Submitted:
15 April 2026
Posted:
16 April 2026
You are already at the latest version
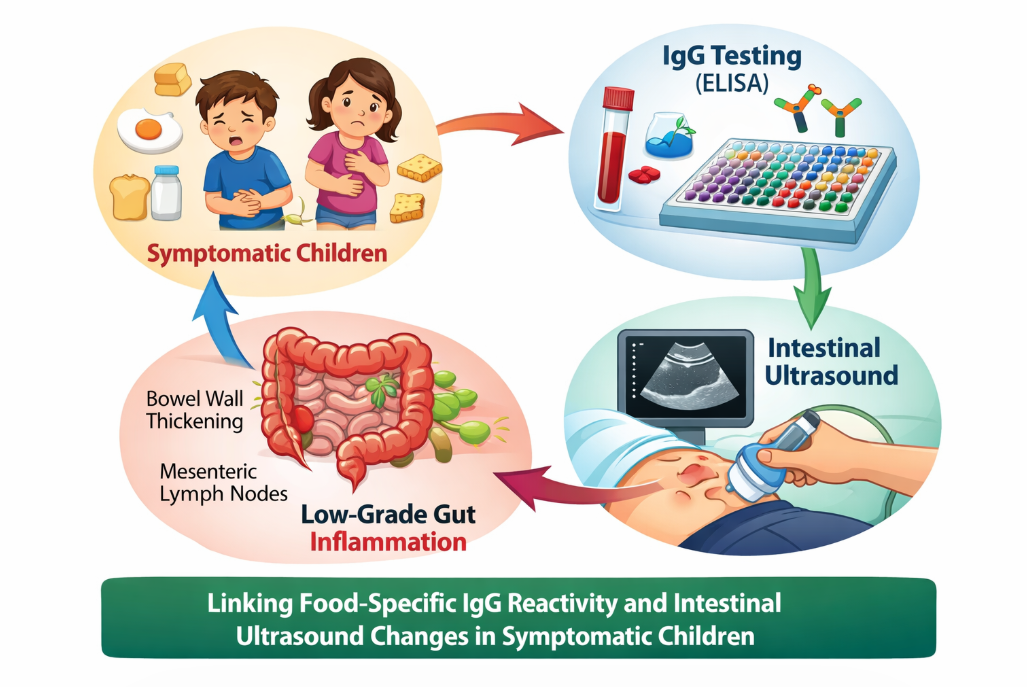
Abstract
Keywords:
1. Introduction
2. Materials and Methods
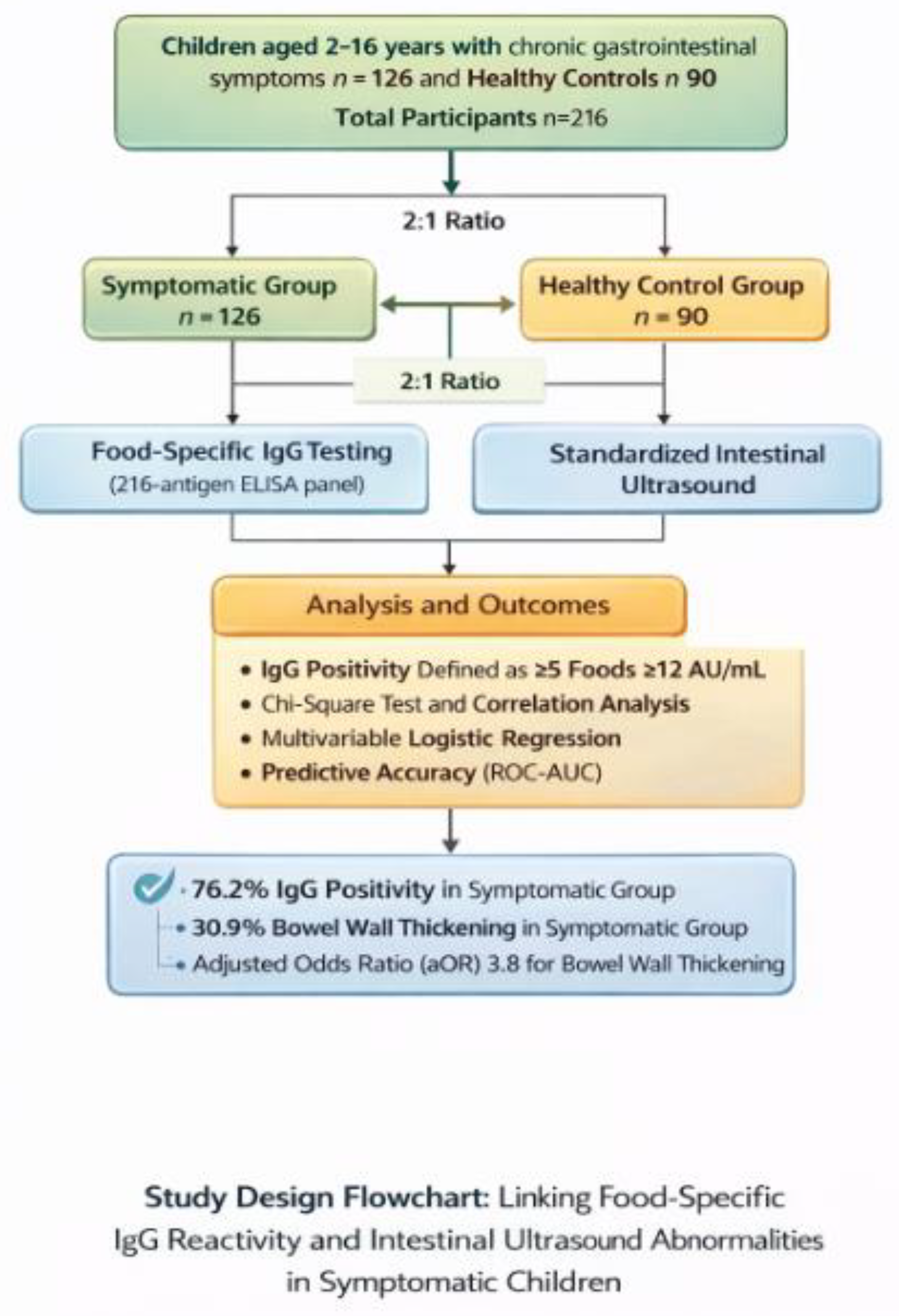
2.1. Study Design and Population
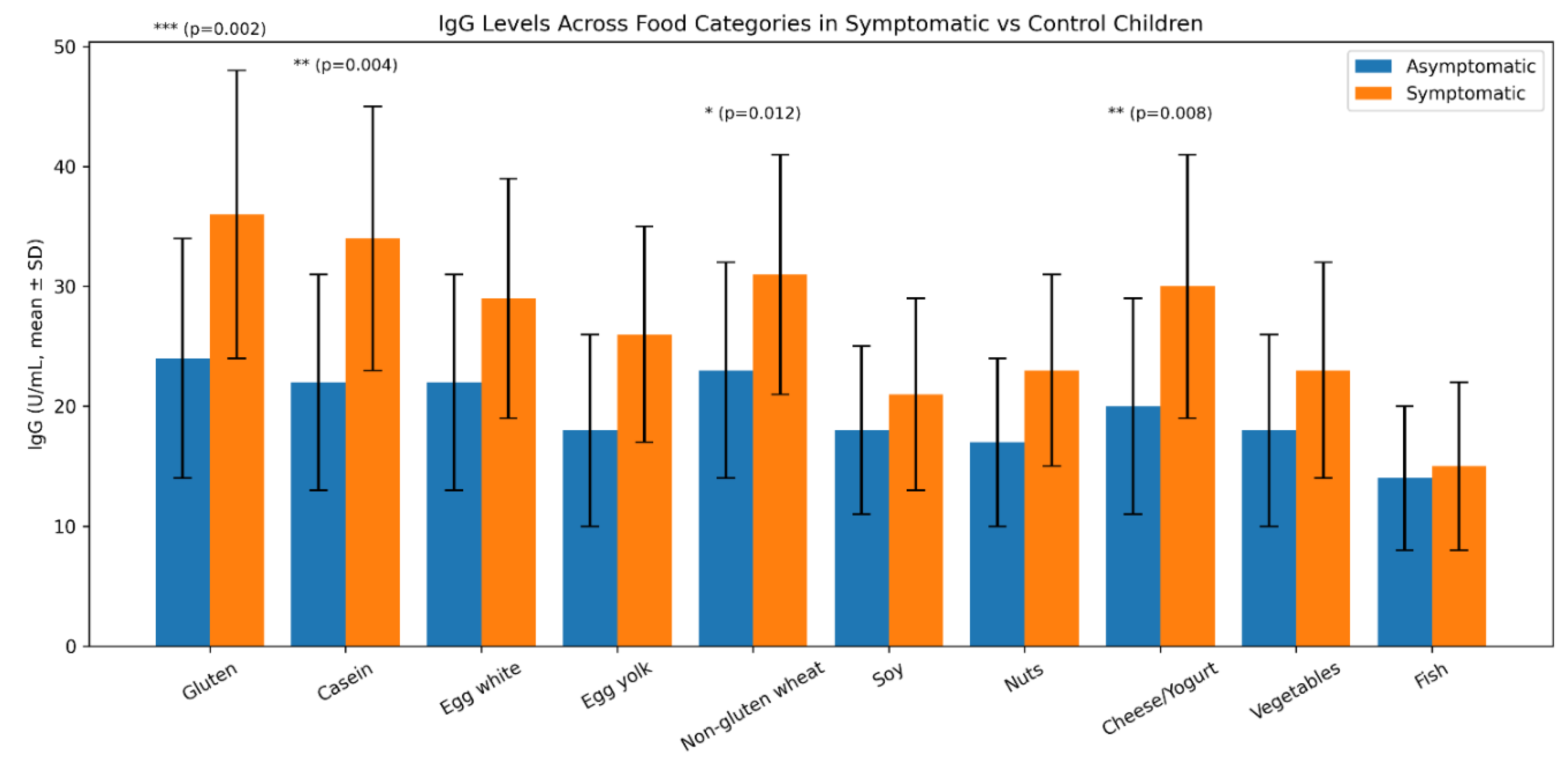
2.2. Food-Specific IgG Antibody Testing
2.3. Abdominal Ultrasound
2.4. Symptom Assessment
2.5. Assessment of Food-Specific IgG Antibodies
2.6. Statistical Analysis
3. Results
3.1. Demographic and Clinical Characteristics of the Study Population
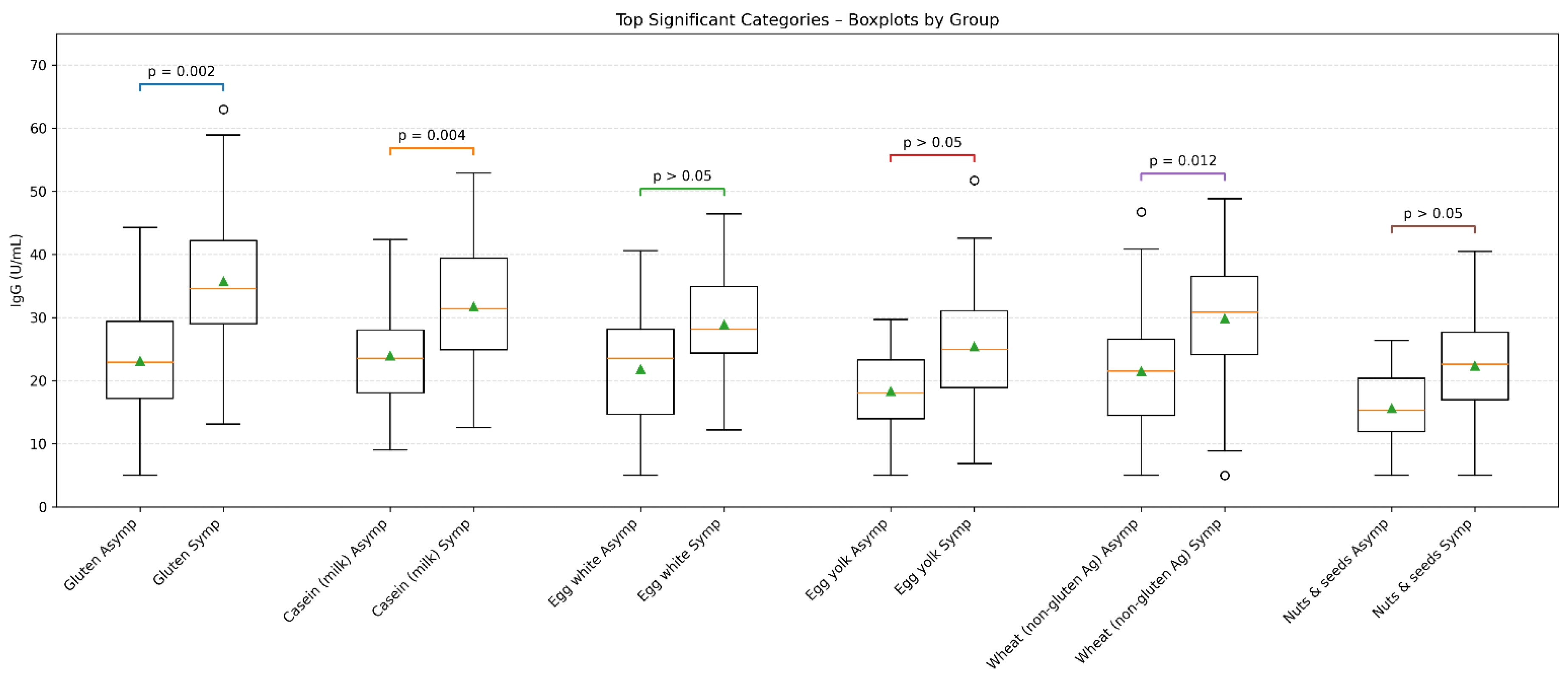
3.2.
3.3.
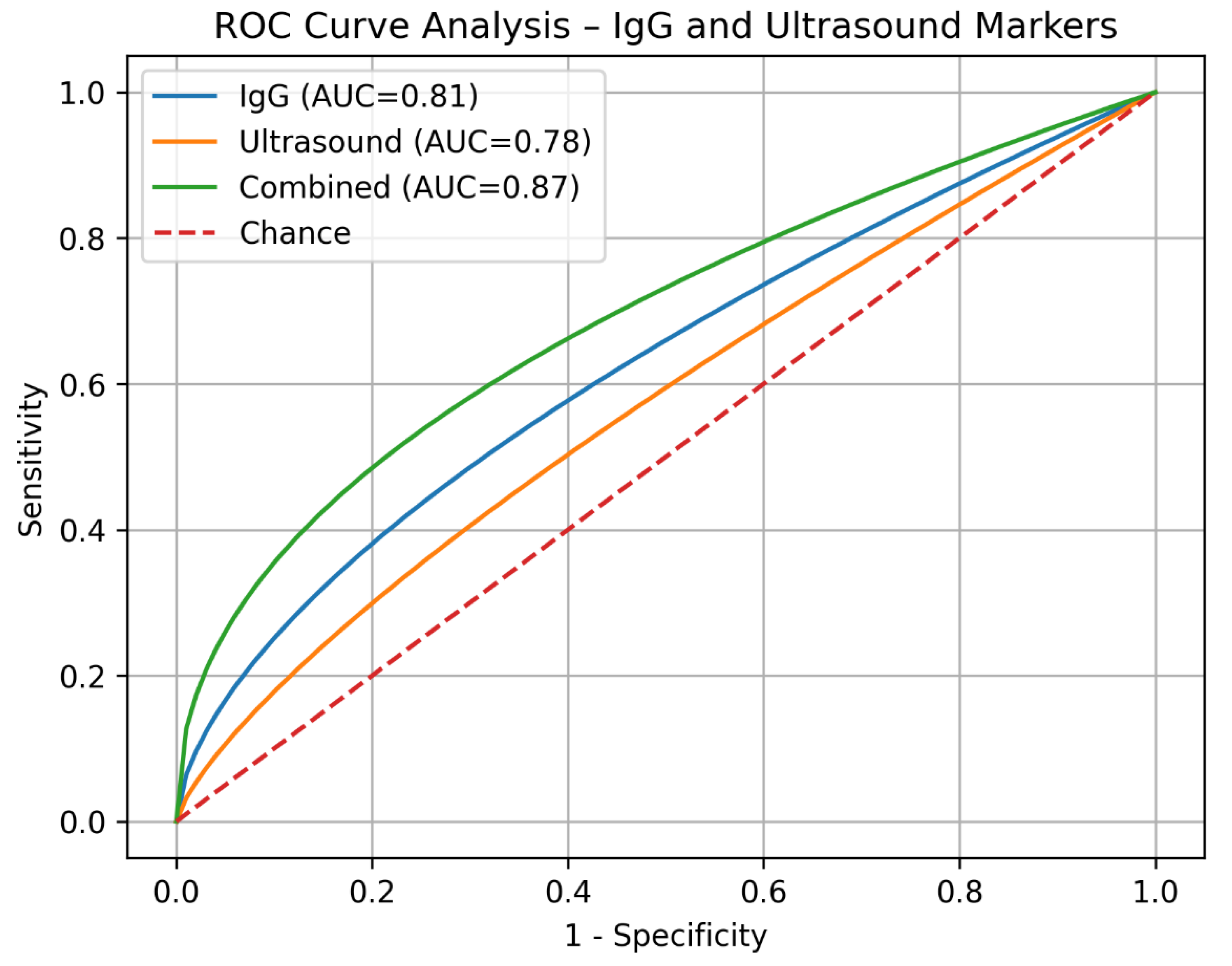
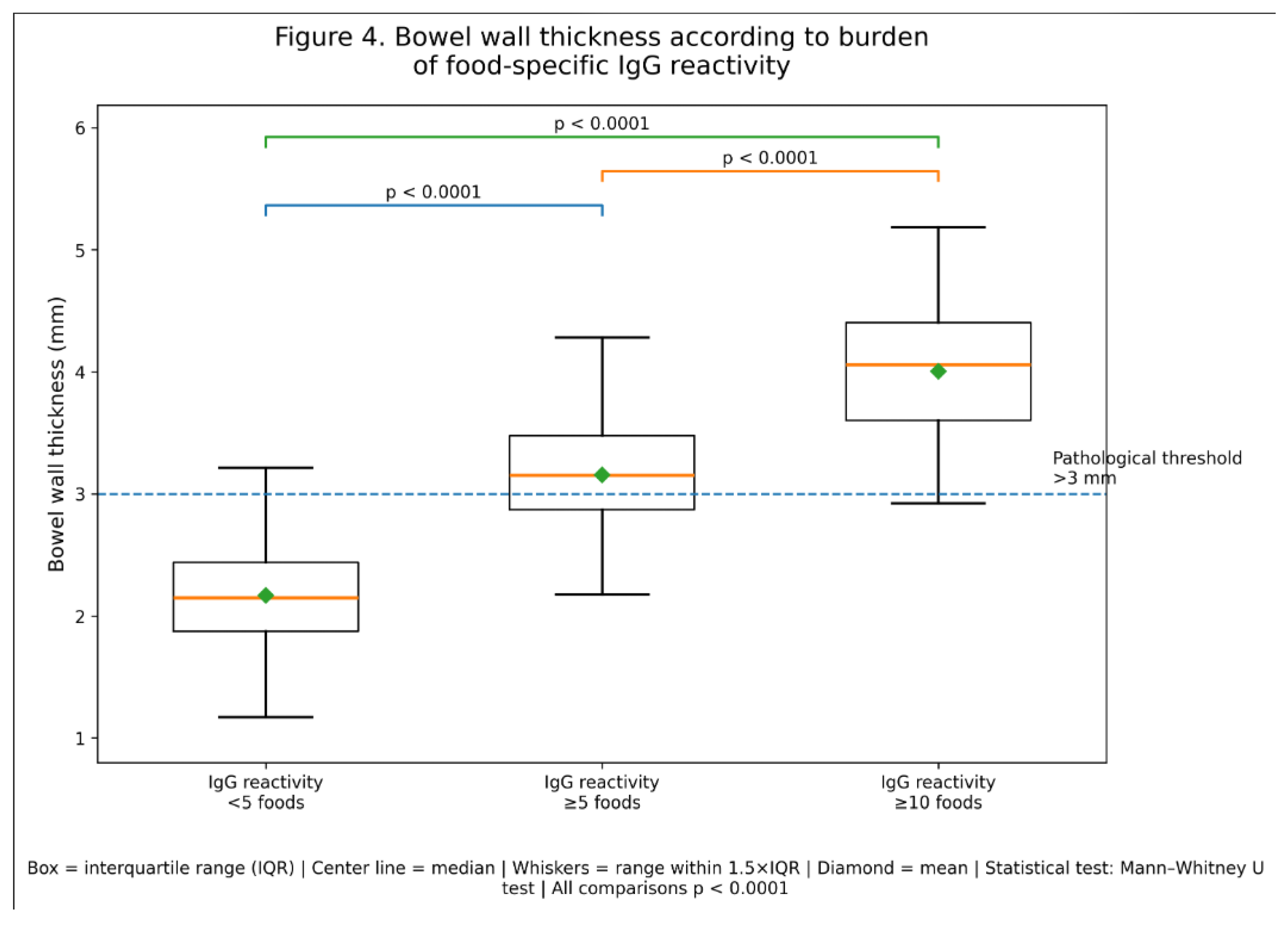
3.4. Ultrasound Findings
4. Discussion
4.1. Integrated Immunological–Imaging Perspective
4.2. Clinical Significance of Food-Specific IgG Antibodies
4.3. Ultrasound as a Marker of Subclinical Inflammation
4.4. Comparison with Recent Literature
4.5. Clinical Implications
4.6. Limitations
4.7. Future Directions
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Patt YS, Lahat A, David P, Patt C, Eyade R, Sharif K. Unraveling the Immunopathological Landscape of Celiac Disease: A Comprehensive Review. Int J Mol Sci. 2023 Oct 23;24(20):15482. PMID: 37895160; PMCID: PMC10607730. [CrossRef]
- Suurmond, J.; Diamond, B. Autoantibodies in systemic autoimmune diseases. J. Clin. Invest. 2015, 125, 2194–2202.
- Tordesillas, L.; Berin, M.C. Mechanisms of oral tolerance. Clin. Rev. Allergy Immunol. 2018, 55, 107–117.
- Leung, D.Y.M.; Sicherer, S.H.; Sampson, H.A. Food allergy: epidemiology and pathogenesis. J. Allergy Clin. Immunol. 2014, 133, 291–307.5.
- Gocki, J.; Bartuzi, Z. Role of immunoglobulin G antibodies in food allergy. Adv. Dermatol. Allergol. 2016, 33, 253–256.
- Zeng, Q.; Dong, S.Y.; Wu, L.X.; Li, H.; Sun, Z.J.; Li, J.B. Variable food-specific IgG levels in adults. PLoS ONE 2013, 8, e53612.
- Xiao, N.; Liu, F.; Zhou, G.; Sun, M.; Ai, F.; Liu, Z. Food-specific IgG in inflammatory bowel disease. Intern. Med. 2018, 57, 2787–2798.
- Santos, A.F.; Riggioni, C.; Agache, I.; Akdis, C.A.; Akdis, M.; Alvarez-Perea, A.; Alvaro-Lozano, M.; Ballmer-Weber, B.; Barni, S.; Beyer, K.; et al. EAACI guidelines on the diagnosis of IgE-mediated food allergy. Allergy 2023, 78, 3057–3076. [Google Scholar] [CrossRef].
- McGowan, E.C.; Medernach, J.; Keshavarz, B.; Workman, L.J.; Li, R.C.; Barnes, B.H.; Sauer, B.; Wilson, J.M.; Platts-Mills, T.A.E. Food antigen consumption and disease activity affect food-specific IgG4 levels in patients with eosinophilic esophagitis (EoE). Clin. Exp. Allergy 2023, 53, 307–315. [Google Scholar] [CrossRef].
- Morales W, Rezaie A, Barlow G, Pimentel M. Second-generation biomarker testing for irritable bowel syndrome using plasma anti-CdtB and anti-vinculin levels. Dig Dis Sci. (2019) 64:3115–21. [CrossRef]
- Vojdani A, Vojdani E, Kharrazian D. Fluctuation of zonulin levels in blood vs stability of antibodies. World J Gastroenterol. (2017) 23:5669–79. [CrossRef]
- Vojdani A, Vojdani E, Herbert M, Kharrazian D. Correlation between antibodies to bacterial lipopolysaccharides and barrier proteins in sera positive for asca and anca. Int J Mol Sci. (2020) 21:1381. [CrossRef]
- Bays JL, DeMali KA. Vinculin in cell-cell and cell-matrix adhesions. Cell Mol Life Sci. (2017) 74:2999–3009. [CrossRef]
- Korterink, J.J.; Diederen, K.; Benninga, M.A. Epidemiology of pediatric FGIDs. J. Pediatr. Gastroenterol. Nutr. 2015, 60, 1–7.
- Robin, S.G.; Keller, C.; Zwiener, R. Prevalence of pediatric FGIDs. Gastroenterology 2018, 154, 1081–1089.
- Hyams, J.S.; Di Lorenzo, C.; Saps, M. Functional disorders in children (Rome IV). Gastroenterology 2016, 150, 1456–1468.17.
- Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)-A metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. (2009) 42:377–81. [CrossRef]
- Harris PA, Taylor R, Minor BL, Elliott V, Fernandez M, O’Neal L, et al. The RED Cap consortium: building an international community of software platform partners. J Biomed Inform. (2019) 95:103208. [CrossRef]
- Peruhova M, Mihova A, Altankova I, Velikova T. Specific immunoglobulin E and G to common food antigens and increased serum zonulin in IBS patients: a single-Center Bulgarian study. Antibodies. (2022) 11:23. [CrossRef]
- Cappelletti M, Tognon E, Vona L, Basello K, Costanzi A, Speciani MC, et al. Food-specific serum IgG and symptom reduction with a personalized, unrestricted- calorie diet of six weeks in irritable bowel syndrome (IBS). Nutr Metab. (2020) 17:101. [CrossRef]
- Xiao N, Liu F, Zhou G, Sun M, Ai F, Liu Z. Food-specific igGs are highly increased in the sera of patients with inflammatory bowel disease and are clinically relevant to the pathogenesis. Intern Med. (2018) 57:2787–98. [CrossRef]
- Xiao YT, Yan WH, Cao Y, Yan JK, Cai W. Neutralization of IL-6 and TNF-α ameliorates intestinal permeability in DSS-induced colitis. Cytokine. (2016) 83:189–92. [CrossRef]
- Schoultz, I.; Keita, Å.V. Intestinal barrier and permeability. Cells 2020, 9, 1909.
- König, J.; Wells, J.; Cani, P.D. Intestinal barrier in health and disease. Clin. Transl. Gastroenterol. 2016, 7, e196.
- Seethaler, B.; Basrai, M.; Neyrinck, A.M. Biomarkers of intestinal permeability. Am. J. Physiol. 2021, 321, G11–G17.
- Fukao, S.; Haniuda, K.; Tamaki, H. IgG response and microbiota interaction. eLife 2021, 10, e72116.
- Mohammad, S.; Thiemermann, C. Metabolic endotoxemia and inflammation. Front. Immunol. 2021, 11, 594150.
- Pimentel, M.; Lembo, A. Microbiome in IBS. Dig. Dis. Sci. 2020, 65, 829–839.26. Fasano A, Not T, Wang W, Uzzau S, Berti I, Tommasini A, et al. Zonulin, a newly discovered modulator of intestinal permeability, and its expression in coeliac disease. Lancet. (2000) 355:1518–9. [CrossRef]
- 27. Sturgeon C, Fasano A. Zonulin, a regulator of epithelial and endothelial barrier functions, and its involvement in chronic inflammatory diseases. Tissue Barriers. (2016) 4:e1251384. [CrossRef]
- Atkinson W, Sheldon TA, Shaath N, Whorwell PJ. Food elimination based on IgG antibodies in irritable bowel syndrome: a randomised controlled trial. Gut. 2004;53(10):1459–1464.
- Zar S, Mincher L, Benson MJ, Kumar D. Food-specific IgG4 antibody-guided exclusion diet improves symptoms in irritable bowel syndrome: a randomized controlled trial. Scand J Gastroenterol. 2005;40(7):800–807.
- Alpay K, Ertaş M, Orhan EK, et al. Diet restriction in migraine, based on IgG against foods: a clinical double-blind, randomised, cross-over trial. Cephalalgia. 2010;30(7):829–837.
- Fasano A. Zonulin and its regulation of intestinal barrier function: the biological door to inflammation. Physiol Rev. 2011;91(1):151–175.
- Camilleri M. Leaky gut: mechanisms, measurement and clinical implications in humans. Gut. 2019;68(8):1516–1526.
- Vighi G, Marcucci F, Sensi L, et al. Allergy and the gastrointestinal system. Clin Exp Immunol. 2008;153(S1):3–6.
- Sethi S, Wadhwa V, LeClair J, et al. IgG-based elimination diets in IBS: a systematic review. Clin Gastroenterol Hepatol. 2015;13(12):2044–2052.
- Ott B, Skurk T, Lagkouvardos L, Fischer S, Büttner J, Lichtenegger M, et al. Short-term overfeeding with dairy cream does not modify gut permeability, the fecal microbiota, or glucose metabolism in young healthy men. J Nutr. (2018) 148:77–85. [CrossRef]
- Janeway CA, Travers P, Walport M, Shlomchik M. Autoimmune Responses are Directed Against Self Antigens.,” Immunobiology: The Immune System in Health and Disease. 5th ed. New York, NY: Garland Science (2001). p. 892.
- Cole CR, Frem JC, Schmotzer B, Gewirtz AT, Meddings JB, Gold BD, et al. The rate of bloodstream infection is high in infants with short bowel syndrome: relationship with small bowel bacterial overgrowth, enteral feeding, and inflammatory and immune responses. J Pediatr. (2010) 156: 941–47.e1. [CrossRef]
- Yang, B.; Yu, H.; Yao, W.; Diao, R.; Li, B.; Wang, Y.; Li, T.; Ge, L.; Hu, Y.; Wang, H. Food-specific IgG4-guided diet elimination improves allergy symptoms in children. Front. Immunol. 2024, 15, 1281741. [Google Scholar] [CrossRef].
- American Asociation of Allergy, Astham and Immunoplogy. Available online: https://www.aaaai.org/tools-for-the-public/conditions-library/allergies/igg-food-test (accessed on 23 February 2026).
- McDonald CM, Manji KP, Gosselin K, Tran H, Liu E, Kisenge R, et al. Elevations in serum anti-flagellin and anti-LPS Igs are related to growth faltering in young Tanzanian children. Am J Clin Nutr. (2016) 103:1548–54. [CrossRef]
- Fukao S, Haniuda K, Tamaki H, Kitamura D. Protein kinase cδ is essential for the igg response against T cell-independent type 2 antigens and commensal bacteria. Elife. (2021) 10:e72116. [CrossRef]
- Madhavan R, Porkodi R, Panchapakesa Rajendran C, Chandrasekaran AN, Umadevi KR, Alamelu R. IgM, IgG, and IgA response to enterobacteria in patients with ankylosing spondylitis in southern India. Ann N Y Acad Sci. (2002) 958:408– 11. [CrossRef]
- Chen MX, Chen Y, Fu R, Mao GQ, Liu SY, Shen TB. Rab5a promotes cytolethal distending toxin b-induced cytotoxicity and inflammation. Infect Immun. (2020) 88:e132–120. [CrossRef]
- Mohammad S, Thiemermann C. Role of metabolic endotoxemia in systemic inflammation and potential interventions. Front Immunol. (2021) 11:594150. [CrossRef]
- Pokkunuri V, Pimentel M, Morales W, Jee SR, Alpern J, Weitsman S, et al. Role of cytolethal distending toxin in altered stool form and bowel phenotypes in a rat model of post-infectious irritable bowel syndrome. J Neurogastroenterol Motil. (2012) 18:434–42. [CrossRef]
- Pimentel M, Lembo A. Microbiome and its role in irritable bowel syndrome. Dig Dis Sci. (2020) 65:829–39. [CrossRef]
- Rezaie A, Park SC, Morales W, Marsh E, Lembo A, Kim JH, et al. Assessment of anti-vinculin and anti-cytolethal distending toxin B antibodies in subtypes of irritable bowel syndrome. Dig Dis Sci. (2017) 62:1480–5. [CrossRef]
- Pons BJ, Vignard J, Mirey G. Cytolethal distending toxin subunit B: a review of structure-function relationship. Toxins (Basel). (2019) 11:595. [CrossRef]
- Varon C, Mocan I, Mihi B, Péré-Védrenne C, Aboubacar A, Moraté C, et al. Helicobacter pullorum cytolethal distending toxin targets vinculin and cortactin and triggers formation of lamellipodia in intestinal epithelial cells. J Infect Dis. (2014) 209:588–99. [CrossRef]
- Groetch, M.; Venter, C.; Meyer, R. Clinical Presentation and Nutrition Management of Non-IgE-Mediated Food Allergy in Children. Clin. Exp. Allergy 2025, 55, 213–225. [Google Scholar] [CrossRef].
- Guo H, Jiang T, Wang J, Chang Y, Guo H, Zhang WH. The value of eliminating foods according to food-specific immunoglobulin G antibodies in irritable bowel syndrome with diarrhoea. J Int Med Res. (2012) 40:204–10. [CrossRef]
- Ostrowska L, Wasiluk D, Lieners CFJ, Gał ̨ecka M, Bartnicka A, Tveiten D. Igg food antibody guided elimination-rotation diet was more effective than FODMAP diet and control diet in the treatment of women with mixed ibs– results from an open label study. J Clin Med. (2021) 10:4317. [CrossRef]
- König J, Wells J, Cani PD, García-Ródenas CL, MacDonald T, Mercenier A, et al. Human intestinal barrier function in health and disease. Clin Transl Gastroenterol. (2016) 7:e196. [CrossRef]
- Hoshiko H, Feskens EJM, Oosterink E, Ariens RMC, Mes JJ, De Wit NJW. Identification of leaky gut-related markers as indicators of metabolic health in dutch adults: the nutrition questionnaires plus (NQplus) study. PLoS One. (2021) 16:e0252936. [CrossRef]
- Seethaler B, Basrai M, Neyrinck AM, Nazare JA, Walter J, Delzenne NM, et al. Biomarkers for assessment of intestinal permeability in clinical practice. Am J Physiol Gastrointest Liver Physiol. (2021) 321:G11–7. [CrossRef]
- . Pei R, Dimarco DM, Putt KK, Martin DA, Gu Q, Chitchumroonchokchai C, et al. Low-fat yogurt consumption reduces biomarkers of chronic inflammation and inhibits markers of endotoxin exposure in healthy premenopausal women: a randomised controlled trial. Br J Nutr. (2017) 118:1043–51. [CrossRef]
- Garmendia, J.V.; De Sanctis, J.B.; García, A.H. Food-Specific IgG Antibodies: Decoding Their Dual Role in Immune Tolerance and Food Intolerance. Immuno 2025, 5, 25. [CrossRef]
- Burk, C.M.; Shreffler, W.G. Triggers for eosinophilic esophagitis (EoE): The intersection of food allergy and EoE. J. Allergy Clin. Immunol. 2024, 153, 1500–1509. [Google Scholar] [CrossRef] [PubMed].
- Zhang Y, Li X, Wang Z, et al.Gut microbiota and intestinal barrier function in pediatric gastrointestinal disorders.Nutrients. 2024;16(5):789.
| Variable | Symptomatic (n=126) | Controls (n=90) | p-value | OR (95% CI) |
|---|---|---|---|---|
| Abdominal pain (%) | 84.6% | 52.1% | 0.003 | 4.9 (2.0–11.8) |
| Altered bowel habits (%) | 61.5% | 34.3% | 0.020 | 3.0 (1.4–6.4) |
| Bloating (%) | 58.7% | 29.8% | <0.001 | 3.4 (1.8–6.6) |
| Nausea/Vomiting (%) | 32.5% | 14.4% | 0.004 | 2.9 (1.4–6.0) |
| Dermatological symptoms (%) | 28.5% | 12.2% | 0.006 | 2.8 (1.3–6.1) |
| Fatigue (%) | 36.5% | 18.9% | 0.009 | 2.4 (1.2–4.8) |
| IgG ≥5 foods (%) | 76.2% | 28.9% | <0.0001 | 7.9 (4.1–15.2) |
| IgG ≥10 foods (%) | 42.0% | 9.8% | <0.0001 | 6.7 (3.0–14.8) |
| Variable | IgG Positive (n=142) | IgG Negative (n=74) | p-value | OR (95% CI) |
|---|---|---|---|---|
| Bowel wall thickening (%) | 34% | 11% | <0.0001 | 4.2 (2.0–8.8) |
| Terminal ileum involvement (%) | 22% | 6% | 0.001 | 4.3 (1.7–10.7) |
| Right colon involvement (%) | 18% | 5% | 0.002 | 4.1 (1.5–11.2) |
| Mesenteric lymph nodes (%) | 15% | 10% | 0.280 | NS |
| Abdominal pain (%) | 84.6% | 52.1% | 0.003 | 4.9 (2.0–11.8) |
| Altered bowel habits (%) | 61.5% | 34.3% | 0.020 | 3.0 (1.4–6.4) |
| Variables Compared | Correlation (r) | p-value | Interpretation |
| IgG positivity vs bowel wall thickening | 0.41 | <0.01 | Moderate positive correlation |
| IgG positivity vs abdominal pain | 0.38 | <0.01 | Moderate correlation |
| IgG positivity vs altered bowel habits | 0.29 | 0.02 | Weak–moderate correlation |
| Bowel thickening vs abdominal pain | 0.45 | <0.001 | Strongest association |
| Bowel thickening vs altered bowel habits | 0.33 | 0.01 | Moderate correlation |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




