossRef]
- Kumar, A.; Agrawal, A.K. Existence of Hopf-bifurcation in a 6-dimensional system. Communications in Applied Analysis 2017, 21, 119–126. [Google Scholar]
- Chatterjee, A.N.; Abraha, T.; Al Basir, F.; Torres, D.F.M. A model for the dynamics of COVID-19 infection transmission in human with latent delay. Afrika Matematika 2025, 36, 1–23. [Google Scholar] [CrossRef]
- Chatterjee, A.N.; Basir, F.A.; Biswas, D.; Abraha, T. Global dynamics of SARS-CoV-2 infection with antibody response and the impact of impulsive drug therapy. Vaccines 2022, 10, 1846. [Google Scholar] [CrossRef]
- Ghosh, I. Within host dynamics of SARS-CoV-2 in humans: Modeling immune responses and antiviral treatments. SN Computer Science 2021, 2, 482. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




