Submitted:
15 April 2026
Posted:
16 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Intervention
2.4. Study Procedures and Assessments
- Baseline (T0)
- Week 4 (T1)
- Week 8 (T2, end of treatment)
- Geriatric Depression Scale – 15 items (GDS-15)
- Clinical Global Impression – Severity (CGI-S)
- Montreal Cognitive Assessment (MoCA)
- Apathy Evaluation Scale – Informant (AES-I)
2.5. Statistical Analysis
3. Results
3.1. Participant Characteristics
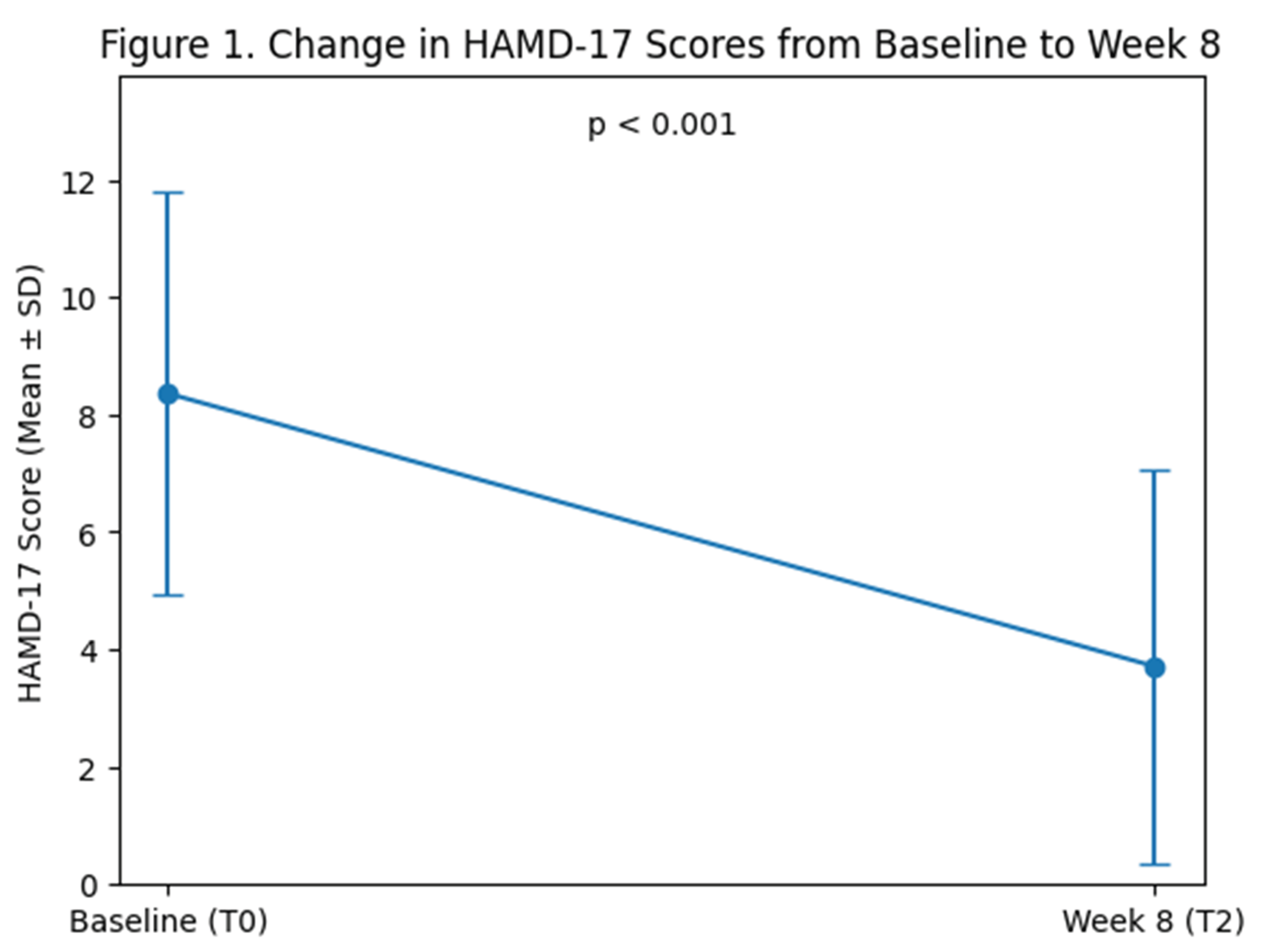
3.2. Primary Outcome
3.3. Secondary Outcomes
3.4. Safety and Tolerability
4. Discussion
4.1. Effects on Depressive Symptoms
4.2. Cognitive Function and Apathy
4.3. Clinical Implications
4.4. Limitations
4.5. Future Directions
5. Conclusion
Authors’ contributions
Funding
Consent for publication
Ethics statement
Data availability statement
Acknowledgments
Competing interests
References
- Volz, H.P.; Stirnweiß, J.; Kasper, S.; Möller, H.J.; Seifritz, E. Subthreshold depression - concept, operationalisation and epidemiological data. A scoping review. Int J Psychiatry Clin Pract. 2023, 27(1), 92–106. [Google Scholar] [CrossRef]
- Judd, L.L.; Rapaport, M.H.; Paulus, M.P.; Brown, J.L. Subsyndromal symptomatic depression: a new mood disorder? J Clin Psychiatry 1994, 55, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Granata, N.; Vercesi, M.; Bonfanti, A.; Mencacci, C.; Coco, I.; Mangrella, M.; Piazza, R.; Cerveri, G. Choline Alphoscerate: A Therapeutic Option for the Management of Subthreshold Depression in the Older Population. Geriatrics (Basel) 2025, 10(2), 32. [Google Scholar] [CrossRef] [PubMed]
- Boyle, X.; Zhang, L.; Sáenz, A.A.; Zhang, X.; Sun, J.; Zhong, Q.; Cheng, Y.; Jia, Y. Prevalence of subthreshold depression in older adults: A systematic review and meta-analysis. Asian J Psychiatr. 2024, 102, 104253. [Google Scholar] [CrossRef]
- Xiang, X.; Leggett, A.; Himle, J.A.; Kales, H.C. Major Depression and Subthreshold Depression among Older Adults Receiving Home Care. Am J Geriatr Psychiatry 2018, 26(9), 939–949. [Google Scholar] [CrossRef] [PubMed]
- Beekman, A.T.; Deeg, D.J.; Braam, A.W.; Smit, J.H.; Van Tilburg, W. Consequences of major and minor depression in later life: a study of disability, well-being and service utilization. Psychol Med. 1997, 27(6), 1397–409. [Google Scholar] [CrossRef]
- Hybels, C.F.; Pieper, C.F.; Blazer, D.G. The complex relationship between depressive symptoms and functional limitations in community-dwelling older adults: the impact of subthreshold depression. Psychol Med. 2009, 39(10), 1677–88. [Google Scholar] [CrossRef]
- Conradi, H.J.; Ormel, J.; De Jonge, P. Presence of individual (residual) symptoms during depressive episodes and periods of remission: a 3-year prospective study. Psychol Med. 2011, 41(6), 1165–74. [Google Scholar] [CrossRef]
- Han, L.; McCusker, J.; Cole, M.; Abrahamowicz, M.; Capek, R. 12-month cognitive outcomes of major and minor depression in older medical patients. Am J Geriatr Psychiatry 2008, 16(9), 742–51. [Google Scholar] [CrossRef]
- Boyle, L.L.; Porsteinsson, A.P.; Cui, X.; King, D.A.; Lyness, J.M. Depression predicts cognitive disorders in older primary care patients. J Clin Psychiatry 2010, 71(1), 74–9. [Google Scholar] [CrossRef]
- Szymkowicz, S.M.; Gerlach, A.R.; Homiack, D.; Taylor, W.D. Biological factors influencing depression in later life: role of aging processes and treatment implications. Transl Psychiatry 2023, 13(1), 160. [Google Scholar] [CrossRef]
- Barbui, C.; Cipriani, A.; Patel, V.; Ayuso-Mateos, J.L.; van Ommeren, M. Efficacy of antidepressants and benzodiazepines in minor depression: systematic review and meta-analysis. Br J Psychiatry 2011, 198(1), 11–6, sup 1. [Google Scholar] [CrossRef] [PubMed]
- Coupland, C.; Dhiman, P.; Morriss, R.; Arthur, A.; Barton, G.; Hippisley-Cox, J. Antidepressant use and risk of adverse outcomes in older people: population based cohort study. BMJ. 2011, 343, d4551. [Google Scholar] [CrossRef]
- Bolton, J.M.; Metge, C.; Lix, L.; Prior, H.; Sareen, J.; Leslie, W.D. Fracture risk from psychotropic medications: a population-based analysis. J Clin Psychopharmacol. 2008, 28(4), 384–91. [Google Scholar] [CrossRef]
- Hegerl, U.; Schönknecht, P.; Mergl, R. Are antidepressants useful in the treatment of minor depression: a critical update of the current literature. Curr Opin Psychiatry 2012, 25(1), 1–6. [Google Scholar] [CrossRef]
- Gatti, G.; Barzaghi, N.; Acuto, G.; Abbiati, G.; Fossati, T.; Perucca, E. A comparative study of free plasma choline levels following intramuscular administration of L-alpha-glycerylphosphorylcholine and citicoline in normal volunteers. Int J Clin Pharmacol Ther Toxicol 1992, 30(9), 331–5. [Google Scholar]
- De Jesus Moreno Moreno, M. Cognitive improvement in mild to moderate Alzheimer’s dementia after treatment with the acetylcholine precursor choline alfoscerate: a multicenter, double-blind, randomized, placebo-controlled trial. Clin Ther. 2003, 25, 178–93. [Google Scholar] [CrossRef] [PubMed]
- Amenta, F.; Carotenuto, A.; Fasanaro, A.M.; et al. The ASCOMALVA (Association between the Cholinesterase Inhibitor Donepezil and the Cholinergic Pre- cursor Choline Alphoscerate in Alzheimer’s Disease) Trial: interim results after two years of treatment. J Alzheimers Dis. 2014, 42 (Suppl. 3), S281–8. [Google Scholar] [CrossRef] [PubMed]
- Tayebati, S.K.; Tomassoni, D.; Nwankwo, I.E.; Di Stefano, A.; Sozio, P.; Cerasa, L.S.; Amenta, F. Modulation of monoaminergic transporters by choline-containing phospholipids in rat brain. CNS Neurol Disord Drug Targets 2013, 12(1), 94–103. [Google Scholar] [CrossRef]
- Rea, R.; Carotenuto, A.; Traini, E.; Fasanaro, A.M.; Manzo, V.; Amenta, F. Apathy Treatment in Alzheimer’s Disease: Interim Results of the ASCOMALVA Trial. J Alzheimers Dis. 2015, 48(2), 377–83. [Google Scholar] [CrossRef]
- Carotenuto, A. Association Between the Cholinesterase Inhibitor Donepezil and the Cholinergic Precursor Choline Alphoscerate in the Treatment of Depression in Patients with Alzheimer’s Disease. J Alzheimers Dis Rep. 2022, 6(1), 235–243. [Google Scholar] [CrossRef] [PubMed]
- Adriani, B.; Arena, J.F.; Fioretti, A.; Mancino, S.; Sarno, F.; Ferracuti, S.; Del Casale, A. Current Diagnostic challenges in late-life depression and neurocognitive disorders. Psychiatry Int. 2024, 5, 904–916. [Google Scholar] [CrossRef]
- Sagaro, G.G.; Amenta, F. Comparison of the effects of choline alphoscerate and citicoline in patients with dementia disorders: a systematic review and meta-analysis. Front Neurol. 2025, 16, 1649661. [Google Scholar] [CrossRef]
- Julious, A. Sample size of 12 per group rule of thumb for a pilot study. Pharmaceut Statis 2005, 4, 287–91. [Google Scholar] [CrossRef]
- Hertzog, M.A. Considerations in determining sample size for pilot studies. Res Nurs Health 2008, 31, 180–91. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Total Sample (N=17), mean ± SD |
|---|---|
| Age (years) | 76.0 ± 7.95 |
| Sex, n (%) | |
|
11 (65%) |
|
6 (35%) |
| Education (years) | 10.65 ± 4.53 |
| MMSE score | 27.02 ± 1.78 |
| HAMD-17 score | 8.47 ± 3.13 |
| GDS-15 score | 6.82 ± 2.58 |
| CGI-S score | 2.76 ± 0.83 |
| MoCA score | 22.29 ± 4.65 |
| Outcome Measure | n | T0, mean ± SD | T2, mean ± SD | Mean change (T2–T0) | p-value |
|---|---|---|---|---|---|
| GDS-15 | 15 | 6.82 ± 2.58 | 5.00 ± 3.06 | −1.82 | <0.05 |
| CGI-S* | 15 | 2.76 ± 0.83 | 1.87 ± 0.91 | −0.89 | <0.05 |
| MoCA | 14 | 22.29 ± 4.65 | 25.00 ± 3.78 | +2.71 | n.s. |
| AES-I | 15 | 48.27 ± 5.18 | 50.07 ± 6.65 | +1.80 | n.s. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




