Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
Patients and Study Design
Data Collection
Pathological and Genetic Analyses
Treatment and Response Assessment
Statistical Analysis
Results
Patient Characteristics
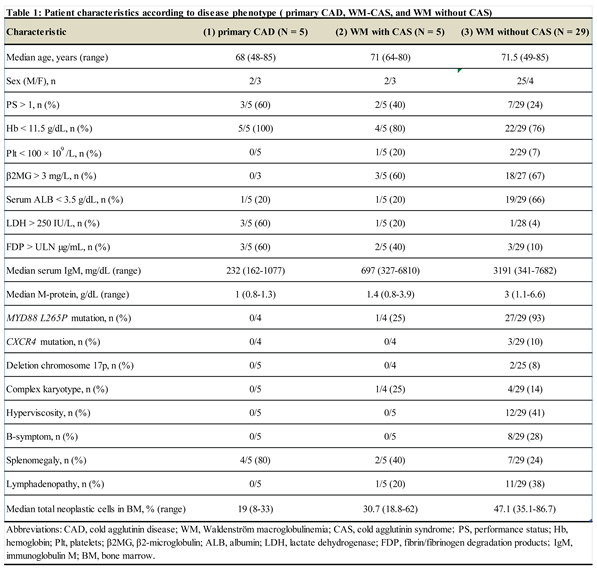 |
Individual Characteristics of CAH
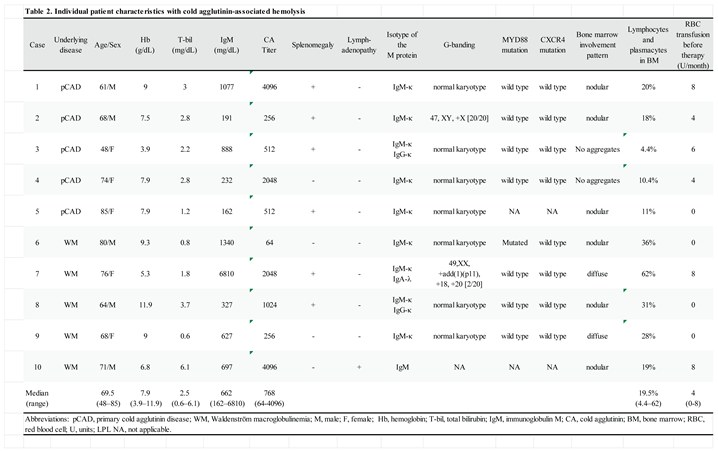 |
Treatment Regimens
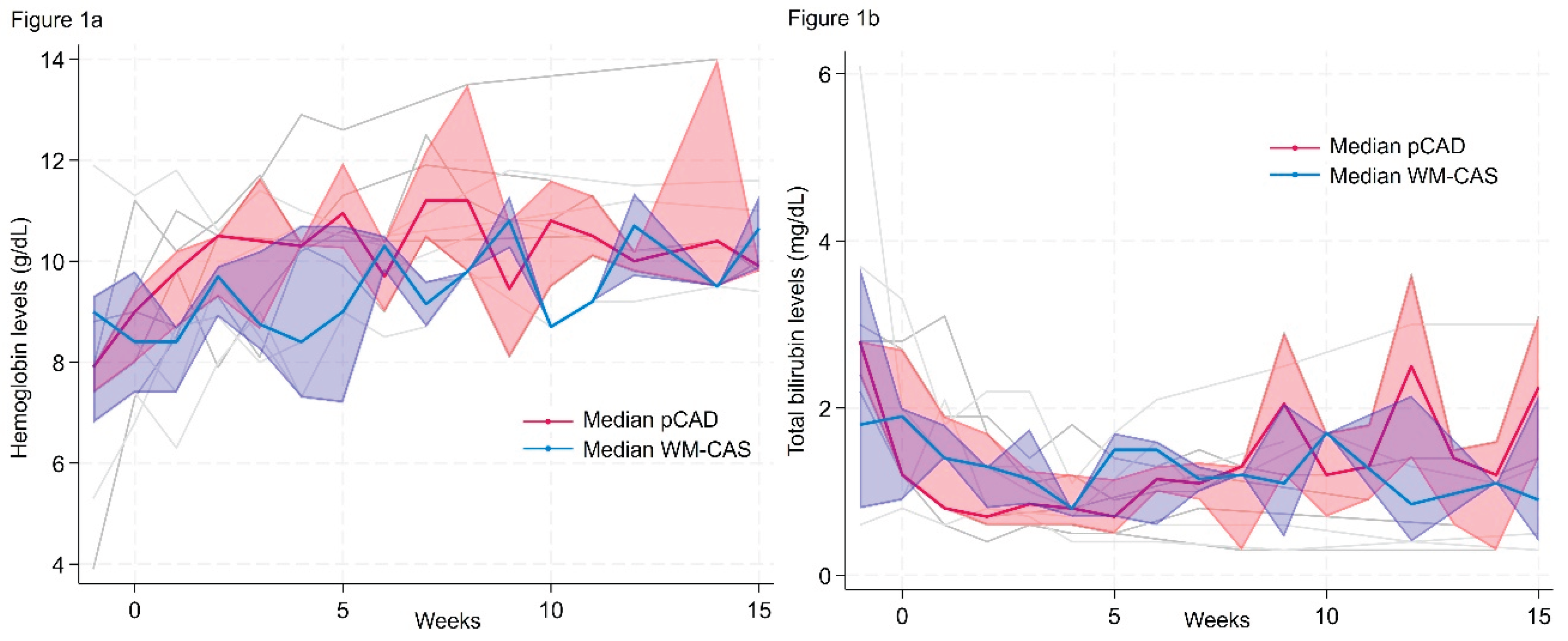
Response of Hemolytic Anemia Assessed by CAH Response Criteria
Tumor Response Assessed by IWWM Response Criteria
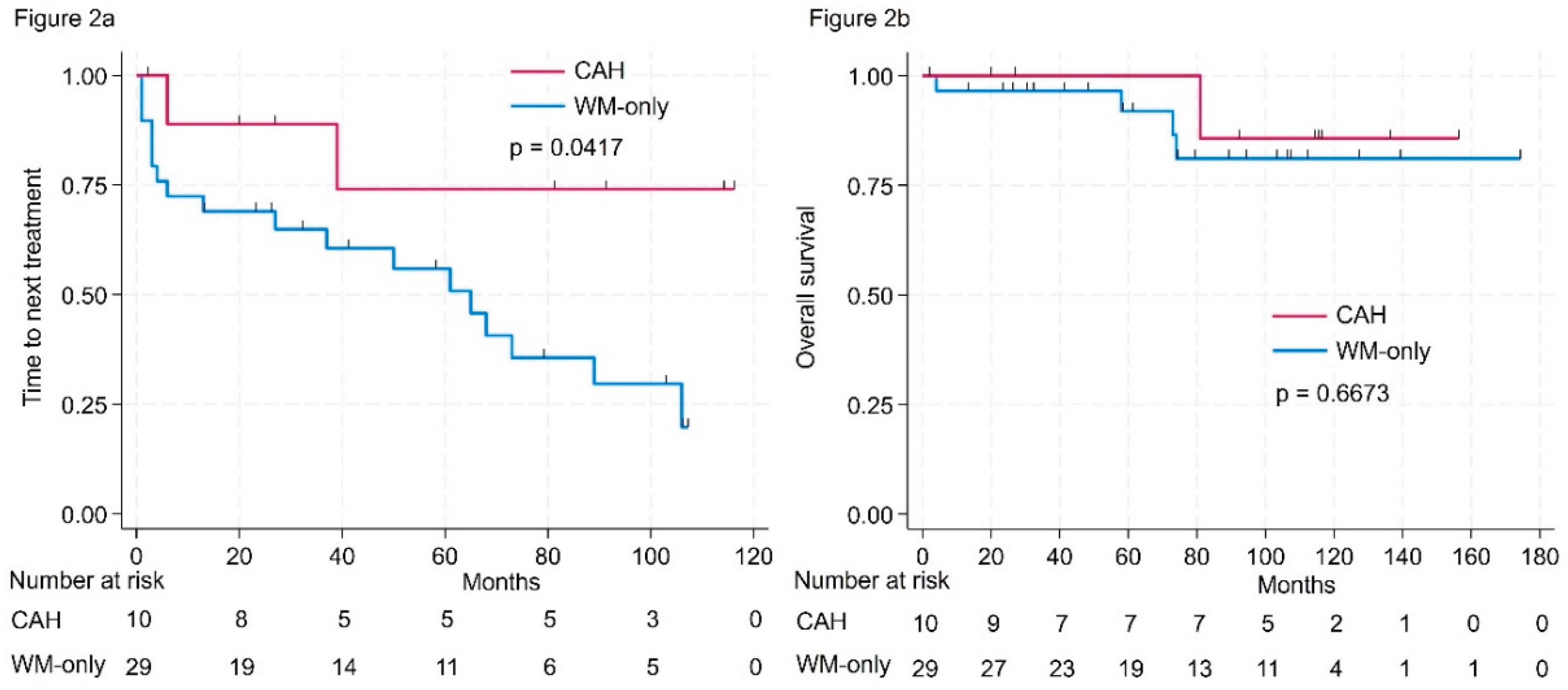
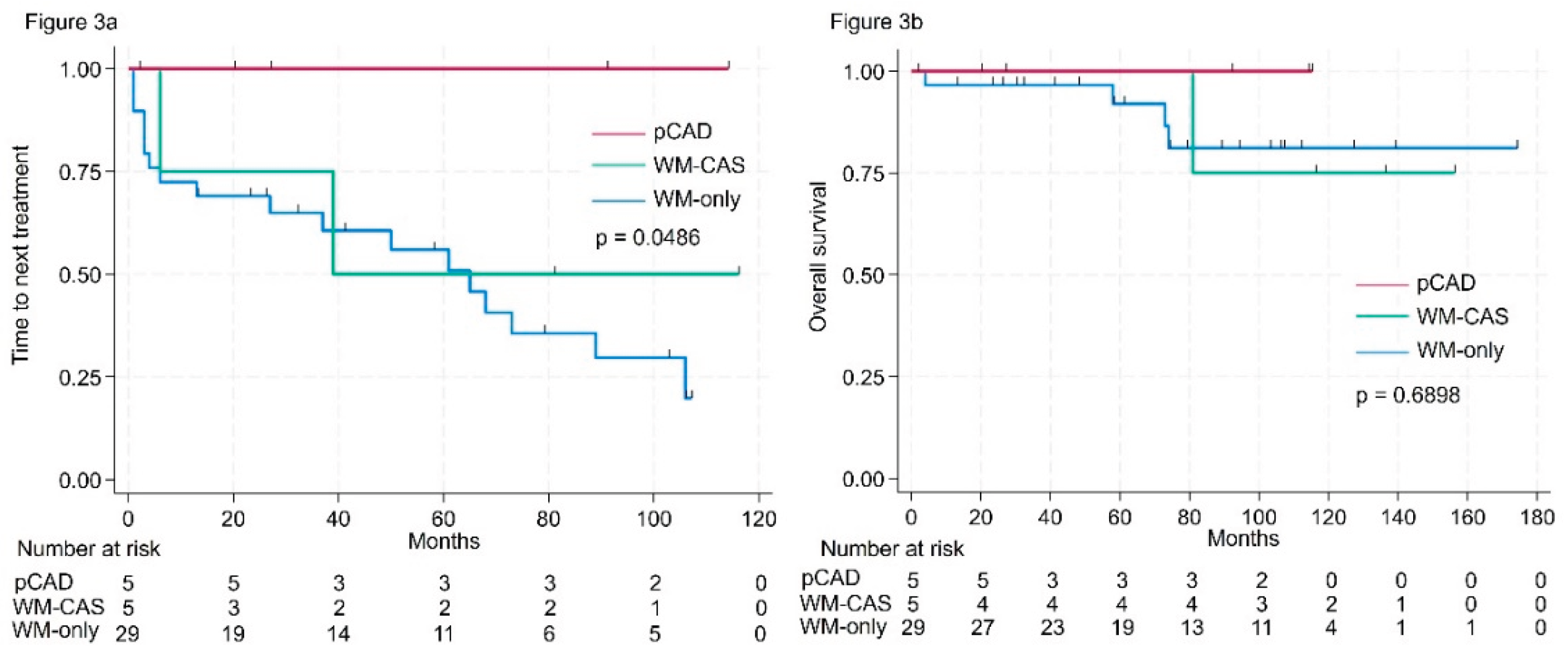
Survival Outcomes
AE
Discussion
Limitations
Conclusion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Berentsen S, Ulvestad E, Langholm R, et al. Primary chronic cold agglutinin disease: a population based clinical study of 86 patients. Haematologica. 2006;91(4):460-6.
- Crisp D, Pruzanski W. B-cell neoplasms with homogeneous cold-reacting antibodies (cold agglutinins). Am J Med. 1982;72(6):915-22. [CrossRef]
- Berentsen S, Tjønnfjord GE. Diagnosis and treatment of cold agglutinin mediated autoimmune hemolytic anemia. Blood Rev. 2012;26(3):107-15. [CrossRef]
- Barcellini W, Fattizzo B. The Changing Landscape of Autoimmune Hemolytic Anemia. Front Immunol. 2020;11:946. [CrossRef]
- Berentsen S, Röth A, Randen U, Jilma B, Tjønnfjord GE. Cold agglutinin disease: current challenges and future prospects. J Blood Med. 2019;10:93-103. [CrossRef]
- Zidaru L, Barbu S, Draghici R, Timotei SN, Coriu D, Badelita SN. Clinical diversity and treatment outcomes in IgM monoclonal gammopathy associated with cold agglutinin disease and cryoglobulinemia: a single center study. Documenta Haematologica - Revista Romana de Hematologie. 2025;3(2):61-66. [CrossRef]
- Broome CM, Cunningham JM, Mullins M, et al. Increased risk of thrombotic events in cold agglutinin disease: A 10-year retrospective analysis. Res Pract Thromb Haemost. 2020;4(4):628-35. [CrossRef]
- Delvasto-Nuñez L, Jongerius I, Zeerleder S. It takes two to thrombosis: Hemolysis and complement. Blood Rev. 2021;50:100834. [CrossRef]
- Kamesaki T, Nishimura J, Wada H, et al. Demographic characteristics, thromboembolism risk, and treatment patterns for patients with cold agglutinin disease in Japan. Int J Hematol. 2020; 112(3): 307-315. [CrossRef]
- Neresh KN, Barentsen S, Chen X, Randen U, Rossi D. Cold agglutinin disease. In: The WHO Classification of Tumours Editorial Board, Eds. World Health Organization classification of tumours. 5th ed. Haematolymphoid tumours. Lyon, IARC Press. 2024; pp.599–601.
- Berentsen S, Randen U, Tjønnfjord GE. Cold agglutinin-mediated autoimmune hemolytic anemia. Hematol Oncol Clin North Am. 2015;29(3):455-71. [CrossRef]
- Treon SP, Xu L, Yang G, et al. MYD88 L265P somatic mutation in Waldenström’s macroglobulinemia. N Engl J Med. 2012;367(9):826–33.
- Małecka A, Trøen G, Tierens A, et al. Immunoglobulin heavy and light chain gene features are correlated with primary cold agglutinin disease onset and activity. Haematologica. 2016;101(9):e361-4. [CrossRef]
- Randen U, Trøen G, Tierens A, et al. Primary cold agglutinin-associated lymphoproliferative disease: a B-cell lymphoma of the bone marrow distinct from lymphoplasmacytic lymphoma. Haematologica. 2014;99(3):497-504. [CrossRef]
- Leblond V, Kastritis E, Advani R, et al. Treatment recommendations from the Eighth International Workshop on Waldenström’s Macroglobulinemia. Blood. 2016;128(10):1321-8. [CrossRef]
- Buske C, Castillo JJ, Abeykoon JP, et al. Report of consensus panel 1 from the 11(th) International Workshop on Waldenstrom’s Macroglobulinemia on management of symptomatic, treatment-naïve patients. Semin Hematol. 2023;60(2):73-9. [CrossRef]
- Berentsen S, Ulvestad E, Gjertsen BT, et al. Rituximab for primary chronic cold agglutinin disease: a prospective study of 37 courses of therapy in 27 patients. Blood. 2004;103(8):2925-2928. [CrossRef]
- Berentsen S, Randen U, Vågan AM, et al. High response rate and durable remissions following fludarabine and rituximab combination therapy for chronic cold agglutinin disease. Blood. 2010;116(17):3180-4. [CrossRef]
- Berentsen S, Randen U, Oksman M, et al. Bendamustine plus rituximab for chronic cold agglutinin disease: results of a Nordic prospective multicenter trial. Blood. 2017;130(4):537-41. [CrossRef]
- Rossi G, Gramegna D, Paoloni F, et al. Short course of bortezomib in anemic patients with relapsed cold agglutinin disease: a phase 2 prospective GIMEMA study. Blood. 2018;132(5):547-50. [CrossRef]
- Jia MN, Qiu Y, Wu YY, et al. Rituximab-containing therapy for cold agglutinin disease: a retrospective study of 16 patients. Sci Rep. 2020;10(1):12694. [CrossRef]
- Ito K, Kitagawa T, Motohashi S, Hirano K, Sekiguchi N. Durable responses and favorable cost-effectiveness of rituximab-based chemoimmunotherapy for primary cold agglutinin disease. Blood Res. 2025;60(1):59. [CrossRef]
- Jalink M, Berentsen S, Castillo JJ, et al. Effect of ibrutinib treatment on hemolytic anemia and acrocyanosis in cold agglutinin disease/cold agglutinin syndrome. Blood. 2021;138(20):2002-5. [CrossRef]
- Jalink M, Jacobs CF, Khwaja J, et al. Daratumumab monotherapy in refractory warm autoimmune hemolytic anemia and cold agglutinin disease. Blood Adv. 2024;8(11):2622-2634. Blood Adv. 2024;8(24):6267. [CrossRef]
- Röth A, Berentsen S, Barcellini W, et al. Sutimlimab in patients with cold agglutinin disease: results of the randomized placebo-controlled phase 3 CADENZA trial. Blood. 2022;140(9):980-91. [CrossRef]
- Montes- Moreno S, Geddie WR, Kersten MJ, et al. “Lymphoplasmacytic Lymphoma,” in World Health Organization Classification of Tumours, 5th ed., ed. The WHO Classification of Tumours Editorial Board (Haematolymphoid tumours. IARC Press, 2024), 394–398.
- Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFad den ET, Carbone PP (1982) Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 5(6):649–655.
- National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE), version 5.0. Bethesda (MD): U.S. Department of Health and Human Services; 2017.
- Treon SP, Tedeschi A, San-Miguel J, Garcia-Sanz R, Anderson KC, Kimby E, et al. Report of consensus Panel 4 from the 11th International Workshop on Waldenstrom’s macroglobulinemia on diagnostic and response criteria. Semin Hematol. 2023;60(2):97-106. [CrossRef]
- Ito K, Kitagawa T, Harada K, Hirano K, Sekiguchi N. Impact of chromosomal aberrations detected by chromosome banding analysis in symptomatic Waldenström’s macroglobulinemia. Ann Hematol. 2025;104(1):713–20. [CrossRef]
- Treon SP, Tedeschi A, San-Miguel J, Garcia-Sanz R, Anderson KC, Kimby E, et al. Report of consensus Panel 4 from the 11th International Workshop on Waldenstrom’s macroglobulinemia on diagnostic and response criteria. Semin Hematol. 2023;60(2):97-106. [CrossRef]
- Sekiguchi N, Hamano A, Kitagawa T, et al. Impact of rituximab and half-dose CHOP as primary therapy for untreated symptomatic Waldenström Macroglobulinemia: review of a combined regimen of rituximab with an alkylating agent. Blood Res. 2018;53(2):117-22. [CrossRef]
- Berentsen S, Barcellini W, D’Sa S, et al. Cold agglutinin disease revisited: a multinational, observational study of 232 patients. Blood. 2020;136(4):480-8. [CrossRef]
- Bozzi S, Umarje S, Hawaldar K, et al. Prevalence and incidence of primary autoimmune hemolytic anemia and cold agglutinin disease in the United States, 2016-2023. PLoS One. 2025;20(6):e0323843. [CrossRef]
- Ito S, Wang D, Purcell A, et al. Cost-effectiveness of sutimlimab in cold agglutinin disease. Am J Hematol. 2024;99(8):1475-84. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



