Submitted:
13 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
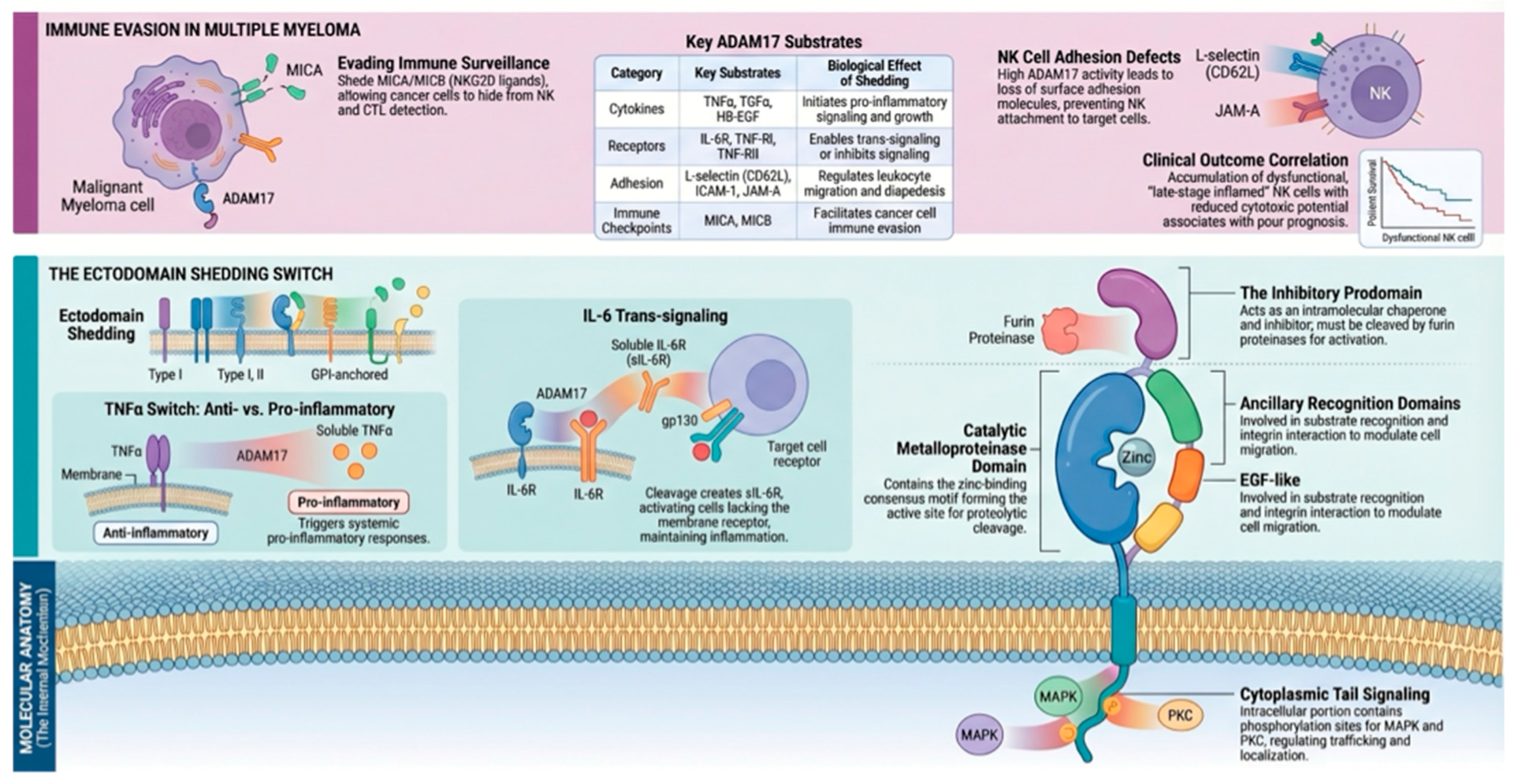
Introduction
Microenvironmental Dependency and Proteolytic Control
Angiogenesis: A Prototypical ADAM17-Driven Circuit
Inflammatory Amplification and IL-6 Trans-Signaling
Immune Dysfunction: A Context-Dependent Role
ADAM17 in Adhesion Remodeling, Dissemination, and Biomarker Potential
A Unifying Model: The Myeloma “Sheddome”
Therapeutic Implications and Challenges
Concluding Perspective
Author Contributions
Acknowledgement and Funding
Data Availability Statement
Conflict of Interest
References
- Black, R.A.; Rauch, C.T.; Kozlosky, C.J.; Peschon, J.J.; Slack, J.L.; Wolfson, M.F.; Castner, B.J.; Stocking, K.L.; Reddy, P.; Srinivasan, S.; Nelson, N.; Boiani, N.; Schooley, K.A.; Gerhart, M.; Davis, R.; Fitzner, J.N.; Johnson, R.S.; Paxton, R.J.; March, C.J.; Cerretti, D.P. A metalloproteinase disintegrin that releases tumour-necrosis factor-alpha from cells. Nature 1997, 385(6618), 729–33. [Google Scholar] [CrossRef] [PubMed]
- Gooz, M. ADAM-17: the enzyme that does it all. Crit Rev Biochem Mol Biol. 2010, 45(2), 146–69. [Google Scholar] [CrossRef]
- Scheller, J.; Chalaris, A.; Garbers, C.; Rose-John, S. ADAM17: a molecular switch to control inflammation and tissue regeneration. Trends Immunol 2011, 32(8), 380–7. [Google Scholar] [CrossRef]
- Solimando, A.G.; Malerba, E.; Leone, P.; Prete, M.; Terragna, C.; Cavo, M.; Racanelli, V. Drug resistance in multiple myeloma: Soldiers and weapons in the bone marrow niche. Front Oncol. 2022, 12, 973836. [Google Scholar] [CrossRef]
- Gullà, A.; Dhodapkar, M.V.; Einsele, H.; Raab, M.S.; Solimando, A.G.; Botta, C.; Turi, M.; Sester, L.S.; Portuguese, A.J.; Steinbrunn, T.; Anderson, K.C. Rethinking Multiple Myeloma Treatment: The Biological and Clinical Insights Guiding Immune-Based Combinations. Blood Cancer Discov. 2026, 7(2), 176–194. [Google Scholar] [CrossRef]
- Evers, M.; Stühmer, T.; Schreder, M.; Steinbrunn, T.; Rudelius, M.; Jundt, F.; Ebert, R.; Hartmann, T.N.; Bargou, R.C.; Rosenwald, A.; Leich, E. Association of ADAM family members with proliferation signaling and disease progression in multiple myeloma. Blood Cancer J 2024, 14(1), 156. [Google Scholar] [CrossRef]
- Evers, M.; Schreder, M.; Stühmer, T.; Jundt, F.; Ebert, R.; Hartmann, T.N.; Altenbuchinger, M.; Rudelius, M.; Kuric, M.; Rindt, W.D.; Steinbrunn, T.; Langer, C.; Heredia-Guerrero, S.C.; Einsele, H.; Bargou, R.C.; Rosenwald, A.; Leich, E. Prognostic value of extracellular matrix gene mutations and expression in multiple myeloma. Blood Cancer J 2023, 13(1), 43. [Google Scholar] [CrossRef]
- Marchica, V.; Toscani, D.; Corcione, A.; Bolzoni, M.; Storti, P.; Vescovini, R.; Ferretti, E.; Dalla Palma, B.; Vicario, E.; Accardi, F.; Mancini, C.; Martella, E.; Ribatti, D.; Vacca, A.; Pistoia, V.; Giuliani, N. Bone Marrow CX3CL1/Fractalkine is a New Player of the Pro-Angiogenic Microenvironment in Multiple Myeloma Patients. Cancers (Basel) 2019, 11(3), 321. [Google Scholar] [CrossRef] [PubMed]
- Solimando, A.G.; Da Vià, M.C.; Leone, P.; Borrelli, P.; Croci, G.A.; Tabares, P.; Brandl, A.; Di Lernia, G.; Bianchi, F.P.; Tafuri, S.; Steinbrunn, T.; Balduini, A.; Melaccio, A.; De Summa, S.; Argentiero, A.; Rauert-Wunderlich, H.; Frassanito, M.A.; Ditonno, P.; Henke, E.; Klapper, W.; Ria, R.; Terragna, C.; Rasche, L.; Rosenwald, A.; Kortüm, M.K.; Cavo, M.; Ribatti, D.; Racanelli, V.; Einsele, H.; Vacca, A.; Beilhack, A. Halting the vicious cycle within the multiple myeloma ecosystem: blocking JAM-A on bone marrow endothelial cells restores angiogenic homeostasis and suppresses tumor progression. Haematologica 2021, 106(7), 1943–1956. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, N.; Rose-John, S. ADAM17 Activity and IL-6 Trans-Signaling in Inflammation and Cancer. Cancers (Basel) 2019, 11(11), 1736. [Google Scholar] [CrossRef]
- Schumacher, N.; Rose-John, S. ADAM17 orchestrates Interleukin-6, TNFα and EGF-R signaling in inflammation and cancer. Biochim Biophys Acta Mol Cell Res. 2022, 1869(1), 119141. [Google Scholar] [CrossRef]
- Wang, Y.; Herrera, A.H.; Li, Y.; Belani, K.K.; Walcheck, B. Regulation of mature ADAM17 by redox agents for L-selectin shedding. J Immunol. 2009, 182(4), 2449–57. [Google Scholar] [CrossRef]
- Qu, J.; Li, Y.; Li, C.; Wu, D.; Cao, Y.; Xiao, S.; Bao, X.; Li, Q.; Chen, Z.; Chen, L. Longitudinal profiling of serum ADAM17 across clinical stages in multiple myeloma: a dynamic biomarker and its association with T cell alterations. Front Mol Biosci. 2026, 13, 1768193. [Google Scholar] [CrossRef]
- Lauko, A.; Mu, Z.; Gutmann, D.H.; Naik, U.P.; Lathia, J.D. Junctional Adhesion Molecules in Cancer: A Paradigm for the Diverse Functions of Cell-Cell Interactions in Tumor Progression. Cancer Res. 2020, 80(22), 4878–4885. [Google Scholar] [CrossRef]
- Koenen, R.R.; Pruessmeyer, J.; Soehnlein, O.; Fraemohs, L.; Zernecke, A.; Schwarz, N.; Reiss, K.; Sarabi, A.; Lindbom, L.; Hackeng, T.M.; Weber, C.; Ludwig, A. Regulated release and functional modulation of junctional adhesion molecule A by disintegrin metalloproteinases. Blood 2009, 113(19), 4799–809. [Google Scholar] [CrossRef]
- Solimando, A.G.; Brandl, A.; Mattenheimer, K.; Graf, C.; Ritz, M.; Ruckdeschel, A.; Stühmer, T.; Mokhtari, Z.; Rudelius, M.; Dotterweich, J.; Bittrich, M.; Desantis, V.; Ebert, R.; Trerotoli, P.; Frassanito, M.A.; Rosenwald, A.; Vacca, A.; Einsele, H.; Jakob, F.; Beilhack, A. JAM-A as a prognostic factor and new therapeutic target in multiple myeloma. Leukemia 2018, 32(3), 736–743. [Google Scholar] [CrossRef]
- Kummer, D.; Steinbacher, T.; Thölmann, S.; Schwietzer, M.F.; Hartmann, C.; Horenkamp, S.; Demuth, S.; Peddibhotla, S.S.D.; Brinkmann, F.; Kemper, B.; Schnekenburger, J.; Brandt, M.; Betz, T.; Liashkovich, I.; Kouzel, I.U.; Shahin, V.; Corvaia, N.; Rottner, K.; Tarbashevich, K.; Raz, E.; Greune, L.; Schmidt, M.A.; Gerke, V.; Ebnet, K. A JAM-A-tetraspanin-αvβ5 integrin complex regulates contact inhibition of locomotion. J Cell Biol. 2022, 221(4), e202105147. [Google Scholar] [CrossRef] [PubMed]
- Moser-Katz, T.; Joseph, N.S.; Dhodapkar, M.V.; Lee, K.P.; Boise, L.H. Game of Bones: How Myeloma Manipulates Its Microenvironment. Front Oncol 2021, 10, 625199. [Google Scholar] [CrossRef] [PubMed]
- Martello, M.; Solli, V.; Mazzocchetti, G.; Solimando, A.G.; Bezzi, D.; Taurisano, B.; Kanapari, A.; Poletti, A.; Borsi, E.; Armuzzi, S.; Vigliotta, I.; Pistis, I.; Desantis, V.; Marzocchi, G.; Rizzello, I.; Pantani, L.; Mancuso, K.; Tacchetti, P.; Testoni, N.; Nanni, C.; Zamagni, E.; Cavo, M.; Terragna, C. High level of circulating cell-free tumor DNA at diagnosis correlates with disease spreading and defines multiple myeloma patients with poor prognosis. Blood Cancer J 2024, 14(1), 208. [Google Scholar] [CrossRef]
- Solimando, A.G.; Di Palma, F.; Desantis, V.; Vacca, A.; Svelto, M.; Pisani, F. Tunneling nanotubes between bone marrow stromal cells support transmitophagy and resistance to apoptosis in myeloma. Blood Cancer J 2025, 15(1), 3. [Google Scholar] [CrossRef] [PubMed]
- Pilcher, W.C.; Yao, L.; Gonzalez-Kozlova, E.; Pita-Juarez, Y.; Karagkouni, D.; Acharya, C.R.; Michaud, M.E.; Hamilton, M.; Nanda, S.; Song, Y.; Sato, K.; Wang, J.T.; Satpathy, S.; Ma, Y.; Schulman, J.; D'Souza, D.; Jayasinghe, R.G.; Ohlstrom, D.; Ferguson, K.E.; Cheloni, G.; Bakhtiari, M.; Pabustan, N.; Nie, K.; Foltz, J.A.; Saldarriaga, I.; Alaaeldin, R.; Lepisto, E.; Chen, R.; Fiala, M.A.; Thomas, B.E.; Cook, A.; Santos, J.V.D.; I-Ling, C.; Figueiredo, I.; Fortier, J.; Slade, M.; Oh, S.T.; Rettig, M.P.; Anderson, E.; Li, Y.; Dasari, S.; Strausbauch, M.A.; Simon, V.A.; Radkevich, E.; Rahman, A.H.; Chen, Z.; Lagana, A.; DiPersio, J.F.; Rosenblatt, J.; Kim-Schulze, S.; Lonial, S.; Kumar, S.; Bhasin, S.S.; Kourelis, T.; Dhodapkar, M.V.; Vij, R.; Avigan, D.; Cho, H.J.; Mulligan, G.; Ding, L.; Gnjatic, S.; Vlachos, I.S.; Bhasin, M.; Immune Atlas Consortium. A single-cell atlas characterizes dysregulation of the bone marrow immune microenvironment associated with outcomes in multiple myeloma. Nat Cancer 2026, 7(1), 224–246. [Google Scholar] [CrossRef] [PubMed]
- Merz, M.; Hu, Q.; Merz, A.M.A.; Wang, J.; Hutson, N.; Rondeau, C.; Celotto, K.; Belal, A.; Alberico, R.; Block, A.W.; Mohammadpour, H.; Wallace, P.K.; Tario, J.; Luce, J.; Glenn, S.T.; Singh, P.; Samur, M.; Munshi, N.; Liu, S.; McCarthy, P.L.; Wei, L.; Hillengass, J. Spatiotemporal assessment of immunogenomic heterogeneity in multiple myeloma. Blood Adv. 2023, 7(5), 718–733. [Google Scholar] [CrossRef]
- Lutz, R.; Poos, A.M.; Solé-Boldo, L.; John, L.; Wagner, J.; Prokoph, N.; Baertsch, M.A.; Vonficht, D.; Palit, S.; Brobeil, A.; Mechtersheimer, G.; Hildenbrand, N.; Hemmer, S.; Steiger, S.; Horn, S.; Pepke, W.; Spranz, D.M.; Rehnitz, C.; Sant, P.; Mallm, J.P.; Friedrich, M.J.; Reichert, P.; Huhn, S.; Trumpp, A.; Rippe, K.; Haghverdi, L.; Fröhling, S.; Müller-Tidow, C.; Hübschmann, D.; Goldschmidt, H.; Willimsky, G.; Sauer, S.; Raab, M.S.; Haas, S.; Weinhold, N. Bone marrow breakout lesions act as key sites for tumor-immune cell diversification in multiple myeloma. Sci Immunol 2025, 10(104), eadp6667. [Google Scholar] [CrossRef]
- Brandl, A.; Solimando, A.G.; Mokhtari, Z.; Tabares, P.; Medler, J.; Manz, H.; Da Vià, M.C.; Croci, G.A.; Kurzwart, M.; Thusek, S.; Schneider, T.; Ebert, R.; Jakob, F.; Einsele, H.; Beilhack, A. Junctional adhesion molecule C expression specifies a CD138low/neg multiple myeloma cell population in mice and humans. Blood Adv. 2022, 6(7), 2195–2206. [Google Scholar] [CrossRef]
- Vogel, L.K.; Saebø, M.; Skjelbred, C.F.; Abell, K.; Pedersen, E.D.; Vogel, U.; Kure, E.H. The ratio of Matriptase/HAI-1 mRNA is higher in colorectal cancer adenomas and carcinomas than corresponding tissue from control individuals. BMC Cancer 2006, 6, 176. [Google Scholar] [CrossRef] [PubMed]
- Armistead, J.; Hatzold, J.; van Roye, A.; Fahle, E.; Hammerschmidt, M. Entosis and apical cell extrusion constitute a tumor-suppressive mechanism downstream of Matriptase. J Cell Biol. 2020, 219(2), e201905190. [Google Scholar] [CrossRef] [PubMed]
- Zhong, S.; Khalil, R.A. A Disintegrin and Metalloproteinase (ADAM) and ADAM with thrombospondin motifs (ADAMTS) family in vascular biology and disease. Biochem Pharmacol 2019, 164, 188–204. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.; Yu, R. Pan-cancer analysis of ADAMs: A promising biomarker for prognosis and response to chemotherapy and immunotherapy. Front Genet. 2023, 14, 1105900. [Google Scholar] [CrossRef]
- Le Gall, S.M.; Maretzky, T.; Issuree, P.D.; Niu, X.D.; Reiss, K.; Saftig, P.; Khokha, R.; Lundell, D.; Blobel, C.P. ADAM17 is regulated by a rapid and reversible mechanism that controls access to its catalytic site. J Cell Sci. 2010, 123 Pt 22, 3913–22. [Google Scholar] [CrossRef]
- Zhang, W.; Xiang, Y.; Lu, C.; Wang, F.; Ren, H.; Wu, H.; Yan, M. The extracellular matrix in inflammation and cancer. Mol Biomed. 2026, 7(1), 21. [Google Scholar] [CrossRef]
- Liu, L.; Tian, E.; Quan, S.; Su, C.; Zhou, J.; Hu, S.; Bian, N.; Du, S.; Li, J. ADAM17 as a promising therapeutic target: from structural basis to inhibitor discovery in human diseases. Front Pharmacol 2025, 16, 1640090. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Xuan, Z.; Liu, X.; Zheng, M.; Yang, C.; Wang, H. Immunomodulatory role of metalloproteinase ADAM17 in tumor development. Front Immunol 2022, 13, 1059376. [Google Scholar] [CrossRef] [PubMed]
- de Jong, M.M.E.; Kellermayer, Z.; Papazian, N.; Tahri, S.; Hofste Op Bruinink, D.; Hoogenboezem, R.; Sanders, M.A.; van de Woestijne, P.C.; Bos, P.K.; Khandanpour, C.; Vermeulen, J.; Moreau, P.; van Duin, M.; Broijl, A.; Sonneveld, P.; Cupedo, T. The multiple myeloma microenvironment is defined by an inflammatory stromal cell landscape. Nat Immunol 2021, 22(6), 769–780. [Google Scholar] [CrossRef]
- Du, J.; Lin, Z.; Fu, X.H.; Gu, X.R.; Lu, G.; Hou, J. Research progress of the chemokine/chemokine receptor axes in the oncobiology of multiple myeloma (MM). Cell Commun Signal 2024, 22(1), 177. [Google Scholar] [CrossRef] [PubMed]
- Düsterhöft, S.; Lokau, J.; Garbers, C. The metalloprotease ADAM17 in inflammation and cancer. Pathol Res Pract. 2019, 215(6), 152410. [Google Scholar] [CrossRef]
- Sisto, M.; Ribatti, D.; Lisi, S. ADAM 17 and Epithelial-to-Mesenchymal Transition: The Evolving Story and Its Link to Fibrosis and Cancer. J Clin Med. 2021, 10(15), 3373. [Google Scholar] [CrossRef]
- Miao, L.; Zhou, R.; Zhang, L.; Feng, L.; Wang, Y.; Song, P.; Lv, D.; Shen, T.M. Unraveling the dual roles of tumor-infiltrating antibodies in solid tumors: friend or foe in the tumor microenvironment? Front Immunol 2025, 16, 1671839. [Google Scholar] [CrossRef]
- Fokkema, C.; Bertamini, L.; de Jong, M.M.E.; Tahri, S.; Hofste Op Bruinink, D.; Kellermayer, Z.; Papazian, N.; den Hollander, C.; Vermeulen, M.P.W.; Stoetman, E.C.G.; van Beek, G.; Hoogenboezem, R.; van der Velden, V.H.J.; Hulin, C.; Perrot, A.; Moreau, P.; Rowe, M.; Vieyra, D.; Carson, R.; van Duin, M.; Sanders, M.A.; Broijl, A.; Sonneveld, P.; Cupedo, T. Circulating tumor cells in myeloma are a compound biomarker for bone marrow high-risk genomic alterations and tumor load. Blood 2026, 147(9), 932–945. [Google Scholar] [CrossRef]
- Da Vià, M.C.; Lazzaroni, F.; Matera, A.; Marella, A.; Maeda, A.; De Magistris, C.; Pettine, L.; Solimando, A.G.; Desantis, V.; Peretti, G.M.; Mangiavini, L.; Giorgino, R.; Fabris, S.; Pioggia, S.; Marchetti, A.; Barbieri, M.; Lonati, S.; Cattaneo, A.; Tornese, M.; Scopetti, M.; Calvi, E.; Latifinavid, N.; Castellano, G.; Torricelli, F.; Neri, A.; Fokkema, C.; Cupedo, T.; Lionetti, M.; Passamonti, F.; Bolli, N. Aberrant single-cell phenotype and clinical implications of genotypically defined polyclonal plasma cells in myeloma. Blood 2025, 145(26), 3124–3138. [Google Scholar] [CrossRef]
- Desantis, V.; Frassanito, M.A.; Tamma, R.; Saltarella, I.; Di Marzo, L.; Lamanuzzi, A.; Solimando, A.G.; Ruggieri, S.; Annese, T.; Nico, B.; Vacca, A.; Ribatti, D. Rhu-Epo down-regulates pro-tumorigenic activity of cancer-associated fibroblasts in multiple myeloma. Ann Hematol. 2018, 97(7), 1251–1258. [Google Scholar] [CrossRef] [PubMed]
- Rao, L.; Giannico, D.; Leone, P.; Solimando, A.G.; Maiorano, E.; Caporusso, C.; Duda, L.; Tamma, R.; Mallamaci, R.; Susca, N.; Buonavoglia, A.; Da Vià, M.C.; Ribatti, D.; De Re, V.; Vacca, A.; Racanelli, V. HB-EGF-EGFR Signaling in Bone Marrow Endothelial Cells Mediates Angiogenesis Associated with Multiple Myeloma. Cancers (Basel) 2020, 12(1), 173. [Google Scholar] [CrossRef]
- Koenen, R.R.; Pruessmeyer, J.; Soehnlein, O.; Fraemohs, L.; Zernecke, A.; Schwarz, N.; Reiss, K.; Sarabi, A.; Lindbom, L.; Hackeng, T.M.; Weber, C.; Ludwig, A. Regulated release and functional modulation of junctional adhesion molecule A by disintegrin metalloproteinases. Blood 2009, 113(19), 4799–809. [Google Scholar] [CrossRef]
- Kummer, D.; Ebnet, K. Junctional Adhesion Molecules (JAMs): The JAM-Integrin Connection. Cells 2018, 7(4), 25. [Google Scholar] [CrossRef]
- Ferguson, I.D.; Patiño-Escobar, B.; Tuomivaara, S.T.; Lin, Y.T.; Nix, M.A.; Leung, K.K.; Kasap, C.; Ramos, E.; Nieves Vasquez, W.; Talbot, A.; Hale, M.; Naik, A.; Kishishita, A.; Choudhry, P.; Lopez-Girona, A.; Miao, W.; Wong, S.W.; Wolf, J.L.; Martin, T.G., 3rd; Shah, N.; Vandenberg, S.; Prakash, S.; Besse, L.; Driessen, C.; Posey, A.D., Jr.; Mullins, R.D.; Eyquem, J.; Wells, J.A.; Wiita, A.P. The surfaceome of multiple myeloma cells suggests potential immunotherapeutic strategies and protein markers of drug resistance. Nat Commun. 2022, 13(1), 4121. [Google Scholar] [CrossRef] [PubMed]
- Tu, C.; Van der Vreken, A.; Meeus, F.; van den Broecke, L.; Brons, J.; De Veirman, K.; Vanderkerken, K.; De Bruyne, E.; Breckpot, K.; Menu, E. Hypoxia promotes BCMA loss and a suppressive secretome thereby hindering CAR T cell therapy in multiple myeloma. Exp Hematol Oncol 2026, 15(1), 1. [Google Scholar] [CrossRef]
- Wada, A.; Ito, A.; Iitsuka, H.; Tsuneyama, K.; Miyazono, T.; Murakami, J.; Shibahara, N.; Sakurai, H.; Saiki, I.; Nakayama, T.; Yoshie, O.; Koizumi, K.; Sugiyama, T. Role of chemokine CX3CL1 in progression of multiple myeloma via CX3CR1 in bone microenvironments. Oncol Rep. 2015, 33(6), 2935–9. [Google Scholar] [CrossRef]
- Göoz, P.; Göoz, M.; Baldys, A.; Hoffman, S. ADAM-17 regulates endothelial cell morphology, proliferation, and in vitro angiogenesis. Biochem Biophys Res Commun 2009, 380(1), 33–8. [Google Scholar] [CrossRef]
- Xia, S.; Menden, H.L.; Mabry, S.M.; Sampath, V. HDAC6 and ERK/ADAM17 Regulate VEGF-Induced NOTCH Signaling in Lung Endothelial Cells. Cells 2023, 12(18), 2231. [Google Scholar] [CrossRef] [PubMed]
- Garbicz, F.; Kaszkowiak, M.; Dudkiewicz-Garbicz, J.; Dorfman, D.M.; Ostrowska, J.; Barankiewicz, J.; Salomon-Perzyński, A.; Lech-Marańda, E.; Nguyen, T.; Juszczyński, P.; Carrasco, R.D.; Misiewicz-Krzeminska, I. Characterization and Experimental Use of Multiple Myeloma Bone Marrow Endothelial Cells and Progenitors. Int J Mol Sci. 2024, 25(22), 12047. [Google Scholar] [CrossRef] [PubMed]
- Ling, W.; Zangari, M.; Van Rhee, F.; Barlogie, B.; Yaccoby, S. Altered mesenchymal and endothelial subsets in interstitial bone marrow and focal lesions in myeloma patients and SCID-hu mice. Haematologica 2025, 110(11), 2740–2751. [Google Scholar] [CrossRef] [PubMed]
- Waldschmidt, J.M.; Simon, A.; Wider, D.; Müller, S.J.; Follo, M.; Ihorst, G.; Decker, S.; Lorenz, J.; Chatterjee, M.; Azab, A.K.; Duyster, J.; Wäsch, R.; Engelhardt, M. CXCL12 and CXCR7 are relevant targets to reverse cell adhesion-mediated drug resistance in multiple myeloma. Br J Haematol 2017, 179(1), 36–49. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, R.; Lima, R.N.; Domingues, R.R.; Pauletti, B.A.; Meirelles, G.V.; Assis, M.; Figueira, A.C.; Paes Leme, A.F. Deciphering the role of the ADAM17-dependent secretome in cell signaling. J Proteome Res. 2014, 13(4), 2080–93. [Google Scholar] [CrossRef]
- Kuroki, W.; Kitadate, A.; Takahashi, Y.; Iwama, S.; Yamada, M.; Kobayashi, T.; Ikeda, S.; Narita, K.; Matsue, K.; Takahashi, N. Multiple myeloma with 1q gain/amplification exhibits reduced CD38 expression via interleukin-6 receptor overexpression. Br J Haematol. 2025, 206(6), 1615–1626. [Google Scholar] [CrossRef]
- Wu, S.; Cao, Z.; Lu, R.; Zhang, Z.; Sethi, G.; You, Y. Interleukin-6 (IL-6)-associated tumor microenvironment remodelling and cancer immunotherapy. Cytokine Growth Factor Rev. 2025, 85, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Chalaris, A.; Gewiese, J.; Paliga, K.; Fleig, L.; Schneede, A.; Krieger, K.; Rose-John, S.; Scheller, J. ADAM17-mediated shedding of the IL6R induces cleavage of the membrane stub by gamma-secretase. Biochim Biophys Acta 2010, 1803(2), 234–45. [Google Scholar] [CrossRef] [PubMed]
- Rose-John, S. IL-6 trans-signaling via the soluble IL-6 receptor: importance for the pro-inflammatory activities of IL-6. Int J Biol Sci. 2012, 8(9), 1237–47. [Google Scholar] [CrossRef]
- Müllberg, J.; Schooltink, H.; Stoyan, T.; Günther, M.; Graeve, L.; Buse, G.; Mackiewicz, A.; Heinrich, P.C.; Rose-John, S. The soluble interleukin-6 receptor is generated by shedding. Eur J Immunol 1993, 23(2), 473–80. [Google Scholar] [CrossRef]
- Gu, J.; Huang, X.; Zhang, Y.; Bao, C.; Zhou, Z.; Jin, J. Cytokine profiles in patients with newly diagnosed multiple myeloma: Survival is associated with IL-6 and IL-17A levels. Cytokine 2021, 138, 155358. [Google Scholar] [CrossRef]
- Fisher, D.T.; Appenheimer, M.M.; Evans, S.S. The two faces of IL-6 in the tumor microenvironment. Semin Immunol. 2014, 26(1), 38–47. [Google Scholar] [CrossRef]
- Schumacher, N.; Meyer, D.; Mauermann, A.; von der Heyde, J.; Wolf, J.; Schwarz, J.; Knittler, K.; Murphy, G.; Michalek, M.; Garbers, C.; Bartsch, J.W.; Guo, S.; Schacher, B.; Eickholz, P.; Chalaris, A.; Rose-John, S.; Rabe, B. Shedding of Endogenous Interleukin-6 Receptor (IL-6R) Is Governed by A Disintegrin and Metalloproteinase (ADAM) Proteases while a Full-length IL-6R Isoform Localizes to Circulating Microvesicles. J Biol Chem. 2015, 290(43), 26059–71. [Google Scholar] [CrossRef]
- Robinson, M.H.; Villa, N.Y.; Jaye, D.L.; Nooka, A.K.; Duffy, A.; McCachren, S.S.; Manalo, J.; Switchenko, J.M.; Barnes, S.; Potdar, S.; Azeem, M.I.; Horvat, A.A.; Parihar, V.C.; Gong, J.; Liang, Y.; Smith, G.H.; Gupta, V.A.; Boise, L.H.; Kaufman, J.L.; Hofmeister, C.C.; Joseph, N.S.; Lonial, S.; Dhodapkar, K.M.; Dhodapkar, M.V. Regulation of antigen-specific T cell infiltration and spatial architecture in multiple myeloma and premalignancy. J Clin Invest. 2023, 133(15), e167629. [Google Scholar] [CrossRef] [PubMed]
- Desantis, V.; Andriano, A.; Düking, T.; Hartwig, O.; Ingravallo, G.; Biondo, M.; Botta, C.; Ria, R.; Vacca, A.; Solimando, A.G. Spatial imaging unlocks the potential of charting multiple myeloma and extramedullary disease. J Hematol Oncol. 2025, 18(1), 47. [Google Scholar] [CrossRef] [PubMed]
- Yip, R.K.H.; Er, J.; Qin, L.; Nguyen, Q.H.; Motyer, A.; Rimes, J.S.; Light, A.; Mishi, R.D.; Ling, L.; Anttila, C.J.A.; Tsui, E.; Amann-Zalcenstein, D.; Dowling, M.R.; Rogers, K.L.; Bowden, R.; Chen, Y.; Harrison, S.J.; Hawkins, E.D. Profiling the spatial architecture of multiple myeloma in human bone marrow trephine biopsy specimens with spatial transcriptomics. Blood 2025, 146(15), 1837–1849. [Google Scholar] [CrossRef] [PubMed]
- Saad, M.I.; Jenkins, B.J. The protease ADAM17 at the crossroads of disease: revisiting its significance in inflammation, cancer, and beyond. FEBS J 2024, 291(1), 10–24. [Google Scholar] [CrossRef] [PubMed]
- D'Souza, C.; Keam, S.P.; Yeang, H.X.A.; Neeson, M.; Richardson, K.; Hsu, A.K.; Canfield, R.; Bezman, N.; Robbins, M.; Quach, H.; Ritchie, D.S.; Harrison, S.J.; Trapani, J.A.; Prince, H.M.; Beavis, P.A.; Darcy, P.K.; Neeson, P.J. Myeloma natural killer cells are exhausted and have impaired regulation of activation. Haematologica 2021, 106(9), 2522–2526. [Google Scholar] [CrossRef]
- Bilotta, M.T.; Solimando, A.G.; Desantis, V.; Di Marzo, L.; Sabatini, A.; Forcelloni, S.; Andriano, A.; Morizio, A.; Argentiero, A.; Ria, R.; Quatrini, L.; Moretta, L.; Vacca, P.; Tumino, N. Progressive NK-cell dysfunction and ILC imbalance favor immune evasion in multiple myeloma. Blood Adv. 2025, 9(24), 6246–6251. [Google Scholar] [CrossRef]
- Saltarella, I.; Link, A.; Lamanuzzi, A.; Reichen, C.; Robinson, J.; Altamura, C.; Melaccio, A.; Solimando, A.G.; Ria, R.; Mariggiò, M.A.; Vacca, A.; Frassanito, M.A.; Desaphy, J.F. Improvement of daratumumab- or elotuzumab-mediated NK cell activity by the bi-specific 4-1BB agonist, DARPin α-FAPx4-1BB: A preclinical study in multiple myeloma. Biomed Pharmacother 2024, 176, 116877. [Google Scholar] [CrossRef]
- Pabst, A.; Brock, N.; Hardt, S.; Grannemann, C.C.F.; Kubiza, H.; Thiebes, A.L.; Ludwig, A.; Babendreyer, A. Mechanoresponsive ion channels Piezo1 and TRPV4 stimulate ADAM10 and ADAM17 with differential impact on endothelial migration and proliferation. Cell Commun Signal 2026, 24(1), 52. [Google Scholar] [CrossRef]
- Yang, S.; Liu, F.; Wang, Q.J.; Rosenberg, S.A.; Morgan, R.A. The shedding of CD62L (L-selectin) regulates the acquisition of lytic activity in human tumor reactive T lymphocytes. PLoS One 2011, 6(7), e22560. [Google Scholar] [CrossRef]
- Link, M.A.; Lücke, K.; Schmid, J.; Schumacher, V.; Eden, T.; Rose-John, S.; Mittrücker, H.W. The role of ADAM17 in the T-cell response against bacterial pathogens. PLoS One 2017, 12(9), e0184320. [Google Scholar] [CrossRef]
- Mahnke, K.; Useliene, J.; Ring, S.; Kage, P.; Jendrossek, V.; Robson, S.C.; Bylaite-Bucinskiene, M.; Steinbrink, K.; Enk, A.H. Down-Regulation of CD62L Shedding in T Cells by CD39+ Regulatory T Cells Leads to Defective Sensitization in Contact Hypersensitivity Reactions. J Invest Dermatol. 2017, 137(1), 106–114. [Google Scholar] [CrossRef]
- Li, Y.; Brazzell, J.; Herrera, A.; Walcheck, B. ADAM17 deficiency by mature neutrophils has differential effects on L-selectin shedding. Blood 2006, 108(7), 2275–9. [Google Scholar] [CrossRef] [PubMed]
- Mishra, H.K.; Dixon, K.J.; Pore, N.; Felices, M.; Miller, J.S.; Walcheck, B. Activation of ADAM17 by IL-15 Limits Human NK Cell Proliferation. Front Immunol 2021, 12, 711621. [Google Scholar] [CrossRef]
- Romee, R.; Foley, B.; Lenvik, T.; Wang, Y.; Zhang, B.; Ankarlo, D.; Luo, X.; Cooley, S.; Verneris, M.; Walcheck, B.; Miller, J. NK cell CD16 surface expression and function is regulated by a disintegrin and metalloprotease-17 (ADAM17). Blood 2013, 121(18), 3599–608. [Google Scholar] [CrossRef]
- Srpan, K.; Ambrose, A.; Karampatzakis, A.; Saeed, M.; Cartwright, A.N.R.; Guldevall, K.; De Matos, G.D.S.C.; Önfelt, B.; Davis, D.M. Shedding of CD16 disassembles the NK cell immune synapse and boosts serial engagement of target cells. J Cell Biol. 2018, 217(9), 3267–3283. [Google Scholar] [CrossRef]
- Pomeroy, E.J.; Hunzeker, J.T.; Kluesner, M.G.; Lahr, W.S.; Smeester, B.A.; Crosby, M.R.; Lonetree, C.L.; Yamamoto, K.; Bendzick, L.; Miller, J.S.; Geller, M.A.; Walcheck, B.; Felices, M.; Webber, B.R.; Starr, T.K.; Moriarity, B.S. A Genetically Engineered Primary Human Natural Killer Cell Platform for Cancer Immunotherapy. Mol Ther. 2020, 28(1), 52–63. [Google Scholar] [CrossRef] [PubMed]
- John, M.; Helal, M.; Duell, J.; Mattavelli, G.; Stanojkovska, E.; Afrin, N.; Leipold, A.M.; Steinhardt, M.J.; Zhou, X.; Žihala, D.; Anilkumar Sithara, A.; Mersi, J.; Waldschmidt, J.M.; Riedhammer, C.; Kadel, S.K.; Truger, M.; Werner, R.A.; Haferlach, C.; Einsele, H.; Kretzschmar, K.; Jelínek, T.; Rosenwald, A.; Kortüm, K.M.; Riedel, A.; Rasche, L. Spatial transcriptomics reveals profound subclonal heterogeneity and T-cell dysfunction in extramedullary myeloma. Blood 2024, 144(20), 2121–2135. [Google Scholar] [CrossRef]
- Zanwar, S.; Novak, J.; Gonsalves, W.; Howe, M.; Braggio, E.; Rajkumar, S.V.; Binder, M.; Jevremovic, D.; Dasari, S.; Kumar, S. Extramedullary myeloma is genomically complex and characterized by near-universal MAPK pathway alterations. Blood Adv. 2025, 9(15), 3979–3987. [Google Scholar] [CrossRef]
- García-Ortiz, A.; Rodríguez-García, Y.; Encinas, J.; Maroto-Martín, E.; Castellano, E.; Teixidó, J.; Martínez-López, J. The Role of Tumor Microenvironment in Multiple Myeloma Development and Progression. Cancers (Basel) 2021, 13(2), 217. [Google Scholar] [CrossRef]
- Glavey, S.V.; Naba, A.; Manier, S.; Clauser, K.; Tahri, S.; Park, J.; Reagan, M.R.; Moschetta, M.; Mishima, Y.; Gambella, M.; Rocci, A.; Sacco, A.; O'Dwyer, M.E.; Asara, J.M.; Palumbo, A.; Roccaro, A.M.; Hynes, R.O.; Ghobrial, I.M. Proteomic characterization of human multiple myeloma bone marrow extracellular matrix. Leukemia 2017, 31(11), 2426–2434. [Google Scholar] [CrossRef]
- Gooding, S.; Olechnowicz, S.W.Z.; Morris, E.V.; Armitage, A.E.; Arezes, J.; Frost, J.; Repapi, E.; Edwards, J.R.; Ashley, N.; Waugh, C.; Gray, N.; Martinez-Hackert, E.; Lim, P.J.; Pasricha, S.R.; Knowles, H.; Mead, A.J.; Ramasamy, K.; Drakesmith, H.; Edwards, C.M. Transcriptomic profiling of the myeloma bone-lining niche reveals BMP signalling inhibition to improve bone disease. Nat Commun. 2019, 10(1), 4533. [Google Scholar] [CrossRef] [PubMed]
- Sborov, D.W.; Nuovo, G.J.; Stiff, A.; Mace, T.; Lesinski, G.B.; Benson, D.M., Jr.; Efebera, Y.A.; Rosko, A.E.; Pichiorri, F.; Grever, M.R.; Hofmeister, C.C. A phase I trial of single-agent reolysin in patients with relapsed multiple myeloma. Clin Cancer Res. 2014, 20(23), 5946–55. [Google Scholar] [CrossRef]
- Kelly, K.R.; Espitia, C.M.; Zhao, W.; Wendlandt, E.; Tricot, G.; Zhan, F.; Carew, J.S.; Nawrocki, S.T. Junctional adhesion molecule-A is overexpressed in advanced multiple myeloma and determines response to oncolytic reovirus. Oncotarget 2015, 6(38), 41275–89. [Google Scholar] [CrossRef]
- Stiff, A.; Caserta, E.; Sborov, D.W.; Nuovo, G.J.; Mo, X.; Schlotter, S.Y.; Canella, A.; Smith, E.; Badway, J.; Old, M.; Jaime-Ramirez, A.C.; Yan, P.; Benson, D.M.; Byrd, J.C.; Baiocchi, R.; Kaur, B.; Hofmeister, C.C.; Pichiorri, F. Histone Deacetylase Inhibitors Enhance the Therapeutic Potential of Reovirus in Multiple Myeloma. Mol Cancer Ther 2016, 15(5), 830–41. [Google Scholar] [CrossRef]
- Liu, K.; Yang, H.; Xiong, R.; Shen, Y.; Song, G.; Yang, J.; Wang, Z. Generation and characterization of mAb 61H9 against junctional adhesion molecule-a with potent antitumor activity. PeerJ 2024, 12, e17088. [Google Scholar] [CrossRef]
- Leech, A.O.; Vellanki, S.H.; Rutherford, E.J.; Keogh, A.; Jahns, H.; Hudson, L.; O'Donovan, N.; Sabri, S.; Abdulkarim, B.; Sheehan, K.M.; Kay, E.W.; Young, L.S.; Hill, A.D.K.; Smith, Y.E.; Hopkins, A.M. Cleavage of the extracellular domain of junctional adhesion molecule-A is associated with resistance to anti-HER2 therapies in breast cancer settings. Breast Cancer Res. 2018, 20(1), 140. [Google Scholar] [CrossRef]
- Thölmann, S.; Seebach, J.; Otani, T.; Florin, L.; Schnittler, H.; Gerke, V.; Furuse, M.; Ebnet, K. JAM-A interacts with α3β1 integrin and tetraspanins CD151 and CD9 to regulate collective cell migration of polarized epithelial cells. Cell Mol Life Sci. 2022, 79(2), 88. [Google Scholar] [CrossRef]
- Tian, Y.; Tian, Y.; Zhang, W.; Wei, F.; Yang, J.; Luo, X.; Zhou, T.; Hou, B.; Qian, S.; Deng, X.; Qiu, Y.; Yao, K. Junctional adhesion molecule-A, an epithelial-mesenchymal transition inducer, correlates with metastasis and poor prognosis in human nasopharyngeal cancer. Carcinogenesis 2015, 36(1), 41–8. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.P.; Sun, Y.F.; Fang, Y.; Song, Q.; Yan, Z.X.; Chen, Y.; Jiang, X.F.; Fei, X.C.; Zhao, Y.; Leboeuf, C.; Li, B.; Wang, C.F.; Janin, A.; Wang, L.; Zhao, W.L. JAM-A overexpression is related to disease progression in diffuse large B-cell lymphoma and downregulated by lenalidomide. Sci Rep. 2017, 7(1), 7433. [Google Scholar] [CrossRef]
- Zunke, F.; Rose-John, S. The shedding protease ADAM17: Physiology and pathophysiology. Biochim Biophys Acta Mol Cell Res. 2017, 1864 11 Pt B, 2059–2070. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, S.; Schumacher, N.; Schwarz, J.; Tangermann, S.; Kenner, L.; Schlederer, M.; Sibilia, M.; Linder, M.; Altendorf-Hofmann, A.; Knösel, T.; Gruber, E.S.; Oberhuber, G.; Bolik, J.; Rehman, A.; Sinha, A.; Lokau, J.; Arnold, P.; Cabron, A.S.; Zunke, F.; Becker-Pauly, C.; Preaudet, A.; Nguyen, P.; Huynh, J.; Afshar-Sterle, S.; Chand, A.L.; Westermann, J.; Dempsey, P.J.; Garbers, C.; Schmidt-Arras, D.; Rosenstiel, P.; Putoczki, T.; Ernst, M.; Rose-John, S. ADAM17 is required for EGF-R-induced intestinal tumors via IL-6 trans-signaling. J Exp Med. 2018, 215(4), 1205–1225. [Google Scholar] [CrossRef]
- Qu, J.; Li, Y.; Li, C.; Wu, D.; Cao, Y.; Xiao, S.; Bao, X.; Li, Q.; Chen, Z.; Chen, L. Longitudinal profiling of serum ADAM17 across clinical stages in multiple myeloma: a dynamic biomarker and its association with T cell alterations. Front Mol Biosci. 2026, 13, 1768193. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, R.N.; Wehenkel, S.C.; Galkina, E.V.; Yates, E.K.; Preece, G.; Newman, A.; Watson, H.A.; Ohme, J.; Bridgeman, J.S.; Durairaj, R.R.P.; Moon, O.R.; Ladell, K.; Miners, K.L.; Dolton, G.; Troeberg, L.; Kashiwagi, M.; Murphy, G.; Nagase, H.; Price, D.A.; Matthews, R.J.; Knäuper, V.; Ager, A. ADAM17-dependent proteolysis of L-selectin promotes early clonal expansion of cytotoxic T cells. Sci Rep. 2019, 9(1), 5487. [Google Scholar] [CrossRef]
- de Queiroz, T.M.; Lakkappa, N.; Lazartigues, E. ADAM17-Mediated Shedding of Inflammatory Cytokines in Hypertension. Front Pharmacol 2020, 11, 1154. [Google Scholar] [CrossRef]
- Le Gall, S.M.; Bobé, P.; Reiss, K.; Horiuchi, K.; Niu, X.D.; Lundell, D.; Gibb, D.R.; Conrad, D.; Saftig, P.; Blobel, C.P. ADAMs 10 and 17 represent differentially regulated components of a general shedding machinery for membrane proteins such as transforming growth factor alpha, L-selectin, and tumor necrosis factor alpha. Mol Biol Cell 2009, 20(6), 1785–94. [Google Scholar] [CrossRef]
- Zhou, Y.; Heitmann, J.S.; Kropp, K.N.; Hinterleitner, M.; Koch, A.; Hartkopf, A.D.; Salih, H.R.; Hinterleitner, C.; Maurer, S. Regulation of Platelet-Derived ADAM17: A Biomarker Approach for Breast Cancer? Diagnostics (Basel) 2021, 11(7), 1188. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, X.; Roozbahani, G.M.; Guan, X. Rapid and sensitive detection of the activity of ADAM17 using a graphene oxide-based fluorescence sensor. Analyst 2019, 144(5), 1825–1830. [Google Scholar] [CrossRef] [PubMed]
- Khattab, H.; Solujic, J.; Saad, M.I. ADAM proteases in cytokine biology: Modulators of immune signaling, inflammation and cancer. Biochim Biophys Acta Mol Cell Res. 2026, 1873(4), 120137. [Google Scholar] [CrossRef]
- Garbers, C.; Jänner, N.; Chalaris, A.; Moss, M.L.; Floss, D.M.; Meyer, D.; Koch-Nolte, F.; Rose-John, S.; Scheller, J. Species specificity of ADAM10 and ADAM17 proteins in interleukin-6 (IL-6) trans-signaling and novel role of ADAM10 in inducible IL-6 receptor shedding. J Biol Chem. 2011, 286(17), 14804–11. [Google Scholar] [CrossRef] [PubMed]
- Hayashida, K.; Bartlett, A.H.; Chen, Y.; Park, P.W. Molecular and cellular mechanisms of ectodomain shedding. Anat Rec (Hoboken) 2010, 293(6), 925–37. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Zarbock, A.; Gomez, I.; Wilson, C.L.; Lefort, C.T.; Stadtmann, A.; Bell, B.; Huang, L.C.; Ley, K.; Raines, E.W. Adam17-dependent shedding limits early neutrophil influx but does not alter early monocyte recruitment to inflammatory sites. Blood 2011, 118(3), 786–94. [Google Scholar] [CrossRef] [PubMed]
- Haining, E.J.; Yang, J.; Bailey, R.L.; Khan, K.; Collier, R.; Tsai, S.; Watson, S.P.; Frampton, J.; Garcia, P.; Tomlinson, M.G. The TspanC8 subgroup of tetraspanins interacts with A disintegrin and metalloprotease 10 (ADAM10) and regulates its maturation and cell surface expression. J Biol Chem. 2012, 287(47), 39753–65. [Google Scholar] [CrossRef]
- Gnosa, S.P.; Puig Blasco, L.; Piotrowski, K.B.; Freiberg, M.L.; Savickas, S.; Madsen, D.H.; Auf dem Keller, U.; Kronqvist, P.; Kveiborg, M. ADAM17-mediated EGFR ligand shedding directs macrophage-promoted cancer cell invasion. JCI Insight 2022, 7(18), e155296. [Google Scholar] [CrossRef]
- Sun, C.; Hu, A.; Wang, S.; Tian, B.; Jiang, L.; Liang, Y.; Wang, H.; Dong, J. ADAM17-regulated CX3CL1 expression produced by bone marrow endothelial cells promotes spinal metastasis from hepatocellular carcinoma. Int J Oncol 2020, 57(1), 249–263. [Google Scholar] [CrossRef]
- Lajoie, L.; Congy-Jolivet, N.; Bolzec, A.; Gouilleux-Gruart, V.; Sicard, E.; Sung, H.C.; Peiretti, F.; Moreau, T.; Vié, H.; Clémenceau, B.; Thibault, G. ADAM17-mediated shedding of FcγRIIIA on human NK cells: identification of the cleavage site and relationship with activation. J Immunol. 2014, 192(2), 741–51. [Google Scholar] [CrossRef]
- Scharfenberg, F.; Helbig, A.; Sammel, M.; Benzel, J.; Schlomann, U.; Peters, F.; Wichert, R.; Bettendorff, M.; Schmidt-Arras, D.; Rose-John, S.; Moali, C.; Lichtenthaler, S.F.; Pietrzik, C.U.; Bartsch, J.W.; Tholey, A.; Becker-Pauly, C. Degradome of soluble ADAM10 and ADAM17 metalloproteases. Cell Mol Life Sci. 2020, 77(2), 331–350. [Google Scholar] [CrossRef] [PubMed]
- Moss, M.L.; Minond, D. Recent Advances in ADAM17 Research: A Promising Target for Cancer and Inflammation. Mediators Inflamm. 2017, 2017, 9673537. [Google Scholar] [CrossRef] [PubMed]
- Zatovicova, M.; Kajanova, I.; Takacova, M.; Jelenska, L.; Sedlakova, O.; Labudova, M.; Pastorekova, S. ADAM10 mediates shedding of carbonic anhydrase IX ectodomain non-redundantly to ADAM17. Oncol Rep. 2023, 49(2), 27. [Google Scholar] [CrossRef] [PubMed]
- Zegeye, M.M.; Andersson, B.; Sirsjö, A.; Ljungberg, L.U. IL-6 trans-Signaling Impairs Sprouting Angiogenesis by Inhibiting Migration, Proliferation and Tube Formation of Human Endothelial Cells. Cells 2020, 9(6), 1414. [Google Scholar] [CrossRef] [PubMed]
- Calligaris, M.; Cuffaro, D.; Bonelli, S.; Spanò, D.P.; Rossello, A.; Nuti, E.; Scilabra, S.D. Strategies to Target ADAM17 in Disease: From its Discovery to the iRhom Revolution. Molecules 2021, 26(4), 944. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



