Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Search Strategy
| MPS type | Syndrome name | Enzyme deficiency | OMIM |
|---|---|---|---|
| 1 (H, HS, S) | Hurler, Hurler-Scheie, Scheie | Dermatan sulphate, Heparan sulphate | 607014, 607015, 607016 |
| II | Hunter | Dermatan sulphate, Heparan sulphate | 309900 |
| III (A,B,C,D) | Sanfilippo | Heparan sulphate | 252900, 252920, 252930, 252940 |
| IVa | Morquio | Keratan Sulphate, Chondroitin sulphate | 253000 |
| VI | Maroteaux-Lamy | Dermatan sulphate, Chondroitin sulphate | 253200 |
| VII | Sly | Dermatan sulphate, Heparan sulphate, Chondroitin sulphate | 253220 |
| IX | Natowicz | Hyaluronidase | 601492 |
| X | MPS10 | Mutation in gene ARSK | 619698 |
| MPS Plus Syndrome | MPSPS | Mutation in the VPS33A gene | 617303 |
2.2. Relevant Section
2.3. Disease-Targeting Therapies
2.4. Surgical Interventions
2.5. Supportive Therapies
3. Insomnia
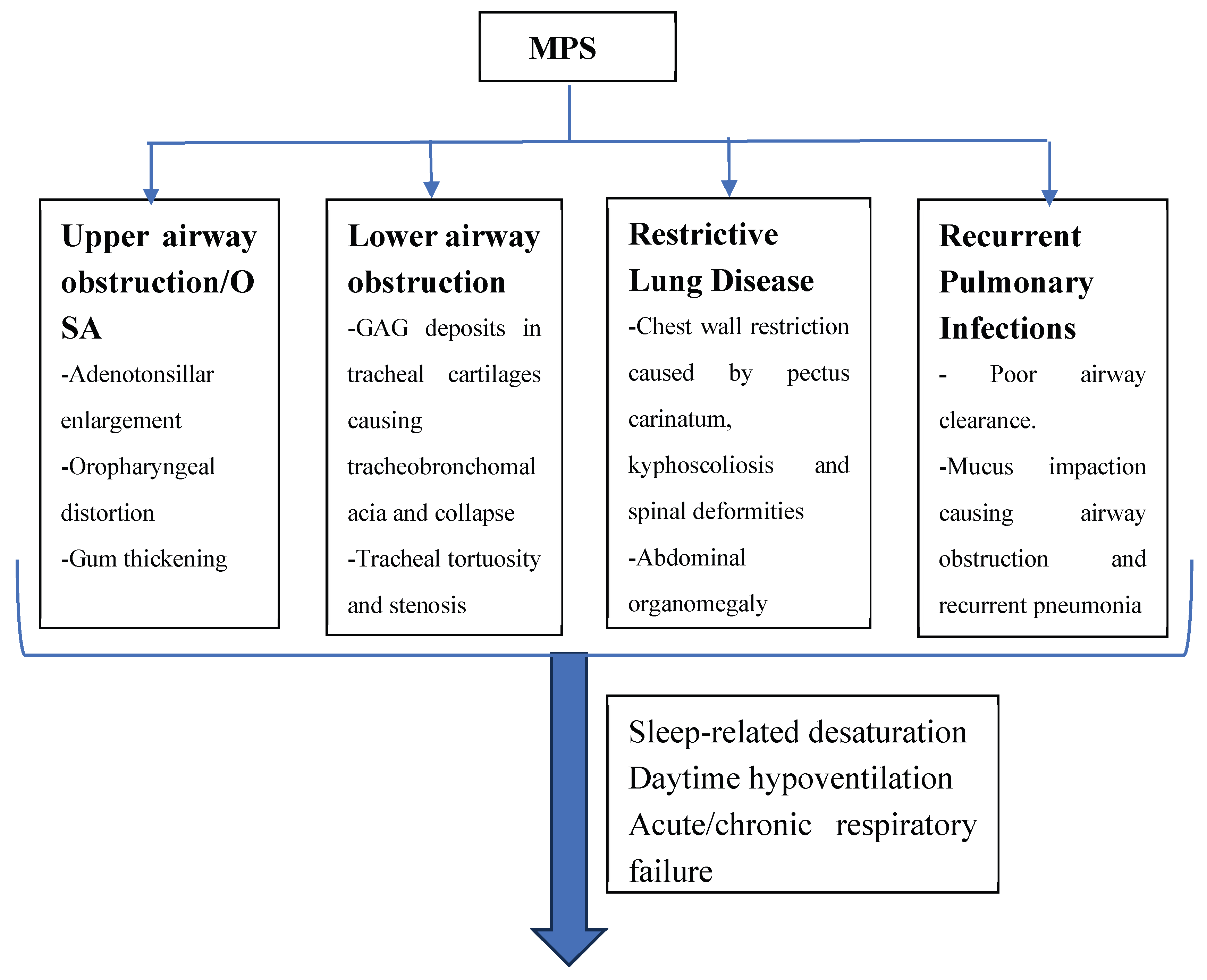
| Pulmonary Manifestation | Description | MPS types involved | Management Strategies |
|---|---|---|---|
| Upper airway obstruction | Adenotonsillar enlargement, oropharyngeal distortion, leading to sleep disordered breathing. Abnormal teeth protrusion, mouth opening, high Mallampati class affects airway. |
MPS 1, II, IVA, VI, VII Minimal involvement in MPS III |
Adenotonsillectomy, nasal steroid (more effective in young patients), tracheostomy (in recalcitrant cases), ERT, HSCT |
| Lower Airway Obstruction | GAG accumulation in tracheal cartilages causing collapse and tracheobronchomalacia. Tracheal stenosis and tortuosity MPS IVA has a peculiarity for severely narrowed airways due to tracheobronchial distortion. | MPS 1, 11, IVA Minimal involvement in MPS III and other subtypes of MPS IV |
PAP therapy particularly at night, use of tracheal and bronchial stents in very selected patients, inhaled steroids (airway inflammation), ERT/HSCT (less effective compared to upper airway obstruction), tracheostomy (for severe cases) |
| Restrictive Lung Disease | Chest wall restriction caused by pectus carinatum, broad spatulate ribs, reduced intercostal space, kyphoscoliosis and spinal deformities. Reduced lung volumes due to abdominal organomegaly. Atelectasis with resultant hypoxemia | MPS IV, VI Minimal involvement in MPS III |
Spinal and chest wall surgeries (if applicable), ERT, HSCT (less effective for chest wall disorders compared to upper airway obstruction). |
| Sleep Disordered Breathing | Obstructive sleep apnea (OSA) and sleep hypoxemia/hypoventilation | MPS I. II | Weight loss, adenotonsillectomy, CPAP or Bilevel PAP devices, supplemental oxygen (may worsen hypoventilation), ERT, HSCT. Tracheostomy (in severe cases) |
| Recurrent Pulmonary Infection | Poor airway clearance causing upper respiratory infections, bronchitis and pneumonia. Reduced immunity. | All MPS types | Appropriately recommended vaccinations, use of mucolytics and cough assistance devices and maneuvers. |
Importance of a Multidisciplinary Approach- The MPS Team
| MPS Team Member | Role in MPS Management |
|---|---|
| Pulmonologist | Provide respiratory care to MPS patients which may include positive airway pressure devices, airway clearance and cough assist devices, prescription of medications to address airway inflammation and help with secretion mobilization. Assessment and monitoring of lung function. |
| Otolaryngologist | Addressing complicated airway obstruction, performing adenotonsillectomy for sleep disordered breathing, airway planning prior to surgical interventions, management of recurrent otitis media and hearing issues |
| Anesthesiologist | Pre- and Peri-operative airway assessment plans and management |
| Neurologist | Evaluation and management of neurological complications, including and nerve impingement affecting overall mobility and functionality |
| Orthopedic/Spinal Surgeon | Management of skeletal deformities, chest wall and restrictive thoracic disorders, addressing spinal cord compression to improve functionality |
| Clinical Geneticist | Diagnosis of MPS type, understanding disease progression and implications for treatment |
| Cardiologist | Optimizing cardiac health to prevent complications particularly during vulnerable periods such as during anesthesia for surgical interventions |
4. Limitations
5. Conclusions
6. Gaps and Future Directions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
List of Abbreviations
| MPS | mucopolysaccharidosis |
| GAG | Glycosaminoglycans |
| ERT | enzyme replacement therapy |
| HSCT | hematopoietic stem cell therapy |
| OSA | obstructive sleep apnea |
| CPAP | continuous positive airway pressure |
| PAP | positive airway pressure |
| FEV1 | forced vital capacity in first second |
| FVC | forced vital capacity |
| 6 MWT | 6-minute walk test |
| BMT | bone marrow transplant |
| AT | adenotonsillectomy |
| LSD | lysosomal storage disease |
| AHI | apnea hypopnea index |
| SDB | sleep disordered breathing |
References
- Muenzer J. Overview of the mucopolysaccharidoses. Rheumatology (Oxford). Dec 2011;50 Suppl 5:v4-12. [CrossRef]
- Kubaski F, de Oliveira Poswar F, Michelin-Tirelli K, et al. Mucopolysaccharidosis Type I. Diagnostics (Basel). Mar 16 2020;10(3)doi:10.3390/diagnostics10030161.
- Beck M. Mucopolysaccharidosis Type II (Hunter Syndrome): clinical picture and treatment. Curr Pharm Biotechnol. Jun 2011;12(6):861-6. [CrossRef]
- Hunter C. A Rare Disease in Two Brothers. Proc R Soc Med. 1917;10(Sect Study Dis Child):104-16. [CrossRef]
- Żuber Z, Kieć-Wilk B, Kałużny Ł, Wierzba J, Tylki-Szymańska A. Diagnosis and Management of Mucopolysaccharidosis Type II (Hunter Syndrome) in Poland. Biomedicines. Jun 08 2023;11(6)doi:10.3390/biomedicines11061668.
- Northover H, Cowie RA, Wraith JE. Mucopolysaccharidosis type IVA (Morquio syndrome): a clinical review. J Inherit Metab Dis. 1996;19(3):357-65. [CrossRef]
- Beck M, Muenzer J, Scarpa M. Evaluation of disease severity in mucopolysaccharidoses. J Pediatr Rehabil Med. 2010;3(1):39-46. [CrossRef]
- Raymond GV, Pasquali M, Polgreen LE, et al. Elevated cerebral spinal fluid biomarkers in children with mucopolysaccharidosis I-H. Sci Rep. Dec 02 2016;6:38305. [CrossRef]
- MAROTEAUX P, LEVEQUE B, MARIE J, LAMY M. [A NEW DYSOSTOSIS WITH URINARY ELIMINATION OF CHONDROITIN SULFATE B]. Presse Med (1893). Sep 25 1963;71:1849-52.
- Natowicz MR, Short MP, Wang Y, et al. Clinical and biochemical manifestations of hyaluronidase deficiency. N Engl J Med. Oct 03 1996;335(14):1029-33. [CrossRef]
- Sjøgren P, Pedersen T, Steinmetz H. Mucopolysaccharidoses and anaesthetic risks. Acta Anaesthesiol Scand. Apr 1987;31(3):214-8. [CrossRef]
- Shih SL, Lee YJ, Lin SP, Sheu CY, Blickman JG. Airway changes in children with mucopolysaccharidoses. Acta Radiol. Jan 2002;43(1):40-3. [CrossRef]
- John A, Fagondes S, Schwartz I, et al. Sleep abnormalities in untreated patients with mucopolysaccharidosis type VI. Am J Med Genet A. Jul 2011;155A(7):1546-51. [CrossRef]
- Muhlebach MS, Wooten W, Muenzer J. Respiratory manifestations in mucopolysaccharidoses. Paediatr Respir Rev. Jun 2011;12(2):133-8. [CrossRef]
- Rutten M, Ciet P, van den Biggelaar R, et al. Severe tracheal and bronchial collapse in adults with type II mucopolysaccharidosis. Orphanet J Rare Dis. Apr 26 2016;11:50. [CrossRef]
- Gadepalli C, Stepien KM, Sharma R, Jovanovic A, Tol G, Bentley A. Airway Abnormalities in Adult Mucopolysaccharidosis and Development of Salford Mucopolysaccharidosis Airway Score. J Clin Med. Jul 24 2021;10(15)doi:10.3390/jcm10153275.
- Lee CL, Lee KS, Chuang CK, et al. Otorhinolaryngological Management in Taiwanese Patients with Mucopolysaccharidoses. Int J Med Sci. 2021;18(15):3373-3379. [CrossRef]
- Lee YH, Su CH, Lin CY, et al. Endoscopic and Image Analysis of the Airway in Patients with Mucopolysaccharidosis Type IVA. J Pers Med. Mar 09 2023;13(3)doi:10.3390/jpm13030494.
- Ufuk F. Tracheobronchial Involvement of Mucopolysaccharidosis Type VI. Pediatr Pulmonol. Feb 2025;60(2):e71007. [CrossRef]
- Dodsworth C, Burton BK. Increased incidence of neonatal respiratory distress in infants with mucopolysaccharidosis type II (MPS II, Hunter syndrome). Mol Genet Metab. Feb 2014;111(2):203-4. [CrossRef]
- Rapoport DM, Mitchell JJ. Pathophysiology, evaluation, and management of sleep disorders in the mucopolysaccharidoses. Mol Genet Metab. Dec 2017;122S:49-54. [CrossRef]
- Asir N, Al-Naimi AR. Polysomnographic Analysis of Sleep-Disordered Breathing In Children With Mucopolysaccharidoses in Qatar: A Retrospective Study. Cureus. Feb 2025;17(2):e79773. [CrossRef]
- Berger KI, Fagondes SC, Giugliani R, et al. Respiratory and sleep disorders in mucopolysaccharidosis. J Inherit Metab Dis. Mar 2013;36(2):201-10. [CrossRef]
- Wooten WI, Muenzer J, Vaughn BV, Muhlebach MS. Relationship of sleep to pulmonary function in mucopolysaccharidosis II. J Pediatr. Jun 2013;162(6):1210-5. [CrossRef]
- Pal AR, Langereis EJ, Saif MA, et al. Sleep disordered breathing in mucopolysaccharidosis I: a multivariate analysis of patient, therapeutic and metabolic correlators modifying long term clinical outcome. Orphanet J Rare Dis. Apr 10 2015;10:42. [CrossRef]
- Kasapkara Ç, Tümer L, Aslan AT, et al. Home sleep study characteristics in patients with mucopolysaccharidosis. Sleep Breath. Mar 2014;18(1):143-9. [CrossRef]
- Ademhan Tural D, Emiralioglu N, Dogru D, et al. Evaluation of sleep-disordered breathing and its relationship with respiratory parameters in children with mucopolysaccharidosis Type IVA and VI. Am J Med Genet A. Aug 2021;185(8):2306-2314. [CrossRef]
- MURRAY JF. Pulmonary disability in the Hurler syndrome (lipochondrodystrophy): a study of two cases. N Engl J Med. Aug 20 1959;261:378-82. [CrossRef]
- Mitchell J, Berger KI, Borgo A, et al. Unique medical issues in adult patients with mucopolysaccharidoses. Eur J Intern Med. Oct 2016;34:2-10. [CrossRef]
- Giugliani R, Federhen A, Muñoz Rojas MV, et al. [Enzyme replacement therapy for mucopolysaccharidoses I, II and VI: recommendations from a group of Brazilian F experts]. Rev Assoc Med Bras (1992). 2010;56(3):271-7. [CrossRef]
- Harmatz PR, Lampe C, Parini R, et al. Enzyme replacement therapy outcomes across the disease spectrum: Findings from the mucopolysaccharidosis VI Clinical Surveillance Program. J Inherit Metab Dis. May 2019;42(3):519-526. [CrossRef]
- Muenzer J. Early initiation of enzyme replacement therapy for the mucopolysaccharidoses. Mol Genet Metab. Feb 2014;111(2):63-72. [CrossRef]
- Politei J, Schenone A, Blanco M, Szlago M. [Mucopolysaccharidosis type VI: clinical aspects, diagnosis and treatment with enzyme replacement therapy]. Arch Argent Pediatr. Jun 2014;112(3):258-62. [CrossRef]
- Brunelli MJ, Atallah Á, da Silva EM. Enzyme replacement therapy with galsulfase for mucopolysaccharidosis type VI. Cochrane Database Syst Rev. Mar 04 2016;3:CD009806. [CrossRef]
- Wraith JE. The first 5 years of clinical experience with laronidase enzyme replacement therapy for mucopolysaccharidosis I. Expert Opin Pharmacother. Mar 2005;6(3):489-506. [CrossRef]
- Nan H, Park C, Maeng S. Mucopolysaccharidoses I and II: Brief Review of Therapeutic Options and Supportive/Palliative Therapies. Biomed Res Int. 2020;2020:2408402. [CrossRef]
- Lee CL, Chuang CK, Chiu HC, et al. Clinical Utility of Elosulfase Alfa in the Treatment of Morquio A Syndrome. Drug Des Devel Ther. 2022;16:143-154. [CrossRef]
- Parini R, Rigoldi M, Tedesco L, et al. Enzymatic replacement therapy for Hunter disease: Up to 9 years experience with 17 patients. Mol Genet Metab Rep. Jun 2015;3:65-74. [CrossRef]
- Horovitz DD, Magalhães TS, Acosta A, et al. Enzyme replacement therapy with galsulfase in 34 children younger than five years of age with MPS VI. Mol Genet Metab. May 2013;109(1):62-9. [CrossRef]
- Tomanin R, Zanetti A, D'Avanzo F, et al. Clinical efficacy of enzyme replacement therapy in paediatric Hunter patients, an independent study of 3.5 years. Orphanet J Rare Dis. Sep 18 2014;9:129. [CrossRef]
- Broomfield A, Sims J, Mercer J, et al. The evolution of pulmonary function in childhood onset Mucopolysaccharidosis type I. Mol Genet Metab. Feb 2021;132(2):94-99. [CrossRef]
- Bernardo Figueirêdo B, Reinaux C, Oliveira TG, Cavalcanti G, Fernandes J, Dornelas DE Andrade A. Respiratory muscular strength in children with mucopolysacaridosis: comparison with predictive equations. Minerva Pediatr (Torino). Aug 2024;76(4):507-516. [CrossRef]
- Rochester DF, Arora NS. Respiratory muscle failure. Med Clin North Am. May 1983;67(3):573-97. [CrossRef]
- Mitchell JJ, Burton BK, Bober MB, et al. Findings from the Morquio A Registry Study (MARS) after 6 years: Long-term outcomes of MPS IVA patients treated with elosulfase alfa. Mol Genet Metab. 2022;137(1-2):164-172. [CrossRef]
- Hendriksz CJ, Berger KI, Parini R, et al. Impact of long-term elosulfase alfa treatment on respiratory function in patients with Morquio A syndrome. J Inherit Metab Dis. Nov 2016;39(6):839-847. [CrossRef]
- Clarke LA, Wraith JE, Beck M, et al. Long-term efficacy and safety of laronidase in the treatment of mucopolysaccharidosis I. Pediatrics. Jan 2009;123(1):229-40. [CrossRef]
- Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A. GeneReviews. 1993.
- Burrow TA, Leslie ND. Review of the use of idursulfase in the treatment of mucopolysaccharidosis II. Biologics. Jun 2008;2(2):311-20.
- Allen JL. Treatment of respiratory system (not just lung!) abnormalities in Mucopolysaccharidosis I. J Pediatr. May 2004;144(5):561-2. [CrossRef]
- Kılıç M, Dursun A, Coşkun T, et al. Genotypic-phenotypic features and enzyme replacement therapy outcome in patients with mucopolysaccharidosis VI from Turkey. Am J Med Genet A. Nov 2017;173(11):2954-2967. [CrossRef]
- Muenzer J, Wraith JE, Beck M, et al. A phase II/III clinical study of enzyme replacement therapy with idursulfase in mucopolysaccharidosis II (Hunter syndrome). Genet Med. Aug 2006;8(8):465-73. [CrossRef]
- Wraith JE, Clarke LA, Beck M, et al. Enzyme replacement therapy for mucopolysaccharidosis I: a randomized, double-blinded, placebo-controlled, multinational study of recombinant human alpha-L-iduronidase (laronidase). J Pediatr. May 2004;144(5):581-8. [CrossRef]
- Burton BK, Harmatz PR, Horvathova V, et al. Long-term enzyme replacement therapy: Findings from the mucopolysaccharidosis VI clinical surveillance program after 15 years follow-up. Mol Genet Metab. Jul 2025;145(3):109135. [CrossRef]
- Lampe C, Bosserhoff AK, Burton BK, et al. Long-term experience with enzyme replacement therapy (ERT) in MPS II patients with a severe phenotype: an international case series. J Inherit Metab Dis. Sep 2014;37(5):823-9. [CrossRef]
- Tomatsu S, Alméciga-Díaz CJ, Montaño AM, et al. Therapies for the bone in mucopolysaccharidoses. Mol Genet Metab. Feb 2015;114(2):94-109. [CrossRef]
- Rowan DJ, Tomatsu S, Grubb JH, et al. Long circulating enzyme replacement therapy rescues bone pathology in mucopolysaccharidosis VII murine model. Mol Genet Metab. Sep 2012;107(1-2):161-72. [CrossRef]
- Okuyama T, Eto Y, Sakai N, et al. A Phase 2/3 Trial of Pabinafusp Alfa, IDS Fused with Anti-Human Transferrin Receptor Antibody, Targeting Neurodegeneration in MPS-II. Mol Ther. Feb 03 2021;29(2):671-679. [CrossRef]
- Clarke LA, Harmatz P, Fong EW. Implementing evidence-driven individualized treatment plans within Morquio A Syndrome. Mol Genet Metab. Feb 2016;117(2):217. [CrossRef]
- Kirkpatrick K, Ellwood J, Walker RW. Mucopolysaccharidosis type I (Hurler syndrome) and anesthesia: the impact of bone marrow transplantation, enzyme replacement therapy, and fiberoptic intubation on airway management. Paediatr Anaesth. Aug 2012;22(8):745-51. [CrossRef]
- Rodgers NJ, Kaizer AM, Miller WP, Rudser KD, Orchard PJ, Braunlin EA. Mortality after hematopoietic stem cell transplantation for severe mucopolysaccharidosis type I: the 30-year University of Minnesota experience. J Inherit Metab Dis. Mar 2017;40(2):271-280. [CrossRef]
- Selvanathan A, Ellaway C, Wilson C, Owens P, Shaw PJ, Bhattacharya K. Effectiveness of Early Hematopoietic Stem Cell Transplantation in Preventing Neurocognitive Decline in Mucopolysaccharidosis Type II: A Case Series. JIMD Rep. 2018;41:81-89. [CrossRef]
- Tomatsu S, Mackenzie WG, Theroux MC, et al. Current and emerging treatments and surgical interventions for Morquio A syndrome: a review. Res Rep Endocr Disord. Dec 2012;2012(2):65-77. [CrossRef]
- Chen J, Jiang H, Dong L, et al. [Treatment of 2 children with mucopolysaccharidosis by allogeneic hematopoietic stem cell transplantation]. Zhonghua Yi Xue Yi Chuan Xue Za Zhi. Dec 2008;25(6):675-7.
- Orchard PJ, Gupta AO, Eisengart JB, et al. Hematopoietic stem cell transplant for Hurler syndrome: does using bone marrow or umbilical cord blood make a difference? Blood Adv. Dec 13 2022;6(23):6023-6027. [CrossRef]
- Boelens JJ, Aldenhoven M, Purtill D, et al. Outcomes of transplantation using various hematopoietic cell sources in children with Hurler syndrome after myeloablative conditioning. Blood. May 09 2013;121(19):3981-7. [CrossRef]
- Walker R, Belani KG, Braunlin EA, et al. Anaesthesia and airway management in mucopolysaccharidosis. J Inherit Metab Dis. Mar 2013;36(2):211-9. [CrossRef]
- Frawley G, Fuenzalida D, Donath S, Yaplito-Lee J, Peters H. A retrospective audit of anesthetic techniques and complications in children with mucopolysaccharidoses. Paediatr Anaesth. Aug 2012;22(8):737-44. [CrossRef]
- Yabe H, Tanaka A, Chinen Y, et al. Hematopoietic stem cell transplantation for Morquio A syndrome. Mol Genet Metab. Feb 2016;117(2):84-94. [CrossRef]
- Theroux MC, Nerker T, Ditro C, Mackenzie WG. Anesthetic care and perioperative complications of children with Morquio syndrome. Paediatr Anaesth. Sep 2012;22(9):901-7. [CrossRef]
- Bidone J, Schuh RS, Farinon M, et al. Intra-articular nonviral gene therapy in mucopolysaccharidosis I mice. Int J Pharm. Sep 05 2018;548(1):151-158. [CrossRef]
- Schuh RS, Gonzalez EA, Tavares AMV, et al. Neonatal nonviral gene editing with the CRISPR/Cas9 system improves some cardiovascular, respiratory, and bone disease features of the mucopolysaccharidosis I phenotype in mice. Gene Ther. Feb 2020;27(1-2):74-84. [CrossRef]
- Wood SR, Bigger BW. Delivering gene therapy for mucopolysaccharide diseases. Front Mol Biosci. 2022;9:965089. [CrossRef]
- Wang D, Tai PWL, Gao G. Adeno-associated virus vector as a platform for gene therapy delivery. Nat Rev Drug Discov. May 2019;18(5):358-378. [CrossRef]
- Broeders M, Herrero-Hernandez P, Ernst MPT, van der Ploeg AT, Pijnappel WWMP. Sharpening the Molecular Scissors: Advances in Gene-Editing Technology. iScience. Jan 24 2020;23(1):100789. [CrossRef]
- Gentner B, Tucci F, Galimberti S, et al. Hematopoietic Stem- and Progenitor-Cell Gene Therapy for Hurler Syndrome. N Engl J Med. Nov 18 2021;385(21):1929-1940. [CrossRef]
- Mendelsohn NJ, Harmatz P, Bodamer O, et al. Importance of surgical history in diagnosing mucopolysaccharidosis type II (Hunter syndrome): data from the Hunter Outcome Survey. Genet Med. Dec 2010;12(12):816-22. [CrossRef]
- Keilmann A, Läßig AK, Pollak-Hainz A, Mann WJ, Beck M, Hainz M. Adenoids of patients with mucopolysaccharidoses demonstrate typical alterations. Int J Pediatr Otorhinolaryngol. Feb 2015;79(2):115-8. [CrossRef]
- Gönüldaş B, Yılmaz T, Sivri HS, et al. Mucopolysaccharidosis: Otolaryngologic findings, obstructive sleep apnea and accumulation of glucosaminoglycans in lymphatic tissue of the upper airway. Int J Pediatr Otorhinolaryngol. Jun 2014;78(6):944-9. [CrossRef]
- Galluzzi F, Garavello W. Tracheostomy in children with mucopolysaccharidosis: A systematic review. Int J Pediatr Otorhinolaryngol. Dec 2024;187:112167. [CrossRef]
- Bianchi PM, Gaini R, Vitale S. ENT and mucopolysaccharidoses. Ital J Pediatr. Nov 16 2018;44(Suppl 2):127. [CrossRef]
- Pizarro C, Davies RR, Theroux M, Spurrier EA, Averill LW, Tomatsu S. Surgical Reconstruction for Severe Tracheal Obstruction in Morquio A Syndrome. Ann Thorac Surg. Oct 2016;102(4):e329-31. [CrossRef]
- Hack H, Chapman I, Finucane K, Barber C. Anaesthesia and orphan disease: Tracheal reconstruction in two children with Morquio disease. Eur J Anaesthesiol. Feb 2020;37(2):132-137. [CrossRef]
- Kenth JJ, Thompson G, Fullwood C, Wilkinson S, Jones S, Bruce IA. The characterisation of pulmonary function in patients with mucopolysaccharidoses IVA: A longitudinal analysis. Mol Genet Metab Rep. Sep 2019;20:100487. [CrossRef]
- Karl R, Carola S, Regina E, Thomas N, Huber RM. Tracheobronchial stents in mucopolysaccharidosis. Int J Pediatr Otorhinolaryngol. Apr 2016;83:187-92. [CrossRef]
- Lao HC, Lin YC, Liang ML, et al. The Anesthetic Strategy for Patients with Mucopolysaccharidoses: A Retrospective Cohort Study. J Pers Med. Aug 21 2022;12(8)doi:10.3390/jpm12081343.
- Machado A, Rodrigues D, Ferreira A, Dias J, Santos P. Anaesthetic Management in Mucopolysaccharidoses Patients: Clinical Experience in a Tertiary Hospital. Cureus. Jul 2022;14(7):e27474. [CrossRef]
- Belani KG, Krivit W, Carpenter BL, et al. Children with mucopolysaccharidosis: perioperative care, morbidity, mortality, and new findings. J Pediatr Surg. Mar 1993;28(3):403-8; discussion 408-10. [CrossRef]
- Moretto A, Bosatra MG, Marchesini L, Tesoro S. Anesthesiological risks in mucopolysaccharidoses. Ital J Pediatr. Nov 16 2018;44(Suppl 2):116. [CrossRef]
- Tong BK, Cistulli PA, Ledger S, Chan ASL. A novel three-dimensional-printed customized nasal mask for improving CPAP adherence and satisfaction for the treatment of obstructive sleep apnea. J Clin Sleep Med. Jan 01 2025;21(1):9-16. [CrossRef]
- Fraser J, Wraith JE, Delatycki MB. Sleep disturbance in mucopolysaccharidosis type III (Sanfilippo syndrome): a survey of managing clinicians. Clin Genet. Nov 2002;62(5):418-21. [CrossRef]
- Fraser J, Gason AA, Wraith JE, Delatycki MB. Sleep disturbance in Sanfilippo syndrome: a parental questionnaire study. Arch Dis Child. Dec 2005;90(12):1239-42. [CrossRef]
- Guerrero JM, Pozo D, Diaz-Rodriguez JL, Martinez-Cruz F, Vela-Campos F. Impairment of the melatonin rhythm in children with Sanfilippo syndrome. J Pineal Res. Mar 2006;40(2):192-3. [CrossRef]
- Escolar ML, Jones SA, Shapiro EG, Horovitz DDG, Lampe C, Amartino H. Practical management of behavioral problems in mucopolysaccharidoses disorders. Mol Genet Metab. Dec 2017;122S:35-40. [CrossRef]
- Stepien KM, Bentley A, Chen C, et al. Non-cardiac Manifestations in Adult Patients With Mucopolysaccharidosis. Front Cardiovasc Med. 2022;9:839391. [CrossRef]
- Ramos BCF, Aranda CS, Cardona RSB, et al. Vaccination strategies for people living with inborn errors of metabolism in Brazil. J Pediatr (Rio J). 2023;99 Suppl 1(Suppl 1):S70-S80. [CrossRef]
- Gnasso R, Corrado B, Iommazzo I, et al. Assessment, pharmacological therapy and rehabilitation management of musculoskeletal pain in children with mucopolysaccharidoses: a scoping review. Orphanet J Rare Dis. Jul 08 2022;17(1):255. [CrossRef]
- Nelson J, Thomas PS. Clinical findings in 12 patients with MPS IV A (Morquio's disease). Further evidence for heterogeneity. Part III: Odontoid dysplasia. Clin Genet. Feb 1988;33(2):126-30. [CrossRef]
- Pantel T, Lindschau M, Luebke AM, et al. Spinal cord compression in patients with mucopolysaccharidosis. Eur Spine J. Jul 2022;31(7):1693-1699. [CrossRef]
- Akyol MU, Alden TD, Amartino H, et al. Recommendations for the management of MPS VI: systematic evidence- and consensus-based guidance. Orphanet J Rare Dis. May 29 2019;14(1):118. [CrossRef]
- Solano VM, Mandujano CYC, Avila-Rejon CA, Espin VH, Montaño HPQ. Disease burden, management patterns and multidisciplinary clinical approaches for patients with MPS IVA and VI in selected Latin American Countries. Mol Genet Metab Rep. Sep 2021;28:100769. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




