Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
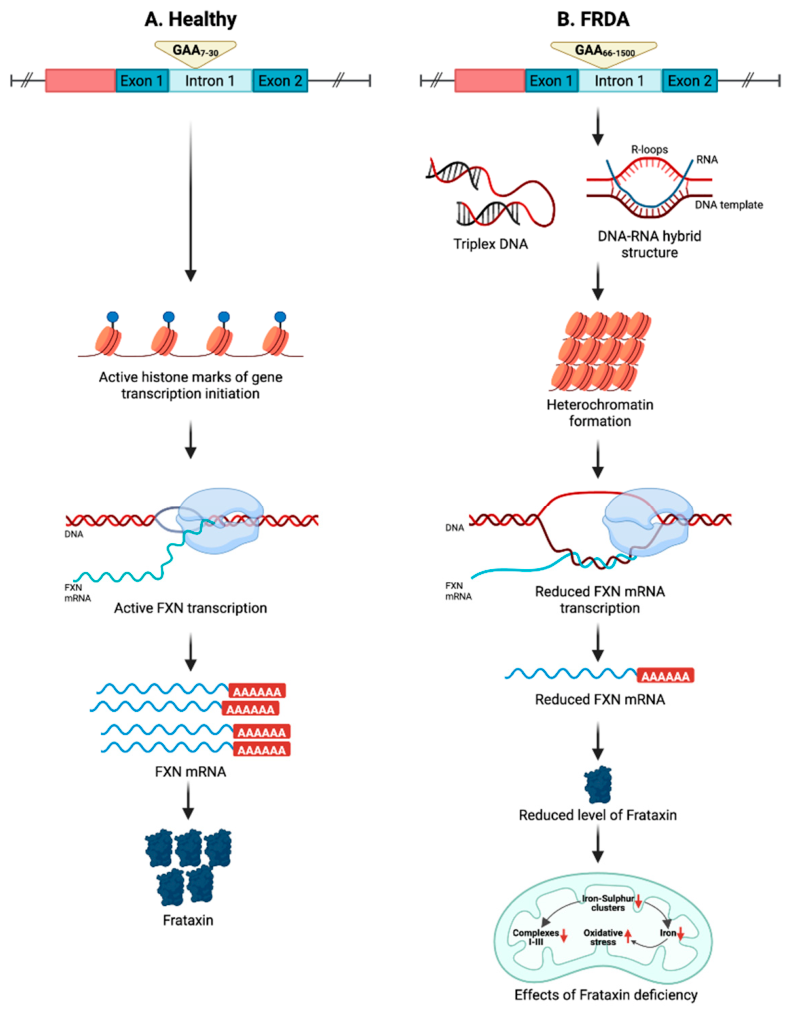
2. Genetics of FRDA
3. Frataxin Biology and Consequences of Its Deficiency
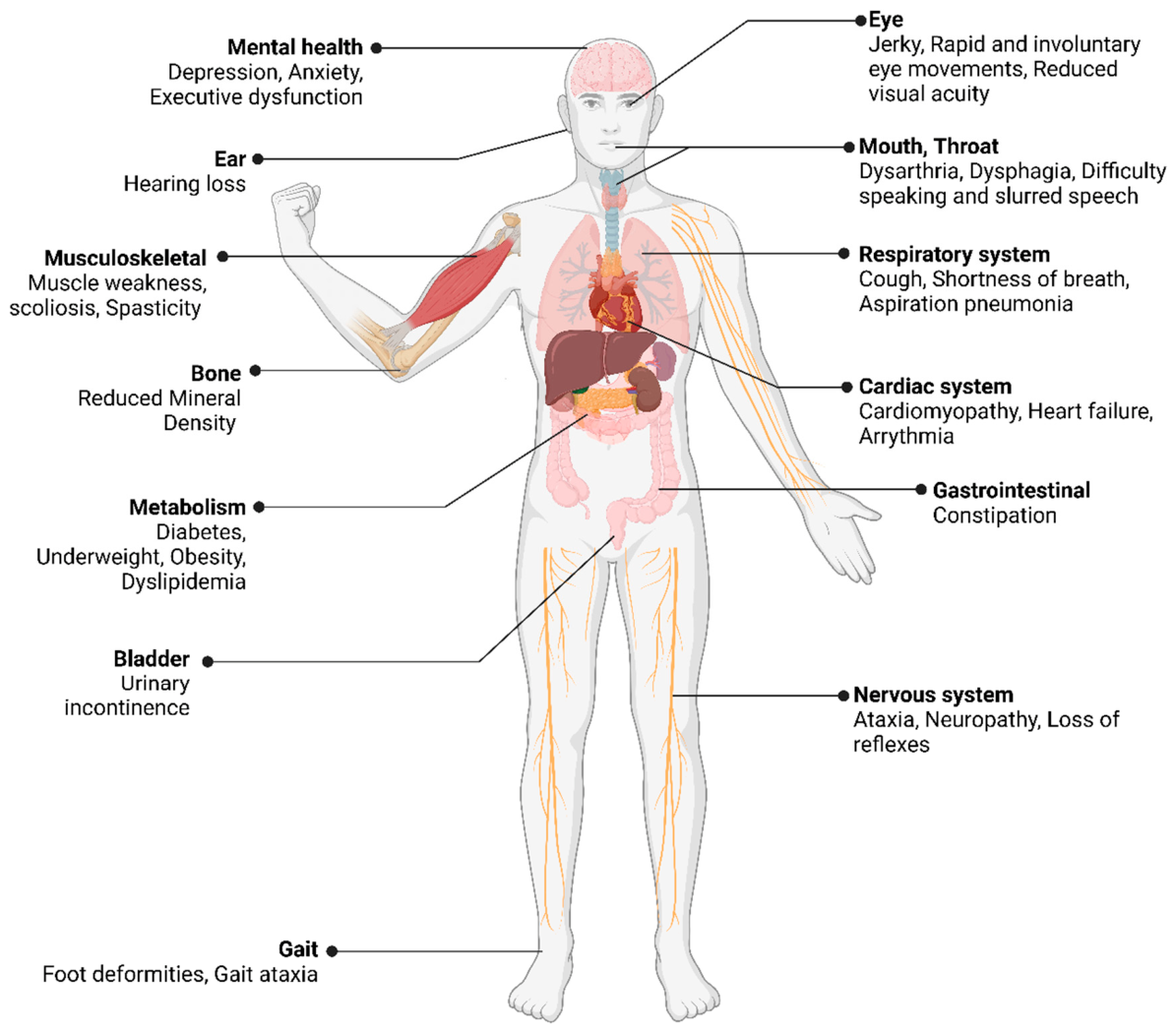
4. Multisystem Involvement of the Disease
4.1. Neurological and Psychiatric Symptoms
4.2. Musculoskeletal Symptoms
4.3. Cardiac Symptoms
4.4. Metabolic Symptoms
5. Cellular & Animal Models of FRDA
5.1. Cellular Models
5.2. Mouse Models
6. Bio Marks & Outcome Measures
7. Therapeutic Development
7.1. Approved Therapy
7.2. Therapies Targeting Oxidative Stress and Mitochondrial Dysfunction
7.3. Therapies to Increase Frataxin Expression and to Modulate Frataxin-Controlled Metabolic Pathways
7.4. Therapies Modulating Lipid Metabolism
7.5. Gene Therapies
7.6. Antisense Oligonucleotide (ASO)-Based Therapeutic Strategies
7.7. CRISPR/Cas-Based Therapeutic Strategies
8. Current Challenges
9. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Campuzano, V.; Montermini, L.; Molto, M.D.; Pianese, L.; Cossee, M.; Cavalcanti, F.; Monros, E.; Rodius, F.; Duclos, F.; Monticelli, A.; et al. Friedreich's ataxia: autosomal recessive disease caused by an intronic GAA triplet repeat expansion. Science 1996, 271, 1423-1427. [CrossRef]
- Pandolfo, M. Friedreich ataxia: Detection of GAA repeat expansions and frataxin point mutations. Methods Mol Med 2006, 126, 197-216. [CrossRef]
- Pandolfo, M. Friedreich Ataxia: An (Almost) 30-Year History After Gene Discovery. Neurol Genet 2025, 11, e200236. [CrossRef]
- Koeppen, A.H. Friedreich's ataxia: pathology, pathogenesis, and molecular genetics. J Neurol Sci 2011, 303, 1-12. [CrossRef]
- Montermini, L.; Rodius, F.; Pianese, L.; Moltò, M.D.; Cossée, M.; Campuzano, V.; Cavalcanti, F.; Monticelli, A.; Palau, F.; Gyapay, G.; et al. The Friedreich ataxia critical region spans a 150-kb interval on chromosome 9q13. Am J Hum Genet 1995, 57, 1061-1067.
- Galea, C.A.; Huq, A.; Lockhart, P.J.; Tai, G.; Corben, L.A.; Yiu, E.M.; Gurrin, L.C.; Lynch, D.R.; Gelbard, S.; Durr, A.; et al. Compound heterozygous FXN mutations and clinical outcome in friedreich ataxia. Ann Neurol 2016, 79, 485-495. [CrossRef]
- Shen, M.M.; Rummey, C.; Lynch, D.R. Phenotypic variation of FXN compound heterozygotes in a Friedreich ataxia cohort. Ann Clin Transl Neurol 2024, 11, 1110-1121. [CrossRef]
- Reetz, K.; Lischewski, S.A.; Dogan, I.; Didszun, C.; Pishnamaz, M.; Konrad, K.; Marx-Schutt, K.; Farmer, J.; Lynch, D.R.; Corben, L.A.; et al. Friedreich's ataxia-a rare multisystem disease. Lancet Neurol 2025, 24, 614-624. [CrossRef]
- Reetz, K.; Dogan, I.; Costa, A.S.; Dafotakis, M.; Fedosov, K.; Giunti, P.; Parkinson, M.H.; Sweeney, M.G.; Mariotti, C.; Panzeri, M.; et al. Biological and clinical characteristics of the European Friedreich's Ataxia Consortium for Translational Studies (EFACTS) cohort: a cross-sectional analysis of baseline data. Lancet Neurol 2015, 14, 174-182. [CrossRef]
- Reetz, K.; Dogan, I.; Hilgers, R.D.; Giunti, P.; Mariotti, C.; Durr, A.; Boesch, S.; Klopstock, T.; de Rivera, F.J.R.; Schols, L.; et al. Progression characteristics of the European Friedreich's Ataxia Consortium for Translational Studies (EFACTS): a 2 year cohort study. Lancet Neurol 2016, 15, 1346-1354. [CrossRef]
- Rummey, C.; Farmer, J.M.; Lynch, D.R. Predictors of loss of ambulation in Friedreich's ataxia. EClinicalMedicine 2020, 18, 100213. [CrossRef]
- Schulz, J.B.; Boesch, S.; Burk, K.; Durr, A.; Giunti, P.; Mariotti, C.; Pousset, F.; Schols, L.; Vankan, P.; Pandolfo, M. Diagnosis and treatment of Friedreich ataxia: a European perspective. Nat Rev Neurol 2009, 5, 222-234. [CrossRef]
- Scott, V.; Delatycki, M.B.; Tai, G.; Corben, L.A. New and Emerging Drug and Gene Therapies for Friedreich Ataxia. CNS Drugs 2024, 38, 791-805. [CrossRef]
- Montermini, L.; Andermann, E.; Labuda, M.; Richter, A.; Pandolfo, M.; Cavalcanti, F.; Pianese, L.; Iodice, L.; Farina, G.; Monticelli, A.; et al. The Friedreich ataxia GAA triplet repeat: premutation and normal alleles. Hum Mol Genet 1997, 6, 1261-1266. [CrossRef]
- Burnett, R.; Melander, C.; Puckett, J.W.; Son, L.S.; Wells, R.D.; Dervan, P.B.; Gottesfeld, J.M. DNA sequence-specific polyamides alleviate transcription inhibition associated with long GAA.TTC repeats in Friedreich's ataxia. Proc Natl Acad Sci U S A 2006, 103, 11497-11502. [CrossRef]
- Ohshima, K.; Montermini, L.; Wells, R.D.; Pandolfo, M. Inhibitory effects of expanded GAA.TTC triplet repeats from intron I of the Friedreich ataxia gene on transcription and replication in vivo. J Biol Chem 1998, 273, 14588-14595. [CrossRef]
- McDaniel, D.O.; Keats, B.; Vedanarayanan, V.V.; Subramony, S.H. Sequence variation in GAA repeat expansions may cause differential phenotype display in Friedreich's ataxia. Mov Disord 2001, 16, 1153-1158. [CrossRef]
- Monros, E.; Molto, M.D.; Martinez, F.; Canizares, J.; Blanca, J.; Vilchez, J.J.; Prieto, F.; de Frutos, R.; Palau, F. Phenotype correlation and intergenerational dynamics of the Friedreich ataxia GAA trinucleotide repeat. Am J Hum Genet 1997, 61, 101-110. [CrossRef]
- De Biase, I.; Rasmussen, A.; Endres, D.; Al-Mahdawi, S.; Monticelli, A.; Cocozza, S.; Pook, M.; Bidichandani, S.I. Progressive GAA expansions in dorsal root ganglia of Friedreich's ataxia patients. Ann Neurol 2007, 61, 55-60. [CrossRef]
- Long, A.; Napierala, J.S.; Polak, U.; Hauser, L.; Koeppen, A.H.; Lynch, D.R.; Napierala, M. Somatic instability of the expanded GAA repeats in Friedreich's ataxia. PLoS One 2017, 12, e0189990. [CrossRef]
- Sakamoto, N.; Chastain, P.D.; Parniewski, P.; Ohshima, K.; Pandolfo, M.; Griffith, J.D.; Wells, R.D. Sticky DNA: self-association properties of long GAA.TTC repeats in R.R.Y triplex structures from Friedreich's ataxia. Mol Cell 1999, 3, 465-475. [CrossRef]
- Groh, M.; Lufino, M.M.; Wade-Martins, R.; Gromak, N. R-loops associated with triplet repeat expansions promote gene silencing in Friedreich ataxia and fragile X syndrome. PLoS Genet 2014, 10, e1004318. [CrossRef]
- Soragni, E.; Miao, W.; Iudicello, M.; Jacoby, D.; De Mercanti, S.; Clerico, M.; Longo, F.; Piga, A.; Ku, S.; Campau, E.; et al. Epigenetic therapy for Friedreich ataxia. Ann Neurol 2014, 76, 489-508. [CrossRef]
- Al-Mahdawi, S.; Pinto, R.M.; Ismail, O.; Varshney, D.; Lymperi, S.; Sandi, C.; Trabzuni, D.; Pook, M. The Friedreich ataxia GAA repeat expansion mutation induces comparable epigenetic changes in human and transgenic mouse brain and heart tissues. Hum Mol Genet 2008, 17, 735-746. [CrossRef]
- Wang, Y.; Juranek, S.; Li, H.; Sheng, G.; Tuschl, T.; Patel, D.J. Structure of an argonaute silencing complex with a seed-containing guide DNA and target RNA duplex. Nature 2008, 456, 921-926. [CrossRef]
- Greene, E.; Mahishi, L.; Entezam, A.; Kumari, D.; Usdin, K. Repeat-induced epigenetic changes in intron 1 of the frataxin gene and its consequences in Friedreich ataxia. Nucleic Acids Res 2007, 35, 3383-3390. [CrossRef]
- Rodden, L.N.; Chutake, Y.K.; Gilliam, K.; Lam, C.; Soragni, E.; Hauser, L.; Gilliam, M.; Wiley, G.; Anderson, M.P.; Gottesfeld, J.M.; et al. Methylated and unmethylated epialleles support variegated epigenetic silencing in Friedreich ataxia. Hum Mol Genet 2021, 29, 3818-3829. [CrossRef]
- Chutake, Y.K.; Lam, C.; Costello, W.N.; Anderson, M.; Bidichandani, S.I. Epigenetic promoter silencing in Friedreich ataxia is dependent on repeat length. Ann Neurol 2014, 76, 522-528. [CrossRef]
- Delatycki, M.B.; Bidichandani, S.I. Friedreich ataxia- pathogenesis and implications for therapies. Neurobiol Dis 2019, 132, 104606. [CrossRef]
- Ercanbrack, W.S.; Ramirez, M.; Dungan, A.; Gaul, E.; Ercanbrack, S.J.; Wingert, R.A. Frataxin deficiency and the pathology of Friedreich's Ataxia across tissues. Tissue Barriers 2025, 13, 2462357. [CrossRef]
- Schmucker, S.; Argentini, M.; Carelle-Calmels, N.; Martelli, A.; Puccio, H. The in vivo mitochondrial two-step maturation of human frataxin. Hum Mol Genet 2008, 17, 3521-3531. [CrossRef]
- Castro, I.H.; Pignataro, M.F.; Sewell, K.E.; Espeche, L.D.; Herrera, M.G.; Noguera, M.E.; Dain, L.; Nadra, A.D.; Aran, M.; Smal, C.; et al. Frataxin Structure and Function. Subcell Biochem 2019, 93, 393-438. [CrossRef]
- Campuzano, V.; Montermini, L.; Lutz, Y.; Cova, L.; Hindelang, C.; Jiralerspong, S.; Trottier, Y.; Kish, S.J.; Faucheux, B.; Trouillas, P.; et al. Frataxin is reduced in Friedreich ataxia patients and is associated with mitochondrial membranes. Hum Mol Genet 1997, 6, 1771-1780. [CrossRef]
- Tsai, C.L.; Barondeau, D.P. Human frataxin is an allosteric switch that activates the Fe-S cluster biosynthetic complex. Biochemistry 2010, 49, 9132-9139. [CrossRef]
- Martelli, A.; Wattenhofer-Donze, M.; Schmucker, S.; Bouvet, S.; Reutenauer, L.; Puccio, H. Frataxin is essential for extramitochondrial Fe-S cluster proteins in mammalian tissues. Hum Mol Genet 2007, 16, 2651-2658. [CrossRef]
- Adamec, J.; Rusnak, F.; Owen, W.G.; Naylor, S.; Benson, L.M.; Gacy, A.M.; Isaya, G. Iron-dependent self-assembly of recombinant yeast frataxin: implications for Friedreich ataxia. Am J Hum Genet 2000, 67, 549-562. [CrossRef]
- Llorens, J.V.; Soriano, S.; Calap-Quintana, P.; Gonzalez-Cabo, P.; Molto, M.D. The Role of Iron in Friedreich's Ataxia: Insights From Studies in Human Tissues and Cellular and Animal Models. Front Neurosci 2019, 13, 75. [CrossRef]
- Yoon, T.; Cowan, J.A. Frataxin-mediated iron delivery to ferrochelatase in the final step of heme biosynthesis. J Biol Chem 2004, 279, 25943-25946. [CrossRef]
- Jasoliya, M.J.; McMackin, M.Z.; Henderson, C.K.; Perlman, S.L.; Cortopassi, G.A. Frataxin deficiency impairs mitochondrial biogenesis in cells, mice and humans. Hum Mol Genet 2017, 26, 2627-2633. [CrossRef]
- Das, D.; Patra, S.; Bridwell-Rabb, J.; Barondeau, D.P. Mechanism of frataxin "bypass" in human iron-sulfur cluster biosynthesis with implications for Friedreich's ataxia. J Biol Chem 2019, 294, 9276-9284. [CrossRef]
- Pandey, A.; Gordon, D.M.; Pain, J.; Stemmler, T.L.; Dancis, A.; Pain, D. Frataxin directly stimulates mitochondrial cysteine desulfurase by exposing substrate-binding sites, and a mutant Fe-S cluster scaffold protein with frataxin-bypassing ability acts similarly. J Biol Chem 2013, 288, 36773-36786. [CrossRef]
- Rotig, A.; de Lonlay, P.; Chretien, D.; Foury, F.; Koenig, M.; Sidi, D.; Munnich, A.; Rustin, P. Aconitase and mitochondrial iron-sulphur protein deficiency in Friedreich ataxia. Nat Genet 1997, 17, 215-217. [CrossRef]
- Martelli, A.; Schmucker, S.; Reutenauer, L.; Mathieu, J.R.R.; Peyssonnaux, C.; Karim, Z.; Puy, H.; Galy, B.; Hentze, M.W.; Puccio, H. Iron regulatory protein 1 sustains mitochondrial iron loading and function in frataxin deficiency. Cell Metab 2015, 21, 311-323. [CrossRef]
- Babcock, M.; de Silva, D.; Oaks, R.; Davis-Kaplan, S.; Jiralerspong, S.; Montermini, L.; Pandolfo, M.; Kaplan, J. Regulation of mitochondrial iron accumulation by Yfh1p, a putative homolog of frataxin. Science 1997, 276, 1709-1712. [CrossRef]
- Marmolino, D.; Manto, M.; Acquaviva, F.; Vergara, P.; Ravella, A.; Monticelli, A.; Pandolfo, M. PGC-1alpha down-regulation affects the antioxidant response in Friedreich's ataxia. PLoS One 2010, 5, e10025. [CrossRef]
- Chantrel-Groussard, K.; Geromel, V.; Puccio, H.; Koenig, M.; Munnich, A.; Rotig, A.; Rustin, P. Disabled early recruitment of antioxidant defenses in Friedreich's ataxia. Hum Mol Genet 2001, 10, 2061-2067. [CrossRef]
- Koeppen, A.H.; Ramirez, R.L.; Becker, A.B.; Bjork, S.T.; Levi, S.; Santambrogio, P.; Parsons, P.J.; Kruger, P.C.; Yang, K.X.; Feustel, P.J.; et al. The pathogenesis of cardiomyopathy in Friedreich ataxia. PLoS One 2015, 10, e0116396. [CrossRef]
- Pianese, L.; Turano, M.; Lo Casale, M.S.; De Biase, I.; Giacchetti, M.; Monticelli, A.; Criscuolo, C.; Filla, A.; Cocozza, S. Real time PCR quantification of frataxin mRNA in the peripheral blood leucocytes of Friedreich ataxia patients and carriers. J Neurol Neurosurg Psychiatry 2004, 75, 1061-1063. [CrossRef]
- Li, Y.; Lu, Y.; Polak, U.; Lin, K.; Shen, J.; Farmer, J.; Seyer, L.; Bhalla, A.D.; Rozwadowska, N.; Lynch, D.R.; et al. Expanded GAA repeats impede transcription elongation through the FXN gene and induce transcriptional silencing that is restricted to the FXN locus. Hum Mol Genet 2015, 24, 6932-6943. [CrossRef]
- Burk, K. Friedreich Ataxia: current status and future prospects. Cerebellum Ataxias 2017, 4, 4. [CrossRef]
- Reetz, K.; Dogan, I.; Hohenfeld, C.; Didszun, C.; Giunti, P.; Mariotti, C.; Durr, A.; Boesch, S.; Klopstock, T.; Rodriguez de Rivera Garrido, F.J.; et al. Nonataxia symptoms in Friedreich Ataxia: Report from the Registry of the European Friedreich's Ataxia Consortium for Translational Studies (EFACTS). Neurology 2018, 91, e917-e930. [CrossRef]
- Corben, L.A.; Blomfield, E.; Tai, G.; Bilal, H.; Harding, I.H.; Georgiou-Karistianis, N.; Delatycki, M.B.; Vogel, A.P. The Role of Verbal Fluency in the Cerebellar Cognitive Affective Syndrome Scale in Friedreich Ataxia. Cerebellum 2024, 23, 1975-1980. [CrossRef]
- Patel, M.; Isaacs, C.J.; Seyer, L.; Brigatti, K.; Gelbard, S.; Strawser, C.; Foerster, D.; Shinnick, J.; Schadt, K.; Yiu, E.M.; et al. Progression of Friedreich ataxia: quantitative characterization over 5 years. Ann Clin Transl Neurol 2016, 3, 684-694. [CrossRef]
- Harding, I.H.; Lynch, D.R.; Koeppen, A.H.; Pandolfo, M. Central Nervous System Therapeutic Targets in Friedreich Ataxia. Hum Gene Ther 2020, 31, 1226-1236. [CrossRef]
- Koeppen, A.H.; Becker, A.B.; Qian, J.; Gelman, B.B.; Mazurkiewicz, J.E. Friedreich Ataxia: Developmental Failure of the Dorsal Root Entry Zone. J Neuropathol Exp Neurol 2017, 76, 969-977. [CrossRef]
- Koeppen, A.H.; Becker, A.B.; Qian, J.; Feustel, P.J. Friedreich Ataxia: Hypoplasia of Spinal Cord and Dorsal Root Ganglia. J Neuropathol Exp Neurol 2017, 76, 101-108. [CrossRef]
- Koeppen, A.H.; Mazurkiewicz, J.E. Friedreich ataxia: neuropathology revised. J Neuropathol Exp Neurol 2013, 72, 78-90. [CrossRef]
- Koeppen, A.H.; Davis, A.N.; Morral, J.A. The cerebellar component of Friedreich's ataxia. Acta Neuropathol 2011, 122, 323-330. [CrossRef]
- Keage, M.J.; Delatycki, M.B.; Gupta, I.; Corben, L.A.; Vogel, A.P. Dysphagia in Friedreich Ataxia. Dysphagia 2017, 32, 626-635. [CrossRef]
- Lad, M.; Parkinson, M.H.; Rai, M.; Pandolfo, M.; Bogdanova-Mihaylova, P.; Walsh, R.A.; Murphy, S.; Emmanuel, A.; Panicker, J.; Giunti, P. Urinary, bowel and sexual symptoms in a cohort of patients with Friedreich's ataxia. Orphanet J Rare Dis 2017, 12, 158. [CrossRef]
- Durr, A.; Cossee, M.; Agid, Y.; Campuzano, V.; Mignard, C.; Penet, C.; Mandel, J.L.; Brice, A.; Koenig, M. Clinical and genetic abnormalities in patients with Friedreich's ataxia. N Engl J Med 1996, 335, 1169-1175. [CrossRef]
- Rance, G.; Fava, R.; Baldock, H.; Chong, A.; Barker, E.; Corben, L.; Delatycki, M.B. Speech perception ability in individuals with Friedreich ataxia. Brain 2008, 131, 2002-2012. [CrossRef]
- Seyer, L.A.; Galetta, K.; Wilson, J.; Sakai, R.; Perlman, S.; Mathews, K.; Wilmot, G.R.; Gomez, C.M.; Ravina, B.; Zesiewicz, T.; et al. Analysis of the visual system in Friedreich ataxia. J Neurol 2013, 260, 2362-2369. [CrossRef]
- Perez-Flores, J.; Hernandez-Torres, A.; Monton, F.; Nieto, A. Health-related quality of life and depressive symptoms in Friedreich ataxia. Qual Life Res 2020, 29, 413-420. [CrossRef]
- Nieto, A.; Hernandez-Torres, A.; Perez-Flores, J.; Monton, F. Depressive symptoms in Friedreich ataxia. Int J Clin Health Psychol 2018, 18, 18-26. [CrossRef]
- Indelicato, E.; Wanschitz, J.; Loscher, W.; Boesch, S. Skeletal Muscle Involvement in Friedreich Ataxia. Int J Mol Sci 2024, 25. [CrossRef]
- Nachbauer, W.; Boesch, S.; Reindl, M.; Eigentler, A.; Hufler, K.; Poewe, W.; Loscher, W.; Wanschitz, J. Skeletal muscle involvement in friedreich ataxia and potential effects of recombinant human erythropoietin administration on muscle regeneration and neovascularization. J Neuropathol Exp Neurol 2012, 71, 708-715. [CrossRef]
- Gallagher, C.L.; Waclawik, A.J.; Beinlich, B.R.; Harding, C.O.; Pauli, R.M.; Poirer, J.; Pandolfo, M.; Salamat, M.S. Friedreich's ataxia associated with mitochondrial myopathy: clinicopathologic report. J Child Neurol 2002, 17, 453-456. [CrossRef]
- Indelicato, E.; Kirchmair, A.; Amprosi, M.; Steixner, S.; Nachbauer, W.; Eigentler, A.; Wahl, N.; Apostolova, G.; Krogsdam, A.; Schneider, R.; et al. Skeletal muscle transcriptomics dissects the pathogenesis of Friedreich's ataxia. Hum Mol Genet 2023, 32, 2241-2250. [CrossRef]
- Indelicato, E.; Nachbauer, W.; Eigentler, A.; Amprosi, M.; Matteucci Gothe, R.; Giunti, P.; Mariotti, C.; Arpa, J.; Durr, A.; Klopstock, T.; et al. Onset features and time to diagnosis in Friedreich's Ataxia. Orphanet J Rare Dis 2020, 15, 198. [CrossRef]
- Rummey, C.; Flynn, J.M.; Corben, L.A.; Delatycki, M.B.; Wilmot, G.; Subramony, S.H.; Bushara, K.; Duquette, A.; Gomez, C.M.; Hoyle, J.C.; et al. Scoliosis in Friedreich's ataxia: longitudinal characterization in a large heterogeneous cohort. Ann Clin Transl Neurol 2021, 8, 1239-1250. [CrossRef]
- Milbrandt, T.A.; Kunes, J.R.; Karol, L.A. Friedreich's ataxia and scoliosis: the experience at two institutions. J Pediatr Orthop 2008, 28, 234-238. [CrossRef]
- Pousset, F.; Legrand, L.; Monin, M.L.; Ewenczyk, C.; Charles, P.; Komajda, M.; Brice, A.; Pandolfo, M.; Isnard, R.; Tezenas du Montcel, S.; et al. A 22-Year Follow-up Study of Long-term Cardiac Outcome and Predictors of Survival in Friedreich Ataxia. JAMA Neurol 2015, 72, 1334-1341. [CrossRef]
- Norrish, G.; Rance, T.; Montanes, E.; Field, E.; Brown, E.; Bhole, V.; Stuart, G.; Uzun, O.; McLeod, K.A.; Ilina, M.; et al. Friedreich's ataxia-associated childhood hypertrophic cardiomyopathy: a national cohort study. Arch Dis Child 2022, 107, 450-455. [CrossRef]
- Tsou, A.Y.; Paulsen, E.K.; Lagedrost, S.J.; Perlman, S.L.; Mathews, K.D.; Wilmot, G.R.; Ravina, B.; Koeppen, A.H.; Lynch, D.R. Mortality in Friedreich ataxia. J Neurol Sci 2011, 307, 46-49. [CrossRef]
- Weidemann, F.; Stork, S.; Liu, D.; Hu, K.; Herrmann, S.; Ertl, G.; Niemann, M. Cardiomyopathy of Friedreich ataxia. J Neurochem 2013, 126 Suppl 1, 88-93. [CrossRef]
- Peverill, R.E.; Romanelli, G.; Donelan, L.; Hassam, R.; Corben, L.A.; Delatycki, M.B. Left ventricular structural and functional changes in Friedreich ataxia - Relationship with body size, sex, age and genetic severity. PLoS One 2019, 14, e0225147. [CrossRef]
- Legrand, L.; Heuze, C.; Diallo, A.; Monin, M.L.; Ewenczyk, C.; Vicaut, E.; Montalescot, G.; Isnard, R.; Durr, A.; Pousset, F. Prognostic value of longitudinal strain and ejection fraction in Friedreich's ataxia. Int J Cardiol 2021, 330, 259-265. [CrossRef]
- Rojsajjakul, T.; Wu, L.; Grady, C.B.; Hwang, W.T.; Mesaros, C.; Lynch, D.R.; Blair, I.A. Liquid Chromatography-Mass Spectrometry Analysis of Frataxin Proteoforms in Whole Blood as Biomarkers of the Genetic Disease Friedreich's Ataxia. Anal Chem 2023, 95, 4251-4260. [CrossRef]
- Lynch, D.R.; Schadt, K.; Kichula, E.; McCormack, S.; Lin, K.Y. Friedreich Ataxia: Multidisciplinary Clinical Care. J Multidiscip Healthc 2021, 14, 1645-1658. [CrossRef]
- Yoda, M.; El-Banayosy, A.; Arusoglu, L.; Tendrich, G.; Minami, K.; Korfer, R. Permanent use of a ventricle assist device for dilated cardiomyopathy in Friedreich's ataxia. J Heart Lung Transplant 2006, 25, 251-252. [CrossRef]
- Sedlak, T.L.; Chandavimol, M.; Straatman, L. Cardiac transplantation: a temporary solution for Friedreich's ataxia-induced dilated cardiomyopathy. J Heart Lung Transplant 2004, 23, 1304-1306. [CrossRef]
- Legrand, L.; Weinsaft, J.W.; Pousset, F.; Ewenczyk, C.; Charles, P.; Hatem, S.; Heinzmann, A.; Biet, M.; Durr, A.; Redheuil, A. Characterizing cardiac phenotype in Friedreich's ataxia: The CARFA study. Arch Cardiovasc Dis 2022, 115, 17-28. [CrossRef]
- Isnard, R.; Kalotka, H.; Durr, A.; Cossee, M.; Schmitt, M.; Pousset, F.; Thomas, D.; Brice, A.; Koenig, M.; Komajda, M. Correlation between left ventricular hypertrophy and GAA trinucleotide repeat length in Friedreich's ataxia. Circulation 1997, 95, 2247-2249. [CrossRef]
- Friedman, L.S.; Schadt, K.A.; Regner, S.R.; Mark, G.E.; Lin, K.Y.; Sciascia, T.; St John Sutton, M.; Willi, S.; Lynch, D.R. Elevation of serum cardiac troponin I in a cross-sectional cohort of asymptomatic subjects with Friedreich ataxia. Int J Cardiol 2013, 167, 1622-1624. [CrossRef]
- Ivak, P.; Zumrova, A.; Netuka, I. Friedreich's ataxia and advanced heart failure: An ethical conundrum in decision-making. J Heart Lung Transplant 2016, 35, 1144-1145. [CrossRef]
- Coskun, K.O.; Popov, A.F.; Schmitto, J.D.; Coskun, S.T.; Brandes, I.; Zenker, D.; Melnychenko, I.; Schoendube, F.A.; Ruschewski, W. Feasibility of implantable cardioverter defibrillator treatment in five patients with familial Friedreich's ataxia--a case series. Artif Organs 2010, 34, 1061-1065. [CrossRef]
- Feingold, B.; Mahle, W.T.; Auerbach, S.; Clemens, P.; Domenighetti, A.A.; Jefferies, J.L.; Judge, D.P.; Lal, A.K.; Markham, L.W.; Parks, W.J.; et al. Management of Cardiac Involvement Associated With Neuromuscular Diseases: A Scientific Statement From the American Heart Association. Circulation 2017, 136, e200-e231. [CrossRef]
- Cnop, M.; Igoillo-Esteve, M.; Rai, M.; Begu, A.; Serroukh, Y.; Depondt, C.; Musuaya, A.E.; Marhfour, I.; Ladriere, L.; Moles Lopez, X.; et al. Central role and mechanisms of beta-cell dysfunction and death in friedreich ataxia-associated diabetes. Ann Neurol 2012, 72, 971-982. [CrossRef]
- Tamaroff, J.; DeDio, A.; Wade, K.; Wells, M.; Park, C.; Leavens, K.; Rummey, C.; Kelly, A.; Lynch, D.R.; McCormack, S.E. Friedreich's Ataxia related Diabetes: Epidemiology and management practices. Diabetes Res Clin Pract 2022, 186, 109828. [CrossRef]
- Chakraborty, P.P.; Ray, S.; Bhattacharjee, R.; Ghosh, S.; Mukhopadhyay, P.; Mukhopadhyay, S.; Chowdhury, S. First Presentation of Diabetes as Diabetic Ketoacidosis in a Case of Friedreich's Ataxia. Clin Diabetes 2015, 33, 84-86. [CrossRef]
- Pappa, A.; Hausler, M.G.; Veigel, A.; Tzamouranis, K.; Pfeifer, M.W.; Schmidt, A.; Bokamp, M.; Haberland, H.; Wagner, S.; Bruckel, J.; et al. Diabetes mellitus in Friedreich Ataxia: A case series of 19 patients from the German-Austrian diabetes mellitus registry. Diabetes Res Clin Pract 2018, 141, 229-236. [CrossRef]
- Lynch, D.R.; Willi, S.M.; Wilson, R.B.; Cotticelli, M.G.; Brigatti, K.W.; Deutsch, E.C.; Kucheruk, O.; Shrader, W.; Rioux, P.; Miller, G.; et al. A0001 in Friedreich ataxia: biochemical characterization and effects in a clinical trial. Mov Disord 2012, 27, 1026-1033. [CrossRef]
- Igoillo-Esteve, M.; Gurgul-Convey, E.; Hu, A.; Romagueira Bichara Dos Santos, L.; Abdulkarim, B.; Chintawar, S.; Marselli, L.; Marchetti, P.; Jonas, J.C.; Eizirik, D.L.; et al. Unveiling a common mechanism of apoptosis in beta-cells and neurons in Friedreich's ataxia. Hum Mol Genet 2015, 24, 2274-2286. [CrossRef]
- McCormick, A.; Farmer, J.; Perlman, S.; Delatycki, M.; Wilmot, G.; Matthews, K.; Yoon, G.; Hoyle, C.; Subramony, S.H.; Zesiewicz, T.; et al. Impact of diabetes in the Friedreich ataxia clinical outcome measures study. Ann Clin Transl Neurol 2017, 4, 622-631. [CrossRef]
- Patel, M.; McCormick, A.; Tamaroff, J.; Dunn, J.; Mitchell, J.A.; Lin, K.Y.; Farmer, J.; Rummey, C.; Perlman, S.L.; Delatycki, M.B.; et al. Body Mass Index and Height in the Friedreich Ataxia Clinical Outcome Measures Study. Neurol Genet 2021, 7, e638. [CrossRef]
- Wolny, S.; McFarland, R.; Chinnery, P.; Cheetham, T. Abnormal growth in mitochondrial disease. Acta Paediatr 2009, 98, 553-554. [CrossRef]
- Wortmann, S.B.; Zweers-van Essen, H.; Rodenburg, R.J.; van den Heuvel, L.P.; de Vries, M.C.; Rasmussen-Conrad, E.; Smeitink, J.A.; Morava, E. Mitochondrial energy production correlates with the age-related BMI. Pediatr Res 2009, 65, 103-108. [CrossRef]
- Pomplun, D.; Voigt, A.; Schulz, T.J.; Thierbach, R.; Pfeiffer, A.F.; Ristow, M. Reduced expression of mitochondrial frataxin in mice exacerbates diet-induced obesity. Proc Natl Acad Sci U S A 2007, 104, 6377-6381. [CrossRef]
- Shinnick, J.E.; Schadt, K.; Strawser, C.; Wilcox, N.; Perlman, S.L.; Wilmot, G.R.; Gomez, C.M.; Mathews, K.D.; Yoon, G.; Zesiewicz, T.; et al. Comorbid Medical Conditions in Friedreich Ataxia: Association With Inflammatory Bowel Disease and Growth Hormone Deficiency. J Child Neurol 2016, 31, 1161-1165. [CrossRef]
- Nachun, D.; Gao, F.; Isaacs, C.; Strawser, C.; Yang, Z.; Dokuru, D.; Van Berlo, V.; Sears, R.; Farmer, J.; Perlman, S.; et al. Peripheral blood gene expression reveals an inflammatory transcriptomic signature in Friedreich's ataxia patients. Hum Mol Genet 2018, 27, 2965-2977. [CrossRef]
- Sciarretta, F.; Zaccaria, F.; Ninni, A.; Ceci, V.; Turchi, R.; Apolloni, S.; Milani, M.; Della Valle, I.; Tiberi, M.; Chiurchiu, V.; et al. Frataxin deficiency shifts metabolism to promote reactive microglia via glucose catabolism. Life Sci Alliance 2024, 7. [CrossRef]
- Weber, D.R. Bone Health in Childhood Chronic Disease. Endocrinol Metab Clin North Am 2020, 49, 637-650. [CrossRef]
- Perdomini, M.; Hick, A.; Puccio, H.; Pook, M.A. Animal and cellular models of Friedreich ataxia. J Neurochem 2013, 126 Suppl 1, 65-79. [CrossRef]
- Calmels, N.; Schmucker, S.; Wattenhofer-Donze, M.; Martelli, A.; Vaucamps, N.; Reutenauer, L.; Messaddeq, N.; Bouton, C.; Koenig, M.; Puccio, H. The first cellular models based on frataxin missense mutations that reproduce spontaneously the defects associated with Friedreich ataxia. PLoS One 2009, 4, e6379. [CrossRef]
- Jauslin, M.L.; Wirth, T.; Meier, T.; Schoumacher, F. A cellular model for Friedreich Ataxia reveals small-molecule glutathione peroxidase mimetics as novel treatment strategy. Hum Mol Genet 2002, 11, 3055-3063. [CrossRef]
- Wong, A.; Yang, J.; Cavadini, P.; Gellera, C.; Lonnerdal, B.; Taroni, F.; Cortopassi, G. The Friedreich's ataxia mutation confers cellular sensitivity to oxidant stress which is rescued by chelators of iron and calcium and inhibitors of apoptosis. Hum Mol Genet 1999, 8, 425-430. [CrossRef]
- Jauslin, M.L.; Meier, T.; Smith, R.A.; Murphy, M.P. Mitochondria-targeted antioxidants protect Friedreich Ataxia fibroblasts from endogenous oxidative stress more effectively than untargeted antioxidants. FASEB J 2003, 17, 1972-1974. [CrossRef]
- Liu, J.; Verma, P.J.; Evans-Galea, M.V.; Delatycki, M.B.; Michalska, A.; Leung, J.; Crombie, D.; Sarsero, J.P.; Williamson, R.; Dottori, M.; et al. Generation of induced pluripotent stem cell lines from Friedreich ataxia patients. Stem Cell Rev Rep 2011, 7, 703-713. [CrossRef]
- Hick, A.; Wattenhofer-Donze, M.; Chintawar, S.; Tropel, P.; Simard, J.P.; Vaucamps, N.; Gall, D.; Lambot, L.; Andre, C.; Reutenauer, L.; et al. Neurons and cardiomyocytes derived from induced pluripotent stem cells as a model for mitochondrial defects in Friedreich's ataxia. Dis Model Mech 2013, 6, 608-621. [CrossRef]
- Khan, W.; Corben, L.A.; Bilal, H.; Vivash, L.; Delatycki, M.B.; Egan, G.F.; Harding, I.H. Neuroinflammation in the Cerebellum and Brainstem in Friedreich Ataxia: An [18F]-FEMPA PET Study. Mov Disord 2022, 37, 218-224. [CrossRef]
- Dionisi, C.; Chazalon, M.; Rai, M.; Keime, C.; Imbault, V.; Communi, D.; Puccio, H.; Schiffmann, S.N.; Pandolfo, M. Proprioceptors-enriched neuronal cultures from induced pluripotent stem cells from Friedreich ataxia patients show altered transcriptomic and proteomic profiles, abnormal neurite extension, and impaired electrophysiological properties. Brain Commun 2023, 5, fcad007. [CrossRef]
- Mazzara, P.G.; Muggeo, S.; Luoni, M.; Massimino, L.; Zaghi, M.; Valverde, P.T.; Brusco, S.; Marzi, M.J.; Palma, C.; Colasante, G.; et al. Frataxin gene editing rescues Friedreich's ataxia pathology in dorsal root ganglia organoid-derived sensory neurons. Nat Commun 2020, 11, 4178. [CrossRef]
- Correia, A.R.; Pastore, C.; Adinolfi, S.; Pastore, A.; Gomes, C.M. Dynamics, stability and iron-binding activity of frataxin clinical mutants. FEBS J 2008, 275, 3680-3690. [CrossRef]
- Delatycki, M.B.; Knight, M.; Koenig, M.; Cossee, M.; Williamson, R.; Forrest, S.M. G130V, a common FRDA point mutation, appears to have arisen from a common founder. Hum Genet 1999, 105, 343-346. [CrossRef]
- Puccio, H.; Simon, D.; Cossee, M.; Criqui-Filipe, P.; Tiziano, F.; Melki, J.; Hindelang, C.; Matyas, R.; Rustin, P.; Koenig, M. Mouse models for Friedreich ataxia exhibit cardiomyopathy, sensory nerve defect and Fe-S enzyme deficiency followed by intramitochondrial iron deposits. Nat Genet 2001, 27, 181-186. [CrossRef]
- Payne, R.M.; Pride, P.M.; Babbey, C.M. Cardiomyopathy of Friedreich's ataxia: use of mouse models to understand human disease and guide therapeutic development. Pediatr Cardiol 2011, 32, 366-378. [CrossRef]
- Simon, D.; Seznec, H.; Gansmuller, A.; Carelle, N.; Weber, P.; Metzger, D.; Rustin, P.; Koenig, M.; Puccio, H. Friedreich ataxia mouse models with progressive cerebellar and sensory ataxia reveal autophagic neurodegeneration in dorsal root ganglia. J Neurosci 2004, 24, 1987-1995. [CrossRef]
- Ristow, M.; Mulder, H.; Pomplun, D.; Schulz, T.J.; Muller-Schmehl, K.; Krause, A.; Fex, M.; Puccio, H.; Muller, J.; Isken, F.; et al. Frataxin deficiency in pancreatic islets causes diabetes due to loss of beta cell mass. J Clin Invest 2003, 112, 527-534. [CrossRef]
- Thierbach, R.; Schulz, T.J.; Isken, F.; Voigt, A.; Mietzner, B.; Drewes, G.; von Kleist-Retzow, J.C.; Wiesner, R.J.; Magnuson, M.A.; Puccio, H.; et al. Targeted disruption of hepatic frataxin expression causes impaired mitochondrial function, decreased life span and tumor growth in mice. Hum Mol Genet 2005, 14, 3857-3864. [CrossRef]
- Miranda, C.J.; Santos, M.M.; Ohshima, K.; Smith, J.; Li, L.; Bunting, M.; Cossee, M.; Koenig, M.; Sequeiros, J.; Kaplan, J.; et al. Frataxin knockin mouse. FEBS Lett 2002, 512, 291-297. [CrossRef]
- Al-Mahdawi, S.; Pinto, R.M.; Varshney, D.; Lawrence, L.; Lowrie, M.B.; Hughes, S.; Webster, Z.; Blake, J.; Cooper, J.M.; King, R.; et al. GAA repeat expansion mutation mouse models of Friedreich ataxia exhibit oxidative stress leading to progressive neuronal and cardiac pathology. Genomics 2006, 88, 580-590. [CrossRef]
- Sahdeo, S.; Scott, B.D.; McMackin, M.Z.; Jasoliya, M.; Brown, B.; Wulff, H.; Perlman, S.L.; Pook, M.A.; Cortopassi, G.A. Dyclonine rescues frataxin deficiency in animal models and buccal cells of patients with Friedreich's ataxia. Hum Mol Genet 2014, 23, 6848-6862. [CrossRef]
- Chutake, Y.K.; Costello, W.N.; Lam, C.C.; Parikh, A.C.; Hughes, T.T.; Michalopulos, M.G.; Pook, M.A.; Bidichandani, S.I. FXN Promoter Silencing in the Humanized Mouse Model of Friedreich Ataxia. PLoS One 2015, 10, e0138437. [CrossRef]
- Pook, M.A.; Al-Mahdawi, S.; Carroll, C.J.; Cossee, M.; Puccio, H.; Lawrence, L.; Clark, P.; Lowrie, M.B.; Bradley, J.L.; Cooper, J.M.; et al. Rescue of the Friedreich's ataxia knockout mouse by human YAC transgenesis. Neurogenetics 2001, 3, 185-193. [CrossRef]
- Guo, L.; Wang, Q.; Weng, L.; Hauser, L.A.; Strawser, C.J.; Mesaros, C.; Lynch, D.R.; Blair, I.A. Characterization of a new N-terminally acetylated extra-mitochondrial isoform of frataxin in human erythrocytes. Sci Rep 2018, 8, 17043. [CrossRef]
- Clay, A.; Obrochta, K.M.; Soon, R.K., Jr.; Russell, C.B.; Lynch, D.R. Neurofilament light chain as a potential biomarker of disease status in Friedreich ataxia. J Neurol 2020, 267, 2594-2598. [CrossRef]
- Marty, B.; Naeije, G.; Bourguignon, M.; Wens, V.; Jousmaki, V.; Lynch, D.R.; Gaetz, W.; Goldman, S.; Hari, R.; Pandolfo, M.; et al. Evidence for genetically determined degeneration of proprioceptive tracts in Friedreich ataxia. Neurology 2019, 93, e116-e124. [CrossRef]
- Santoro, L.; Perretti, A.; Lanzillo, B.; Coppola, G.; De Joanna, G.; Manganelli, F.; Cocozza, S.; De Michele, G.; Filla, A.; Caruso, G. Influence of GAA expansion size and disease duration on central nervous system impairment in Friedreich's ataxia: contribution to the understanding of the pathophysiology of the disease. Clin Neurophysiol 2000, 111, 1023-1030. [CrossRef]
- Dogan, I.; Romanzetti, S.; Didszun, C.; Mirzazade, S.; Timmann, D.; Saft, C.; Schols, L.; Synofzik, M.; Giordano, I.A.; Klockgether, T.; et al. Structural characteristics of the central nervous system in Friedreich ataxia: an in vivo spinal cord and brain MRI study. J Neurol Neurosurg Psychiatry 2019, 90, 615-617. [CrossRef]
- Dogan, I.; Tinnemann, E.; Romanzetti, S.; Mirzazade, S.; Costa, A.S.; Werner, C.J.; Heim, S.; Fedosov, K.; Schulz, S.; Timmann, D.; et al. Cognition in Friedreich's ataxia: a behavioral and multimodal imaging study. Ann Clin Transl Neurol 2016, 3, 572-587. [CrossRef]
- Oz, G.; Harding, I.H.; Krahe, J.; Reetz, K. MR imaging and spectroscopy in degenerative ataxias: toward multimodal, multisite, multistage monitoring of neurodegeneration. Curr Opin Neurol 2020, 33, 451-461. [CrossRef]
- Di Letto, P.; De Leonibus, C.; Palmieri, F.P.; Zanobio, M.; Scarpato, M.; Cetrangolo, V.; Rahman, S.I.; Selicorni, A.; Mariani, M.; D'Arrigo, S.; et al. Reanalysis of Undiagnosed Neurodevelopmental Disorder Cases: From RNU4-2 Variants to Clinical Phenotypes. Neurol Genet 2025, 11, e200312. [CrossRef]
- Rummey, C.; Harding, I.H.; Delatycki, M.B.; Tai, G.; Rezende, T.; Corben, L.A. Harmonizing results of ataxia rating scales: mFARS, SARA, and ICARS. Ann Clin Transl Neurol 2022, 9, 2041-2046. [CrossRef]
- Jacobi, H.; Rakowicz, M.; Rola, R.; Fancellu, R.; Mariotti, C.; Charles, P.; Durr, A.; Kuper, M.; Timmann, D.; Linnemann, C.; et al. Inventory of Non-Ataxia Signs (INAS): validation of a new clinical assessment instrument. Cerebellum 2013, 12, 418-428. [CrossRef]
- Reetz, K.; Dogan, I.; Hilgers, R.D.; Giunti, P.; Parkinson, M.H.; Mariotti, C.; Nanetti, L.; Durr, A.; Ewenczyk, C.; Boesch, S.; et al. Progression characteristics of the European Friedreich's Ataxia Consortium for Translational Studies (EFACTS): a 4-year cohort study. Lancet Neurol 2021, 20, 362-372. [CrossRef]
- Schmitz-Hubsch, T.; Giunti, P.; Stephenson, D.A.; Globas, C.; Baliko, L.; Sacca, F.; Mariotti, C.; Rakowicz, M.; Szymanski, S.; Infante, J.; et al. SCA Functional Index: a useful compound performance measure for spinocerebellar ataxia. Neurology 2008, 71, 486-492. [CrossRef]
- Lee, A. Omaveloxolone: First Approval. Drugs 2023, 83, 725-729. [CrossRef]
- Lynch, D.R.; Chin, M.P.; Delatycki, M.B.; Subramony, S.H.; Corti, M.; Hoyle, J.C.; Boesch, S.; Nachbauer, W.; Mariotti, C.; Mathews, K.D.; et al. Safety and Efficacy of Omaveloxolone in Friedreich Ataxia (MOXIe Study). Ann Neurol 2021, 89, 212-225. [CrossRef]
- Lynch, D.R.; Goldsberry, A.; Rummey, C.; Farmer, J.; Boesch, S.; Delatycki, M.B.; Giunti, P.; Hoyle, J.C.; Mariotti, C.; Mathews, K.D.; et al. Propensity matched comparison of omaveloxolone treatment to Friedreich ataxia natural history data. Ann Clin Transl Neurol 2024, 11, 4-16. [CrossRef]
- Gunther, K.; Profeta, V.; Keita, M.; Park, C.; Wells, M.; Sharma, S.; Schadt, K.; Lynch, D.R. Safety Monitoring of Omaveloxolone in Friedreich Ataxia: Results from One Year of Clinical Treatment. Neurol Ther 2025, 14, 1105-1114. [CrossRef]
- Shrader, W.D.; Amagata, A.; Barnes, A.; Enns, G.M.; Hinman, A.; Jankowski, O.; Kheifets, V.; Komatsuzaki, R.; Lee, E.; Mollard, P.; et al. alpha-Tocotrienol quinone modulates oxidative stress response and the biochemistry of aging. Bioorg Med Chem Lett 2011, 21, 3693-3698. [CrossRef]
- SOURCE PTC Therapeutics, I. PTC Therapeutics Announces Positive Results from Long-Term Treatment Studies and Updates on Regulatory Progress for Vatiquinone Friedreich Ataxia Program. Available online: https://ir.ptcbio.com/news-releases/news-release-details/ptc-therapeutics-announces-positive-results-long-term-treatment (accessed on.
- Venturelli, S.; Berger, A.; Bocker, A.; Busch, C.; Weiland, T.; Noor, S.; Leischner, C.; Schleicher, S.; Mayer, M.; Weiss, T.S.; et al. Resveratrol as a pan-HDAC inhibitor alters the acetylation status of histone [corrected] proteins in human-derived hepatoblastoma cells. PLoS One 2013, 8, e73097. [CrossRef]
- Yiu, E.M.; Tai, G.; Peverill, R.E.; Lee, K.J.; Croft, K.D.; Mori, T.A.; Scheiber-Mojdehkar, B.; Sturm, B.; Praschberger, M.; Vogel, A.P.; et al. An open-label trial in Friedreich ataxia suggests clinical benefit with high-dose resveratrol, without effect on frataxin levels. J Neurol 2015, 262, 1344-1353. [CrossRef]
- Edzeamey, F.J.; Ramchunder, Z.; Pourzand, C.; Anjomani Virmouni, S. Emerging antioxidant therapies in Friedreich's ataxia. Front Pharmacol 2024, 15, 1359618. [CrossRef]
- Pharaoh, G.; Kamat, V.; Kannan, S.; Stuppard, R.S.; Whitson, J.; Martin-Perez, M.; Qian, W.J.; MacCoss, M.J.; Villen, J.; Rabinovitch, P.; et al. The mitochondrially targeted peptide elamipretide (SS-31) improves ADP sensitivity in aged mitochondria by increasing uptake through the adenine nucleotide translocator (ANT). Geroscience 2023, 45, 3529-3548. [CrossRef]
- Pencina, K.M.; Lavu, S.; Dos Santos, M.; Beleva, Y.M.; Cheng, M.; Livingston, D.; Bhasin, S. MIB-626, an Oral Formulation of a Microcrystalline Unique Polymorph of beta-Nicotinamide Mononucleotide, Increases Circulating Nicotinamide Adenine Dinucleotide and its Metabolome in Middle-Aged and Older Adults. J Gerontol A Biol Sci Med Sci 2023, 78, 90-96. [CrossRef]
- Clayton, R.; Galas, T.; Scherer, N.; Farmer, J.; Ruiz, N.; Hamdani, M.; Schecter, D.; Bettoun, D. Safety, pharmacokinetics, and pharmacodynamics of nomlabofusp (CTI-1601) in Friedreich's ataxia. Ann Clin Transl Neurol 2024, 11, 540-553. [CrossRef]
- Larimar Therapeutics, I. Larimar Therapeutics Reports Positive Top-line Data from Phase 2 Dose Exploration Study from 25 mg and 50 mg Cohorts of Nomlabofusp in Patients with Friedreich’s Ataxia. Available online: https://investors.larimartx.com/news-releases/news-release-details/larimar-therapeutics-reports-positive-top-line-data-phase-2-dose (accessed on 04-11).
- Jasoliya, M.; Sacca, F.; Sahdeo, S.; Chedin, F.; Pane, C.; Brescia Morra, V.; Filla, A.; Pook, M.; Cortopassi, G. Dimethyl fumarate dosing in humans increases frataxin expression: A potential therapy for Friedreich's Ataxia. PLoS One 2019, 14, e0217776. [CrossRef]
- Pane, C.; Marra, A.M.; Aliberti, L.; Campanile, M.; Coscetta, F.; Crisci, G.; D'Assante, R.; Marsili, A.; Puorro, G.; Salzano, A.; et al. Rationale and protocol of a double-blind, randomized, placebo-controlled trial to test the efficacy, safety, and tolerability of dimethyl fumarate in Friedreich Ataxia (DMF-FA-201). Front Neurosci 2023, 17, 1260977. [CrossRef]
- Rodriguez-Pascau, L.; Britti, E.; Calap-Quintana, P.; Dong, Y.N.; Vergara, C.; Delaspre, F.; Medina-Carbonero, M.; Tamarit, J.; Pallardo, F.V.; Gonzalez-Cabo, P.; et al. PPAR gamma agonist leriglitazone improves frataxin-loss impairments in cellular and animal models of Friedreich Ataxia. Neurobiol Dis 2021, 148, 105162. [CrossRef]
- Pandolfo, M.; Reetz, K.; Darling, A.; Rodriguez de Rivera, F.J.; Henry, P.G.; Joers, J.; Lenglet, C.; Adanyeguh, I.; Deelchand, D.; Mochel, F.; et al. Efficacy and Safety of Leriglitazone in Patients With Friedreich Ataxia: A Phase 2 Double-Blind, Randomized Controlled Trial (FRAMES). Neurol Genet 2022, 8, e200034. [CrossRef]
- Khorkova, O.; Stahl, J.; Joji, A.; Volmar, C.H.; Wahlestedt, C. Amplifying gene expression with RNA-targeted therapeutics. Nat Rev Drug Discov 2023, 22, 539-561. [CrossRef]
- Design Therapeutics, I. Design Therapeutics Outlines Progress Across GeneTAC™ Platform and Announces Fourth Quarter and Full Year 2023 Financial Results. Available online: https://designtx.gcs-web.com/news-releases/news-release-details/design-therapeutics-outlines-progress-across-genetactm-platform/ (accessed on 04-11).
- Rufini, A.; Malisan, F.; Condo, I.; Testi, R. Drug Repositioning in Friedreich Ataxia. Front Neurosci 2022, 16, 814445. [CrossRef]
- Alfedi, G.; Luffarelli, R.; Condo, I.; Pedini, G.; Mannucci, L.; Massaro, D.S.; Benini, M.; Toschi, N.; Alaimo, G.; Panarello, L.; et al. Drug repositioning screening identifies etravirine as a potential therapeutic for friedreich's ataxia. Mov Disord 2019, 34, 323-334. [CrossRef]
- Paparella, G.; Straga, C.; Pesenti, N.; Dal Molin, V.; Martorel, G.A.; Merotto, V.; Genova, C.; Piazza, A.; Piccoli, G.; Panzeri, E.; et al. A Pilot Phase 2 Randomized Trial to Evaluate the Safety and Potential Efficacy of Etravirine in Friedreich Ataxia Patients. Children (Basel) 2024, 11. [CrossRef]
- Yang, X.; Zheng, Y.; Liu, L.; Huang, J.; Wang, F.; Zhang, J. Progress on the study of the anticancer effects of artesunate. Oncol Lett 2021, 22, 750. [CrossRef]
- Drecourt, A.; Babdor, J.; Dussiot, M.; Petit, F.; Goudin, N.; Garfa-Traore, M.; Habarou, F.; Bole-Feysot, C.; Nitschke, P.; Ottolenghi, C.; et al. Impaired Transferrin Receptor Palmitoylation and Recycling in Neurodegeneration with Brain Iron Accumulation. Am J Hum Genet 2018, 102, 266-277. [CrossRef]
- Petit, F.; Drecourt, A.; Dussiot, M.; Zangarelli, C.; Hermine, O.; Munnich, A.; Rotig, A. Defective palmitoylation of transferrin receptor triggers iron overload in Friedreich ataxia fibroblasts. Blood 2021, 137, 2090-2102. [CrossRef]
- Ba, Q.; Zhou, N.; Duan, J.; Chen, T.; Hao, M.; Yang, X.; Li, J.; Yin, J.; Chu, R.; Wang, H. Dihydroartemisinin exerts its anticancer activity through depleting cellular iron via transferrin receptor-1. PLoS One 2012, 7, e42703. [CrossRef]
- Herman, D.; Jenssen, K.; Burnett, R.; Soragni, E.; Perlman, S.L.; Gottesfeld, J.M. Histone deacetylase inhibitors reverse gene silencing in Friedreich's ataxia. Nat Chem Biol 2006, 2, 551-558. [CrossRef]
- Rai, M.; Soragni, E.; Jenssen, K.; Burnett, R.; Herman, D.; Coppola, G.; Geschwind, D.H.; Gottesfeld, J.M.; Pandolfo, M. HDAC inhibitors correct frataxin deficiency in a Friedreich ataxia mouse model. PLoS One 2008, 3, e1958. [CrossRef]
- Punga, T.; Buhler, M. Long intronic GAA repeats causing Friedreich ataxia impede transcription elongation. EMBO Mol Med 2010, 2, 120-129. [CrossRef]
- Codazzi, F.; Hu, A.; Rai, M.; Donatello, S.; Salerno Scarzella, F.; Mangiameli, E.; Pelizzoni, I.; Grohovaz, F.; Pandolfo, M. Friedreich ataxia-induced pluripotent stem cell-derived neurons show a cellular phenotype that is corrected by a benzamide HDAC inhibitor. Hum Mol Genet 2016, 25, 4847-4855. [CrossRef]
- Libri, V.; Yandim, C.; Athanasopoulos, S.; Loyse, N.; Natisvili, T.; Law, P.P.; Chan, P.K.; Mohammad, T.; Mauri, M.; Tam, K.T.; et al. Epigenetic and neurological effects and safety of high-dose nicotinamide in patients with Friedreich's ataxia: an exploratory, open-label, dose-escalation study. Lancet 2014, 384, 504-513. [CrossRef]
- Zesiewicz, T.; Heerinckx, F.; De Jager, R.; Omidvar, O.; Kilpatrick, M.; Shaw, J.; Shchepinov, M.S. Randomized, clinical trial of RT001: Early signals of efficacy in Friedreich's ataxia. Mov Disord 2018, 33, 1000-1005. [CrossRef]
- Lynch, D.R.; Mathews, K.D.; Perlman, S.; Zesiewicz, T.; Subramony, S.; Omidvar, O.; Vogel, A.P.; Krtolica, A.; Litterman, N.; van der Ploeg, L.; et al. Double blind trial of a deuterated form of linoleic acid (RT001) in Friedreich ataxia. J Neurol 2023, 270, 1615-1623. [CrossRef]
- Lexeo Therapeutics, I. Lexeo Therapeutics Provides Update on Cardiac Portfolio and Reports Third Quarter 2024 Financial Results. Available online: https://ir.lexeotx.com/news-releases/news-release-details/lexeo-therapeutics-provides-update-cardiac-portfolio-and-reports (accessed on 04-11).
- Chang, J.C.; Ryan, M.R.; Stark, M.C.; Liu, S.; Purushothaman, P.; Bolan, F.; Johnson, C.A.; Champe, M.; Meng, H.; Lawlor, M.W.; et al. AAV8 gene therapy reverses cardiac pathology and prevents early mortality in a mouse model of Friedreich's ataxia. Mol Ther Methods Clin Dev 2024, 32, 101193. [CrossRef]
- Li, L.; Matsui, M.; Corey, D.R. Activating frataxin expression by repeat-targeted nucleic acids. Nat Commun 2016, 7, 10606. [CrossRef]
- Li, L.; Shen, X.; Liu, Z.; Norrbom, M.; Prakash, T.P.; O'Reilly, D.; Sharma, V.K.; Damha, M.J.; Watts, J.K.; Rigo, F.; et al. Activation of Frataxin Protein Expression by Antisense Oligonucleotides Targeting the Mutant Expanded Repeat. Nucleic Acid Ther 2018, 28, 23-33. [CrossRef]
- Li, Y.; Li, J.; Wang, J.; Lynch, D.R.; Shen, X.; Corey, D.R.; Parekh, D.; Bhat, B.; Woo, C.; Cherry, J.J.; et al. Targeting 3' and 5' untranslated regions with antisense oligonucleotides to stabilize frataxin mRNA and increase protein expression. Nucleic Acids Res 2021, 49, 11560-11574. [CrossRef]
- Mozafari, N.; Milagres, S.; Umek, T.; Rocha, C.S.J.; Vargiu, C.M.; Freyberger, F.; Saher, O.; Napierala, M.; Napierala, J.S.; Blomberg, P.; et al. Anti-gene oligonucleotides targeting Friedreich's ataxia expanded GAA⋅TTC repeats increase Frataxin expression. Mol Ther Nucleic Acids 2025, 36, 102541. [CrossRef]
- Yameogo, P.; Aguilar, S.; Prakash, T.P.; Rigo, F.; Lynch, D.R.; Napierala, J.S.; Napierala, M. Antisense oligonucleotide therapy for patients with Friedreich's ataxia carrying the c.165+5G>C splicing mutation. Mol Ther Nucleic Acids 2025, 36, 102617. [CrossRef]
- Li, Y.; Li, J.; Wang, J.; Zhang, S.; Giles, K.; Prakash, T.P.; Rigo, F.; Napierala, J.S.; Napierala, M. Premature transcription termination at the expanded GAA repeats and aberrant alternative polyadenylation contributes to the Frataxin transcriptional deficit in Friedreich's ataxia. Hum Mol Genet 2022, 31, 3539-3557. [CrossRef]
- Shen, X.; Beasley, S.; Putman, J.N.; Li, Y.; Prakash, T.P.; Rigo, F.; Napierala, M.; Corey, D.R. Efficient electroporation of neuronal cells using synthetic oligonucleotides: identifying duplex RNA and antisense oligonucleotide activators of human frataxin expression. RNA 2019, 25, 1118-1129. [CrossRef]
- Shen, X.; Wong, J.; Prakash, T.P.; Rigo, F.; Li, Y.; Napierala, M.; Corey, D.R. Progress towards drug discovery for Friedreich's Ataxia: Identifying synthetic oligonucleotides that more potently activate expression of human frataxin protein. Bioorg Med Chem 2020, 28, 115472. [CrossRef]
- Rocca, C.J.; Rainaldi, J.N.; Sharma, J.; Shi, Y.; Haquang, J.H.; Luebeck, J.; Mali, P.; Cherqui, S. CRISPR-Cas9 Gene Editing of Hematopoietic Stem Cells from Patients with Friedreich's Ataxia. Mol Ther Methods Clin Dev 2020, 17, 1026-1036. [CrossRef]
- Mishra, P.; Sivakumar, A.; Johnson, A.; Pernaci, C.; Warden, A.S.; El-Hachem, L.R.; Hansen, E.; Badell-Grau, R.A.; Khare, V.; Ramirez, G.; et al. Gene editing improves endoplasmic reticulum-mitochondrial contacts and unfolded protein response in Friedreich's ataxia iPSC-derived neurons. Front Pharmacol 2024, 15, 1323491. [CrossRef]
- Li, Y.; Polak, U.; Bhalla, A.D.; Rozwadowska, N.; Butler, J.S.; Lynch, D.R.; Dent, S.Y.R.; Napierala, M. Excision of Expanded GAA Repeats Alleviates the Molecular Phenotype of Friedreich's Ataxia. Mol Ther 2015, 23, 1055-1065. [CrossRef]
- Matuszek, Z.; Arbab, M.; Kesavan, M.; Hsu, A.; Roy, J.C.L.; Zhao, J.; Yu, T.; Weisburd, B.; Newby, G.A.; Doherty, N.J.; et al. Base editing of trinucleotide repeats that cause Huntington's disease and Friedreich's ataxia reduces somatic repeat expansions in patient cells and in mice. Nat Genet 2025, 57, 1437-1451. [CrossRef]
- Rummey, C.; Corben, L.A.; Delatycki, M.; Wilmot, G.; Subramony, S.H.; Corti, M.; Bushara, K.; Duquette, A.; Gomez, C.; Hoyle, J.C.; et al. Natural History of Friedreich Ataxia: Heterogeneity of Neurologic Progression and Consequences for Clinical Trial Design. Neurology 2022, 99, e1499-e1510. [CrossRef]
- Corben, L.A.; Collins, V.; Milne, S.; Farmer, J.; Musheno, A.; Lynch, D.; Subramony, S.; Pandolfo, M.; Schulz, J.B.; Lin, K.; et al. Clinical management guidelines for Friedreich ataxia: best practice in rare diseases. Orphanet J Rare Dis 2022, 17, 415. [CrossRef]
| Category | Therapy | Mechanism of action | Clinical status/ Pre-clinical status/ Key findings |
| Approved Therapy | Omaveloxolone (Skyclarys™) | Activates Nrf2 → improves redox balance, reduces oxidative stress, supports mitochondrial function | FDA (2023), EU (2024); improves mFARS; slows progression (~55% over 3 years); mild side effects (↑ liver enzymes, cholesterol) |
| Oxidative Stress & Mitochondrial Dysfunction | Vatiquinone (PTC-743) | Inhibits 15-lipoxygenase → reduces oxidative stress & ferroptosis | Phase 3 (MOVE-FA); no primary endpoint met; improved fatigue & stability; ~50% progression delay (extension) |
| Resveratrol | Antioxidant; improves mitochondrial function | Phase 2 completed; mixed results; high dose limited by GI side effects | |
| Elamipretide (SS-31) | Binds cardiolipin → improves mitochondrial respiration & ATP | Ongoing Phase 1/2 (ELViS-FA); targeting vision & cardiac function | |
| MIB-626 (NMN) | Increases NAD⁺ → enhances mitochondrial metabolism | Phase 2 (open-label); improved bioenergetics; further trials planned | |
| Frataxin Restoration / Metabolic Modulation | Nomlabofusp (CTI-1601) | Frataxin replacement protein → mitochondrial delivery | Phase 2; ↑ frataxin (~⅓ normal); well tolerated; frequent dosing needed |
| Dimethyl fumarate (DMF) | ↑ FXN expression + activates Nrf2 | Ongoing Phase 2 trial | |
| Leriglitazone (MIN-102) | PPARγ agonist → improves mitochondrial function & metabolism | Phase 2; modest biomarker improvements; well tolerated | |
| DT-216 | GeneTAC™ → bypasses GAA transcription block | Phase 1; ↑ FXN mRNA; optimized version in development | |
| Etravirine | Repurposed HIV drug → increases frataxin (unclear mechanism) | Phase 2 pilot; transient neurological improvement | |
| Artesunate | Restores TfR1 trafficking → reduces iron overload | Early clinical investigation | |
| RGFP109 (HDAC inhibitor) | Reverses FXN gene silencing via histone acetylation | Phase 1; ↑ FXN mRNA; no proven clinical benefit yet | |
| Lipid Metabolism | RT001 | Stabilizes lipids → reduces peroxidation | Phase 2; no significant clinical benefit |
| Gene Therapy | LX2006 (AAVrh.10hFXN) | AAV-based FXN delivery to heart | Phase 1/2; ↑ cardiac frataxin (~5×); well tolerated |
| ASP2016 (AAV8-FXN) | Gene replacement targeting cardiomyocytes | Preclinical success; Phase 1 planned | |
| ASO-Based Therapies | Multiple ASO strategies | Target GAA repeat, stabilize mRNA, anti-gene DNA targeting, splice correction | Strong preclinical data; challenges: delivery, PK, off-target effects |
| CRISPR/Cas Approaches | Gene editing strategies | Repeat excision, base editing, ex vivo HSPC correction | Preclinical stage; restores FXN expression; promising but early |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




