Submitted:
16 April 2026
Posted:
16 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and methods
2.1. Materials
2.2. Methods
2.2.1. Co-amorphous system formation screening and solvent selection
2.2.2. Co-amorphous Drug Delivery System Preparation
2.2.3. Scanning Electron Microscopy
2.2.4. Powder X-Ray Diffraction
2.2.5. Differential Scanning Calorimetry-Thermogravimetric Analysis
2.2.6. Modulated Differential Scanning Calorimetry-MDSC Analysis
2.2.7. Fourier-Transform Infrared Spectroscopy
2.2.8. Dissolution Study
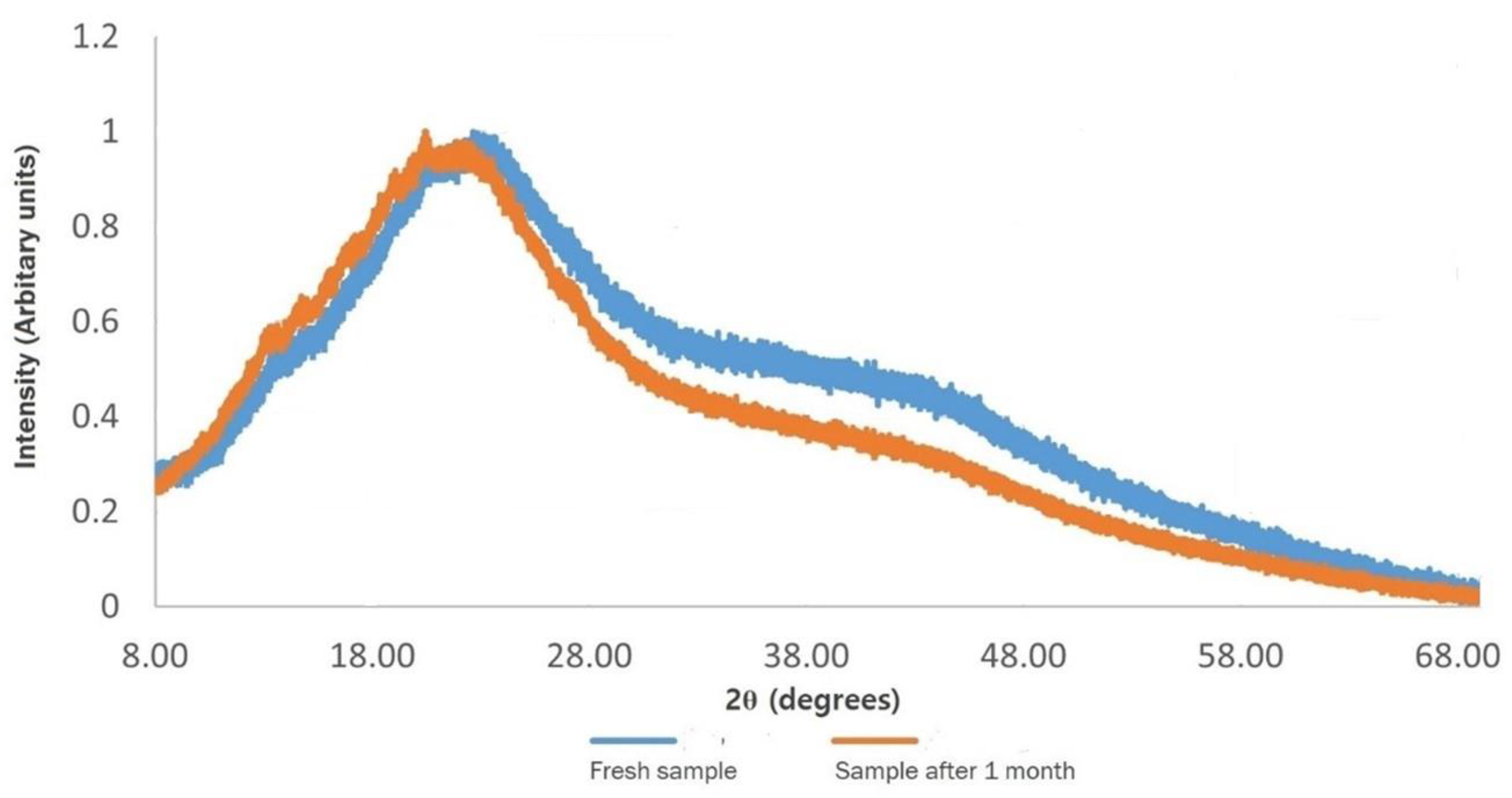
2.2.9. Accelerated Stability Study
2.2.10. Statistical Analysis
3. Results and Discussion
3.1. Morphology and Solid-State Characterization
3.1.1. Morphology by Scanning Electron Microscopy
3.1.2. Powder X-Ray Diffraction Patterns
3.1.3. Differential Scanning Calorimetry-Thermogravimetric Analysis Results
3.1.4. FTIR Spectra Results
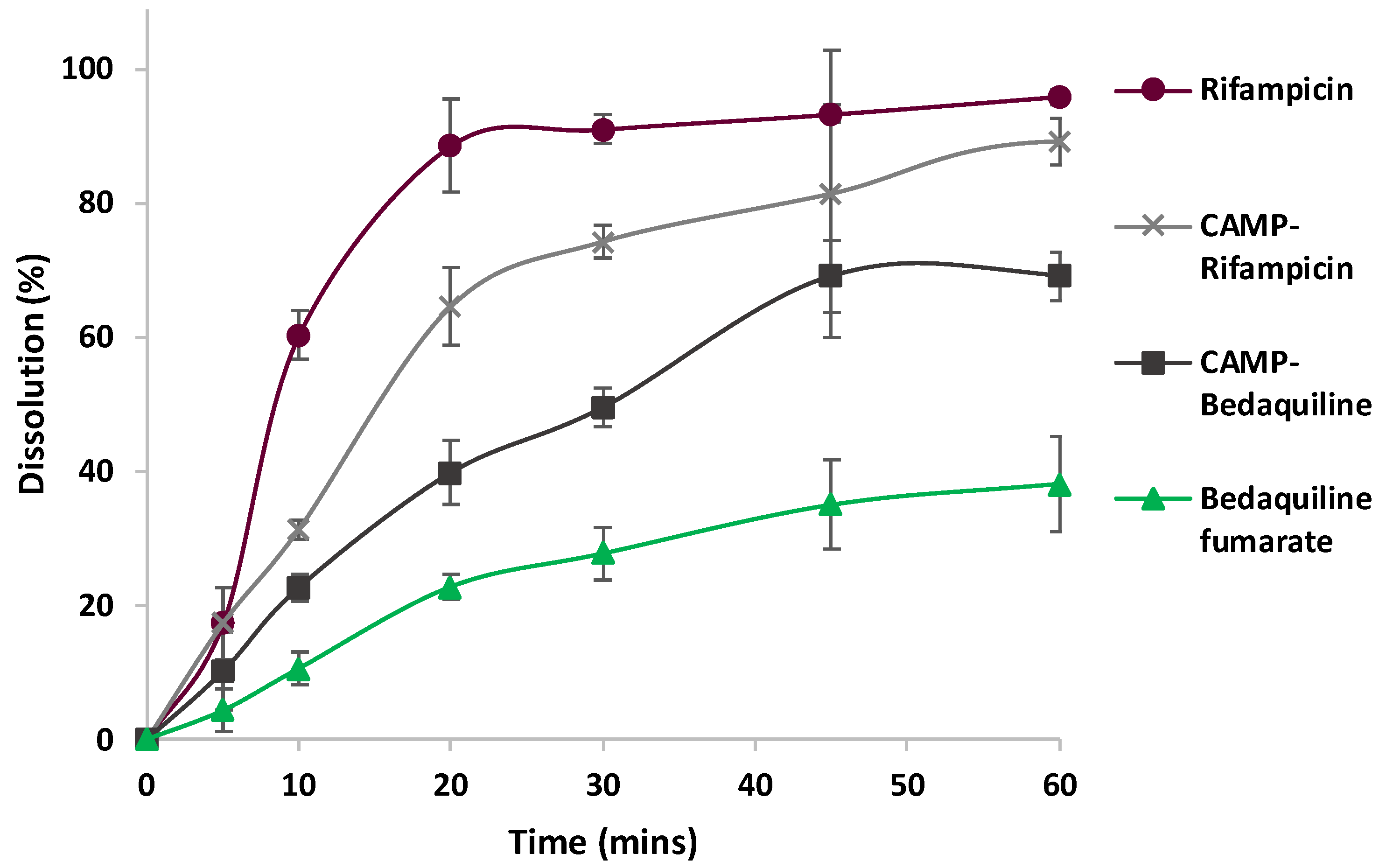
3.2. Dissolution
3.3. Accelerated Stability Study Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- WHO (World Health Organization). Global tuberculosis report 2023; Geneva, 2023. [Google Scholar]
- Pallett, S.; Houston, A. Tuberculosis. Medicine 2021, vol. 49(no. 12), 751–755. [Google Scholar] [CrossRef]
- Furin, J.; Cox, H.; Pai, M. Tuberculosis. The Lancet 2019, vol. 393(no. 10181), 1642–1656. [Google Scholar] [CrossRef]
- Martínez-Campreciós, J.; Espinosa-Pereiro, J.; Sánchez-Montalvá, A. Update on the treatment of tuberculosis. Medicina Clínica (English Edition) 2024, vol. 163(no. 5), 245–252. [Google Scholar] [CrossRef]
- Desikan, P.; et al. Trends of drug resistance in M. tuberculosis in a reference laboratory in Central India: Forging ahead towards TB elimination. Indian J Med Microbiol 2024, vol. 51, 100701. [Google Scholar] [CrossRef] [PubMed]
- Zumla, A. I. , New antituberculosis drugs, regimens, and adjunct therapies: needs, advances, and future prospects. Lancet Infect Dis 2014, vol. 14(no. 4), 327–340. [Google Scholar] [CrossRef]
- Sarathy, J. P.; Gruber, G.; Dick, T. Re-Understanding the Mechanisms of Action of the Anti-Mycobacterial Drug Bedaquiline. Antibiotics 2019, vol. 8(no. 4), 261. [Google Scholar] [CrossRef]
- Okezue, M. , Crystal structures of salts of bedaquiline. Acta Crystallogr C Struct Chem 2020, vol. 76(no. 11), 1010–1023. [Google Scholar] [CrossRef] [PubMed]
- Khoshnood, S. , Bedaquiline: Current status and future perspectives. J Glob Antimicrob Resist 2021, vol. 25, 48–59. [Google Scholar] [CrossRef] [PubMed]
- Unissa, A. Nusrath; Hanna, L. E. Molecular mechanisms of action, resistance, detection to the first-line anti tuberculosis drugs: Rifampicin and pyrazinamide in the post whole genome sequencing era. Tuberculosis 2017, vol. 105, 96–107. [Google Scholar] [CrossRef] [PubMed]
- Wehrli, W. Rifampin: Mechanisms of Action and Resistance. Clinical Infectious Diseases 1983, vol. 5, no. Supplement_3, S407–S411. [Google Scholar] [CrossRef] [PubMed]
- Wallis, R. S.; et al. Mycobactericidal activity of bedaquiline plus rifabutin or rifampin in ex vivo whole blood cultures of healthy volunteers: A randomized controlled trial. PLoS One 2018, vol. 13(no. 5), e0196756. [Google Scholar] [CrossRef]
- Svensson, E. M.; Murray, S.; Karlsson, M. O.; Dooley, K. E. Rifampicin and rifapentine significantly reduce concentrations of bedaquiline, a new anti-TB drug. Journal of Antimicrobial Chemotherapy 2015, vol. 70(no. 4), 1106–1114. [Google Scholar] [CrossRef]
- Younoussa, A.; et al. Protocol, rationale and design of BE-PEOPLE (Bedaquiline enhanced exposure prophylaxis for LEprosy in the Comoros): a cluster randomized trial on effectiveness of rifampicin and bedaquiline as post-exposure prophylaxis of leprosy contacts. BMC Infect Dis 2023, vol. 23(no. 1), 310. [Google Scholar] [CrossRef] [PubMed]
- Campbell, P. O.; Douglas, N. M.; Chambers, S. T. A Review of the Efficacy, Safety, and Feasibility of Rifamycin-Based Post-Exposure Chemoprophylaxis for Leprosy. Trop Med Infect Dis 2025, vol. 10(no. 4), 84. [Google Scholar] [CrossRef]
- Buya, A. B.; et al. Application of Lipid-Based Nanocarriers for Antitubercular Drug Delivery: A Review. Pharmaceutics 2021, vol. 13(no. 12), 2041. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Grohganz, H.; Löbmann, K.; Rades, T.; Hempel, N.-J. Co-Amorphous Drug Formulations in Numbers: Recent Advances in Co-Amorphous Drug Formulations with Focus on Co-Formability, Molar Ratio, Preparation Methods, Physical Stability, In Vitro and In Vivo Performance, and New Formulation Strategies. Pharmaceutics 2021, vol. 13(no. 3), 389. [Google Scholar] [CrossRef]
- FUKUOKA, E.; MAKITA, M.; YAMAMURA, S. Glassy State of Pharmaceuticals. III. : Thermal Properties and Stability of Glassy Pharmaceuticals and Their Binary Glass Systems. Chem Pharm Bull (Tokyo) 1989, vol. 37(no. 4), 1047–1050. [Google Scholar] [CrossRef]
- Saberi, A. , Development, recent advances, and updates in binary, ternary co-amorphous systems, and ternary solid dispersions. J Drug Deliv Sci Technol 2023, vol. 86, 104746. [Google Scholar] [CrossRef]
- Wang, S.-H.; et al. Hansen solubility parameter analysis on the dispersion of zirconia nanocrystals. J Colloid Interface Sci 2013, vol. 407, 140–147. [Google Scholar] [CrossRef]
- Mohammad, M. A.; Alhalaweh, A.; Velaga, S. P. Hansen solubility parameter as a tool to predict cocrystal formation. Int J Pharm 2011, vol. 407(no. 1), 63–71. [Google Scholar] [CrossRef]
- Salem, A.; Nagy, S.; Pál, S.; Széchenyi, A. Reliability of the Hansen solubility parameters as co-crystal formation prediction tool. Int J Pharm 2019, vol. 558, 319–327. [Google Scholar] [CrossRef]
- Peña, M. Á; Martínez, F. Hansen Solubility Parameters: A Tool for Solvent Selection in Drugs. Pharm Sci 2023, vol. 29(no. 2), 133–134. [Google Scholar] [CrossRef]
- Korshin, G.; Chow, C. W. K.; Fabris, R.; Drikas, M. Absorbance spectroscopy-based examination of effects of coagulation on the reactivity of fractions of natural organic matter with varying apparent molecular weights. Water Res 2009, vol. 43(no. 6), 1541–1548. [Google Scholar] [CrossRef]
- International Council for Harmonization. Guidance for Industry Q1A(R2) Stability Testing of New Drug Substances and Products; Nov 2003. [Google Scholar]
- Singh, C.; Bhatt, T. D.; Gill, M. S.; Suresh, S. Novel rifampicin–phospholipid complex for tubercular therapy: Synthesis, physicochemical characterization and in-vivo evaluation. Int J Pharm 2014, vol. 460(no. 1–2), 220–227. [Google Scholar] [CrossRef] [PubMed]
- Rangnekar, B.; Momin, M. A. M.; Eedara, B. B.; Sinha, S.; Das, S. C. Bedaquiline containing triple combination powder for inhalation to treat drug-resistant tuberculosis. Int J Pharm 2019, vol. 570, 118689. [Google Scholar] [CrossRef] [PubMed]
- Parvathaneni, V.; et al. Repurposing Bedaquiline for Effective Non-Small Cell Lung Cancer (NSCLC) Therapy as Inhalable Cyclodextrin-Based Molecular Inclusion Complexes. Int J Mol Sci 2021, vol. 22(no. 9), 4783. [Google Scholar] [CrossRef]
- Ivanisevic, *!!! REPLACE !!!*; McClurg, R. B.; Schields, P. J. Uses of X -Ray Powder Diffraction In the Pharmaceutical Industry. In Pharmaceutical Sciences Encyclopedia; Wiley, 2010; pp. 1–42. [Google Scholar] [CrossRef]
- Khadka, P.; Hill, P. C.; Zhang, B.; Katare, R.; Dummer, J.; Das, S. C. A study on polymorphic forms of rifampicin for inhaled high dose delivery in tuberculosis treatment. Int J Pharm 2020, vol. 587, 119602. [Google Scholar] [CrossRef]
- Alves, R.; Reis, T. V. da S.; da Silva, L. C. C.; Storpírtis, S.; Mercuri, L. P.; Matos, J. do R. Thermal behavior and decomposition kinetics of rifampicin polymorphs under isothermal and non-isothermal conditions. Brazilian Journal of Pharmaceutical Sciences 2010, vol. 46(no. 2), 343–351. [Google Scholar] [CrossRef]
- Pardhi, V. P.; Jain, K. Impact of binary/ternary solid dispersion utilizing poloxamer 188 and TPGS to improve pharmaceutical attributes of bedaquiline fumarate. J Drug Deliv Sci Technol 2021, vol. 62, 102349. [Google Scholar] [CrossRef]
- Agrawal, S.; Ashokraj, Y.; Bharatam, P. V.; Pillai, O.; Panchagnula, R. Solid-state characterization of rifampicin samples and its biopharmaceutic relevance. European Journal of Pharmaceutical Sciences 2004, vol. 22(no. 2–3), 127–144. [Google Scholar] [CrossRef] [PubMed]
- Jinyi XULiang ZhangXiangyang ZhangXinzeng WANGJian CHAIHongying LUOZhiqing YANG, “Crystalline forms of bedaquiline fumarate and preparation methods therefor,” EP3366676B1, 2021.

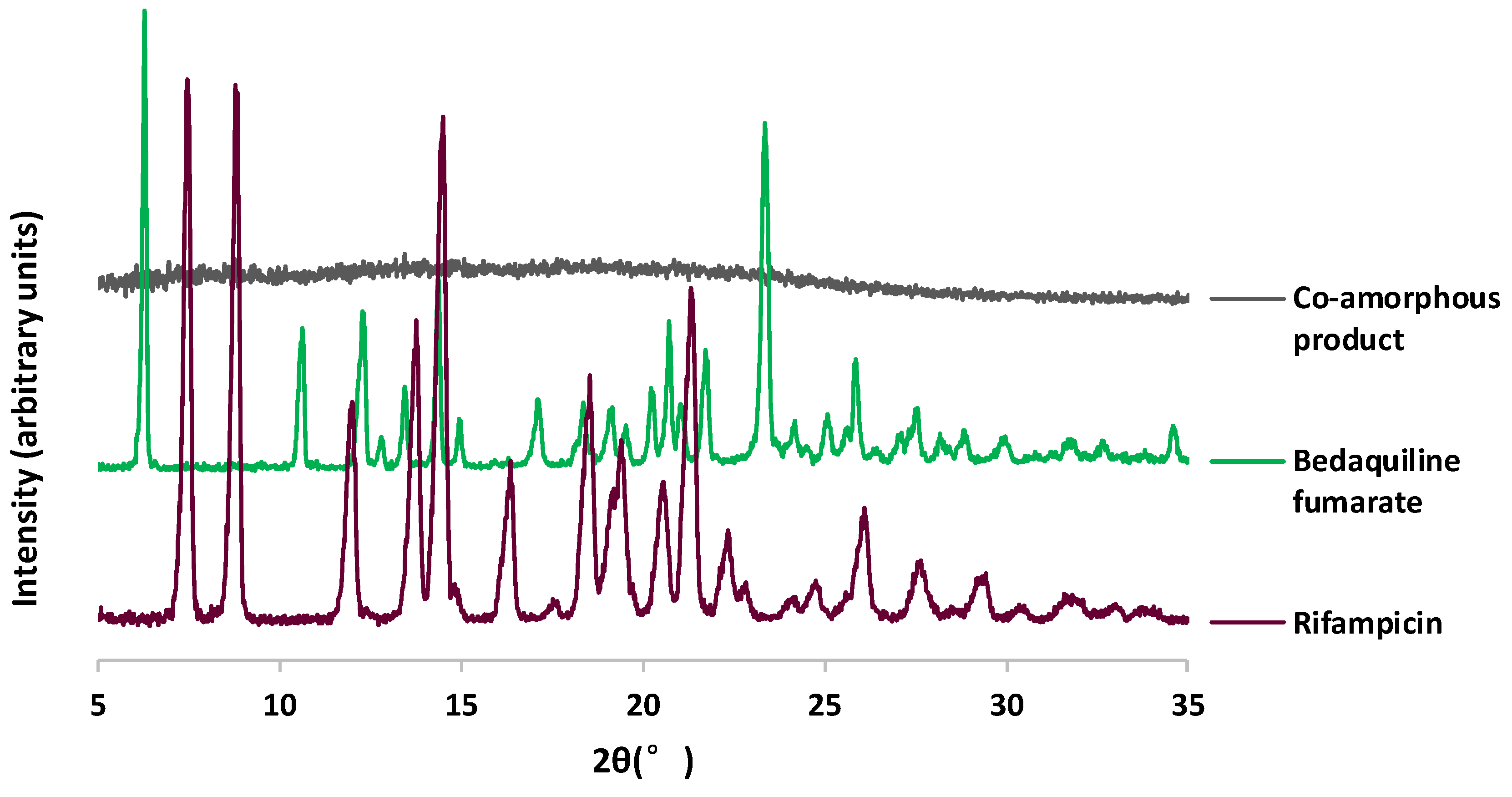
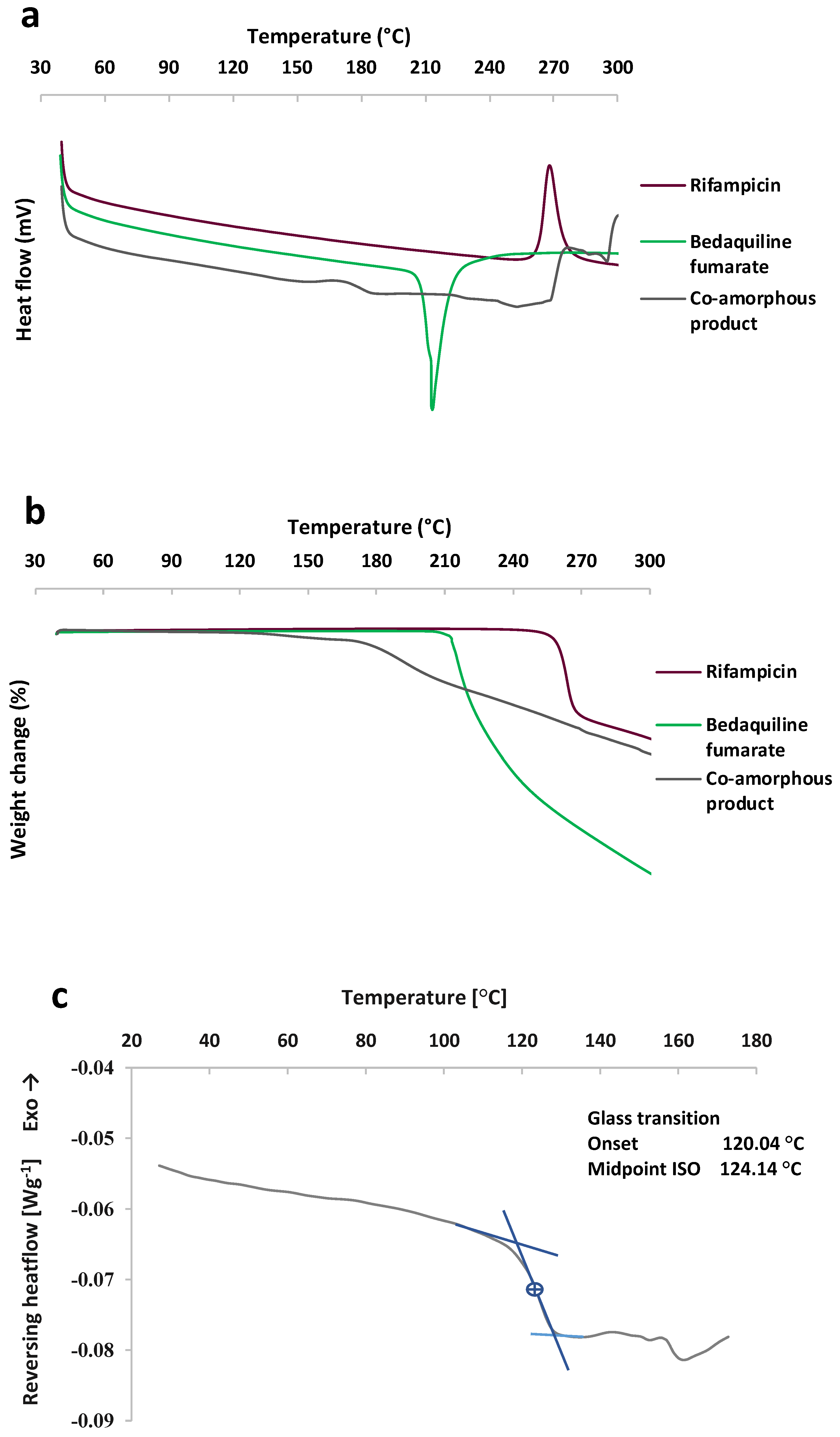
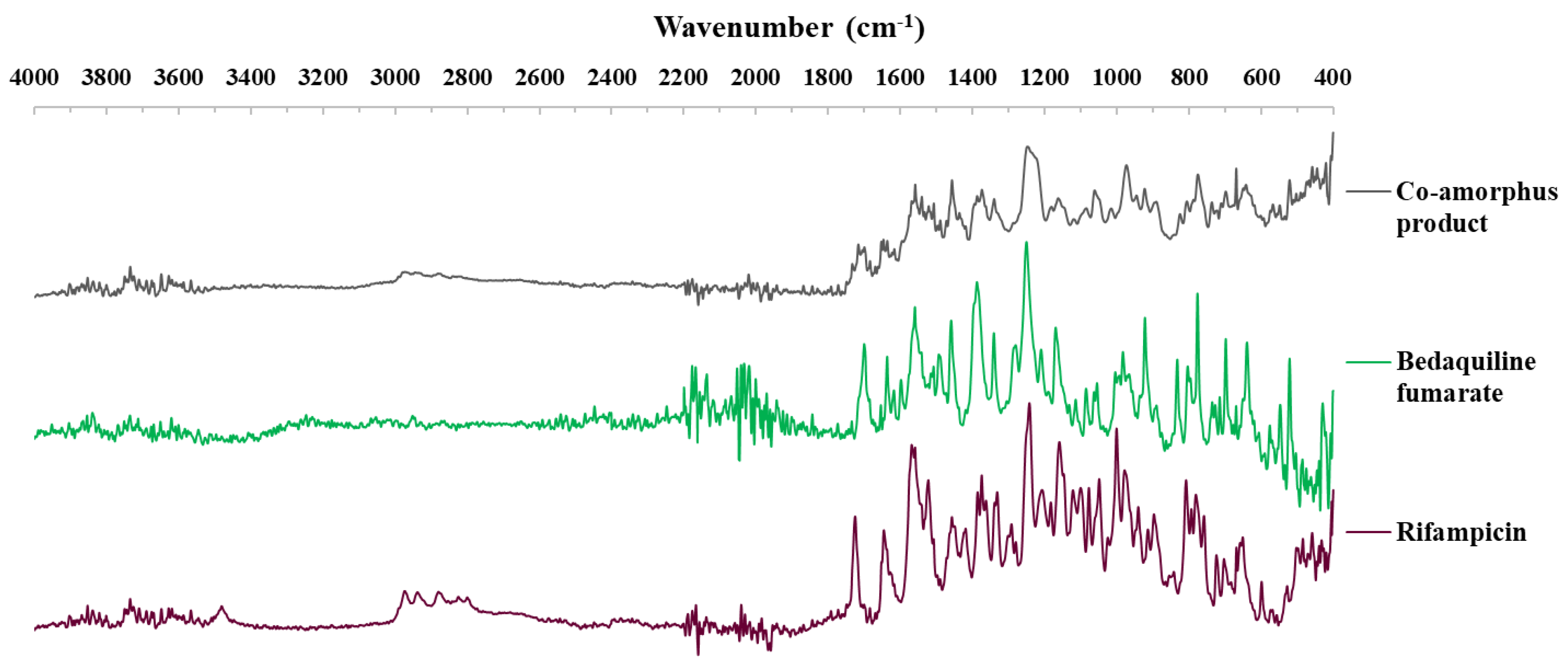
| API | δd | δp | δh | δ | δhDon | δhAcc | Ra | Δδ | Δδtot |
| Rifampicin | 20.0 | 3.4 | 3.8 | 20.6 | 0.3 | 1 | - | - | - |
| Bedaquiline | 20.5 | 3.4 | 5.6 | 21.5 | 1.5 | 5.8 | 2.0 | 0.9 | 1.8 |
| Fumaric acid | 18.16 | 10.78 | 23.02 | 30.2 | 18.8 | 13.2 | 20.9 | 10.6 | 20.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




