Submitted:
15 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
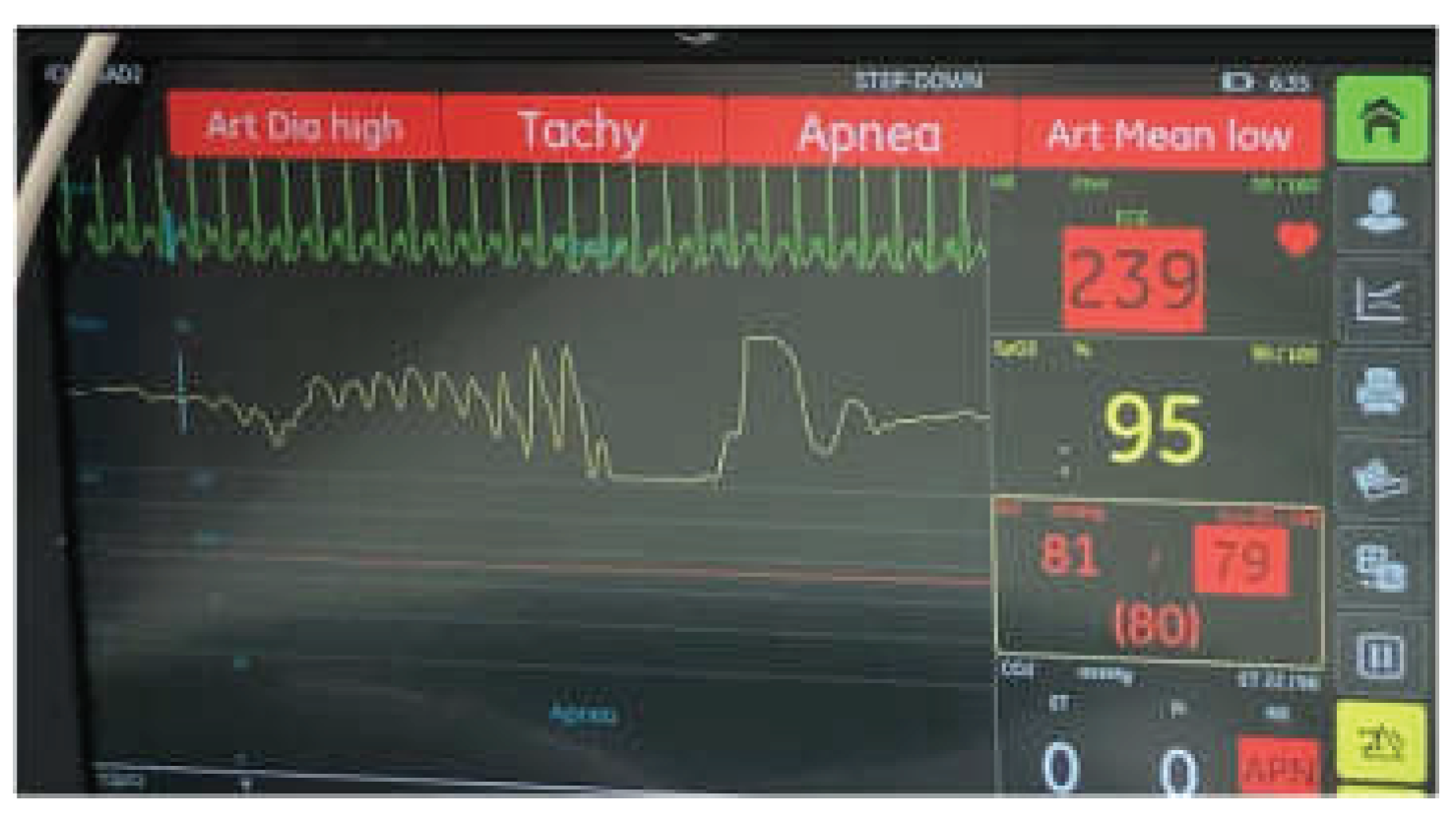
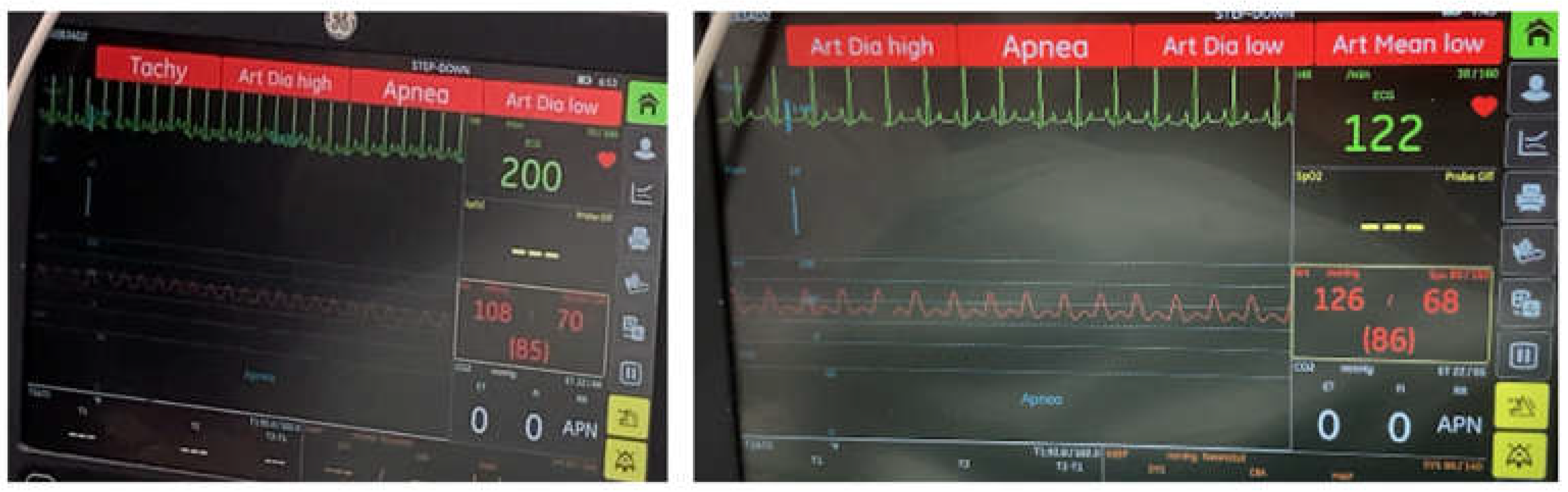
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| The following abbreviations are used in this manuscript |
References
- Kukanich, B.; Clark, T.P. The history and pharmacology of fentanyl: relevance to a novel, long-acting transdermal fentanyl solution newly approved for use in dogs. J Vet Pharmacol Ther. 2012, 35, Suppl 2:3–19. [Google Scholar] [CrossRef] [PubMed]
- Wegner, K.; Horais, K.A.; Tozier, N.A.; Rathbun, M.L.; Shtaerman, Y.; Yaksh, T.L. Development of a canine nociceptive thermal escape model. J Neurosci Methods 2008, 168, 88–97. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ilkiw, J.E. Balanced anesthetic techniques in dogs and cats. Clin Tech Small Anim Pract. 1999, 1, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.R.; Hug, C.C., Jr. The anesthetic potency of fentanyl in terms of its reduction of enflurane MAC. Anesthesiology 1982, 57, 485–8. [Google Scholar] [CrossRef] [PubMed]
- Becker, W.M.; Mama, K.R.; Rao, S.; Palmer, R.H.; Egger, E.L. Prevalence of dysphoria after fentanyl in dogs undergoing stifle surgery. Vet Surg. 2013, 42, 302–7. [Google Scholar] [CrossRef] [PubMed]
- Jarosinski, S.K.; Simon, B.T.; Baetge, C.L.; Parry, S.; Araos, J. The Effects of Prophylactic Dexmedetomidine Administration on General Anesthesia Recovery Quality in Healthy Dogs Anesthetized With Sevoflurane and a Fentanyl Constant Rate Infusion Undergoing Elective Orthopedic Procedures. Front Vet Sci. 2021, 28, 8:722038. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Copland, V.S.; Haskins, S.C.; Patz, J. Naloxone reversal of oxymorphone effects in dogs. Am J Vet Res. 1989, 50, 1854–8. [Google Scholar] [CrossRef] [PubMed]
- Lemke, K.A.; Tranquilli, W.J.; Thurmon, J.C.; Benson, G.J.; Olson, W.A. Ability of flumazenil, butorphanol, and naloxone to reverse the anesthetic effects of oxymorphone-diazepam in dogs. J Am Vet Med Assoc. 1996, 15, 776–9. [Google Scholar] [CrossRef] [PubMed]
- Burkitt-Creedon, J.M.; Boller, M.; Fletcher, D.J.; Brainard, B.M.; Buckley, G.J.; Epstein, S.E.; Fausak, E.D.; Hopper, K.; Lane, S.L.; Rozanski, E.A.; Wolf, J. 2024 RECOVER Guidelines: Updated treatment recommendations for CPR in dogs and cats. J Vet Emerg Crit Care (San Antonio) 2024, 34, 104–123. [Google Scholar] [CrossRef] [PubMed]
- KuKanich, B.; Papich, M.G. Opioid analgesic drugs. In Veterinary Pharmacology and Therapeutics, 10th ed.; Riviere, J.E., Papich, M.G., Eds.; John Wiley & Sons Inc.: New Jersey, USA, 2018; pp. 305–347. [Google Scholar]
- Mills, C.A.; Flacke, J.W.; Miller, J.D.; Davis, L.J.; Bloor, B.C.; Flacke, W.E. Cardiovascular effects of fentanyl reversal by naloxone at varying arterial carbon dioxide tensions in dogs. Anesth Analg. 1988, 67, 730–6. [Google Scholar] [CrossRef] [PubMed]
- Huse, K.; Hartung, E.; Nadjmabadi, M. H. Wirkungen von Naloxone (Narcan) auf Kreislauf und Atmung nach Neurolept-Anaesthesie für neurochirurgische Operationen [The effects of naloxone (Narcan) on circulation and respiration after neurolept anaesthesia for neurosurgical operations (author's transl)]; Anaesthesist.: German, 1974; Volume 23, pp. 493–9. [Google Scholar] [PubMed]
- Neale, J.; Strang, J. Naloxone--does over-antagonism matter? Evidence of iatrogenic harm after emergency treatment of heroin/opioid overdose. Addiction 2015, 110, 1644–52. [Google Scholar] [CrossRef] [PubMed]
- Pariaut, R.; Reynolds, C. Bradyarrhythmias and conduction abnormalities. In Small Animal Critical Care Medicine, 2nd ed.; Silverstein, D.C., Hopper, K., Eds.; Saunders Elsevier: Philadelphia, USA, 2014; pp. 246–249. [Google Scholar]
- Congdon, J. M.; Cardiovascular Disease. Canine and Feline Anesthesia and Co-Existing Disease, 2nd ed.; Johnson, R.A., Snyder, L.B.C., Schroeder, C.A., Eds.; Wiley Blackwell: New Jersey, USA, 2022; pp. 1–85. [Google Scholar]
- Rzasa Lynn, R.; Galinkin, J.L. Naloxone dosage for opioid reversal: current evidence and clinical implications. Ther Adv Drug Saf. 2018, 9, 63–88. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Azar, I.; Turndorf, H. Severe hypertension and multiple atrial premature contractions following naloxone administration. Anesth Analg. 1979, 58, 524–5. [Google Scholar] [CrossRef] [PubMed]
- Flacke, J.W.; Flacke, W.E.; Williams, G.D. Acute pulmonary edema following naloxone reversal of high-dose morphine anesthesia. Anesthesiology 1977, 47, 376–8. [Google Scholar] [CrossRef] [PubMed]
- Lameijer, H.; Azizi, N.; Ligtenberg, J.J.; Ter Maaten, J.C. Ventricular Tachycardia After Naloxone Administration: a Drug Related Complication? Case Report and Literature Review. Drug Saf Case Rep. 2014, 1, 2. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Vazquez, J.M.; Seaman, R.W., Jr.; Caldron, D.; Collins, G.T. Naloxone reversal of the cardiorespiratory depressant effects of opioids and mixtures of opioids and stimulants in rats. Front Pharmacol. 2025, 22, 1654791. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Paddleford, R.R.; Short, C.E. An evaluation of naloxone as a narcotic antagonist in the dog. J Am Vet Med Assoc. 1973, 163, 144–6. [Google Scholar] [CrossRef] [PubMed]
- Freise, K.J.; Newbound, G.C.; Tudan, C.; Clark, T.P. Naloxone reversal of an overdose of a novel, long-acting transdermal fentanyl solution in laboratory Beagles. J Vet Pharmacol Ther. 2012, 35, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Flacke, J.W.; Flacke, W.E.; Bloor, B.C.; Olewine, S. Effects of fentanyl, naloxone, and clonidine on hemodynamics and plasma catecholamine levels in dogs. Anesth Analg. 1983, 62, 305–13. [Google Scholar] [CrossRef] [PubMed]
- Estilo, A.E.; Cottrell, J.E. Hemodynamic and catecholamine changes after administration of naloxone. Anesth Analg. 1982, 61, 349–53. [Google Scholar] [CrossRef] [PubMed]
- Iizuka, T.; Nishimura, R. Context-sensitive half-time of fentanyl in dogs. J Vet Med Sci. 2015, 77, 615–7. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hughes, M.A.; Glass, P.S.; Jacobs, J.R. Context-sensitive half-time in multicompartment pharmacokinetic models for intravenous anesthetic drugs. Anesthesiology 1992, 76, 334–41. [Google Scholar] [CrossRef] [PubMed]
- Sano, T.; Nishimura, R.; Kanazawa, H.; Igarashi, E.; Nagata, Y.; Mochizuki, M.; Sasaki, N. Pharmacokinetics of fentanyl after single intravenous injection and constant rate infusion in dogs. Vet Anaesth Analg. 2006, 33, 266–73. [Google Scholar] [CrossRef] [PubMed]
- Simões, C.R.; Monteiro, E.R.; Rangel, J.P.; Nunes-Junior, J.S.; Campagnol, D. Effects of a prolonged infusion of fentanyl, with or without atropine, on the minimum alveolar concentration of isoflurane in dogs. Vet Anaesth Analg. 2016, 43, 136–44. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.R.; Hug, C.C., Jr.; McClain, D.A. Dose-independent pharmacokinetics of fentanyl. Anesthesiology 1983, 59, 537–40. [Google Scholar] [CrossRef] [PubMed]
- Hofmeister, E. H. Anesthetic Emergencies, Resuscitation, and Adverse Events. In Lumb and Jones Veterinary Anesthesia and Analgesia, 6th ed.; Lamont, L., Grimm, K., Robertson, S., Love, L., Eds.; Wiley Blackwell: New Jersey, USA, 2024; pp. 54–73. [Google Scholar]
- Hay Kraus, B.L.; Greenblatt, D.J.; Venkatakrishnan, K.; Court, M.H. Evidence for propofol hydroxylation by cytochrome P4502B11 in canine liver microsomes: breed and gender differences. Xenobiotica 2000, 30, 575–88. [Google Scholar] [CrossRef] [PubMed]
- Court, M.H.; Hay-Kraus, B.L.; Hill, D.W.; Kind, A.J.; Greenblatt, D.J. Propofol hydroxylation by dog liver microsomes: assay development and dog breed differences. Drug Metab Dispos. 1999, 27, 1293–9. [Google Scholar] [CrossRef] [PubMed]
- Mise, M.; Yadera, S.; Matsuda, M.; Hashizume, T.; Matsumoto, S.; Terauchi, Y.; Fujii, T. Polymorphic expression of CYP1A2 leading to interindividual variability in metabolism of a novel benzodiazepine receptor partial inverse agonist in dogs. Drug Metab Dispos. 2004, 32, 240–5. [Google Scholar] [CrossRef] [PubMed]
- Mealey, K.L.; Meurs, K.M. Breed distribution of the ABCB1-1Delta (multidrug sensitivity) polymorphism among dogs undergoing ABCB1 genotyping. J Am Vet Med Assoc. 2008, 233, 921–4. [Google Scholar] [CrossRef] [PubMed]
- Wengrowski, A.M.; Wang, X.; Tapa, S.; Posnack, N.G.; Mendelowitz, D.; Kay, M.W. Optogenetic release of norepinephrine from cardiac sympathetic neurons alters mechanical and electrical function. Cardiovasc Res. 2015, 105, 143–50. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



