Submitted:
15 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Search Strategies
3. Epidemiology of RLS: Prevalence, Sex Differences, and the Challenge of Under-Recognition
4. Clinical Presentation and Systemic Implications of RLS
4.1. Symptom Spectrum and Functional Burden
4.2. Neuropsychiatric Associations and Network Implications
4.3. Cardiovascular Associations: Signal, Confounding, and Uncertainty
4.4. Periodic Limb Movements of Sleep: Motor Signature or Epiphenomenon?
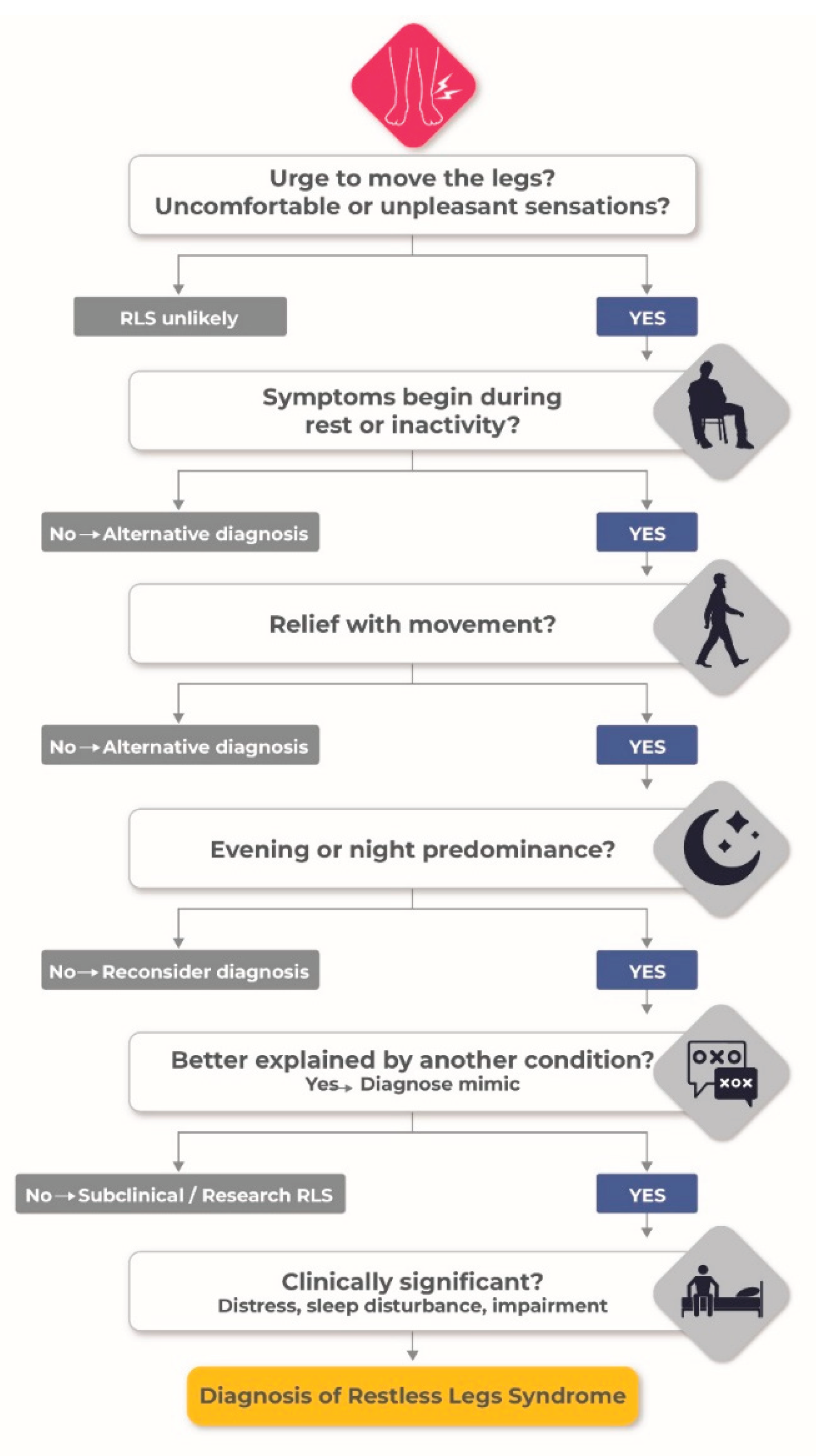
5. Assessment and Diagnosis: From Symptom Recognition to Mechanism-Informed Evaluation
5.1. Clinical Evaluation and Differential Diagnosis
5.2. Laboratory Assessment and Iron Status
5.3. Role of Polysomnography and Objective Testing
5.4. Toward Mechanism-Informed Diagnostic Frameworks
5.5. Limitations and Future Directions
| Domain | Element | Clinical signal | Interpretation (network level) | Evidence |
|---|---|---|---|---|
| Core criteria | Urge to move the legs | Compelling need to move → often with abnormal sensations | Sensorimotor integration disturbance → cortico–striatal dysregulation | [1,2,3,39,40] |
| Rest-induced worsening | Symptoms ↑ during inactivity → sitting or lying | State-dependent network instability → reduced inhibitory tone | [39,41] | |
| Relief with movement | Movement → partial or complete symptom relief | Transient normalization of network excitability | [39,41] | |
| Circadian pattern | Evening/night predominance → symptoms ↑ at night | Circadian neuromodulation → dopamine and iron fluctuation | [1,2,3,39,40] | |
| Exclusion | Alternative causes | Symptoms not explained by neuropathy, cramps, arthritis, or akathisia | Avoids diagnostic misclassification → ensures specificity | [39,41] |
| Clinical significance | Functional impact | Sleep disruption → ↓ sleep efficiency, ↑ awakenings | Sleep–wake instability → network hyperexcitability | [42,43,44] |
| Daytime consequences | Daytime sleepiness → cognitive impairment → reduced function | Propagation from nocturnal disruption to daytime dysfunction | [44,45] | |
| Phenotypic modifiers | Distribution | Legs → arms → trunk (in severe cases) | Spread of network involvement → disease progression | [41,44] |
| Sensory variability | Dysesthesia may be subtle or absent | Risk of under-recognition → variable sensory processing | [44] | |
| Severe disease | Movement relief ↓ or incomplete | Persistent hyperexcitability → advanced dysregulation | [45] | |
| Treatment-related changes | Circadian pattern ↓ or lost during augmentation | Dopaminergic maladaptive plasticity → network destabilisation | [11] | |
| Contextual factors | Iron status | Ferritin ↓ (<75–100 ng/mL) → transferrin saturation ↓ | Central iron deficiency → altered dopamine and glutamate signalling | [1,8,12,46] |
| Pregnancy | Symptoms ↑ across trimesters → resolve postpartum | Hormonal + iron fluctuations → reversible vulnerability | [13,14] | |
| Chronic kidney disease | Increased prevalence of symptoms | Systemic inflammation → iron dysregulation | [47] | |
| Medications | Antidepressants, antipsychotics → symptoms ↑ | Neurotransmitter imbalance → exacerbation of network instability | [48] | |
| Associated physiology | Periodic limb movements during sleep | Repetitive movements → sleep fragmentation → ↑ heart rate | Marker of motor network excitability → not disease-specific | [23,24,26] |
| Diagnostic tools | Clinical history | Circadian pattern + movement relief → key diagnostic anchors | Core diagnostic framework remains clinical | [39,40] |
| Laboratory assessment | Serum ferritin and iron indices | Peripheral markers → indirect proxy of brain iron | [12,46] | |
| Polysomnography | Detects limb movements → evaluates sleep structure | Adjunctive tool → not required for diagnosis | [23,24,25,26,27,49] | |
| Emerging biomarkers | Brain iron imaging | Reduced iron signal in basal ganglia → MRI-based measures | Supports the iron-dependent network model | [1,8] |
| Genetic profile | Risk loci (e.g., MEIS1, BTBD9) | Developmental–metabolic susceptibility | [9,10] | |
| Circadian phenotyping | Temporal variation of symptoms | Biological stratification → future precision medicine | [1,2,3] |
| Diagnostic dimension | Restless legs syndrome | Neurological mimics (peripheral neuropathy, akathisia) | Musculoskeletal mimics (cramps, arthritides) | Contextual mimics (anxiety, positional discomfort) |
|---|---|---|---|---|
| Core symptom | Urge to move → dysesthesia | Neuropathic pain → burning/electric Motor restlessness (akathisia) |
Painful contraction → cramps Joint pain → stiffness |
Inner unease (anxiety) Local pressure (posture) |
| Temporal pattern | Evening/night ↑ (hallmark) | No consistent circadian pattern | Episodic or activity-related | Variable → situational |
| Trigger | Rest/inactivity → symptoms ↑ | Persistent (neuropathy) Drug exposure (akathisia) |
Sudden (cramps) Load/movement (arthritis) |
Stress or posture |
| Relief with movement | Sustained relief → key discriminator | Minimal (neuropathy) Temporary (akathisia) |
Relief after stretching (cramps) Limited (arthritis) |
Immediate repositioning or nonspecific |
| Neurological exam | Normal | Sensory deficits → reflex changes (neuropathy) Motor restlessness (akathisia) |
Normal or joint inflammation signs | Normal |
| Clinical course | Chronic → fluctuating → circadian | Chronic or drug-related | Episodic or chronic structural | Situational or persistent |
| Pathophysiology | Iron ↓ → dopamine modulation ↓ → glutamate ↑ → network hyperexcitability | Peripheral nerve damage Dopamine receptor blockade |
Muscle hyperexcitability Structural joint disease |
Limbic hyperarousal Mechanical compression |
| Key discriminator | Circadian pattern + relief with movement | Objective neurological deficit or medication link | Pain-dominant → structural or muscular origin | Context-dependent → immediate resolution |
| Evidence | [1,2,3,23,24,25,26,27,39,40,49] | [39,40] | [39,40] | [17,50] |
6. Risk Factors and Associated Conditions: Toward Mechanism-Informed Stratification
6.1. Iron Deficiency and Systemic Iron Dysregulation
6.2. Pregnancy: A Model of Reversible Vulnerability
6.3. Opioid Withdrawal and Endogenous Opioid Deficiency
6.4. Adrenergic Modulation and Hyperexcitability in RLS
6.4. Chronic Kidney Disease and Peripheral Neuropathy
6.5. Neurological Comorbidities: Network Convergence
7. Genetic Architecture and Developmental Vulnerability
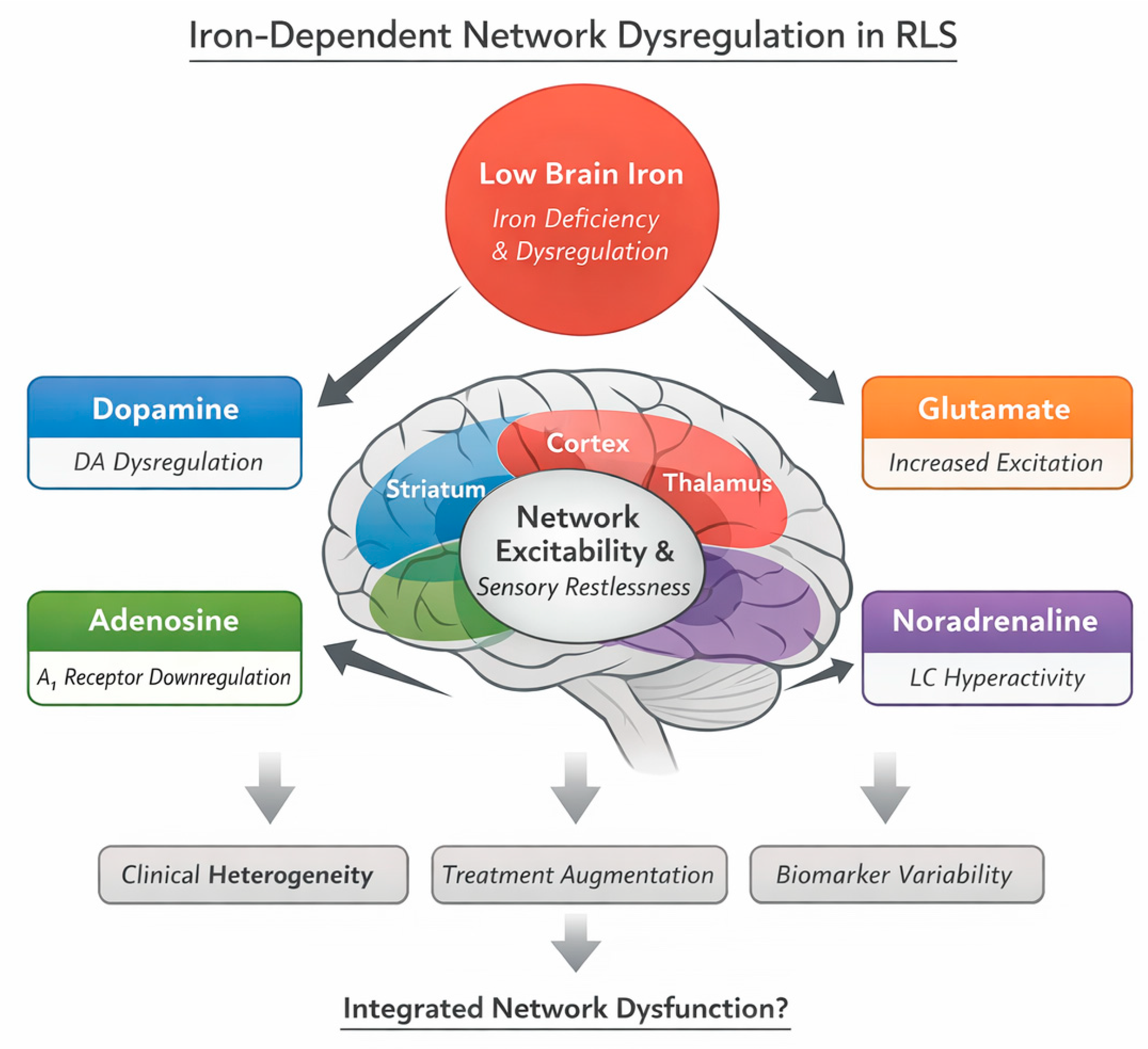
8. Pathophysiology of RLS: From Iron Dysregulation to Network Dysfunction
8.1. Brain Iron Dysregulation and Blood–Brain Barrier Function
8.2. Dopaminergic Dynamics and Circadian Modulation
8.3. Opioidergic and Glutamatergic Contributions
9. Management of RLS: From Symptom Suppression to Mechanism-Based Care
9.1. Initial Management: Addressing Modifiable Drivers
9.2. Iron Supplementation: Foundational Therapy
9.3. Pharmacological Therapy: Individualized and Stratified
| Treatment Class / Agent | Typical RLS Dose Range | CGI-I Responder Difference vs Placebo, % (95% CI) | IRLS Mean Difference vs Placebo (95% CI) | AASM Recommendation (2025) | Common or Clinically Important Adverse Effects | GRADE (Certainty / Recommendation) |
|---|---|---|---|---|---|---|
| Strongly or Conditionally Recommended | ||||||
| Gabapentinoids (α2δ ligands) | ||||||
| Gabapentin | 300–3600 mg/d | — | −8.40 (−12.00 to −4.80) | Strong for | Somnolence (10%–25%), dizziness (15%–19%), cognitive disturbance, mood changes, weight gain | Moderate / Strong |
| Gabapentin enacarbil | 600–1200 mg/d | 34 (24 to 45) | −4.93 (−6.85 to −3.02) | Strong for | Similar to gabapentin | High / Strong |
| Pregabalin | 75–600 mg/d | 26 (17 to 34) | −4.81 (−6.21 to −3.42) | Strong for | Somnolence, dizziness, weight gain | High / Strong |
| Iron Therapy | ||||||
| IV ferric carboxymaltose | 1000–1500 mg (1–2 doses) | 30 (16 to 44) | −7.43 (−11.89 to −2.97) | Strong for | Headache (12%), nausea (5%), hypophosphatemia, and rare hypersensitivity | High / Strong |
| IV ferumoxytol | 1020 mg (1–2 doses) | — | −7.90 (−11.74 to −4.06) | Conditional for | Infusion reactions (rare) | Moderate / Conditional |
| IV low–molecular-weight iron dextran | 1000 mg (single dose) | — | — | Conditional for | Rare anaphylaxis risk | Low–Moderate / Conditional |
| Oral ferrous sulfate | 325–650 mg daily or alternate days | — | −9.20 (−15.23 to −3.17) | Conditional for | Constipation (12%), nausea (11%), diarrhea (8%) | Moderate / Conditional |
| Opioids (Refractory RLS) | ||||||
| Oxycodone (prolonged-release) | 5–40 mg/d | 32 (21 to 43) | −5.60 (−8.18 to −3.02) | Conditional for | Constipation (47%), sedation, pruritus | Moderate–High / Conditional |
| Methadone | 2.5–20 mg/d | — | — | Conditional for | Sedation, constipation | Low–Moderate / Conditional |
| Buprenorphine | 0.5–6 mg/d | — | — | Conditional for | Fatigue, sweating, pruritus | Low–Moderate / Conditional |
| Nonpharmacologic | ||||||
| Peroneal nerve stimulation | Device-based | 34 (21 to 46)ᵉ | −3.40 (−5.02 to −1.78)ᵉ | Conditional for | Local discomfort (28%), skin irritation (9%) | Moderate / Conditional |
| Pharmacologic (Routine Daily Use) | ||||||
| Dopaminergic Agents | ||||||
| Levodopa | 100–200 mg/d | — | — | Conditional against | Augmentation (7%–10% annually), impulse control disorders (10%–20%), nausea, somnolence | High (efficacy) / Conditional against |
| Pramipexole | 0.125–0.5 mg/d | 23 (18 to 27) | −4.86 (−6.20 to −3.52) | Conditional against | High (efficacy) / Conditional against | |
| Ropinirole | 0.25–2 mg/d | 18 (13 to 23) | −3.98 (−5.36 to −2.60) | Conditional against | High (efficacy) / Conditional against | |
| Rotigotine (transdermal) | 1–3 mg/d | 11 (0 to 22) | −4.67 (−6.18 to −3.16) | Conditional against | Application site reactions (34%) | High (efficacy) / Conditional against |
9.3.1. Gabapentinoids (α2δ Ligands)
9.3.2. Dopamine Agonists
9.3.3. Low-Dose Opioids: Refractory and Augmented Disease
9.4. Neuromodulation and Emerging Therapies
9.5. Management of Augmentation
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AASM | American Academy of Sleep Medicine |
| A1R | Adenosine A1 receptor |
| α2δ ligands | Alpha-2-delta ligands (e.g., gabapentinoids) |
| BBB | Blood–brain barrier |
| BTBD9 | BTB domain-containing protein 9 |
| CKD | Chronic kidney disease |
| CSF | Cerebrospinal fluid |
| CVD | Cardiovascular disease |
| fMRI | Functional magnetic resonance imaging |
| GWAS | Genome-wide association studies |
| MEIS1 | Meis homeobox 1 |
| MRI | Magnetic resonance imaging |
| NCS | Nerve conduction studies |
| PLMS | Periodic limb movements of sleep |
| QSM | Quantitative susceptibility mapping |
| RLS | Restless legs syndrome |
References
- Allen, R.P.; Chen, C.; Garcia-Borreguero, D.; Polo, O.; DuBrava, S.; Miceli, J.; Knapp, L.; Winkelman, J.W. Comparison of pregabalin with pramipexole for restless legs syndrome. N Engl J Med 2014, 370, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Manconi, M.; Garcia-Borreguero, D.; Schormair, B.; Videnovic, A.; Berger, K.; Ferri, R.; Dauvilliers, Y. Restless legs syndrome. Nat Rev Dis Primers 2021, 7, 80. [Google Scholar] [CrossRef] [PubMed]
- Trenkwalder, C.; Paulus, W. Restless legs syndrome: pathophysiology, clinical presentation and management. Nat Rev Neurol 2010, 6, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Fulda, S.; Allen, R.P.; Earley, C.J.; Hogl, B.; Garcia-Borreguero, D.; Inoue, Y.; Ondo, W.; Walters, A.S.; Williams, A.M.; Winkelman, J.W. We need to do better: A systematic review and meta-analysis of diagnostic test accuracy of restless legs syndrome screening instruments. Sleep Med Rev 2021, 58, 101461. [Google Scholar] [CrossRef]
- Silber, M.H.; Buchfuhrer, M.J.; Earley, C.J.; Koo, B.B.; Manconi, M.; Winkelman, J.W.; Scientific; Medical Advisory Board of the Restless Legs Syndrome; F. The Management of Restless Legs Syndrome: An Updated Algorithm. Mayo Clin Proc 2021, 96, 1921–1937. [Google Scholar] [CrossRef]
- Winkelmann, J.; Stautner, A.; Samtleben, W.; Trenkwalder, C. Long-term course of restless legs syndrome in dialysis patients after kidney transplantation. Mov Disord 2002, 17, 1072–1076. [Google Scholar] [CrossRef]
- Winkelmann, J.; Allen, R.P.; Hogl, B.; Inoue, Y.; Oertel, W.; Salminen, A.V.; Winkelman, J.W.; Trenkwalder, C.; Sampaio, C. Treatment of restless legs syndrome: Evidence-based review and implications for clinical practice (Revised 2017)( section sign). Mov Disord 2018, 33, 1077–1091. [Google Scholar] [CrossRef]
- Earley, C.J.; Connor, J.; Garcia-Borreguero, D.; Jenner, P.; Winkelman, J.; Zee, P.C.; Allen, R. Altered brain iron homeostasis and dopaminergic function in Restless Legs Syndrome (Willis-Ekbom Disease). Sleep Med 2014, 15, 1288–1301. [Google Scholar] [CrossRef]
- Schormair, B.; Zhao, C.; Bell, S.; Tilch, E.; Salminen, A.V.; Putz, B.; Dauvilliers, Y.; Stefani, A.; Hogl, B.; Poewe, W.; et al. Identification of novel risk loci for restless legs syndrome in genome-wide association studies in individuals of European ancestry: a meta-analysis. Lancet Neurol 2017, 16, 898–907. [Google Scholar] [CrossRef]
- Winkelmann, J.; Czamara, D.; Schormair, B.; Knauf, F.; Schulte, E.C.; Trenkwalder, C.; Dauvilliers, Y.; Polo, O.; Hogl, B.; Berger, K.; et al. Genome-wide association study identifies novel restless legs syndrome susceptibility loci on 2p14 and 16q12.1. PLoS Genet 2011, 7, e1002171. [Google Scholar] [CrossRef]
- Winkelman, J.W.; Berkowski, J.A.; DelRosso, L.M.; Koo, B.B.; Scharf, M.T.; Sharon, D.; Zak, R.S.; Kazmi, U.; Falck-Ytter, Y.; Shelgikar, A.V.; et al. Treatment of restless legs syndrome and periodic limb movement disorder: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med 2025, 21, 137–152. [Google Scholar] [CrossRef]
- Allen, R.P.; Walters, A.S.; Montplaisir, J.; Hening, W.; Myers, A.; Bell, T.J.; Ferini-Strambi, L. Restless legs syndrome prevalence and impact: REST general population study. Arch Intern Med 2005, 165, 1286–1292. [Google Scholar] [CrossRef]
- Cho, Y.W.; Shin, W.C.; Yun, C.H.; Hong, S.B.; Kim, J.H.; Allen, R.P.; Earley, C.J. Epidemiology of restless legs syndrome in Korean adults. Sleep 2008, 31, 219–223. [Google Scholar] [CrossRef]
- Rothdach, A.J.; Trenkwalder, C.; Haberstock, J.; Keil, U.; Berger, K. Prevalence and risk factors of RLS in an elderly population: the MEMO study. Memory and Morbidity in Augsburg Elderly. Neurology 2000, 54, 1064–1068. [Google Scholar] [CrossRef]
- Allen, R.P.; Stillman, P.; Myers, A.J. Physician-diagnosed restless legs syndrome in a large sample of primary medical care patients in western Europe: Prevalence and characteristics. Sleep Med 2010, 11, 31–37. [Google Scholar] [CrossRef]
- Trenkwalder, C.; Allen, R.; Hogl, B.; Paulus, W.; Winkelmann, J. Restless legs syndrome associated with major diseases: A systematic review and new concept. Neurology 2016, 86, 1336–1343. [Google Scholar] [CrossRef] [PubMed]
- Karroum, E.G.; Saini, P.S.; Trotti, L.M.; Rye, D.B. TOX3 gene variant could be associated with painful restless legs. Sleep Med 2020, 65, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Kerr, S.; McKinon, W.; Bentley, A. Descriptors of restless legs syndrome sensations. Sleep Med 2012, 13, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Hening, W.; Walters, A.S.; Allen, R.P.; Montplaisir, J.; Myers, A.; Ferini-Strambi, L. Impact, diagnosis and treatment of restless legs syndrome (RLS) in a primary care population: the REST (RLS epidemiology, symptoms, and treatment) primary care study. Sleep Med 2004, 5, 237–246. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, Y.; Ren, R.; Yang, L.; Shi, Y.; Vitiello, M.V.; Sanford, L.D.; Tang, X. Polysomnographic features of idiopathic restless legs syndrome: a systematic review and meta-analysis of 13 sleep parameters and 23 leg movement parameters. J Clin Sleep Med 2022, 18, 2561–2575. [Google Scholar] [CrossRef]
- Trenkwalder, C.; Hening, W.A.; Walters, A.S.; Campbell, S.S.; Rahman, K.; Chokroverty, S. Circadian rhythm of periodic limb movements and sensory symptoms of restless legs syndrome. Mov Disord 1999, 14, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Miyaguchi, R.; Masuda, F.; Sumi, Y.; Kadotani, H.; Ozeki, Y.; Banno, M.; Kuniyoshi, Y. Prevalence of depression or depressive state in patients with restless legs syndrome: A systematic review and meta-analysis. Sleep Med Rev 2024, 77, 101975. [Google Scholar] [CrossRef] [PubMed]
- Winkelmann, J.; Prager, M.; Lieb, R.; Pfister, H.; Spiegel, B.; Wittchen, H.U.; Holsboer, F.; Trenkwalder, C.; Strohle, A. Anxietas tibiarum". Depression and anxiety disorders in patients with restless legs syndrome. J Neurol 2005, 252, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Winkelman, J.W.; Shahar, E.; Sharief, I.; Gottlieb, D.J. Association of restless legs syndrome and cardiovascular disease in the Sleep Heart Health Study. Neurology 2008, 70, 35–42. [Google Scholar] [CrossRef]
- Batool-Anwar, S.; Malhotra, A.; Forman, J.; Winkelman, J.; Li, Y.; Gao, X. Restless legs syndrome and hypertension in middle-aged women. Hypertension 2011, 58, 791–796. [Google Scholar] [CrossRef]
- Cappuccio, F.P.; Cooper, D.; D'Elia, L.; Strazzullo, P.; Miller, M.A. Sleep duration predicts cardiovascular outcomes: a systematic review and meta-analysis of prospective studies. Eur Heart J 2011, 32, 1484–1492. [Google Scholar] [CrossRef]
- Koo, B.B.; Blackwell, T.; Ancoli-Israel, S.; Stone, K.L.; Stefanick, M.L.; Redline, S.; Osteoporotic Fractures in Men Study, G. Association of incident cardiovascular disease with periodic limb movements during sleep in older men: outcomes of sleep disorders in older men (MrOS) study. Circulation 2011, 124, 1223–1231. [Google Scholar] [CrossRef]
- Ferri, R.; Fulda, S.; Allen, R.P.; Zucconi, M.; Bruni, O.; Chokroverty, S.; Ferini-Strambi, L.; Frauscher, B.; Garcia-Borreguero, D.; Hirshkowitz, M.; et al. World Association of Sleep Medicine (WASM) 2016 standards for recording and scoring leg movements in polysomnograms developed by a joint task force from the International and the European Restless Legs Syndrome Study Groups (IRLSSG and EURLSSG). Sleep Med 2016, 26, 86–95. [Google Scholar] [CrossRef]
- Pennestri, M.H.; Montplaisir, J.; Fradette, L.; Lavigne, G.; Colombo, R.; Lanfranchi, P.A. Blood pressure changes associated with periodic leg movements during sleep in healthy subjects. Sleep Med 2013, 14, 555–561. [Google Scholar] [CrossRef]
- Winkelman, J.W. The evoked heart rate response to periodic leg movements of sleep. Sleep 1999, 22, 575–580. [Google Scholar] [CrossRef]
- Yumino, D.; Wang, H.; Floras, J.S.; Newton, G.E.; Mak, S.; Ruttanaumpawan, P.; Parker, J.D.; Bradley, T.D. Relation of periodic leg movements during sleep and mortality in patients with systolic heart failure. Am J Cardiol 2011, 107, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Frauscher, B.; Gabelia, D.; Mitterling, T.; Biermayr, M.; Bregler, D.; Ehrmann, L.; Ulmer, H.; Hogl, B. Motor events during healthy sleep: a quantitative polysomnographic study. Sleep 2014, 37, 763-773, 773A-773B. [Google Scholar] [CrossRef]
- Scofield, H.; Roth, T.; Drake, C. Periodic limb movements during sleep: population prevalence, clinical correlates, and racial differences. Sleep 2008, 31, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Kolla, B.P.; Mansukhani, M.P.; Bostwick, J.M. The influence of antidepressants on restless legs syndrome and periodic limb movements: A systematic review. Sleep Med Rev 2018, 38, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Song, X.; Chen, R.; Chen, S.; Lu, B. Exploring the Top 50 Drugs Associated with Restless Legs Syndrome Based on the FDA Data from 2004 to 2024. Nat Sci Sleep 2025, 17, 929–946. [Google Scholar] [CrossRef]
- Zhu, X.Y.; Wu, T.T.; Wang, H.M.; Li, X.; Ni, L.Y.; Chen, T.J.; Qiu, M.Y.; Shen, J.; Liu, T.; Ondo, W.G.; et al. Correlates of Nonanemic Iron Deficiency in Restless Legs Syndrome. Front Neurol 2020, 11, 298. [Google Scholar] [CrossRef]
- Lesage, S.; Hening, W.A. The restless legs syndrome and periodic limb movement disorder: a review of management. Semin Neurol 2004, 24, 249–259. [Google Scholar] [CrossRef]
- Ferre, S.; Garcia-Borreguero, D.; Allen, R.P.; Earley, C.J. New Insights into the Neurobiology of Restless Legs Syndrome. Neuroscientist 2019, 25, 113–125. [Google Scholar] [CrossRef]
- Liu, Y.; Du, Q.; Jiang, Y. Prevalence of restless legs syndrome in maintenance hemodialysis patients: A systematic review and meta-analysis. Sleep Med 2024, 114, 15–23. [Google Scholar] [CrossRef]
- Winkelman, J.W.; Wipper, B.; Zackon, J. Long-term Safety, Dose Stability, and Efficacy of Opioids for Patients With Restless Legs Syndrome in the National RLS Opioid Registry. Neurology 2023, 100, e1520–e1528. [Google Scholar] [CrossRef]
- Winkelman, J.W. Opioids and restless legs syndrome: a double-edged sword. J Clin Sleep Med 2023, 19, 641–642. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Jimenez, F.J.; Alonso-Navarro, H.; Garcia-Martin, E.; Agundez, J.A.G. Association between restless legs syndrome and peripheral neuropathy: A systematic review and meta-analysis. Eur J Neurol 2021, 28, 2423–2442. [Google Scholar] [CrossRef] [PubMed]
- Ozdogar, A.T.; Kalron, A. Restless legs syndrome in people with multiple sclerosis: An updated systematic review and meta-analyses. Mult Scler Relat Disord 2021, 56, 103275. [Google Scholar] [CrossRef] [PubMed]
- Stefansson, H.; Rye, D.B.; Hicks, A.; Petursson, H.; Ingason, A.; Thorgeirsson, T.E.; Palsson, S.; Sigmundsson, T.; Sigurdsson, A.P.; Eiriksdottir, I.; et al. A genetic risk factor for periodic limb movements in sleep. N Engl J Med 2007, 357, 639–647. [Google Scholar] [CrossRef]
- Maggi, G.; Barone, A.; Mastromarino, C.; Santangelo, G.; Vitale, C. Prevalence and clinical profile of patients with restless legs syndrome in Parkinson's disease: A meta-analysis. Sleep Med 2024, 121, 275–286. [Google Scholar] [CrossRef]
- Ohayon, M.M.; O'Hara, R.; Vitiello, M.V. Epidemiology of restless legs syndrome: a synthesis of the literature. Sleep Med Rev 2012, 16, 283–295. [Google Scholar] [CrossRef]
- Walters, A.S.; Li, Y.; Koo, B.B.; Ondo, W.G.; Weinstock, L.B.; Champion, D.; Afrin, L.B.; Karroum, E.G.; Bagai, K.; Spruyt, K. Review of the role of the endogenous opioid and melanocortin systems in the restless legs syndrome. Brain 2024, 147, 26–38. [Google Scholar] [CrossRef]
- Srivali, N.; Thongprayoon, C.; Cheungpasitporn, W.; Zinchuk, A.; Koo, B.B. Impact of continuous positive airway pressure therapy on restless legs syndrome in patients with coexistent obstructive sleep apnea: A qualitative systematic review. J Clin Neurosci 2025, 133, 111075. [Google Scholar] [CrossRef]
- Winter, A.C.; Schurks, M.; Glynn, R.J.; Buring, J.E.; Gaziano, J.M.; Berger, K.; Kurth, T. Vascular risk factors, cardiovascular disease, and restless legs syndrome in women. Am J Med 2013, 126, 220-227, 227 e221-222. [Google Scholar] [CrossRef]
- Kim, B.; Kim, T.Y.; Choi, E.J.; Lee, M.; Kim, W.; Lee, S.A. Restless legs syndrome in patients with obstructive sleep apnea: Association between apnea severity and symptoms of depression, insomnia, and daytime sleepiness. Sleep Med 2024, 117, 40–45. [Google Scholar] [CrossRef]
- Auerbach, M.; DeLoughery, T.G.; Tirnauer, J.S. Iron Deficiency in Adults: A Review. JAMA 2025, 333, 1813–1823. [Google Scholar] [CrossRef]
- Allen, R.P.; Auerbach, S.; Bahrain, H.; Auerbach, M.; Earley, C.J. The prevalence and impact of restless legs syndrome on patients with iron deficiency anemia. Am J Hematol 2013, 88, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, G.; Li, X.; Galantucci, S.; Filippi, M.; Cho, Y.W. Brain imaging and networks in restless legs syndrome. Sleep Med 2017, 31, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.J.; Shi, L.; Bao, Y.P.; Sun, Y.K.; Lin, X.; Que, J.Y.; Vitiello, M.V.; Zhou, Y.X.; Wang, Y.Q.; Lu, L. Prevalence of restless legs syndrome during pregnancy: A systematic review and meta-analysis. Sleep Med Rev 2018, 40, 43–54. [Google Scholar] [CrossRef] [PubMed]
- Lepuzanovic, M.; Sinanovic, O.; Aziraj-Smajic, V.; Kapic, D.; Basagic, E.; Muftic, M. Prevalence of restless legs syndrome during pregnancy and postpartum period. J Perinat Med 2024, 52, 852–857. [Google Scholar] [CrossRef]
- Mackie, S.E.; McHugh, R.K.; McDermott, K.; Griffin, M.L.; Winkelman, J.W.; Weiss, R.D. Prevalence of restless legs syndrome during detoxification from alcohol and opioids. J Subst Abuse Treat 2017, 73, 35–39. [Google Scholar] [CrossRef]
- Walters, A.S.; Li, Y.; Karroum, E.G.; Champion, D.; Weinstock, L.B.; Bagai, K.; Afrin, L.B.; Spruyt, K. Exploring the role of the endogenous opiate system in the pathogenesis of anemia in an opiate receptor knock-out model of Restless Legs Syndrome. Med Hypotheses 2022, 167. [Google Scholar] [CrossRef]
- Mir, F.A.; Jha, S.K.; Nehs, C.J. Noradrenergic dysregulation in the locus coeruleus: Implications for neuropsychiatric disease pathophysiology- A systematic review. Neurosci Biobehav Rev 2025, 179, 106442. [Google Scholar] [CrossRef]
- Best, C.; Sayegh, A.L.C.; Janzen, A.; Oertel, W.H.; Kramer, H.H. RLS shows increased resting state sympathetic activity and decreased sympathetic response capability. J Neurol 2025, 272, 545. [Google Scholar] [CrossRef]
- Boutitah-Benyaich, I.; Eixarch, H.; Villacieros-Alvarez, J.; Hervera, A.; Cobo-Calvo, A.; Montalban, X.; Espejo, C. Multiple sclerosis: molecular pathogenesis and therapeutic intervention. Signal Transduct Target Ther 2025, 10, 324. [Google Scholar] [CrossRef]
- Winkelmann, J.; Wetter, T.C.; Collado-Seidel, V.; Gasser, T.; Dichgans, M.; Yassouridis, A.; Trenkwalder, C. Clinical characteristics and frequency of the hereditary restless legs syndrome in a population of 300 patients. Sleep 2000, 23, 597–602. [Google Scholar] [CrossRef]
- Xiong, L.; Jang, K.; Montplaisir, J.; Levchenko, A.; Thibodeau, P.; Gaspar, C.; Turecki, G.; Rouleau, G.A. Canadian restless legs syndrome twin study. Neurology 2007, 68, 1631–1633. [Google Scholar] [CrossRef] [PubMed]
- Schormair, B.; Zhao, C.; Bell, S.; Didriksen, M.; Nawaz, M.S.; Schandra, N.; Stefani, A.; Hogl, B.; Dauvilliers, Y.; Bachmann, C.G.; et al. Genome-wide meta-analyses of restless legs syndrome yield insights into genetic architecture, disease biology and risk prediction. Nat Genet 2024, 56, 1090–1099. [Google Scholar] [CrossRef] [PubMed]
- Lane, J.M.; Liang, J.; Vlasac, I.; Anderson, S.G.; Bechtold, D.A.; Bowden, J.; Emsley, R.; Gill, S.; Little, M.A.; Luik, A.I.; et al. Genome-wide association analyses of sleep disturbance traits identify new loci and highlight shared genetics with neuropsychiatric and metabolic traits. Nat Genet 2017, 49, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Salminen, A.V.; Lam, D.D.; Winkelmann, J. Role of MEIS1 in restless legs syndrome: From GWAS to functional studies in mice. Adv Pharmacol 2019, 84, 175–184. [Google Scholar] [CrossRef]
- Sarayloo, F.; Dion, P.A.; Rouleau, G.A. MEIS1 and Restless Legs Syndrome: A Comprehensive Review. Front Neurol 2019, 10, 935. [Google Scholar] [CrossRef]
- Sorensen, E.; Grau, K.; Berg, T.; Simonsen, A.C.; Magnussen, K.; Erikstrup, C.; Hansen, M.B.; Ullum, H. A genetic risk factor for low serum ferritin levels in Danish blood donors. Transfusion 2012, 52, 2585–2589. [Google Scholar] [CrossRef]
- Earley, C.J.; P, B.B.; Horska, A.; Allen, R.P. MRI-determined regional brain iron concentrations in early- and late-onset restless legs syndrome. Sleep Med 2006, 7, 458–461. [Google Scholar] [CrossRef]
- Godau, J.; Schweitzer, K.J.; Liepelt, I.; Gerloff, C.; Berg, D. Substantia nigra hypoechogenicity: definition and findings in restless legs syndrome. Mov Disord 2007, 22, 187–192. [Google Scholar] [CrossRef]
- Mizuno, S.; Mihara, T.; Miyaoka, T.; Inagaki, T.; Horiguchi, J. CSF iron, ferritin and transferrin levels in restless legs syndrome. J Sleep Res 2005, 14, 43–47. [Google Scholar] [CrossRef]
- Connor, J.R.; Ponnuru, P.; Wang, X.S.; Patton, S.M.; Allen, R.P.; Earley, C.J. Profile of altered brain iron acquisition in restless legs syndrome. Brain 2011, 134, 959–968. [Google Scholar] [CrossRef]
- Mogavero, M.P.; Lanza, G.; Quercia, A.; Palmigiano, A.; Bruni, O.; Ferri, R. Evaluating the adenosine hypothesis of restless legs syndrome and its implications for current and future treatment strategies. Expert Rev Neurother 2026, 26, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.P.; Earley, C.J.; Jones, B.C.; Unger, E.L. Iron-deficiency and dopaminergic treatment effects on RLS-Like behaviors of an animal model with the brain iron deficiency pattern of the restless legs syndrome. Sleep Med 2020, 71, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Earley, C.J.; Kuwabara, H.; Wong, D.F.; Gamaldo, C.; Salas, R.E.; Brasic, J.R.; Ravert, H.T.; Dannals, R.F.; Allen, R.P. Increased synaptic dopamine in the putamen in restless legs syndrome. Sleep 2013, 36, 51–57. [Google Scholar] [CrossRef]
- Earley, C.J.; Hyland, K.; Allen, R.P. Circadian changes in CSF dopaminergic measures in restless legs syndrome. Sleep Med 2006, 7, 263–268. [Google Scholar] [CrossRef]
- Park, K.M.; Kim, K.T.; Kang, K.W.; Park, J.A.; Seo, J.G.; Kim, J.; Chang, H.; Kim, E.Y.; Cho, Y.W.; Society, R.L.S.S.o.t.K.S.R. Alterations of Functional Connectivity in Patients With Restless Legs Syndrome. J Clin Neurol 2022, 18, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Quiroz, C.; Gulyani, S.; Ruiqian, W.; Bonaventura, J.; Cutler, R.; Pearson, V.; Allen, R.P.; Earley, C.J.; Mattson, M.P.; Ferre, S. Adenosine receptors as markers of brain iron deficiency: Implications for Restless Legs Syndrome. Neuropharmacology 2016, 111, 160–168. [Google Scholar] [CrossRef]
- Koo, B.B.; Abdelfattah, A.; Eysa, A.; Lu, L. The Melanocortin and Endorphin Neuropeptides in Patients with Restless Legs Syndrome. Ann Neurol 2024, 95, 688–699. [Google Scholar] [CrossRef]
- Allen, R.P.; Barker, P.B.; Horska, A.; Earley, C.J. Thalamic glutamate/glutamine in restless legs syndrome: increased and related to disturbed sleep. Neurology 2013, 80, 2028–2034. [Google Scholar] [CrossRef]
- Didriksen, M.; Rigas, A.S.; Allen, R.P.; Burchell, B.J.; Di Angelantonio, E.; Nielsen, M.H.; Jennum, P.; Werge, T.; Erikstrup, C.; Pedersen, O.B.; et al. Prevalence of restless legs syndrome and associated factors in an otherwise healthy population: results from the Danish Blood Donor Study. Sleep Med 2017, 36, 55–61. [Google Scholar] [CrossRef]
- Aukerman, M.M.; Aukerman, D.; Bayard, M.; Tudiver, F.; Thorp, L.; Bailey, B. Exercise and restless legs syndrome: a randomized controlled trial. J Am Board Fam Med 2006, 19, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Cederberg, K.L.J.; Sikes, E.M.; Mignot, E. Perceptions of exercise and restless legs syndrome: Results from a nationwide survey. J Sleep Res 2024, 33, e13980. [Google Scholar] [CrossRef] [PubMed]
- Tolkien, Z.; Stecher, L.; Mander, A.P.; Pereira, D.I.; Powell, J.J. Ferrous sulfate supplementation causes significant gastrointestinal side-effects in adults: a systematic review and meta-analysis. PLoS One 2015, 10, e0117383. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Malo, C.; Garcia-Borreguero, D.; Silber, M.H. Efficacy and safety of intravenous iron in patients with restless legs syndrome with normal serum ferritin levels: a stratified subanalysis. J Clin Sleep Med 2025, 21, 2023–2029. [Google Scholar] [CrossRef]
- Trenkwalder, C.; Winkelmann, J.; Oertel, W.; Virgin, G.; Roubert, B.; Mezzacasa, A.; Investigators, F.-R.S. Ferric carboxymaltose in patients with restless legs syndrome and nonanemic iron deficiency: A randomized trial. Mov Disord 2017, 32, 1478–1482. [Google Scholar] [CrossRef]
- Short, V.; Allen, R.; Earley, C.J.; Bahrain, H.; Rineer, S.; Kashi, K.; Gerb, J.; Auerbach, M. A randomized double-blind pilot study to evaluate the efficacy, safety, and tolerability of intravenous iron versus oral iron for the treatment of restless legs syndrome in patients with iron deficiency anemia. Am J Hematol 2024, 99, 1077–1083. [Google Scholar] [CrossRef]
- Garcia-Borreguero, D.; Larrosa, O.; de la Llave, Y.; Verger, K.; Masramon, X.; Hernandez, G. Treatment of restless legs syndrome with gabapentin: a double-blind, cross-over study. Neurology 2002, 59, 1573–1579. [Google Scholar] [CrossRef]
- Qin, Z.; Zhu, Y.; Shi, D.D.; Chen, R.; Li, S.; Wu, J. The gap between statistical and clinical significance: time to pay attention to clinical relevance in patient-reported outcome measures of insomnia. BMC Med Res Methodol 2024, 24, 177. [Google Scholar] [CrossRef]
- Inoue, Y.; Hirata, K.; Kuroda, K.; Fujita, M.; Shimizu, T.; Emura, N.; Uchimura, N.; Kagimura, T.; Sha, K.; Nozawa, T. Efficacy and safety of pramipexole in Japanese patients with primary restless legs syndrome: A polysomnographic randomized, double-blind, placebo-controlled study. Sleep Med 2010, 11, 11–16. [Google Scholar] [CrossRef]
- Scholz, H.; Trenkwalder, C.; Kohnen, R.; Riemann, D.; Kriston, L.; Hornyak, M. Dopamine agonists for restless legs syndrome. Cochrane Database Syst Rev 2011, CD006009. [Google Scholar] [CrossRef]
- Garcia-Borreguero, D.; Allen, R.P.; Kohnen, R.; Hogl, B.; Trenkwalder, C.; Oertel, W.; Hening, W.A.; Paulus, W.; Rye, D.; Walters, A.; et al. Diagnostic standards for dopaminergic augmentation of restless legs syndrome: report from a World Association of Sleep Medicine-International Restless Legs Syndrome Study Group consensus conference at the Max Planck Institute. Sleep Med 2007, 8, 520–530. [Google Scholar] [CrossRef]
- Silver, N.; Allen, R.P.; Senerth, J.; Earley, C.J. A 10-year, longitudinal assessment of dopamine agonists and methadone in the treatment of restless legs syndrome. Sleep Med 2011, 12, 440–444. [Google Scholar] [CrossRef]
- Cornelius, J.R.; Tippmann-Peikert, M.; Slocumb, N.L.; Frerichs, C.F.; Silber, M.H. Impulse control disorders with the use of dopaminergic agents in restless legs syndrome: a case-control study. Sleep 2010, 33, 81–87. [Google Scholar] [CrossRef]
- Iftikhar, I.H.; Alghothani, L.; Trotti, L.M. Gabapentin enacarbil, pregabalin and rotigotine are equally effective in restless legs syndrome: a comparative meta-analysis. Eur J Neurol 2017, 24, 1446–1456. [Google Scholar] [CrossRef]
- Trenkwalder, C.; Benes, H.; Grote, L.; Garcia-Borreguero, D.; Hogl, B.; Hopp, M.; Bosse, B.; Oksche, A.; Reimer, K.; Winkelmann, J.; et al. Prolonged release oxycodone-naloxone for treatment of severe restless legs syndrome after failure of previous treatment: a double-blind, randomised, placebo-controlled trial with an open-label extension. Lancet Neurol 2013, 12, 1141–1150. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Borreguero, D.; Silber, M.H.; Winkelman, J.W.; Hogl, B.; Bainbridge, J.; Buchfuhrer, M.; Hadjigeorgiou, G.; Inoue, Y.; Manconi, M.; Oertel, W.; et al. Guidelines for the first-line treatment of restless legs syndrome/Willis-Ekbom disease, prevention and treatment of dopaminergic augmentation: a combined task force of the IRLSSG, EURLSSG, and the RLS-foundation. Sleep Med 2016, 21, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bogan, R.K.; Roy, A.; Kram, J.; Ojile, J.; Rosenberg, R.; Hudson, J.D.; Scheuller, H.S.; Winkelman, J.W.; Charlesworth, J.D. Efficacy and safety of tonic motor activation (TOMAC) for medication-refractory restless legs syndrome: a randomized clinical trial. Sleep 2023, 46. [Google Scholar] [CrossRef] [PubMed]
- Nardone, R.; Sebastianelli, L.; Versace, V.; Brigo, F.; Golaszewski, S.; Pucks-Faes, E.; Saltuari, L.; Trinka, E. Contribution of transcranial magnetic stimulation in restless legs syndrome: pathophysiological insights and therapeutical approaches. Sleep Med 2020, 71, 124–134. [Google Scholar] [CrossRef]
- Winkelman, J.W. Treating Severe Refractory and Augmented Restless Legs Syndrome. Chest 2022, 162, 693–700. [Google Scholar] [CrossRef]
- Nirenberg, M.J. Dopamine agonist withdrawal syndrome: implications for patient care. Drugs Aging 2013, 30, 587–592. [Google Scholar] [CrossRef]
- Li, Y.; Mirzaei, F.; O'Reilly, E.J.; Winkelman, J.; Malhotra, A.; Okereke, O.I.; Ascherio, A.; Gao, X. Prospective study of restless legs syndrome and risk of depression in women. Am J Epidemiol 2012, 176, 279–288. [Google Scholar] [CrossRef]
- Wilt, T.J.; MacDonald, R.; Ouellette, J.; Khawaja, I.S.; Rutks, I.; Butler, M.; Fink, H.A. Pharmacologic therapy for primary restless legs syndrome: a systematic review and meta-analysis. JAMA Intern Med 2013, 173, 496–505. [Google Scholar] [CrossRef] [PubMed]
- Ondo, W. Restless Legs Syndrome "Patient Odyssey" survey of disease burden on patient and spouses/partners. Sleep Med 2018, 47, 51–53. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.S.; Kim, T.; Lee, S.; Jeon, H.J.; Bang, Y.R.; Yoon, I.Y. Symptom Severity of Restless Legs Syndrome Predicts Its Clinical Course. Am J Med 2016, 129, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, S.; Na, M.; Winkelman, J.W.; Ba, D.; Liu, C.F.; Liu, G.; Gao, X. Association of Restless Legs Syndrome With Risk of Suicide and Self-harm. JAMA Netw Open 2019, 2, e199966. [Google Scholar] [CrossRef]
- Winkelman, J.W.; Chertow, G.M.; Lazarus, J.M. Restless legs syndrome in end-stage renal disease. Am J Kidney Dis 1996, 28, 372–378. [Google Scholar] [CrossRef]
- Gao, X.; Ba, D.M.; Bagai, K.; Liu, G.; Ma, C.; Walters, A.S. Treating Restless Legs Syndrome Was Associated With Low Risk of Cardiovascular Disease: A Cohort Study With 3.4 Years of Follow-Up. J Am Heart Assoc 2021, 10, e018674. [Google Scholar] [CrossRef]
- Muhoberac, B.B.; Vidal, R. Iron, Ferritin, Hereditary Ferritinopathy, and Neurodegeneration. Front Neurosci 2019, 13, 1195. [Google Scholar] [CrossRef]
- Chifman, J.; Laubenbacher, R.; Torti, S.V. A systems biology approach to iron metabolism. Adv Exp Med Biol 2014, 844, 201–225. [Google Scholar] [CrossRef]
- McCarthy, R.C.; Kosman, D.J. Iron transport across the blood-brain barrier: development, neurovascular regulation and cerebral amyloid angiopathy. Cell Mol Life Sci 2015, 72, 709–727. [Google Scholar] [CrossRef]
- Jomova, K.; Alomar, S.Y.; Valko, R.; Nepovimova, E.; Kuca, K.; Valko, M. The role of redox-active iron, copper, manganese, and redox-inactive zinc in toxicity, oxidative stress, and human diseases. EXCLI J 2025, 24, 880–954. [Google Scholar] [CrossRef]
- Feng, S.; Tang, D.; Wang, Y.; Li, X.; Bao, H.; Tang, C.; Dong, X.; Li, X.; Yang, Q.; Yan, Y.; et al. The mechanism of ferroptosis and its related diseases. Mol Biomed 2023, 4, 33. [Google Scholar] [CrossRef]
- Read, A.D.; Bentley, R.E.; Archer, S.L.; Dunham-Snary, K.J. Mitochondrial iron-sulfur clusters: Structure, function, and an emerging role in vascular biology. Redox Biol 2021, 47, 102164. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Shen, Q.; Liu, Y.; Zhang, Y.; Sun, L.; Ma, X.; Song, N.; Xie, J. Homeostasis and metabolism of iron and other metal ions in neurodegenerative diseases. Signal Transduct Target Ther 2025, 10, 31. [Google Scholar] [CrossRef] [PubMed]
- Salvador, G.A. Iron in neuronal function and dysfunction. Biofactors 2010, 36, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Bueno-Carrasco, M.T.; Cuellar, J.; Flydal, M.I.; Santiago, C.; Krakenes, T.A.; Kleppe, R.; Lopez-Blanco, J.R.; Marcilla, M.; Teigen, K.; Alvira, S.; et al. Structural mechanism for tyrosine hydroxylase inhibition by dopamine and reactivation by Ser40 phosphorylation. Nat Commun 2022, 13, 74. [Google Scholar] [CrossRef]
- Ortega, R.; Cloetens, P.; Deves, G.; Carmona, A.; Bohic, S. Iron storage within dopamine neurovesicles revealed by chemical nano-imaging. PLoS One 2007, 2, e925. [Google Scholar] [CrossRef]
- Rodrigues, M.S.; Ferreira, S.G.; Quiroz, C.; Earley, C.J.; Garcia-Borreguero, D.; Cunha, R.A.; Ciruela, F.; Kofalvi, A.; Ferre, S. Brain Iron Deficiency Changes the Stoichiometry of Adenosine Receptor Subtypes in Cortico-Striatal Terminals: Implications for Restless Legs Syndrome. Molecules 2022, 27. [Google Scholar] [CrossRef]
- McAllum, E.J.; Hare, D.J.; Volitakis, I.; McLean, C.A.; Bush, A.I.; Finkelstein, D.I.; Roberts, B.R. Regional iron distribution and soluble ferroprotein profiles in the healthy human brain. Prog Neurobiol 2020, 186, 101744. [Google Scholar] [CrossRef]
- Snyder, A.M.; Connor, J.R. Iron, the substantia nigra and related neurological disorders. Biochim Biophys Acta 2009, 1790, 606–614. [Google Scholar] [CrossRef]
- Wade, Q.W.; Connor, J.R. What Does Iron Mean to an Oligodendrocyte? Glia 2025, 73, 1784–1804. [Google Scholar] [CrossRef]
- McCarthy, R.C.; Sosa, J.C.; Gardeck, A.M.; Baez, A.S.; Lee, C.H.; Wessling-Resnick, M. Inflammation-induced iron transport and metabolism by brain microglia. J Biol Chem 2018, 293, 7853–7863. [Google Scholar] [CrossRef] [PubMed]
- Dong, K.; Liu, B.; Cheng, G.; Li, Y.; Xie, F.; Zhang, J.; Qian, L. Stress-Induced Dysregulation of Brain Iron Metabolism and Its Links to Neurological Disorders. Biology (Basel) 2025, 14. [Google Scholar] [CrossRef] [PubMed]
- Cheng, R.; Dhorajia, V.V.; Kim, J.; Kim, Y. Mitochondrial iron metabolism and neurodegenerative diseases. Neurotoxicology 2022, 88, 88–101. [Google Scholar] [CrossRef] [PubMed]
- Minetti, A.; Montagni, E.; Meneghetti, N.; Macchi, F.; Coulomb, E.; Martello, A.; Tiberi, A.; Capsoni, S.; Mazzoni, A.; Allegra Mascaro, A.L.; et al. Parkinsonism disrupts cortical function by dysregulating oscillatory, network and synaptic activity of parvalbumin positive interneurons. NPJ Parkinsons Dis 2025, 11, 194. [Google Scholar] [CrossRef]
- Berridge, C.W. Noradrenergic modulation of arousal. Brain Res Rev 2008, 58, 1–17. [Google Scholar] [CrossRef]
- Liu, Z.; Guan, R.; Pan, L. Exploration of restless legs syndrome under the new concept: A review. Medicine (Baltimore) 2022, 101, e32324. [Google Scholar] [CrossRef]
- Allen, R.P.; Picchietti, D.; Hening, W.A.; Trenkwalder, C.; Walters, A.S.; Montplaisi, J.; Restless Legs Syndrome, D.; Epidemiology workshop at the National Institutes of; H; International Restless Legs Syndrome Study; G. Restless legs syndrome: diagnostic criteria, special considerations, and epidemiology. A report from the restless legs syndrome diagnosis and epidemiology workshop at the National Institutes of Health. Sleep Med 2003, 4, 101–119. [Google Scholar] [CrossRef]
- Connor, J.R.; Wang, X.S.; Allen, R.P.; Beard, J.L.; Wiesinger, J.A.; Felt, B.T.; Earley, C.J. Altered dopaminergic profile in the putamen and substantia nigra in restless leg syndrome. Brain 2009, 132, 2403–2412. [Google Scholar] [CrossRef]
- Beliveau, V.; Stefani, A.; Birkl, C.; Kremser, C.; Gizewski, E.R.; Hogl, B.; Scherfler, C. Revisiting brain iron deficiency in restless legs syndrome using magnetic resonance imaging. Neuroimage Clin 2022, 34, 103024. [Google Scholar] [CrossRef]
- Akcimen, F.; Chia, R.; Saez-Atienzar, S.; Ruffo, P.; Rasheed, M.; Ross, J.P.; Liao, C.; Ray, A.; Dion, P.A.; Scholz, S.W.; et al. Genomic Analysis Identifies Risk Factors in Restless Legs Syndrome. Ann Neurol 2024, 96, 994–1005. [Google Scholar] [CrossRef]
- Catoire, H.; Sarayloo, F.; Mourabit Amari, K.; Apuzzo, S.; Grant, A.; Rochefort, D.; Xiong, L.; Montplaisir, J.; Earley, C.J.; Turecki, G.; et al. A direct interaction between two Restless Legs Syndrome predisposing genes: MEIS1 and SKOR1. Sci Rep 2018, 8, 12173. [Google Scholar] [CrossRef]
- An, T.; Sun, H.; Yuan, L.; Wu, X.; Lu, B. Associations of anxiety and depression with restless leg syndrome: a systematic review and meta-analysis. Front Neurol 2024, 15, 1366839. [Google Scholar] [CrossRef]
- Tang, M.; Sun, Q.; Zhang, Y.; Li, H.; Wang, D.; Wang, Y.; Wang, Z. Circadian rhythm in restless legs syndrome. Front Neurol 2023, 14, 1105463. [Google Scholar] [CrossRef]
- Verwey, M.; Dhir, S.; Amir, S. Circadian influences on dopamine circuits of the brain: regulation of striatal rhythms of clock gene expression and implications for psychopathology and disease. F1000Res 2016, 5. [Google Scholar] [CrossRef]
- Aggarwal, A.; Negrean, A.; Chen, Y.; Iyer, R.; Reep, D.; Liu, A.; Palutla, A.; Xie, M.E.; MacLennan, B.J.; Hagihara, K.M.; et al. Glutamate indicators with increased sensitivity and tailored deactivation rates. bioRxiv 2025. [Google Scholar] [CrossRef]
- Caestecker, S.; Lescrauwaet, E.; Boon, P.; Carrette, E.; Raedt, R.; Vonck, K. The Locus Coeruleus-Noradrenergic System in the Healthy and Diseased Brain: A Narrative Review. Eur J Neurol 2025, 32, e70337. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




