Submitted:
15 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Psilocybin
3. Mechanism of Action of Psilocybin: Implications for the Aging Brain
4. Anti-Inflammatory Action of Psilocybin
5. Clinical Applications in Inflammation-Linked Conditions
5.1. Depression and Anxiety
5.2. Neurodegenerative Disorders
5.3. Pharmacokinetics of Psilocybin
5.4. Effectiveness of Psilocybin in Older Adults
5.4.1. Neurobiological Considerations
5.4.2. Emerging Clinical Evidence
6. Adverse Effects and Safety Considerations of Psilocybin
7. Perspectives
8. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AD | – Alzheimer’s disease |
| ALS | – amyotrophic lateral sclerosis |
| BBB | – blood-brain barrier |
| BDNF | – brain-derived neurotrophic factor |
| CRP | – C-reactive protein |
| DMN | – default mode network |
| FDA | – Food and Drug Administration |
| HPA | – hypothalamic–pituitary–adrenal |
| IL-1β | – interleukin-1 beta |
| IL-6 | – interleukin-6 |
| LPS | – lipopolysaccharide |
| LSD | – lysergic acid diethylamide |
| MCI | – mild cognitive impairment |
| MDD | – major depressive disorder |
| MS | – multiple sclerosis |
| mTOR | – mammalian target of rapamycin |
| NF-κB | – nuclear factor-kappa B |
| NSAIDs | – nonsteroidal anti-inflammatory drugs |
| PD | – Parkinson’s disease |
| SSRI | – selective serotonin reuptake inhibitor |
| TBI | – traumatic brain injury |
| TNF-α | – tumor necrosis factor-alpha |
| TRD | – treatment-resistant depression |
| TrkB | – tropomyosin receptor kinase B |
| WHO | – World Health Organization |
| 5-HT | – serotonin |
| 5-HT2A | – serotonin 2A receptor |
References
- Mental Health of Older Adults Available online: https://www.who.int/news-room/fact-sheets/detail/mental-health-of-older-adults (accessed on 27 August 2025).
- Elshaikh, U.; Sheik, R.; Saeed, R.K.M.; Chivese, T.; Alsayed Hassan, D. Barriers and Facilitators of Older Adults for Professional Mental Health Help-Seeking: A Systematic Review. BMC Geriatr 2023, 23, 516. [CrossRef]
- Franceschi, C.; Garagnani, P.; Parini, P.; Giuliani, C.; Santoro, A. Inflammaging: A New Immune-Metabolic Viewpoint for Age-Related Diseases. Nat Rev Endocrinol 2018, 14, 576–590. [CrossRef]
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; et al. Chronic Inflammation in the Etiology of Disease across the Life Span. Nat Med 2019, 25, 1822–1832. [CrossRef]
- Blossom, V.; Ullal, S.D.; D’Souza, M.M.; Ranade, A.V.; Kumar, N.A.; Rai, R. Implicating Neuroinflammation in Hippocampus, Prefrontal Cortex and Amygdala with Cognitive Deficit: A Narrative Review. 3 Biotech 2025, 15, 320. [CrossRef]
- Aquino, Y.C.; Cabral, L.M.; Miranda, N.C.; Naccarato, M.C.; Falquetto, B.; Moreira, T.S.; Takakura, A.C. Respiratory Disorders of Parkinson’s Disease. J Neurophysiol 2022, 127, 1–15. [CrossRef]
- Lassmann, H. Multiple Sclerosis Pathology. Cold Spring Harb Perspect Med 2018, 8, a028936. [CrossRef]
- Jia, P.; Peng, Q.; Fan, X.; Zhang, Y.; Xu, H.; Li, J.; Sonita, H.; Liu, S.; Le, A.; Hu, Q.; et al. Immune-Mediated Disruption of the Blood-Brain Barrier after Intracerebral Hemorrhage: Insights and Potential Therapeutic Targets. CNS Neurosci Ther 2024, 30, e14853. [CrossRef]
- de Deus, J.L.; Maia, J.M.; Soriano, R.N.; Amorim, M.R.; Branco, L.G.S. Psychedelics in Neuroinflammation: Mechanisms and Therapeutic Potential. Prog Neuropsychopharmacol Biol Psychiatry 2025, 137, 111278. [CrossRef]
- Wang, S.-M.; Kim, S.; Choi, W.-S.; Lim, H.K.; Woo, Y.S.; Pae, C.-U.; Bahk, W.-M. Current Understanding on Psilocybin for Major Depressive Disorder: A Review Focusing on Clinical Trials. Clin Psychopharmacol Neurosci 2024, 22, 222–231. [CrossRef]
- Lal, R.; Singh, A.; Watts, S.; Chopra, K. Experimental Models of Parkinson’s Disease: Challenges and Opportunities. Eur J Pharmacol 2024, 980, 176819. [CrossRef]
- Zveik, O.; Rechtman, A.; Ganz, T.; Vaknin-Dembinsky, A. The Interplay of Inflammation and Remyelination: Rethinking MS Treatment with a Focus on Oligodendrocyte Progenitor Cells. Mol Neurodegener 2024, 19, 53. [CrossRef]
- Dharmadasa, T.; Pavey, N.; Tu, S.; Menon, P.; Huynh, W.; Mahoney, C.J.; Timmins, H.C.; Higashihara, M.; van den Bos, M.; Shibuya, K.; et al. Novel Approaches to Assessing Upper Motor Neuron Dysfunction in Motor Neuron Disease/Amyotrophic Lateral Sclerosis: IFCN Handbook Chapter. Clin Neurophysiol 2024, 163, 68–89. [CrossRef]
- Negro-Demontel, L.; Maleki, A.F.; Reich, D.S.; Kemper, C. The Complement System in Neurodegenerative and Inflammatory Diseases of the Central Nervous System. Front Neurol 2024, 15, 1396520. [CrossRef]
- Obukohwo, O.M.; Oreoluwa, O.A.; Andrew, U.O.; Williams, U.E. Microglia-Mediated Neuroinflammation in Traumatic Brain Injury: A Review. Mol Biol Rep 2024, 51, 1073. [CrossRef]
- Pitharouli, M.C.; Hagenaars, S.P.; Glanville, K.P.; Coleman, J.R.I.; Hotopf, M.; Lewis, C.M.; Pariante, C.M. Elevated C-Reactive Protein in Patients With Depression, Independent of Genetic, Health, and Psychosocial Factors: Results From the UK Biobank. AJP 2021, 178, 522–529. [CrossRef]
- Jóźwiak-Bębenista, M.; Sokolowska, P.; Wiktorowska-Owczarek, A.; Kowalczyk, E.; Sienkiewcz, M. Ketamine - a New Antidepressant Drug with Anti-Inflammatory Properties. Journal of Pharmacology and Experimental Therapeutics 2023, JPET-MR. [CrossRef]
- Haniff, Z.R.; Bocharova, M.; Mantingh, T.; Rucker, J.J.; Velayudhan, L.; Taylor, D.M.; Young, A.H.; Aarsland, D.; Vernon, A.C.; Thuret, S. Psilocybin for Dementia Prevention? The Potential Role of Psilocybin to Alter Mechanisms Associated with Major Depression and Neurodegenerative Diseases. Pharmacol Ther 2024, 258, 108641. [CrossRef]
- Amorim, M.R.; de Deus, J.L.; Cazuza, R.A.; Mota, C.M.D.; da Silva, L.E.V.; Borges, G.S.; Batalhão, M.E.; Cárnio, E.C.; Branco, L.G.S. Neuroinflammation in the NTS Is Associated with Changes in Cardiovascular Reflexes during Systemic Inflammation. J Neuroinflammation 2019, 16, 125. [CrossRef]
- Bouchet, L.; Sager, Z.; Yrondi, A.; Nigam, K.B.; Anderson, B.T.; Ross, S.; Petridis, P.D.; Beaussant, Y. Older Adults in Psychedelic-Assisted Therapy Trials: A Systematic Review. J Psychopharmacol 2024, 38, 33–48. [CrossRef]
- Haikazian, S.; Chen-Li, D.C.J.; Johnson, D.E.; Fancy, F.; Levinta, A.; Husain, M.I.; Mansur, R.B.; McIntyre, R.S.; Rosenblat, J.D. Psilocybin-Assisted Therapy for Depression: A Systematic Review and Meta-Analysis. Psychiatry Res 2023, 329, 115531. [CrossRef]
- Vollenweider, F.X.; Kometer, M. The Neurobiology of Psychedelic Drugs: Implications for the Treatment of Mood Disorders. Nat Rev Neurosci 2010, 11, 642–651. [CrossRef]
- Nichols, D.E. Psilocybin: From Ancient Magic to Modern Medicine. J Antibiot 2020, 73, 679–686. [CrossRef]
- Tabaac, B.J.; Shinozuka, K.; Arenas, A.; Beutler, B.D.; Cherian, K.; Evans, V.D.; Fasano, C.; Muir, O.S. Psychedelic Therapy: A Primer for Primary Care Clinicians-Historical Perspective and Overview. Am J Ther 2024, 31, e97–e103. [CrossRef]
- Sharma, P.; Nguyen, Q.A.; Matthews, S.J.; Carpenter, E.; Mathews, D.B.; Patten, C.A.; Hammond, C.J. Psilocybin History, Action and Reaction: A Narrative Clinical Review. J Psychopharmacol 2023, 37, 849–865. [CrossRef]
- Siegel, J.S.; Daily, J.E.; Perry, D.A.; Nicol, G.E. Psychedelic Drug Legislative Reform and Legalization in the US. JAMA Psychiatry 2023, 80, 77–83. [CrossRef]
- Nutt, D.; Carhart-Harris, R. The Current Status of Psychedelics in Psychiatry. JAMA Psychiatry 2021, 78, 121–122. [CrossRef]
- MacCallum, C.A.; Lo, L.A.; Pistawka, C.A.; Deol, J.K. Therapeutic Use of Psilocybin: Practical Considerations for Dosing and Administration. Front Psychiatry 2022, 13, 1040217. [CrossRef]
- Lowe, H.; Toyang, N.; Steele, B.; Valentine, H.; Grant, J.; Ali, A.; Ngwa, W.; Gordon, L. The Therapeutic Potential of Psilocybin. Molecules 2021, 26, 2948. [CrossRef]
- Moliner, R.; Girych, M.; Brunello, C.A.; Kovaleva, V.; Biojone, C.; Enkavi, G.; Antenucci, L.; Kot, E.F.; Goncharuk, S.A.; Kaurinkoski, K.; et al. Psychedelics Promote Plasticity by Directly Binding to BDNF Receptor TrkB. Nat Neurosci 2023, 26, 1032–1041. [CrossRef]
- Xue, B.; Waseem, S.M.A.; Zhu, Z.; Alshahrani, M.A.; Nazam, N.; Anjum, F.; Habib, A.H.; Rafeeq, M.M.; Nazam, F.; Sharma, M. Brain-Derived Neurotrophic Factor: A Connecting Link Between Nutrition, Lifestyle, and Alzheimer’s Disease. Front Neurosci 2022, 16, 925991. [CrossRef]
- Ranasinghe, P.V.N.N.; Mapa, M.S.T. Functional Connectivity and Cognitive Decline: A Review of Rs-fMRI, EEG, MEG, and Graph Theory Approaches in Aging and Dementia. Explor Med. 2024, 5, 797–821. [CrossRef]
- Kim, J.; Kim, Y.-K. Crosstalk between Depression and Dementia with Resting-State fMRI Studies and Its Relationship with Cognitive Functioning. Biomedicines 2021, 9, 82. [CrossRef]
- Carhart-Harris, R.L.; Erritzoe, D.; Williams, T.; Stone, J.M.; Reed, L.J.; Colasanti, A.; Tyacke, R.J.; Leech, R.; Malizia, A.L.; Murphy, K.; et al. Neural Correlates of the Psychedelic State as Determined by fMRI Studies with Psilocybin. Proc Natl Acad Sci U S A 2012, 109, 2138–2143. [CrossRef]
- Chisamore, N.; Kaczmarek, E.; Le, G.H.; Wong, S.; Orsini, D.; Mansur, R.; McIntyre, R.; Rosenblat, J. Neurobiology of the Antidepressant Effects of Serotonergic Psychedelics: A Narrative Review. Current Treatment Options in Psychiatry 2024, 11, 1–16. [CrossRef]
- Bysiek, A.; Wojtas, A.; Szpręgiel, I.; Wawrzczak-Bargieła, A.; Maćkowiak, M.; Gołembiowska, K. The Effect of Low-Dose Psilocybin on Brain Neurotransmission and Rat Behavior. Prog Neuropsychopharmacol Biol Psychiatry 2025, 138, 111347. [CrossRef]
- Wojtas, A.; Bysiek, A.; Wawrzczak-Bargiela, A.; Maćkowiak, M.; Gołembiowska, K. Limbic System Response to Psilocybin and Ketamine Administration in Rats: A Neurochemical and Behavioral Study. Int J Mol Sci 2023, 25, 100. [CrossRef]
- Kozlowska, U.; Nichols, C.; Wiatr, K.; Figiel, M. From Psychiatry to Neurology: Psychedelics as Prospective Therapeutics for Neurodegenerative Disorders. J Neurochem 2022, 162, 89–108. [CrossRef]
- Mason, N.L.; Szabo, A.; Kuypers, K.P.C.; Mallaroni, P.A.; de la Torre Fornell, R.; Reckweg, J.T.; Tse, D.H.Y.; Hutten, N.R.P.W.; Feilding, A.; Ramaekers, J.G. Psilocybin Induces Acute and Persisting Alterations in Immune Status in Healthy Volunteers: An Experimental, Placebo-Controlled Study. Brain, Behavior, and Immunity 2023, 114, 299–310. [CrossRef]
- Guida, C.R.; Maia, J.M.; Ferreira, L.F.R.; Rahdar, A.; Branco, L.G.S.; Soriano, R.N. Advancements in Addressing Drug Dependence: A Review of Promising Therapeutic Strategies and Interventions. Progress in Neuro-Psychopharmacology and Biological Psychiatry 2024, 134, 111070. [CrossRef]
- Vargas, M.V.; Dunlap, L.E.; Dong, C.; Carter, S.J.; Tombari, R.J.; Jami, S.A.; Cameron, L.P.; Patel, S.D.; Hennessey, J.J.; Saeger, H.N.; et al. Psychedelics Promote Neuroplasticity through the Activation of Intracellular 5-HT2A Receptors. Science 2023, 379, 700–706. [CrossRef]
- Ornelas, I.M.; Cini, F.A.; Wießner, I.; Marcos, E.; Araújo, D.B.; Goto-Silva, L.; Nascimento, J.; Silva, S.R.B.; Costa, M.N.; Falchi, M.; et al. Nootropic Effects of LSD: Behavioral, Molecular and Computational Evidence. Experimental Neurology 2022, 356, 114148. [CrossRef]
- Szabo, A. Psychedelics and Immunomodulation: Novel Approaches and Therapeutic Opportunities. Front. Immunol. 2015, 6. [CrossRef]
- Kariyawasam, S.H.; Handley, S.L. Quinolinic Acid Inhibits DOI-Induced Head Shakes in Mice. J Psychopharmacol 1999, 13, 284–285. [CrossRef]
- Villalba, S.; González, B.; Junge, S.; Bernardi, A.; González, J.; Fagúndez, C.; Torterolo, P.; Carrera, I.; Urbano, F.J.; Bisagno, V. 5-HT2A Receptor Knockout Mice Show Sex-Dependent Differences Following Acute Noribogaine Administration. International Journal of Molecular Sciences 2024, 25, 687. [CrossRef]
- Gouda, N.A.; Alshammari, S.O.; Abourehab, M.A.S.; Alshammari, Q.A.; Elkamhawy, A. Therapeutic Potential of Natural Products in Inflammation: Underlying Molecular Mechanisms, Clinical Outcomes, Technological Advances, and Future Perspectives. Inflammopharmacol 2023, 31, 2857–2883. [CrossRef]
- VanderZwaag, J.; Halvorson, T.; Dolhan, K.; Šimončičová, E.; Ben-Azu, B.; Tremblay, M.-È. The Missing Piece? A Case for Microglia’s Prominent Role in the Therapeutic Action of Anesthetics, Ketamine, and Psychedelics. Neurochem Res 2023, 48, 1129–1166. [CrossRef]
- Burmester, D.R.; Madsen, M.K.; Szabo, A.; Aripaka, S.S.; Stenbæk, D.S.; Frokjaer, V.G.; Elfving, B.; Mikkelsen, J.D.; Knudsen, G.M.; Fisher, P.M. Subacute Effects of a Single Dose of Psilocybin on Biomarkers of Inflammation in Healthy Humans: An Open-Label Preliminary Investigation. Compr Psychoneuroendocrinol 2023, 13, 100163. [CrossRef]
- Gukasyan, N.; Davis, A.K.; Barrett, F.S.; Cosimano, M.P.; Sepeda, N.D.; Johnson, M.W.; Griffiths, R.R. Efficacy and Safety of Psilocybin-Assisted Treatment for Major Depressive Disorder: Prospective 12-Month Follow-Up. J Psychopharmacol 2022, 36, 151–158. [CrossRef]
- Davis, A.K.; Barrett, F.S.; May, D.G.; Cosimano, M.P.; Sepeda, N.D.; Johnson, M.W.; Finan, P.H.; Griffiths, R.R. Effects of Psilocybin-Assisted Therapy on Major Depressive Disorder. JAMA Psychiatry 2021, 78, 1–9. [CrossRef]
- Carhart-Harris, R.; Giribaldi, B.; Watts, R.; Baker-Jones, M.; Murphy-Beiner, A.; Murphy, R.; Martell, J.; Blemings, A.; Erritzoe, D.; Nutt, D.J. Trial of Psilocybin versus Escitalopram for Depression. N Engl J Med 2021, 384, 1402–1411. [CrossRef]
- Carhart-Harris, R.L.; Bolstridge, M.; Rucker, J.; Day, C.M.J.; Erritzoe, D.; Kaelen, M.; Bloomfield, M.; Rickard, J.A.; Forbes, B.; Feilding, A.; et al. Psilocybin with Psychological Support for Treatment-Resistant Depression: An Open-Label Feasibility Study. The Lancet Psychiatry 2016, 3, 619–627. [CrossRef]
- Goodwin, G.M.; Aaronson, S.T.; Alvarez, O.; Arden, P.C.; Baker, A.; Bennett, J.C.; Bird, C.; Blom, R.E.; Brennan, C.; Brusch, D.; et al. Single-Dose Psilocybin for a Treatment-Resistant Episode of Major Depression. N Engl J Med 2022, 387, 1637–1648. [CrossRef]
- Goodwin, G.M.; Croal, M.; Feifel, D.; Kelly, J.R.; Marwood, L.; Mistry, S.; O’Keane, V.; Peck, S.K.; Simmons, H.; Sisa, C.; et al. Psilocybin for Treatment Resistant Depression in Patients Taking a Concomitant SSRI Medication. Neuropsychopharmacology 2023, 48, 1492–1499. [CrossRef]
- Griffiths, R.R.; Johnson, M.W.; Carducci, M.A.; Umbricht, A.; Richards, W.A.; Richards, B.D.; Cosimano, M.P.; Klinedinst, M.A. Psilocybin Produces Substantial and Sustained Decreases in Depression and Anxiety in Patients with Life-Threatening Cancer: A Randomized Double-Blind Trial. J Psychopharmacol 2016, 30, 1181–1197. [CrossRef]
- Ross, S.; Bossis, A.; Guss, J.; Agin-Liebes, G.; Malone, T.; Cohen, B.; Mennenga, S.E.; Belser, A.; Kalliontzi, K.; Babb, J.; et al. Rapid and Sustained Symptom Reduction Following Psilocybin Treatment for Anxiety and Depression in Patients with Life-Threatening Cancer: A Randomized Controlled Trial. J Psychopharmacol 2016, 30, 1165–1180. [CrossRef]
- Johnson, M.W.; Griffiths, R.R. Potential Therapeutic Effects of Psilocybin. Neurotherapeutics 2017, 14, 734–740. [CrossRef]
- Lashgari, N.-A.; Khalaji, M.; Rana, P.; Badrabadi, F.; Rahnama, M.; Nasoori, H.; Momeni Roudsari, N.; Khosravi Nia, M.M.; Shafaroodi, H. Psychedelics in the Treatment of Neurologic and Psychiatric Disorders: Coincidence or a New Point of View. Mol Neurobiol 2025. [CrossRef]
- Saeger, H.N.; Olson, D.E. Psychedelic-Inspired Approaches for Treating Neurodegenerative Disorders. J Neurochem 2022, 162, 109–127. [CrossRef]
- Kato, K.; Kleinhenz, J.M.; Shin, Y.-J.; Coarfa, C.; Zarrabi, A.J.; Hecker, L. Psilocybin Treatment Extends Cellular Lifespan and Improves Survival of Aged Mice. npj Aging 2025, 11, 55. [CrossRef]
- Bradley, E.R.; Sakai, K.; Fernandes-Osterhold, G.; Szigeti, B.; Ludwig, C.; Ostrem, J.L.; Tanner, C.M.; Bock, M.A.; Llerena, K.; Finley, P.R.; et al. Psilocybin Therapy for Mood Dysfunction in Parkinson’s Disease: An Open-Label Pilot Trial. Neuropsychopharmacology 2025, 50, 1200–1209. [CrossRef]
- Fleury, V.; Tomkova, E.; Catalano Chiuvé, S.; Penzenstadler, L. Psilocybin-Assisted Psychotherapy for Parkinson’s Disease without Depression: A Case-Report. J Parkinsons Dis 2025, 15, 440–444. [CrossRef]
- Jevotovsky, D.S.; Chopra, H.; Wing, C.; Spotswood, C.J.; Castellanos, J. Refractory CRPS Pain Treated with Psilocybin: A Case Report. Clin Case Rep 2024, 12, e9421. [CrossRef]
- Askey, T.; Lasrado, R.; Maiarú, M.; Stephens, G.J. Psilocybin as a Novel Treatment for Chronic Pain. British Journal of Pharmacology n/a. [CrossRef]
- De Gregorio, D.; Aguilar-Valles, A.; Preller, K.H.; Heifets, B.D.; Hibicke, M.; Mitchell, J.; Gobbi, G. Hallucinogens in Mental Health: Preclinical and Clinical Studies on LSD, Psilocybin, MDMA, and Ketamine. J Neurosci 2021, 41, 891–900. [CrossRef]
- Lee, J.; Philip, K.; Wijeysundera, D.N.; Clarke, H.; Pritlove, C.; Katz, J.; Ritvo, P.; Goel, A.; Husain, M.I.; Ladha, K.S. Prospective Preference Assessment for the Psilocybin for Enhanced Analgesia in Chronic nEuropathic PAIN (PEACE-PAIN) Trial. Can J Pain 8, 2406285. [CrossRef]
- Holze, F.; Becker, A.M.; Kolaczynska, K.E.; Duthaler, U.; Liechti, M.E. Pharmacokinetics and Pharmacodynamics of Oral Psilocybin Administration in Healthy Participants. Clin Pharmacol Ther 2023, 113, 822–831. [CrossRef]
- Meshkat, S.; Al-Shamali, H.; Perivolaris, A.; Tullu, T.; Zeifman, R.J.; Zhang, Y.; Burback, L.; Winkler, O.; Greenshaw, A.; Husain, M.I.; et al. Pharmacokinetics of Psilocybin: A Systematic Review. Pharmaceutics 2025, 17, 411. [CrossRef]
- Otto, M.E.; van der Heijden, K.V.; Schoones, J.W.; van Esdonk, M.J.; Borghans, L.G.J.M.; Jacobs, G.E.; van Hasselt, J.G.C. Clinical Pharmacokinetics of Psilocin After Psilocybin Administration: A Systematic Review and Post-Hoc Analysis. Clin Pharmacokinet 2025, 64, 53–66. [CrossRef]
- Thomann, J.; Kolaczynska, K.E.; Stoeckmann, O.V.; Rudin, D.; Vizeli, P.; Hoener, M.C.; Pryce, C.R.; Vollenweider, F.X.; Liechti, M.E.; Duthaler, U. In Vitro and in Vivo Metabolism of Psilocybin’s Active Metabolite Psilocin. Front Pharmacol 2024, 15, 1391689. [CrossRef]
- Pho, H.; Amorim, M.R.; Qiu, Q.; Shin, M.-K.; Kim, L.J.; Anokye-Danso, F.; Jun, J.J.; Ahima, R.S.; Branco, L.G.S.; Kuhn, D.M.; et al. The Effect of Brain Serotonin Deficiency on Breathing Is Magnified by Age. Physiol Rep 2022, 10, e15245. [CrossRef]
- Karrer, T.M.; McLaughlin, C.L.; Guaglianone, C.P.; Samanez-Larkin, G.R. Reduced Serotonin Receptors and Transporters in Normal Aging Adults: A Meta-Analysis of PET and SPECT Imaging Studies. Neurobiol Aging 2019, 80, 1–10. [CrossRef]
- Fidalgo, S.; Yeoman, M.S. Age-Related Changes in Central Nervous System 5-Hydroxytryptamine Signalling and Its Potential Effects on the Regulation of Lifespan. In Biochemistry and Cell Biology of Ageing: Part III Biomedical Science; Harris, J.R., Korolchuk, V.I., Eds.; Subcellular Biochemistry; Springer International Publishing: Cham, 2023; Vol. 102, pp. 379–413 ISBN 978-3-031-21409-7.
- Moses-Kolko, E.L.; Price, J.C.; Shah, N.; Berga, S.; Sereika, S.M.; Fisher, P.M.; Coleman, R.; Becker, C.; Mason, N.S.; Loucks, T.; et al. Age, Sex, and Reproductive Hormone Effects on Brain Serotonin-1A and Serotonin-2A Receptor Binding in a Healthy Population. Neuropsychopharmacol 2011, 36, 2729–2740. [CrossRef]
- Kettner, H.; Roseman, L.; Gazzaley, A.; Carhart-Harris, R.L.; Pasquini, L. Effects of Psychedelics in Older Adults: A Prospective Cohort Study. Am J Geriatr Psychiatry 2024, 32, 1047–1059. [CrossRef]
- Friesen, P. Psychosis and Psychedelics: Historical Entanglements and Contemporary Contrasts. Transcult Psychiatry 2022, 59, 592–609. [CrossRef]
- Bremler, R.; Katati, N.; Shergill, P.; Erritzoe, D.; Carhart-Harris, R.L. Case Analysis of Long-Term Negative Psychological Responses to Psychedelics. Sci Rep 2023, 13, 15998. [CrossRef]
- von Rotz, R.; Schindowski, E.M.; Jungwirth, J.; Schuldt, A.; Rieser, N.M.; Zahoranszky, K.; Seifritz, E.; Nowak, A.; Nowak, P.; Jäncke, L.; et al. Single-Dose Psilocybin-Assisted Therapy in Major Depressive Disorder: A Placebo-Controlled, Double-Blind, Randomised Clinical Trial. EClinicalMedicine 2023, 56, 101809. [CrossRef]
- Hinkle, J.T.; Graziosi, M.; Nayak, S.M.; Yaden, D.B. Adverse Events in Studies of Classic Psychedelics: A Systematic Review and Meta-Analysis. JAMA Psychiatry 2024, 81, 1225–1235. [CrossRef]
- Johnston, C.B.; Mangini, M.; Grob, C.; Anderson, B. The Safety and Efficacy of Psychedelic-Assisted Therapies for Older Adults: Knowns and Unknowns. Am J Geriatr Psychiatry 2023, 31, 44–53. [CrossRef]
- Anderson, B.T.; Danforth, A.; Daroff, P.R.; Stauffer, C.; Ekman, E.; Agin-Liebes, G.; Trope, A.; Boden, M.T.; Dilley, P.J.; Mitchell, J.; et al. Psilocybin-Assisted Group Therapy for Demoralized Older Long-Term AIDS Survivor Men: An Open-Label Safety and Feasibility Pilot Study. EClinicalMedicine 2020, 27, 100538. [CrossRef]
- Perna, J.; Trop, J.; Palitsky, R.; Bosshardt, Z.; Vantine, H.; Dunlop, B.W.; Zarrabi, A.J. Prolonged Adverse Effects from Repeated Psilocybin Use in an Underground Psychedelic Therapy Training Program: A Case Report. BMC Psychiatry 2025, 25, 184. [CrossRef]
- Maia, M.C.B.; Albuquerque, L.L.M.; Oliveira, A.A.; Neto, M.R.F.R.; Abreu, L.M. de; Souza, F.G. de M. e; Bisol, L.W. Psychedelics in Older Adults: Difficulties of a Clear Therapeutic Evidence. The American Journal of Geriatric Psychiatry 2025, 33, 116–117. [CrossRef]
- Abuhasira, R.; Schleider, L.B.-L.; Mechoulam, R.; Novack, V. Epidemiological Characteristics, Safety and Efficacy of Medical Cannabis in the Elderly. Eur J Intern Med 2018, 49, 44–50. [CrossRef]
- Aday, J.S.; Heifets, B.D.; Pratscher, S.D.; Bradley, E.; Rosen, R.; Woolley, J.D. Great Expectations: Recommendations for Improving the Methodological Rigor of Psychedelic Clinical Trials. Psychopharmacology (Berl) 2022, 239, 1989–2010. [CrossRef]
- Schlag, A.K.; Aday, J.; Salam, I.; Neill, J.C.; Nutt, D.J. Adverse Effects of Psychedelics: From Anecdotes and Misinformation to Systematic Science. J Psychopharmacol 2022, 36, 258–272. [CrossRef]
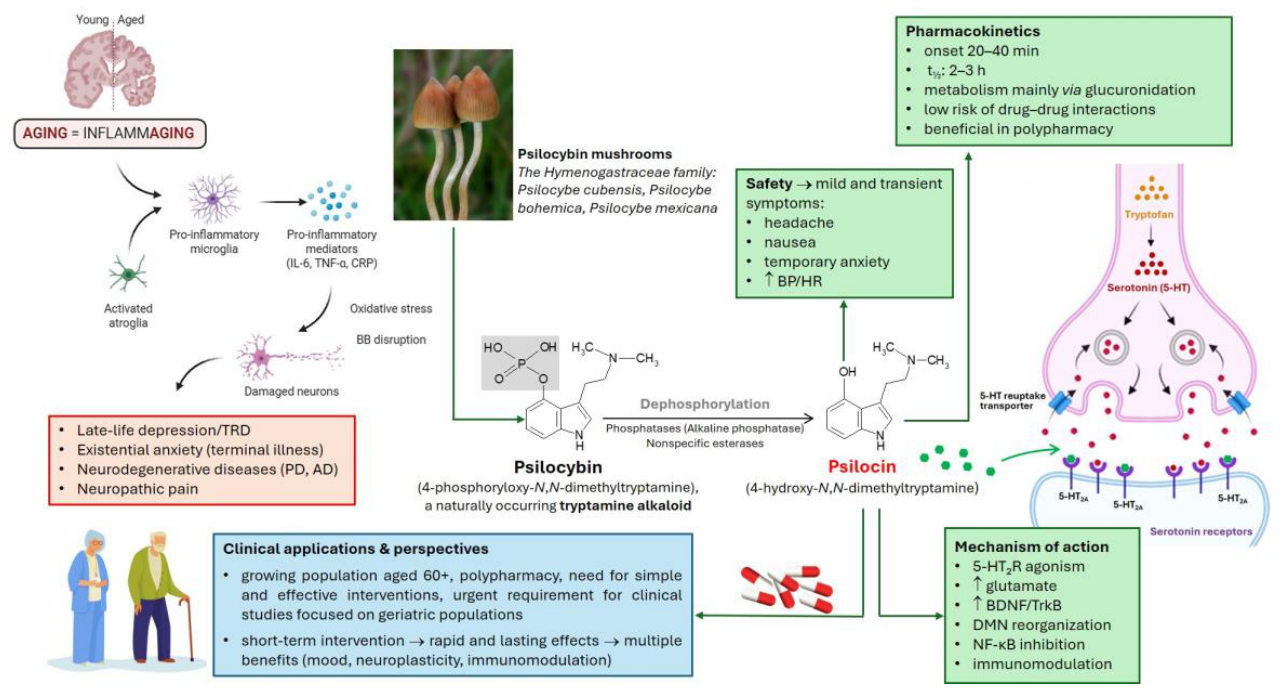
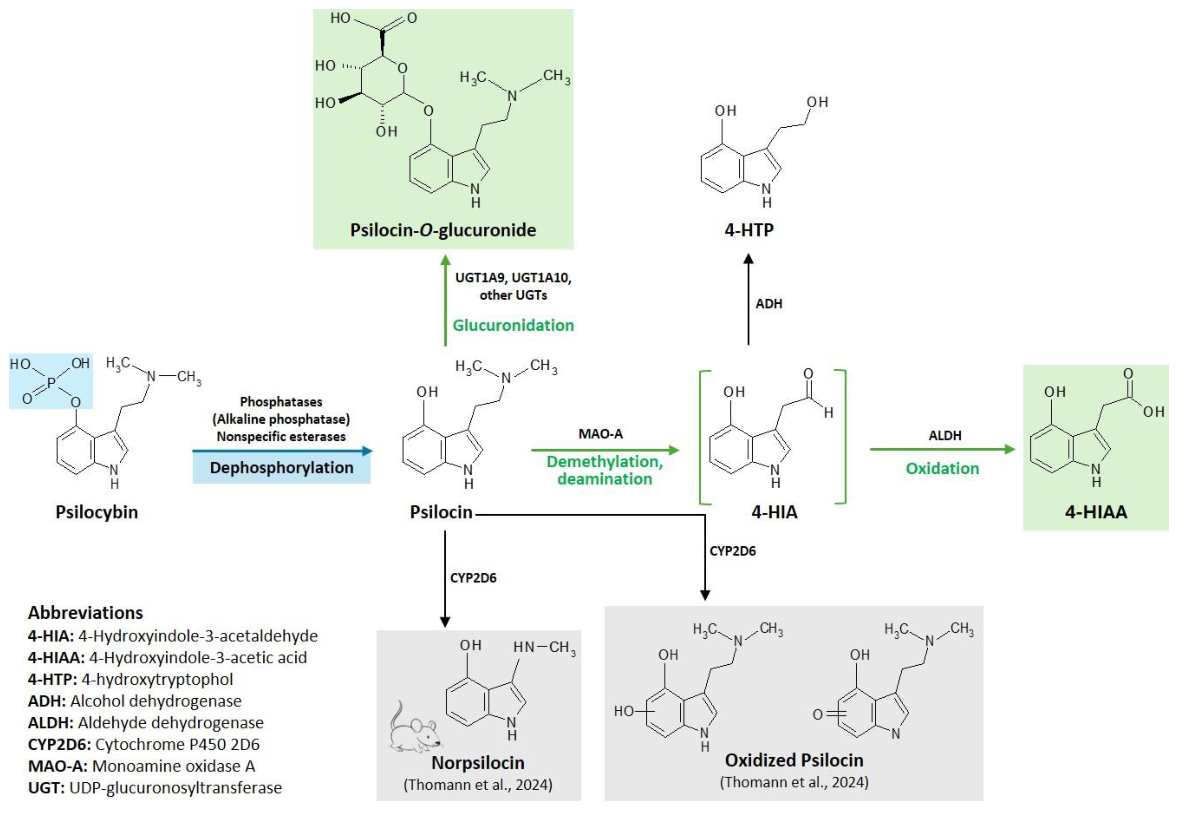
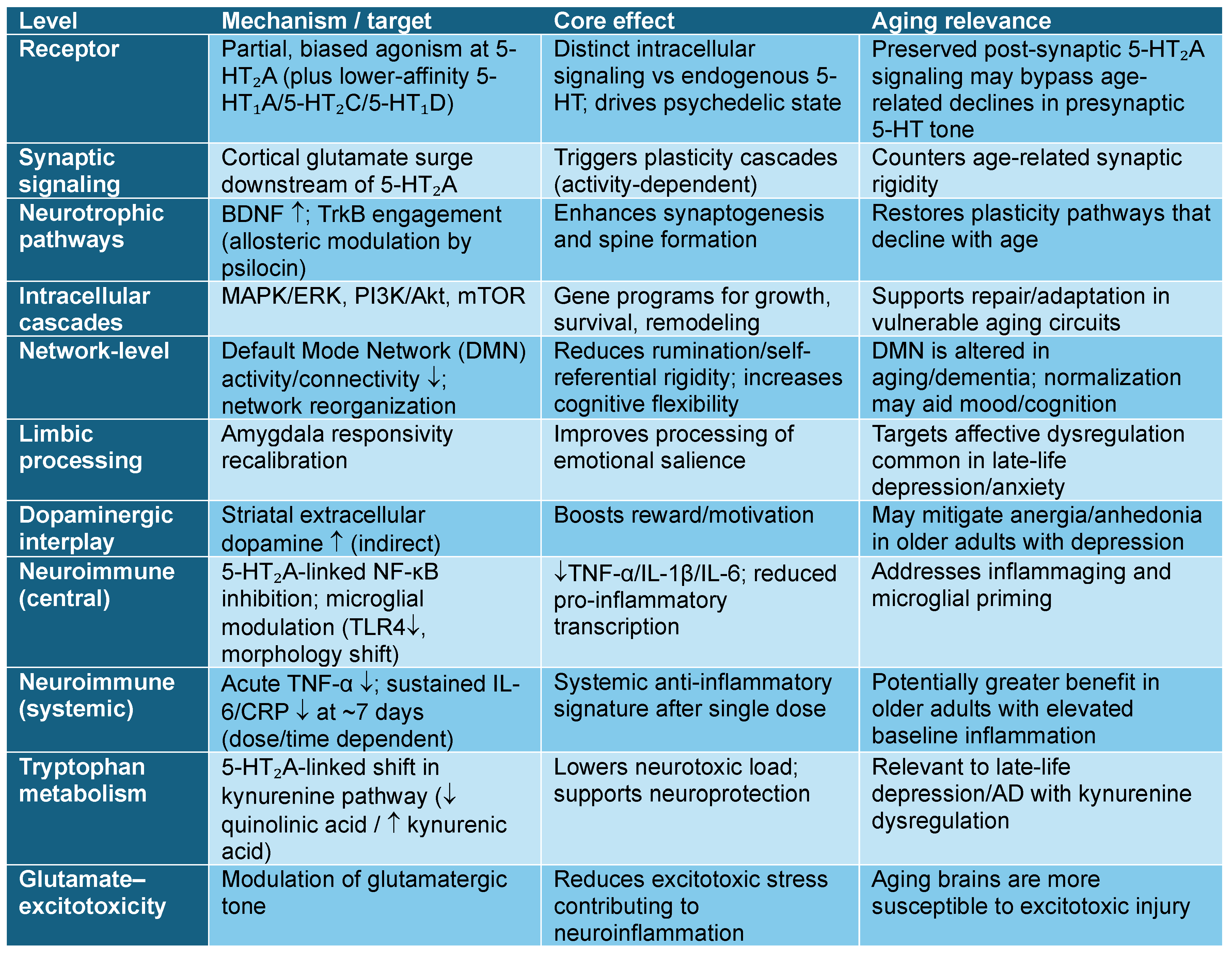 |
| AE category | Typical events reported in older adults |
|---|---|
| Cardiovascular | Transient increases in blood pressure and heart rate during dosing; occasional SBP >140–180 mmHg |
| Gastrointestinal | Nausea, GI upset; occasional vomiting |
| Neurologic / somatic | Headache (including “next-day headache”), dizziness, fatigue |
| Psychological (acute) | Transient anxiety, emotional discomfort, brief confusion; rare session-limited paranoid/psychotic-like content |
| Serious adverse events (SAEs) | None observed; no HPPD or persistent psychosis |
| Drug–drug interactions | SSRIs/MAOIs/TCAs may blunt or alter acute effects; serotonin toxicity risk theoretical in trials. Psilocin primarily glucuronidated via UGT1A9/UGT1A10; inducers (e.g., rifampicin) may lower exposure; inhibitors (e.g., probenecid, diclofenac) may raise exposure |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




