Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Structure and Function of the GJB2 Gene
3. Genetic Epidemiology and Clinical Spectrum of GJB2-Related Hearing Loss
3.1. Genetic Epidemiology
3.2. Clinical Spectrum
3.2.1. Genotype–Phenotype Correlation and Clinical Variability in Nonsyndromic GJB2-Related Hearing Loss
| Genotype class | Nucleotide changes | General phenotypic trend | Sources of variability |
| Truncating coding variants [73,74] | c.35delG, c.235delC, c.167delT, c.71G>A, c.299_300delAT | Often prelingual severe to profound sensorineural hearing loss | Genetic background and cochlear network context can shift severity and age at onset |
| Canonical splice site variants [75,76] | c.-23+1G>A, c.-22-2A>C | Frequently severe, with strong population enrichment in some groups | Expressivity may vary across families and cohorts |
| Non truncating coding variants [53] | c.109G>A, c.101T>C, c.269T>C | Often mild to moderate or context dependent phenotypes | Incomplete penetrance and heterogeneous trajectories are common for recurrent missense alleles |
| Mixed severity biallelic genotypes [67] | c.35delG/c.109G>A; c.35delG/c.101T>C; c.235delC/c.109G>A | Broad spectrum, often intermediate but not predictable | Modifier burden and life course context can move the phenotype in either direction |
| DFNB1 regulatory or structural variants [77,78] |
del(GJB6-D13S1830); del(GJB6-D13S1854); upstream DFNB1 cis-regulatory deletions | Can mimic monoallelic GJB2 results on exon sequencing | Cis regulatory disruption may reduce GJB2 expression and explain unresolved cases |
3.2.2. Syndromic Phenotypes Associated with Specific GJB2 Variants
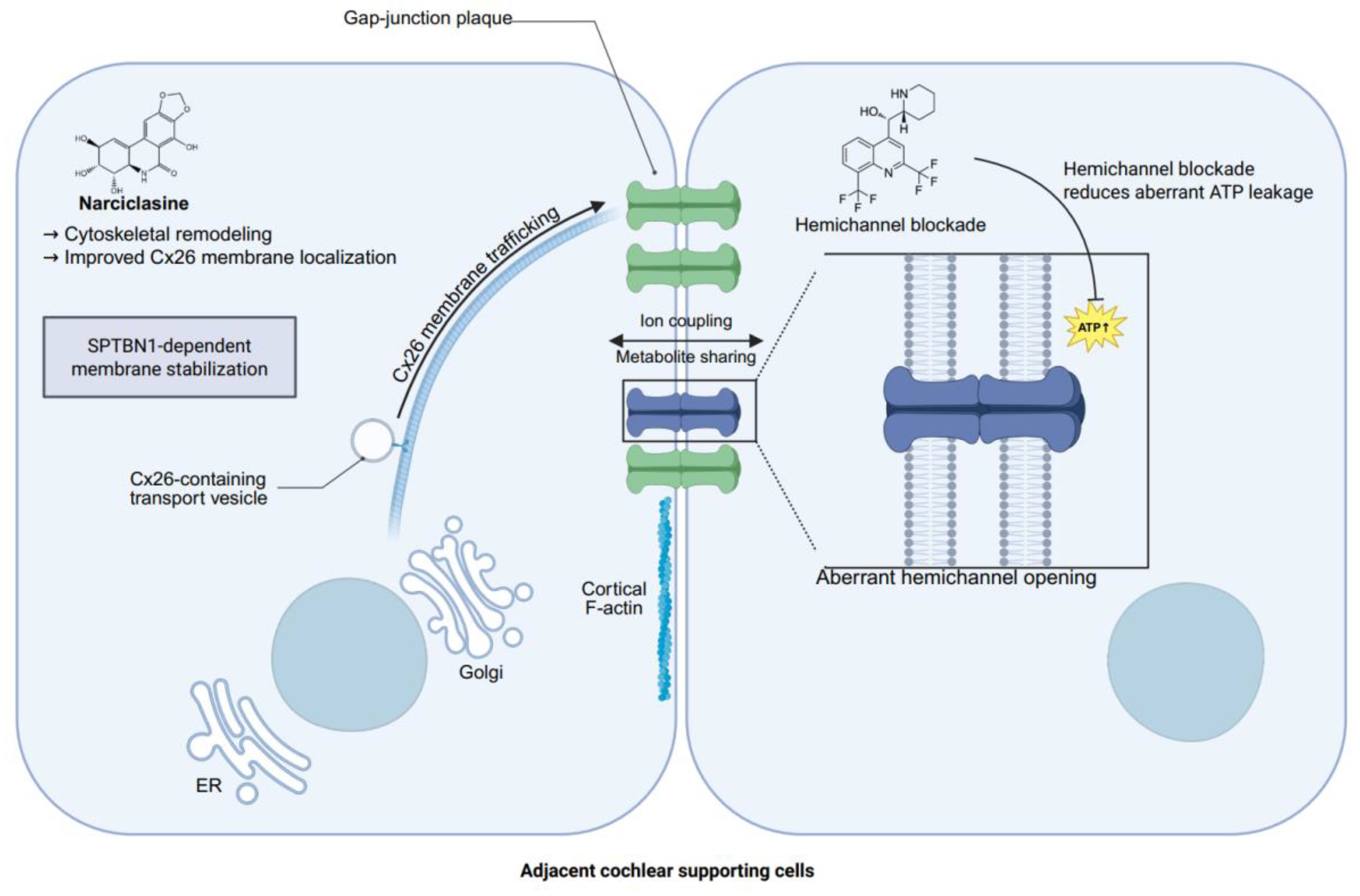
4. Pathogenic Mechanisms and Therapeutic Opportunities in GJB2-Related Hearing Loss
4.1. Therapeutically Relevant Upstream Defects in Cx26 Expression, Trafficking, and Localization
4.1.1. Reduced Cx26 Abundance and Impaired Trafficking, Assembly, and Channel Competence
4.1.2. Therapeutic Rescue of Upstream Cx26 Defects
4.2. Developmental Disruption and Rescue of Cochlear Maturation
4.2.1. Developmental Roles of Cx26 in Cochlear Maturation
4.2.2. Developmental Timing and Opportunities for Early Rescue
4.3. Sensory Epithelial Degeneration and Cell-Protective Interventions
4.3.1. Patterns and Mechanisms of Sensory Epithelial Degeneration
4.3.2. Cell Death Pathways and Protective Interventions
4.4. Cochlear Homeostasis Disruption and Intervention and Targeted Intervention
4.4.1. Homeostatic Breakdown and Ionic Dysregulation in the Cochlear Supporting-Cell Network
4.4.2. Active Amplification Failure and Microenvironment-Targeted Rescue
4.5. Secondary Pathogenic Pathways and their Therapeutic Modulation
4.5.1. Transcriptional Dysregulation and Secondary Injury Amplification
4.5.2. Anti-Inflammatory and Stress-Modulating Therapeutic Opportunities
5. Carrier Screening and Reproductive Prevention Strategies for GJB2-Related Hearing Loss
6. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Livingston, G.; Huntley, J.; Sommerlad, A.; Ames, D.; Ballard, C.; Banerjee, S.; Brayne, C.; Burns, A.; Cohen-Mansfield, J.; Cooper, C.; et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet (London, England) 2020, 396, 413-446. [CrossRef]
- Podury, A.; Jiam, N.T.; Kim, M.; Donnenfield, J.I.; Dhand, A. Hearing and sociality: the implications of hearing loss on social life. Frontiers in neuroscience 2023, 17, 1245434. [CrossRef]
- Wilson, B.S.; Tucci, D.L. Addressing the global burden of hearing loss. Lancet (London, England) 2021, 397, 945-947. [CrossRef]
- Hearing loss prevalence and years lived with disability, 1990-2019: findings from the Global Burden of Disease Study 2019. Lancet (London, England) 2021, 397, 996-1009. [CrossRef]
- Shen, Y.; Zhou, T.; Zou, W.; Zhang, J.; Yan, S.; Ye, H.; Huang, W. Global, regional, and national burden of hearing loss from 1990 to 2021: findings from the 2021 global burden of disease study. Annals of medicine 2025, 57, 2527367. [CrossRef]
- Yin, L.; Liu, J.; Zhang, J.; Tang, L.; He, S.; Shan, C.; Li, X.; Yang, H.; Zhang, J.; Liu, C.; et al. Economic burden due to hearing loss among individuals in Hebei, China. BMC public health 2025, 25, 1080. [CrossRef]
- Snoeckx, R.L.; Huygen, P.L.; Feldmann, D.; Marlin, S.; Denoyelle, F.; Waligora, J.; Mueller-Malesinska, M.; Pollak, A.; Ploski, R.; Murgia, A.; et al. GJB2 mutations and degree of hearing loss: a multicenter study. American journal of human genetics 2005, 77, 945-957. [CrossRef]
- Kenneson, A.; Van Naarden Braun, K.; Boyle, C. GJB2 (connexin 26) variants and nonsyndromic sensorineural hearing loss: a HuGE review. Genetics in medicine : official journal of the American College of Medical Genetics 2002, 4, 258-274. [CrossRef]
- Prasad, K.; Borre, E.D.; Dillard, L.K.; Ayer, A.; Der, C.; Bainbridge, K.E.; McMahon, C.M.; Tucci, D.L.; Wilson, B.S.; Schmidler, G.D.S.; et al. Priorities for hearing loss prevention and estimates of global cause-specific burdens of hearing loss: a systematic rapid review. The Lancet. Global health 2024, 12, e217-e225. [CrossRef]
- Guo, Z.; Ji, W.; Song, P.; Zhao, J.; Yan, M.; Zou, X.; Bai, F.; Wu, Y.; Guo, Z.; Song, L. Global, regional, and national burden of hearing loss in children and adolescents, 1990-2021: a systematic analysis from the Global Burden of Disease Study 2021. BMC public health 2024, 24, 2521. [CrossRef]
- Choe, G.; Park, S.K.; Kim, B.J. Hearing loss in neonates and infants. Clinical and experimental pediatrics 2023, 66, 369-376. [CrossRef]
- Cradeur, A.; Jackson, A.; Ware, E.; Fourrier, T.L.; Mankekar, G. Congenital Cytomegalovirus (cCMV) Infection as a Leading Cause of Pediatric Hearing Loss: Review. Children (Basel, Switzerland) 2025, 12. [CrossRef]
- Yoshimura, H.; Okubo, T.; Shinagawa, J.; Nishio, S.Y.; Takumi, Y.; Usami, S.I. Epidemiology, aetiology and diagnosis of congenital hearing loss via hearing screening of 153 913 newborns. International journal of epidemiology 2024, 53. [CrossRef]
- Walz, K. Reply to Elgoyhen et al. Comment on “De Rosa et al. Hearing Loss: Genetic Testing, Current Advances and the Situation in Latin America. Genes 2024, 15, 178”. Genes 2024, 15. [CrossRef]
- Egilmez, O.K.; Kalcioglu, M.T. Genetics of Nonsyndromic Congenital Hearing Loss. Scientifica 2016, 2016, 7576064. [CrossRef]
- Svidnicki, M.C.; Silva-Costa, S.M.; Ramos, P.Z.; dos Santos, N.Z.; Martins, F.T.; Castilho, A.M.; Sartorato, E.L. Screening of genetic alterations related to non-syndromic hearing loss using MassARRAY iPLEX® technology. BMC medical genetics 2015, 16, 85. [CrossRef]
- Ouyang, X.M.; Yan, D.; Yuan, H.J.; Pu, D.; Du, L.L.; Han, D.Y.; Liu, X.Z. The genetic bases for non-syndromic hearing loss among Chinese. Journal of human genetics 2009, 54, 131-140. [CrossRef]
- Mahdieh, N.; Bagherian, H.; Shirkavand, A.; Sharafi, M.; Zeinali, S. High level of intrafamilial phenotypic variability of non-syndromic hearing loss in a Lur family due to delE120 mutation in GJB2 gene. International journal of pediatric otorhinolaryngology 2010, 74, 1089-1091. [CrossRef]
- Petrova, N.; Tebieva, I.; Kadyshev, V.; Getoeva, Z.; Balinova, N.; Marakhonov, A.; Vasilyeva, T.; Ginter, E.; Kutsev, S.; Zinchenko, R. Hereditary etiology of non-syndromic sensorineural hearing loss in the Republic of North Ossetia-Alania. PeerJ 2023, 11, e14514. [CrossRef]
- Wener, E.R.; McLennan, J.D.; Papsin, B.C.; Cushing, S.L.; Stavropoulos, D.J.; Mendoza-Londono, R.; Quercia, N.; Gordon, K.A. Variants in Genes Associated with Hearing Loss in Children: Prevalence in a Large Canadian Cohort. The Laryngoscope 2024, 134, 3832-3838. [CrossRef]
- Tlili, A.; Mahfood, M.; Al Mutery, A.; Chouchen, J. Genetic analysis of 106 sporadic cases with hearing loss in the UAE population. Human genomics 2024, 18, 59. [CrossRef]
- Lautermann, J.; ten Cate, W.J.; Altenhoff, P.; Grümmer, R.; Traub, O.; Frank, H.; Jahnke, K.; Winterhager, E. Expression of the gap-junction connexins 26 and 30 in the rat cochlea. Cell and tissue research 1998, 294, 415-420. [CrossRef]
- Liu, Y.P.; Zhao, H.B. Cellular characterization of Connexin26 and Connnexin30 expression in the cochlear lateral wall. Cell and tissue research 2008, 333, 395-403. [CrossRef]
- Kikuchi, T.; Adams, J.C.; Miyabe, Y.; So, E.; Kobayashi, T. Potassium ion recycling pathway via gap junction systems in the mammalian cochlea and its interruption in hereditary nonsyndromic deafness. Medical electron microscopy : official journal of the Clinical Electron Microscopy Society of Japan 2000, 33, 51-56. [CrossRef]
- Zhu, Y.; Zhao, H.B. ATP-mediated potassium recycling in the cochlear supporting cells. Purinergic signalling 2010, 6, 221-229. [CrossRef]
- Jagger, D.J.; Forge, A. Compartmentalized and signal-selective gap junctional coupling in the hearing cochlea. The Journal of neuroscience : the official journal of the Society for Neuroscience 2006, 26, 1260-1268. [CrossRef]
- Cohen-Salmon, M.; Ott, T.; Michel, V.; Hardelin, J.P.; Perfettini, I.; Eybalin, M.; Wu, T.; Marcus, D.C.; Wangemann, P.; Willecke, K.; et al. Targeted ablation of connexin26 in the inner ear epithelial gap junction network causes hearing impairment and cell death. Current biology : CB 2002, 12, 1106-1111. [CrossRef]
- Xu, J.; Nicholson, B.J. The role of connexins in ear and skin physiology - functional insights from disease-associated mutations. Biochimica et biophysica acta 2013, 1828, 167-178. [CrossRef]
- Thönnissen, E.; Rabionet, R.; Arbonès, M.L.; Estivill, X.; Willecke, K.; Ott, T. Human connexin26 (GJB2) deafness mutations affect the function of gap junction channels at different levels of protein expression. Human genetics 2002, 111, 190-197. [CrossRef]
- Mani, R.S.; Ganapathy, A.; Jalvi, R.; Srikumari Srisailapathy, C.R.; Malhotra, V.; Chadha, S.; Agarwal, A.; Ramesh, A.; Rangasayee, R.R.; Anand, A. Functional consequences of novel connexin 26 mutations associated with hereditary hearing loss. European journal of human genetics : EJHG 2009, 17, 502-509. [CrossRef]
- Chen, Y.; Wang, Z.; Jiang, Y.; Lin, Y.; Wang, X.; Wang, Z.; Tang, Z.; Wang, Y.; Wang, J.; Gao, Y.; et al. Biallelic p.V37I variant in GJB2 is associated with increasing incidence of hearing loss with age. Genetics in medicine : official journal of the American College of Medical Genetics 2022, 24, 915-923. [CrossRef]
- Meşe, G.; Valiunas, V.; Brink, P.R.; White, T.W. Connexin26 deafness associated mutations show altered permeability to large cationic molecules. American journal of physiology. Cell physiology 2008, 295, C966-974. [CrossRef]
- Meşe, G.; Londin, E.; Mui, R.; Brink, P.R.; White, T.W. Altered gating properties of functional Cx26 mutants associated with recessive non-syndromic hearing loss. Human genetics 2004, 115, 191-199. [CrossRef]
- Cryns, K.; Orzan, E.; Murgia, A.; Huygen, P.L.; Moreno, F.; del Castillo, I.; Chamberlin, G.P.; Azaiez, H.; Prasad, S.; Cucci, R.A.; et al. A genotype-phenotype correlation for GJB2 (connexin 26) deafness. Journal of medical genetics 2004, 41, 147-154. [CrossRef]
- Shahin, H.; Walsh, T.; Sobe, T.; Lynch, E.; King, M.C.; Avraham, K.B.; Kanaan, M. Genetics of congenital deafness in the Palestinian population: multiple connexin 26 alleles with shared origins in the Middle East. Human genetics 2002, 110, 284-289. [CrossRef]
- Gandía, M.; Del Castillo, F.J.; Rodríguez-Álvarez, F.J.; Garrido, G.; Villamar, M.; Calderón, M.; Moreno-Pelayo, M.A.; Moreno, F.; del Castillo, I. A novel splice-site mutation in the GJB2 gene causing mild postlingual hearing impairment. PLoS One 2013, 8, e73566. [CrossRef]
- Liu, W.; Rask-Andersen, H. GJB2 and GJB6 gene transcripts in the human cochlea: A study using RNAscope, confocal, and super-resolution structured illumination microscopy. Frontiers in molecular neuroscience 2022, 15, 973646. [CrossRef]
- Yum, S.W.; Zhang, J.; Valiunas, V.; Kanaporis, G.; Brink, P.R.; White, T.W.; Scherer, S.S. Human connexin26 and connexin30 form functional heteromeric and heterotypic channels. American journal of physiology. Cell physiology 2007, 293, C1032-1048. [CrossRef]
- Xu, J.; Nicholson, B.J. Divergence between Hemichannel and Gap Junction Permeabilities of Connexin 30 and 26. Life (Basel, Switzerland) 2023, 13. [CrossRef]
- Sanchez, H.A.; Kraujaliene, L.; Verselis, V.K. A pore locus in the E1 domain differentially regulates Cx26 and Cx30 hemichannel function. The Journal of general physiology 2024, 156. [CrossRef]
- Naulin, P.A.; Lozano, B.; Fuentes, C.; Liu, Y.; Schmidt, C.; Contreras, J.E.; Barrera, N.P. Polydisperse molecular architecture of connexin 26/30 heteromeric hemichannels revealed by atomic force microscopy imaging. The Journal of biological chemistry 2020, 295, 16499-16509. [CrossRef]
- Harris, A.L. Connexin specificity of second messenger permeation: real numbers at last. The Journal of general physiology 2008, 131, 287-292. [CrossRef]
- Bedner, P.; Niessen, H.; Odermatt, B.; Kretz, M.; Willecke, K.; Harz, H. Selective permeability of different connexin channels to the second messenger cyclic AMP. The Journal of biological chemistry 2006, 281, 6673-6681. [CrossRef]
- Zhao, H.B. Connexin26 is responsible for anionic molecule permeability in the cochlea for intercellular signalling and metabolic communications. The European journal of neuroscience 2005, 21, 1859-1868. [CrossRef]
- Beltramello, M.; Bicego, M.; Piazza, V.; Ciubotaru, C.D.; Mammano, F.; D’Andrea, P. Permeability and gating properties of human connexins 26 and 30 expressed in HeLa cells. Biochemical and biophysical research communications 2003, 305, 1024-1033. [CrossRef]
- Kraujaliene, L.; Kraujalis, T.; Snipas, M.; Verselis, V.K. An Ala/Glu difference in E1 of Cx26 and Cx30 contributes to their differential anionic permeabilities. The Journal of general physiology 2024, 156. [CrossRef]
- Yi, E.; Lee, J.; Lee, C.J. Developmental Role of Anoctamin-1/TMEM16A in Ca(2+)-Dependent Volume Change in Supporting Cells of the Mouse Cochlea. Experimental neurobiology 2013, 22, 322-329. [CrossRef]
- Ortolano, S.; Di Pasquale, G.; Crispino, G.; Anselmi, F.; Mammano, F.; Chiorini, J.A. Coordinated control of connexin 26 and connexin 30 at the regulatory and functional level in the inner ear. Proceedings of the National Academy of Sciences of the United States of America 2008, 105, 18776-18781. [CrossRef]
- Smith, R.J.H.; Azaiez, H.; Booth, K. GJB2-Related Autosomal Recessive Nonsyndromic Hearing Loss. In GeneReviews(®), Adam, M.P., Bick, S., Mirzaa, G.M., Pagon, R.A., Wallace, S.E., Amemiya, A., Eds.; University of Washington, Seattle.
- Copyright © 1993-2026, University of Washington, Seattle. GeneReviews is a registered trademark of the University of Washington, Seattle. All rights reserved.: Seattle (WA), 1993.
- Dai, P.; Yu, F.; Han, B.; Yuan, Y.; Li, Q.; Wang, G.; Liu, X.; He, J.; Huang, D.; Kang, D.; et al. The prevalence of the 235delC GJB2 mutation in a Chinese deaf population. Genetics in medicine : official journal of the American College of Medical Genetics 2007, 9, 283-289. [CrossRef]
- Dai, P.; Yu, F.; Han, B.; Liu, X.; Wang, G.; Li, Q.; Yuan, Y.; Liu, X.; Huang, D.; Kang, D.; et al. GJB2 mutation spectrum in 2,063 Chinese patients with nonsyndromic hearing impairment. J Transl Med 2009, 7, 26. [CrossRef]
- Dahl, H.H.; Tobin, S.E.; Poulakis, Z.; Rickards, F.W.; Xu, X.; Gillam, L.; Williams, J.; Saunders, K.; Cone-Wesson, B.; Wake, M. The contribution of GJB2 mutations to slight or mild hearing loss in Australian elementary school children. Journal of medical genetics 2006, 43, 850-855. [CrossRef]
- Kim, S.Y.; Park, G.; Han, K.H.; Kim, A.; Koo, J.W.; Chang, S.O.; Oh, S.H.; Park, W.Y.; Choi, B.Y. Prevalence of p.V37I variant of GJB2 in mild or moderate hearing loss in a pediatric population and the interpretation of its pathogenicity. PLoS One 2013, 8, e61592. [CrossRef]
- Van Laer, L.; Coucke, P.; Mueller, R.F.; Caethoven, G.; Flothmann, K.; Prasad, S.D.; Chamberlin, G.P.; Houseman, M.; Taylor, G.R.; Van de Heyning, C.M.; et al. A common founder for the 35delG GJB2 gene mutation in connexin 26 hearing impairment. Journal of medical genetics 2001, 38, 515-518. [CrossRef]
- Barashkov, N.A.; Dzhemileva, L.U.; Fedorova, S.A.; Teryutin, F.M.; Posukh, O.L.; Fedotova, E.E.; Lobov, S.L.; Khusnutdinova, E.K. Autosomal recessive deafness 1A (DFNB1A) in Yakut population isolate in Eastern Siberia: extensive accumulation of the splice site mutation IVS1+1G>A in GJB2 gene as a result of founder effect. Journal of human genetics 2011, 56, 631-639. [CrossRef]
- Morell, R.J.; Kim, H.J.; Hood, L.J.; Goforth, L.; Friderici, K.; Fisher, R.; Van Camp, G.; Berlin, C.I.; Oddoux, C.; Ostrer, H.; et al. Mutations in the connexin 26 gene (GJB2) among Ashkenazi Jews with nonsyndromic recessive deafness. N Engl J Med 1998, 339, 1500-1505. [CrossRef]
- Alvarez, A.; del Castillo, I.; Villamar, M.; Aguirre, L.A.; González-Neira, A.; López-Nevot, A.; Moreno-Pelayo, M.A.; Moreno, F. High prevalence of the W24X mutation in the gene encoding connexin-26 (GJB2) in Spanish Romani (gypsies) with autosomal recessive non-syndromic hearing loss. American journal of medical genetics. Part A 2005, 137a, 255-258. [CrossRef]
- Bouwer, S.; Angelicheva, D.; Chandler, D.; Seeman, P.; Tournev, I.; Kalaydjieva, L. Carrier rates of the ancestral Indian W24X mutation in GJB2 in the general Gypsy population and individual subisolates. Genetic testing 2007, 11, 455-458. [CrossRef]
- Brobby, G.W.; Müller-Myhsok, B.; Horstmann, R.D. Connexin 26 R143W mutation associated with recessive nonsyndromic sensorineural deafness in Africa. N Engl J Med 1998, 338, 548-550. [CrossRef]
- Adadey, S.M.; Quaye, O.; Amedofu, G.K.; Awandare, G.A.; Wonkam, A. Screening for GJB2-R143W-Associated Hearing Impairment: Implications for Health Policy and Practice in Ghana. Public health genomics 2020, 23, 184-189. [CrossRef]
- Bliznetz, E.A.; Lalayants, M.R.; Markova, T.G.; Balanovsky, O.P.; Balanovska, E.V.; Skhalyakho, R.A.; Pocheshkhova, E.A.; Nikitina, N.V.; Voronin, S.V.; Kudryashova, E.K.; et al. Update of the GJB2/DFNB1 mutation spectrum in Russia: a founder Ingush mutation del(GJB2-D13S175) is the most frequent among other large deletions. Journal of human genetics 2017, 62, 789-795. [CrossRef]
- Posukh, O.L.; Maslova, E.A.; Danilchenko, V.Y.; Zytsar, M.V.; Orishchenko, K.E. Functional Consequences of Pathogenic Variants of the GJB2 Gene (Cx26) Localized in Different Cx26 Domains. Biomolecules 2023, 13. [CrossRef]
- Chan, D.K.; Chang, K.W. GJB2-associated hearing loss: systematic review of worldwide prevalence, genotype, and auditory phenotype. The Laryngoscope 2014, 124, E34-53. [CrossRef]
- Marlin, S.; Feldmann, D.; Blons, H.; Loundon, N.; Rouillon, I.; Albert, S.; Chauvin, P.; Garabédian, E.N.; Couderc, R.; Odent, S.; et al. GJB2 and GJB6 mutations: genotypic and phenotypic correlations in a large cohort of hearing-impaired patients. Archives of otolaryngology--head & neck surgery 2005, 131, 481-487. [CrossRef]
- Kecskeméti, N.; Szönyi, M.; Gáborján, A.; Küstel, M.; Milley, G.M.; Süveges, A.; Illés, A.; Kékesi, A.; Tamás, L.; Molnár, M.J.; et al. Analysis of GJB2 mutations and the clinical manifestation in a large Hungarian cohort. European archives of oto-rhino-laryngology : official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS) : affiliated with the German Society for Oto-Rhino-Laryngology - Head and Neck Surgery 2018, 275, 2441-2448. [CrossRef]
- Sobe, T.; Vreugde, S.; Shahin, H.; Berlin, M.; Davis, N.; Kanaan, M.; Yaron, Y.; Orr-Urtreger, A.; Frydman, M.; Shohat, M.; et al. The prevalence and expression of inherited connexin 26 mutations associated with nonsyndromic hearing loss in the Israeli population. Human genetics 2000, 106, 50-57. [CrossRef]
- Kenna, M.A.; Feldman, H.A.; Neault, M.W.; Frangulov, A.; Wu, B.L.; Fligor, B.; Rehm, H.L. Audiologic phenotype and progression in GJB2 (Connexin 26) hearing loss. Archives of otolaryngology--head & neck surgery 2010, 136, 81-87. [CrossRef]
- Belintani Piatto, V.; Vasques Moreira, O.A.; Orate Menezes da Silva, M.A.; Victor Maniglia, J.; Coimbra Pereira, M.; Sartorato, E.L. Correlation between audiometric data and the 35delG mutation in ten patients. Brazilian journal of otorhinolaryngology 2007, 73, 777-783. [CrossRef]
- Norris, V.W.; Arnos, K.S.; Hanks, W.D.; Xia, X.; Nance, W.E.; Pandya, A. Does universal newborn hearing screening identify all children with GJB2 (Connexin 26) deafness? Penetrance of GJB2 deafness. Ear and hearing 2006, 27, 732-741. [CrossRef]
- Minami, S.B.; Mutai, H.; Nakano, A.; Arimoto, Y.; Taiji, H.; Morimoto, N.; Sakata, H.; Adachi, N.; Masuda, S.; Sakamoto, H.; et al. GJB2-associated hearing loss undetected by hearing screening of newborns. Gene 2013, 532, 41-45. [CrossRef]
- Li, L.; Lu, J.; Tao, Z.; Huang, Q.; Chai, Y.; Li, X.; Huang, Z.; Li, Y.; Xiang, M.; Yang, J.; et al. The p.V37I exclusive genotype of GJB2: a genetic risk-indicator of postnatal permanent childhood hearing impairment. PLoS One 2012, 7, e36621. [CrossRef]
- Huang, S.; Huang, B.; Wang, G.; Yuan, Y.; Dai, P. The Relationship between the p.V37I Mutation in GJB2 and Hearing Phenotypes in Chinese Individuals. PLoS One 2015, 10, e0129662. [CrossRef]
- Chan, D.K.; Schrijver, I.; Chang, K.W. Connexin-26-associated deafness: phenotypic variability and progression of hearing loss. Genetics in medicine : official journal of the American College of Medical Genetics 2010, 12, 174-181. [CrossRef]
- Sakata, A.; Kashio, A.; Koyama, M.; Urata, S.; Koyama, H.; Yamasoba, T. Hearing and Hearing Loss Progression in Patients with GJB2 Gene Mutations: A Long-Term Follow-Up. International journal of molecular sciences 2023, 24. [CrossRef]
- Teryutin, F.M.; Pshennikova, V.G.; Solovyev, A.V.; Romanov, G.P.; Fedorova, S.A.; Barashkov, N.A. Genotype-phenotype analysis of hearing function in patients with DFNB1A caused by the c.-23+1G>A splice site variant of the GJB2 gene (Cx26). PLoS One 2024, 19, e0309439. [CrossRef]
- Aboagye, E.T.; Adadey, S.M.; Wonkam-Tingang, E.; Amenga-Etego, L.; Awandare, G.A.; Wonkam, A. Global Distribution of Founder Variants Associated with Non-Syndromic Hearing Impairment. Genes 2023, 14. [CrossRef]
- Rodriguez-Paris, J.; Tamayo, M.L.; Gelvez, N.; Schrijver, I. Allele-specific impairment of GJB2 expression by GJB6 deletion del(GJB6-D13S1854). PLoS One 2011, 6, e21665. [CrossRef]
- Cifuentes, G.A.; Diñeiro, M.; Huete, A.R.; Capín, R.; Santiago, A.; Vargas, A.A.R.; Carrero, D.; Martínez, E.L.; Aguiar, B.; Fischer, A.; et al. A Novel Recurrent 200 kb CRYL1 Deletion Underlies DFNB1A Hearing Loss in Patients from Northwestern Spain. Genes 2025, 16. [CrossRef]
- Iossa, S.; Marciano, E.; Franzé, A. GJB2 Gene Mutations in Syndromic Skin Diseases with Sensorineural Hearing Loss. Current genomics 2011, 12, 475-785. [CrossRef]
- Uyguner, O.; Tukel, T.; Baykal, C.; Eris, H.; Emiroglu, M.; Hafiz, G.; Ghanbari, A.; Baserer, N.; Yuksel-Apak, M.; Wollnik, B. The novel R75Q mutation in the GJB2 gene causes autosomal dominant hearing loss and palmoplantar keratoderma in a Turkish family. Clinical genetics 2002, 62, 306-309. [CrossRef]
- Yuan, Y.; Huang, D.; Yu, F.; Zhu, X.; Kang, D.; Yuan, H.; Han, D.; Dai, P. A de novo GJB2 (connexin 26) mutation, R75W, in a Chinese pedigree with hearing loss and palmoplantar keratoderma. American journal of medical genetics. Part A 2009, 149a, 689-692. [CrossRef]
- Maestrini, E.; Korge, B.P.; Ocaña-Sierra, J.; Calzolari, E.; Cambiaghi, S.; Scudder, P.M.; Hovnanian, A.; Monaco, A.P.; Munro, C.S. A missense mutation in connexin26, D66H, causes mutilating keratoderma with sensorineural deafness (Vohwinkel’s syndrome) in three unrelated families. Hum Mol Genet 1999, 8, 1237-1243. [CrossRef]
- Qiu, Y.; Wang, Z.; Chen, N.; Song, Y.; Wang, Z.; Zhang, L. D66H mutation in GJB2 gene in a Chinese family with classical Vohwinkel syndrome. Indian journal of dermatology, venereology and leprology 2012, 78, 640-642. [CrossRef]
- Richard, G.; Brown, N.; Ishida-Yamamoto, A.; Krol, A. Expanding the phenotypic spectrum of Cx26 disorders: Bart-Pumphrey syndrome is caused by a novel missense mutation in GJB2. The Journal of investigative dermatology 2004, 123, 856-863. [CrossRef]
- Richard, G.; Rouan, F.; Willoughby, C.E.; Brown, N.; Chung, P.; Ryynänen, M.; Jabs, E.W.; Bale, S.J.; DiGiovanna, J.J.; Uitto, J.; et al. Missense mutations in GJB2 encoding connexin-26 cause the ectodermal dysplasia keratitis-ichthyosis-deafness syndrome. American journal of human genetics 2002, 70, 1341-1348. [CrossRef]
- Janecke, A.R.; Hennies, H.C.; Günther, B.; Gansl, G.; Smolle, J.; Messmer, E.M.; Utermann, G.; Rittinger, O. GJB2 mutations in keratitis-ichthyosis-deafness syndrome including its fatal form. American journal of medical genetics. Part A 2005, 133a, 128-131. [CrossRef]
- Arita, K.; Akiyama, M.; Aizawa, T.; Umetsu, Y.; Segawa, I.; Goto, M.; Sawamura, D.; Demura, M.; Kawano, K.; Shimizu, H. A novel N14Y mutation in Connexin26 in keratitis-ichthyosis-deafness syndrome: analyses of altered gap junctional communication and molecular structure of N terminus of mutated Connexin26. The American journal of pathology 2006, 169, 416-423. [CrossRef]
- van Geel, M.; van Steensel, M.A.; Küster, W.; Hennies, H.C.; Happle, R.; Steijlen, P.M.; König, A. HID and KID syndromes are associated with the same connexin 26 mutation. The British journal of dermatology 2002, 146, 938-942. [CrossRef]
- Martínez, A.D.; Acuña, R.; Figueroa, V.; Maripillan, J.; Nicholson, B. Gap-junction channels dysfunction in deafness and hearing loss. Antioxidants & redox signaling 2009, 11, 309-322. [CrossRef]
- Zelante, L.; Gasparini, P.; Estivill, X.; Melchionda, S.; D’Agruma, L.; Govea, N.; Milá, M.; Monica, M.D.; Lutfi, J.; Shohat, M.; et al. Connexin26 mutations associated with the most common form of non-syndromic neurosensory autosomal recessive deafness (DFNB1) in Mediterraneans. Hum Mol Genet 1997, 6, 1605-1609. [CrossRef]
- Martin, P.E.; Coleman, S.L.; Casalotti, S.O.; Forge, A.; Evans, W.H. Properties of connexin26 gap junctional proteins derived from mutations associated with non-syndromal heriditary deafness. Hum Mol Genet 1999, 8, 2369-2376. [CrossRef]
- Ambrosi, C.; Walker, A.E.; Depriest, A.D.; Cone, A.C.; Lu, C.; Badger, J.; Skerrett, I.M.; Sosinsky, G.E. Analysis of trafficking, stability and function of human connexin 26 gap junction channels with deafness-causing mutations in the fourth transmembrane helix. PLoS One 2013, 8, e70916. [CrossRef]
- Beach, R.; Abitbol, J.M.; Allman, B.L.; Esseltine, J.L.; Shao, Q.; Laird, D.W. GJB2 Mutations Linked to Hearing Loss Exhibit Differential Trafficking and Functional Defects as Revealed in Cochlear-Relevant Cells. Frontiers in cell and developmental biology 2020, 8, 215. [CrossRef]
- Skerrett, I.M.; Di, W.L.; Kasperek, E.M.; Kelsell, D.P.; Nicholson, B.J. Aberrant gating, but a normal expression pattern, underlies the recessive phenotype of the deafness mutant Connexin26M34T. FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2004, 18, 860-862. [CrossRef]
- Beltramello, M.; Piazza, V.; Bukauskas, F.F.; Pozzan, T.; Mammano, F. Impaired permeability to Ins(1,4,5)P3 in a mutant connexin underlies recessive hereditary deafness. Nature cell biology 2005, 7, 63-69. [CrossRef]
- Yum, S.W.; Zhang, J.; Scherer, S.S. Dominant connexin26 mutants associated with human hearing loss have trans-dominant effects on connexin30. Neurobiology of disease 2010, 38, 226-236. [CrossRef]
- Lee, J.R.; Derosa, A.M.; White, T.W. Connexin mutations causing skin disease and deafness increase hemichannel activity and cell death when expressed in Xenopus oocytes. The Journal of investigative dermatology 2009, 129, 870-878. [CrossRef]
- Press, E.R.; Shao, Q.; Kelly, J.J.; Chin, K.; Alaga, A.; Laird, D.W. Induction of cell death and gain-of-function properties of connexin26 mutants predict severity of skin disorders and hearing loss. The Journal of biological chemistry 2017, 292, 9721-9732. [CrossRef]
- Crispino, G.; Di Pasquale, G.; Scimemi, P.; Rodriguez, L.; Galindo Ramirez, F.; De Siati, R.D.; Santarelli, R.M.; Arslan, E.; Bortolozzi, M.; Chiorini, J.A.; et al. BAAV mediated GJB2 gene transfer restores gap junction coupling in cochlear organotypic cultures from deaf Cx26Sox10Cre mice. PLoS One 2011, 6, e23279. [CrossRef]
- Yu, Q.; Wang, Y.; Chang, Q.; Wang, J.; Gong, S.; Li, H.; Lin, X. Virally expressed connexin26 restores gap junction function in the cochlea of conditional Gjb2 knockout mice. Gene Ther 2014, 21, 71-80. [CrossRef]
- Vitry, S.; Mendia, C.; Maudoux, A.; El-Amraoui, A. Advancing precision ear medicine: leveraging animal models for disease insights and therapeutic innovations. Mammalian genome : official journal of the International Mammalian Genome Society 2025, 36, 417-443. [CrossRef]
- Suzuki, J.; Hashimoto, K.; Xiao, R.; Vandenberghe, L.H.; Liberman, M.C. Cochlear gene therapy with ancestral AAV in adult mice: complete transduction of inner hair cells without cochlear dysfunction. Scientific reports 2017, 7, 45524. [CrossRef]
- Andres-Mateos, E.; Landegger, L.D.; Unzu, C.; Phillips, J.; Lin, B.M.; Dewyer, N.A.; Sanmiguel, J.; Nicolaou, F.; Valero, M.D.; Bourdeu, K.I.; et al. Choice of vector and surgical approach enables efficient cochlear gene transfer in nonhuman primate. Nature communications 2022, 13, 1359. [CrossRef]
- Ishibashi, Y.; Sung, C.Y.W.; Grati, M.; Chien, W. Immune responses in the mammalian inner ear and their implications for AAV-mediated inner ear gene therapy. Hear Res 2023, 432, 108735. [CrossRef]
- Seist, R.; Copeland, J.S.; Tao, L.; Groves, A.K. Rational design of a Lfng-enhancer AAV construct drives specific and efficient gene expression in inner ear supporting cells. Hear Res 2025, 458, 109203. [CrossRef]
- Ni, X.; Liu, H.; Ji, X.; Chen, Z.; Chen, X.; Luo, Y.; Chen, W.; Yang, S. Cochlear inflammation in AAV-mediated gene therapy: serotype-dependent macrophage activation and promoter-driven immunogenicity. Acta oto-laryngologica 2025, 145, 571-579. [CrossRef]
- Ishibashi, Y.; Zhu, J.; Gernoux, G.; Yu, Y.; Suh, M.J.; Isgrig, K.; Grati, M.; Olszewski, R.; Hoa, M.; Liang, C.; et al. AAV-mediated inner ear gene delivery triggers mild host immune responses in the mammalian inner ear. Mol Ther Methods Clin Dev 2025, 33, 101456. [CrossRef]
- Shi, D.; Han, L.; Li, C.; Geng, G.; Jiang, L.; Chen, X.; Yang, F.; Feng, Y.; Luo, J.; Shu, Y. Transcriptomic Profiling Reveals Divergent Immune Responses to AAV1 and AAV-ie in Mice Inner Ear. International journal of medical sciences 2026, 23, 611-619. [CrossRef]
- Iizuka, T.; Kamiya, K.; Gotoh, S.; Sugitani, Y.; Suzuki, M.; Noda, T.; Minowa, O.; Ikeda, K. Perinatal Gjb2 gene transfer rescues hearing in a mouse model of hereditary deafness. Hum Mol Genet 2015, 24, 3651-3661. [CrossRef]
- Guo, J.; Ma, X.; Skidmore, J.M.; Cimerman, J.; Prieskorn, D.M.; Beyer, L.A.; Swiderski, D.L.; Dolan, D.F.; Martin, D.M.; Raphael, Y. GJB2 gene therapy and conditional deletion reveal developmental stage-dependent effects on inner ear structure and function. Mol Ther Methods Clin Dev 2021, 23, 319-333. [CrossRef]
- Wang, X.; Zhang, L.; Chen, S.; Xie, L.; Qiu, Y.; Kong, C.; Yin, G.; Kong, W.; Sun, Y. Viral-Mediated Connexin 26 Expression Combined with Dexamethasone Rescues Hearing in a Conditional Gjb2 Null Mice Model. Advanced science (Weinheim, Baden-Wurttemberg, Germany) 2025, 12, e2406510. [CrossRef]
- Ivanchenko, M.V.; Booth, K.T.A.; Karavitaki, K.D.; Antonellis, L.M.; Nagy, M.A.; Peters, C.W.; Price, S.; Li, Y.; Lytvyn, A.; Ward, A.; et al. Cell-specific delivery of GJB2 restores auditory function in mouse models of DFNB1 deafness and mediates appropriate expression in NHP cochlea. Nature communications 2025, 16, 11157. [CrossRef]
- Zong, Y.J.; Liu, X.Z.; Shi, X.Y.; Zhao, Z.D.; Sun, Y. Promotion of Cx26 mutants located in TM4 region for membrane translocation successfully rescued hearing loss. Theranostics 2025, 15, 5801-5825. [CrossRef]
- Zhu, Y.; Chen, J.; Liang, C.; Zong, L.; Chen, J.; Jones, R.O.; Zhao, H.B. Connexin26 (GJB2) deficiency reduces active cochlear amplification leading to late-onset hearing loss. Neuroscience 2015, 284, 719-729. [CrossRef]
- Wang, Y.; Jin, Y.; Zhang, Q.; Xiong, Y.; Gu, X.; Zeng, S.; Chen, W. Research progress in delineating the pathological mechanisms of GJB2-related hearing loss. Frontiers in cellular neuroscience 2023, 17, 1208406. [CrossRef]
- Zhao, H.B.; Yu, N. Distinct and gradient distributions of connexin26 and connexin30 in the cochlear sensory epithelium of guinea pigs. The Journal of comparative neurology 2006, 499, 506-518. [CrossRef]
- Mammano, F. Inner Ear Connexin Channels: Roles in Development and Maintenance of Cochlear Function. Cold Spring Harbor perspectives in medicine 2019, 9. [CrossRef]
- Zhu, Y.; Zong, L.; Mei, L.; Zhao, H.B. Connexin26 gap junction mediates miRNA intercellular genetic communication in the cochlea and is required for inner ear development. Scientific reports 2015, 5, 15647. [CrossRef]
- Lin, L.; Wang, Y.F.; Wang, S.Y.; Liu, S.F.; Yu, Z.; Xi, L.; Li, H.W. Ultrastructural pathological changes in the cochlear cells of connexin 26 conditional knockout mice. Mol Med Rep 2013, 8, 1029-1036. [CrossRef]
- Chang, Q.; Tang, W.; Kim, Y.; Lin, X. Timed conditional null of connexin26 in mice reveals temporary requirements of connexin26 in key cochlear developmental events before the onset of hearing. Neurobiology of disease 2015, 73, 418-427. [CrossRef]
- Chen, S.; Xie, L.; Xu, K.; Cao, H.Y.; Wu, X.; Xu, X.X.; Sun, Y.; Kong, W.J. Developmental abnormalities in supporting cell phalangeal processes and cytoskeleton in the Gjb2 knockdown mouse model. Disease models & mechanisms 2018, 11. [CrossRef]
- Xie, L.; Chen, S.; Xu, K.; Cao, H.Y.; Du, A.N.; Bai, X.; Sun, Y.; Kong, W.J. Reduced postnatal expression of cochlear Connexin26 induces hearing loss and affects the developmental status of pillar cells in a dose-dependent manner. Neurochemistry international 2019, 128, 196-205. [CrossRef]
- Yang, W.; Vethanayagam, R.R.; Dong, Y.; Cai, Q.; Hu, B.H. Activation of the antigen presentation function of mononuclear phagocyte populations associated with the basilar membrane of the cochlea after acoustic overstimulation. Neuroscience 2015, 303, 1-15. [CrossRef]
- Hirose, K.; Discolo, C.M.; Keasler, J.R.; Ransohoff, R. Mononuclear phagocytes migrate into the murine cochlea after acoustic trauma. The Journal of comparative neurology 2005, 489, 180-194. [CrossRef]
- Wang, Y.; Chang, Q.; Tang, W.; Sun, Y.; Zhou, B.; Li, H.; Lin, X. Targeted connexin26 ablation arrests postnatal development of the organ of Corti. Biochemical and biophysical research communications 2009, 385, 33-37. [CrossRef]
- Liang, C.; Zhu, Y.; Zong, L.; Lu, G.J.; Zhao, H.B. Cell degeneration is not a primary causer for Connexin26 (GJB2) deficiency associated hearing loss. Neuroscience letters 2012, 528, 36-41. [CrossRef]
- Liu, X.; Xie, L.; Jin, Y.; Chen, S.; Shi, X.; Zong, Y.; Zhao, Z.; Kong, W.J.; Sun, Y. Connexin 26 Functions as a Direct Transcriptional Regulator During the Cochlea Development. Advanced science (Weinheim, Baden-Wurttemberg, Germany) 2026, 13, e14136. [CrossRef]
- Hahn, R.; Avraham, K.B. Gene Therapy for Inherited Hearing Loss: Updates and Remaining Challenges. Audiology research 2023, 13, 952-966. [CrossRef]
- Chen, S.; Sun, Y.; Lin, X.; Kong, W. Down regulated connexin26 at different postnatal stage displayed different types of cellular degeneration and formation of organ of Corti. Biochemical and biophysical research communications 2014, 445, 71-77. [CrossRef]
- Li, Q.; Cui, C.; Liao, R.; Yin, X.; Wang, D.; Cheng, Y.; Huang, B.; Wang, L.; Yan, M.; Zhou, J.; et al. The pathogenesis of common Gjb2 mutations associated with human hereditary deafness in mice. Cellular and molecular life sciences : CMLS 2023, 80, 148. [CrossRef]
- Kammen-Jolly, K.; Ichiki, H.; Scholtz, A.W.; Gsenger, M.; Kreczy, A.; Schrott-Fischer, A. Connexin 26 in human fetal development of the inner ear. Hear Res 2001, 160, 15-21. [CrossRef]
- Qu, Y.; Tang, W.; Zhou, B.; Ahmad, S.; Chang, Q.; Li, X.; Lin, X. Early developmental expression of connexin26 in the cochlea contributes to its dominate functional role in the cochlear gap junctions. Biochemical and biophysical research communications 2012, 417, 245-250. [CrossRef]
- Chen, S.; Xu, K.; Xie, L.; Cao, H.Y.; Wu, X.; Du, A.N.; He, Z.H.; Lin, X.; Sun, Y.; Kong, W.J. The spatial distribution pattern of Connexin26 expression in supporting cells and its role in outer hair cell survival. Cell death & disease 2018, 9, 1180. [CrossRef]
- Hosoya, M.; Fujioka, M.; Murayama, A.Y.; Ogawa, K.; Okano, H.; Ozawa, H. Dynamic Spatiotemporal Expression Changes in Connexins of the Developing Primate’s Cochlea. Genes 2021, 12. [CrossRef]
- Hastings, M.L.; Brigande, J.V. Fetal gene therapy and pharmacotherapy to treat congenital hearing loss and vestibular dysfunction. Hear Res 2020, 394, 107931. [CrossRef]
- Gadenstaetter, A.J.; Krumpoeck, P.E.; Landegger, L.D. Inner Ear Gene Therapy: An Overview from Bench to Bedside. Molecular diagnosis & therapy 2025, 29, 161-181. [CrossRef]
- Omichi, R.; Shibata, S.B.; Morton, C.C.; Smith, R.J.H. Gene therapy for hearing loss. Hum Mol Genet 2019, 28, R65-r79. [CrossRef]
- Das, S.; Manor, U. Gene therapy for hearing loss: challenges and the promise of cellular plasticity and epigenetic modulation. Frontiers in neurology 2024, 15, 1511938. [CrossRef]
- Bai, X.; Xu, K.; Xie, L.; Qiu, Y.; Chen, S.; Sun, Y. The Dual Roles of Triiodothyronine in Regulating the Morphology of Hair Cells and Supporting Cells during Critical Periods of Mouse Cochlear Development. International journal of molecular sciences 2023, 24. [CrossRef]
- Minekawa, A.; Abe, T.; Inoshita, A.; Iizuka, T.; Kakehata, S.; Narui, Y.; Koike, T.; Kamiya, K.; Okamura, H.O.; Shinkawa, H.; et al. Cochlear outer hair cells in a dominant-negative connexin26 mutant mouse preserve non-linear capacitance in spite of impaired distortion product otoacoustic emission. Neuroscience 2009, 164, 1312-1319. [CrossRef]
- Anzai, T.; Fukunaga, I.; Hatakeyama, K.; Fujimoto, A.; Kobayashi, K.; Nishikawa, A.; Aoki, T.; Noda, T.; Minowa, O.; Ikeda, K.; et al. Deformation of the Outer Hair Cells and the Accumulation of Caveolin-2 in Connexin 26 Deficient Mice. PLoS One 2015, 10, e0141258. [CrossRef]
- Xu, K.; Chen, S.; Xie, L.; Qiu, Y.; Bai, X.; Liu, X.Z.; Zhang, H.M.; Wang, X.H.; Jin, Y.; Sun, Y.; et al. Local Macrophage-Related Immune Response Is Involved in Cochlear Epithelial Damage in Distinct Gjb2-Related Hereditary Deafness Models. Frontiers in cell and developmental biology 2020, 8, 597769. [CrossRef]
- Zhang, Y.; Zhang, X.; Li, L.; Sun, Y.; Sun, J. Apoptosis Progression in the Hair Cells in the Organ of Corti of GJB2 Conditional Knockout Mice. Clinical and experimental otorhinolaryngology 2012, 5, 132-138. [CrossRef]
- Wang, Y.; Lin, C.; He, Y.; Li, A.; Ni, W.; Sun, S.; Gu, X.; Li, J.; Li, H. Mir-27a promotes apoptosis of cochlear sensory epithelium in Cx26 knockout mice. Frontiers in bioscience (Landmark edition) 2016, 21, 364-373. [CrossRef]
- Wang, X.; Chen, S.; Qiu, Y.; Xu, K.; Bai, X.; Xie, L.; Kong, W.; Sun, Y. PARP inhibitor rescues hearing and hair cell impairment in Cx26-null mice. 2023, 4, 20230066. [CrossRef]
- Wan, G.; Corfas, G. Transient auditory nerve demyelination as a new mechanism for hidden hearing loss. Nature communications 2017, 8, 14487. [CrossRef]
- Kujawa, S.G.; Liberman, M.C. Adding insult to injury: cochlear nerve degeneration after “temporary” noise-induced hearing loss. The Journal of neuroscience : the official journal of the Society for Neuroscience 2009, 29, 14077-14085. [CrossRef]
- Qiu, Y.; Xie, L.; Wang, X.; Xu, K.; Bai, X.; Chen, S.; Sun, Y. Abnormal Innervation, Demyelination, and Degeneration of Spiral Ganglion Neurons as Well as Disruption of Heminodes are Involved in the Onset of Deafness in Cx26 Null Mice. Neuroscience bulletin 2024, 40, 1093-1103. [CrossRef]
- Zhou, X.X.; Chen, S.; Xie, L.; Ji, Y.Z.; Wu, X.; Wang, W.W.; Yang, Q.; Yu, J.T.; Sun, Y.; Lin, X.; et al. Reduced Connexin26 in the Mature Cochlea Increases Susceptibility to Noise-Induced Hearing Lossin Mice. International journal of molecular sciences 2016, 17, 301. [CrossRef]
- Bai, X.; Liao, B.; Hu, W.H.; Wang, M.Q.; Chen, X.B.; Liu, Y.H.; Sun, Y.; Xu, K. Calcium blockers protect against sensory epithelial damage and hearing loss in Cx26-cKO mice. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie 2025, 193, 118829. [CrossRef]
- Zhang, L.; Chen, S.; Sun, Y. Mechanism and Prevention of Spiral Ganglion Neuron Degeneration in the Cochlea. Frontiers in cellular neuroscience 2021, 15, 814891. [CrossRef]
- Takada, Y.; Beyer, L.A.; Swiderski, D.L.; O’Neal, A.L.; Prieskorn, D.M.; Shivatzki, S.; Avraham, K.B.; Raphael, Y. Connexin 26 null mice exhibit spiral ganglion degeneration that can be blocked by BDNF gene therapy. Hear Res 2014, 309, 124-135. [CrossRef]
- Xu, K.; Chen, S.; Xie, L.; Qiu, Y.; Liu, X.Z.; Bai, X.; Jin, Y.; Wang, X.H.; Sun, Y. The protective effects of systemic dexamethasone on sensory epithelial damage and hearing loss in targeted Cx26-null mice. Cell death & disease 2022, 13, 545. [CrossRef]
- Zhao, H.B.; Kikuchi, T.; Ngezahayo, A.; White, T.W. Gap junctions and cochlear homeostasis. The Journal of membrane biology 2006, 209, 177-186. [CrossRef]
- Wu, X.; Zhang, W.; Li, Y.; Lin, X. Structure and Function of Cochlear Gap Junctions and Implications for the Translation of Cochlear Gene Therapies. Frontiers in cellular neuroscience 2019, 13, 529. [CrossRef]
- Zhang, Y.; Tang, W.; Ahmad, S.; Sipp, J.A.; Chen, P.; Lin, X. Gap junction-mediated intercellular biochemical coupling in cochlear supporting cells is required for normal cochlear functions. Proceedings of the National Academy of Sciences of the United States of America 2005, 102, 15201-15206. [CrossRef]
- Zhu, Y.; Liang, C.; Chen, J.; Zong, L.; Chen, G.D.; Zhao, H.B. Active cochlear amplification is dependent on supporting cell gap junctions. Nature communications 2013, 4, 1786. [CrossRef]
- Liu, W.; Boström, M.; Kinnefors, A.; Rask-Andersen, H. Unique expression of connexins in the human cochlea. Hear Res 2009, 250, 55-62. [CrossRef]
- Chang, Q.; Tang, W.; Ahmad, S.; Zhou, B.; Lin, X. Gap junction mediated intercellular metabolite transfer in the cochlea is compromised in connexin30 null mice. PLoS One 2008, 3, e4088. [CrossRef]
- Wang, G.; Simon, D.J.; Wu, Z.; Belsky, D.M.; Heller, E.; O’Rourke, M.K.; Hertz, N.T.; Molina, H.; Zhong, G.; Tessier-Lavigne, M.; et al. Structural plasticity of actin-spectrin membrane skeleton and functional role of actin and spectrin in axon degeneration. eLife 2019, 8. [CrossRef]
- Mohler, P.J.; Davis, J.Q.; Bennett, V. Ankyrin-B coordinates the Na/K ATPase, Na/Ca exchanger, and InsP3 receptor in a cardiac T-tubule/SR microdomain. PLoS biology 2005, 3, e423. [CrossRef]
- Cunha, S.R.; Bhasin, N.; Mohler, P.J. Targeting and stability of Na/Ca exchanger 1 in cardiomyocytes requires direct interaction with the membrane adaptor ankyrin-B. The Journal of biological chemistry 2007, 282, 4875-4883. [CrossRef]
- Chen, P.; Wu, W.; Zhang, J.; Chen, J.; Li, Y.; Sun, L.; Hou, S.; Yang, J. Pathological mechanisms of connexin26-related hearing loss: Potassium recycling, ATP-calcium signaling, or energy supply? Frontiers in molecular neuroscience 2022, 15, 976388. [CrossRef]
- Chen, J.; Chen, J.; Zhu, Y.; Liang, C.; Zhao, H.B. Deafness induced by Connexin 26 (GJB2) deficiency is not determined by endocochlear potential (EP) reduction but is associated with cochlear developmental disorders. Biochemical and biophysical research communications 2014, 448, 28-32. [CrossRef]
- Anselmi, F.; Hernandez, V.H.; Crispino, G.; Seydel, A.; Ortolano, S.; Roper, S.D.; Kessaris, N.; Richardson, W.; Rickheit, G.; Filippov, M.A.; et al. ATP release through connexin hemichannels and gap junction transfer of second messengers propagate Ca2+ signals across the inner ear. Proceedings of the National Academy of Sciences of the United States of America 2008, 105, 18770-18775. [CrossRef]
- Majumder, P.; Crispino, G.; Rodriguez, L.; Ciubotaru, C.D.; Anselmi, F.; Piazza, V.; Bortolozzi, M.; Mammano, F. ATP-mediated cell-cell signaling in the organ of Corti: the role of connexin channels. Purinergic signalling 2010, 6, 167-187. [CrossRef]
- Ceriani, F.; Pozzan, T.; Mammano, F. Critical role of ATP-induced ATP release for Ca2+ signaling in nonsensory cell networks of the developing cochlea. Proceedings of the National Academy of Sciences of the United States of America 2016, 113, E7194-e7201. [CrossRef]
- Mazzarda, F.; D’Elia, A.; Massari, R.; De Ninno, A.; Bertani, F.R.; Businaro, L.; Ziraldo, G.; Zorzi, V.; Nardin, C.; Peres, C.; et al. Organ-on-chip model shows that ATP release through connexin hemichannels drives spontaneous Ca(2+) signaling in non-sensory cells of the greater epithelial ridge in the developing cochlea. Lab on a chip 2020, 20, 3011-3023. [CrossRef]
- Jagger, D.J.; Forge, A. Connexins and gap junctions in the inner ear--it’s not just about K⁺ recycling. Cell and tissue research 2015, 360, 633-644. [CrossRef]
- Schütz, M.; Scimemi, P.; Majumder, P.; De Siati, R.D.; Crispino, G.; Rodriguez, L.; Bortolozzi, M.; Santarelli, R.; Seydel, A.; Sonntag, S.; et al. The human deafness-associated connexin 30 T5M mutation causes mild hearing loss and reduces biochemical coupling among cochlear non-sensory cells in knock-in mice. Hum Mol Genet 2010, 19, 4759-4773. [CrossRef]
- Johnson, S.L.; Ceriani, F.; Houston, O.; Polishchuk, R.; Polishchuk, E.; Crispino, G.; Zorzi, V.; Mammano, F.; Marcotti, W. Connexin-Mediated Signaling in Nonsensory Cells Is Crucial for the Development of Sensory Inner Hair Cells in the Mouse Cochlea. The Journal of neuroscience : the official journal of the Society for Neuroscience 2017, 37, 258-268. [CrossRef]
- Sun, L.; Gao, D.; Chen, J.; Hou, S.; Li, Y.; Huang, Y.; Mammano, F.; Chen, J.; Yang, J. Failure Of Hearing Acquisition in Mice With Reduced Expression of Connexin 26 Correlates With the Abnormal Phasing of Apoptosis Relative to Autophagy and Defective ATP-Dependent Ca(2+) Signaling in Kölliker’s Organ. Frontiers in cellular neuroscience 2022, 16, 816079. [CrossRef]
- Vlajkovic, S.M.; Suzuki-Kerr, H.; Nayagam, B.A. Cochlear Homeostasis in Sensorineural Hearing Loss: Mechanisms, Implications, and Therapeutic Prospects. International journal of molecular sciences 2025, 27. [CrossRef]
- Chen, T.; Rohacek, A.M.; Caporizzo, M.; Nankali, A.; Smits, J.J.; Oostrik, J.; Lanting, C.P.; Kücük, E.; Gilissen, C.; van de Kamp, J.M.; et al. Cochlear supporting cells require GAS2 for cytoskeletal architecture and hearing. Developmental cell 2021, 56, 1526-1540.e1527. [CrossRef]
- Yu, N.; Zhao, H.B. Modulation of outer hair cell electromotility by cochlear supporting cells and gap junctions. PLoS One 2009, 4, e7923. [CrossRef]
- Levic, S.; Lukashkina, V.A.; Simões, P.; Lukashkin, A.N.; Russell, I.J. A Gap-Junction Mutation Reveals That Outer Hair Cell Extracellular Receptor Potentials Drive High-Frequency Cochlear Amplification. The Journal of neuroscience : the official journal of the Society for Neuroscience 2022, 42, 7875-7884. [CrossRef]
- Zhao, H.B. Hypothesis of K(+)-Recycling Defect Is Not a Primary Deafness Mechanism for Cx26 (GJB2) Deficiency. Frontiers in molecular neuroscience 2017, 10, 162. [CrossRef]
- Lin, X.; Li, G.; Zhang, Y.; Zhao, J.; Lu, J.; Gao, Y.; Liu, H.; Li, G.L.; Yang, T.; Song, L.; et al. Hearing consequences in Gjb2 knock-in mice: implications for human p.V37I mutation. Aging 2019, 11, 7416-7441. [CrossRef]
- Xu, K.; Chen, S.; Bai, X.; Xie, L.; Qiu, Y.; Liu, X.Z.; Wang, X.H.; Kong, W.J.; Sun, Y. Degradation of cochlear Connexin26 accelerate the development of age-related hearing loss. Aging cell 2023, 22, e13973. [CrossRef]
- Fetoni, A.R.; Zorzi, V.; Paciello, F.; Ziraldo, G.; Peres, C.; Raspa, M.; Scavizzi, F.; Salvatore, A.M.; Crispino, G.; Tognola, G.; et al. Cx26 partial loss causes accelerated presbycusis by redox imbalance and dysregulation of Nfr2 pathway. Redox biology 2018, 19, 301-317. [CrossRef]
- Sun, L.; Zhang, Y.; Fei, Z.; Wang, X.; Duan, M.; Chen, J.; Yang, J. The cochlear cell atlas of Gjb2-/- mice exhibits alterations accompanied by autophagy-related metabolic imbalances. Free radical biology & medicine 2025, 238, 569-581. [CrossRef]
- Gentile, G.; Paciello, F.; Zorzi, V.; Spampinato, A.G.; Guarnaccia, M.; Crispino, G.; Tettey-Matey, A.; Scavizzi, F.; Raspa, M.; Fetoni, A.R.; et al. miRNA and mRNA Profiling Links Connexin Deficiency to Deafness via Early Oxidative Damage in the Mouse Stria Vascularis. Frontiers in cell and developmental biology 2020, 8, 616878. [CrossRef]
- Mammano, F.; Bortolozzi, M. Ca(2+) signaling, apoptosis and autophagy in the developing cochlea: Milestones to hearing acquisition. Cell calcium 2018, 70, 117-126. [CrossRef]
- Bai, X.; Liao, B.; Hu, W.H.; Wang, M.Q.; Sun, Y.; Chen, X.B.; Xu, K. Cochlear endoplasmic reticulum stress causes connexin 26 degradation is involved in age-related hearing loss. iScience 2025, 28, 113891. [CrossRef]
- Paciello, F.; Pisani, A.; Rolesi, R.; Montuoro, R.; Mohamed-Hizam, V.; Boni, G.; Ripoli, C.; Galli, J.; Sisto, R.; Fetoni, A.R.; et al. Oxidative stress and inflammation cause auditory system damage via glial cell activation and dysregulated expression of gap junction proteins in an experimental model of styrene-induced oto/neurotoxicity. Journal of neuroinflammation 2024, 21, 4. [CrossRef]
- Maniaci, A.; La Via, L.; Lechien, J.R.; Sangiorgio, G.; Iannella, G.; Magliulo, G.; Pace, A.; Mat, Q.; Lavalle, S.; Lentini, M. Hearing Loss and Oxidative Stress: A Comprehensive Review. Antioxidants (Basel, Switzerland) 2024, 13. [CrossRef]
- Liu, Y.C.; Xu, K. Macrophage-related immune responses in inner ear: a potential therapeutic target for sensorineural hearing loss. Frontiers in neuroscience 2023, 17, 1339134. [CrossRef]
- Frye, M.D.; Ryan, A.F.; Kurabi, A. Inflammation associated with noise-induced hearing loss. The Journal of the Acoustical Society of America 2019, 146, 4020. [CrossRef]
- Pan, J.; Wang, K.; Qu, J.; Chen, D.; Chen, A.; You, Y.; Tang, J.; Zhang, H. Activated tissue-resident macrophages contribute to hair cell insults in noise-induced hearing loss in mice. Communications biology 2024, 7, 1078. [CrossRef]
- Paciello, F.; Ripoli, C.; Fetoni, A.R.; Grassi, C. Redox Imbalance as a Common Pathogenic Factor Linking Hearing Loss and Cognitive Decline. Antioxidants (Basel, Switzerland) 2023, 12. [CrossRef]
- Mukherjea, D.; Jajoo, S.; Kaur, T.; Sheehan, K.E.; Ramkumar, V.; Rybak, L.P. Transtympanic administration of short interfering (si)RNA for the NOX3 isoform of NADPH oxidase protects against cisplatin-induced hearing loss in the rat. Antioxidants & redox signaling 2010, 13, 589-598. [CrossRef]
- Coleman, J.K.; Kopke, R.D.; Liu, J.; Ge, X.; Harper, E.A.; Jones, G.E.; Cater, T.L.; Jackson, R.L. Pharmacological rescue of noise induced hearing loss using N-acetylcysteine and acetyl-L-carnitine. Hear Res 2007, 226, 104-113. [CrossRef]
- Fetoni, A.R.; Ralli, M.; Sergi, B.; Parrilla, C.; Troiani, D.; Paludetti, G. Protective effects of N-acetylcysteine on noise-induced hearing loss in guinea pigs. Acta otorhinolaryngologica Italica : organo ufficiale della Societa italiana di otorinolaringologia e chirurgia cervico-facciale 2009, 29, 70-75.
- Ding, D.; Jiang, H.; Chen, G.D.; Longo-Guess, C.; Muthaiah, V.P.; Tian, C.; Sheppard, A.; Salvi, R.; Johnson, K.R. N-acetyl-cysteine prevents age-related hearing loss and the progressive loss of inner hair cells in γ-glutamyl transferase 1 deficient mice. Aging 2016, 8, 730-750. [CrossRef]
- Bai, X.; Chen, S.; Xu, K.; Jin, Y.; Niu, X.; Xie, L.; Qiu, Y.; Liu, X.Z.; Sun, Y. N-Acetylcysteine Combined With Dexamethasone Treatment Improves Sudden Sensorineural Hearing Loss and Attenuates Hair Cell Death Caused by ROS Stress. Frontiers in cell and developmental biology 2021, 9, 659486. [CrossRef]
- Eshraghi, A.A.; Shahal, D.; Davies, C.; Mittal, J.; Shah, V.; Bulut, E.; Garnham, C.; Sinha, P.; Mishra, D.; Marwede, H.; et al. Evaluating the Efficacy of L-N-acetylcysteine and Dexamethasone in Combination to Provide Otoprotection for Electrode Insertion Trauma. J Clin Med 2020, 9. [CrossRef]
- Eshraghi, A.A.; Roell, J.; Shaikh, N.; Telischi, F.F.; Bauer, B.; Guardiola, M.; Bas, E.; Van De Water, T.; Rivera, I.; Mittal, J. A novel combination of drug therapy to protect residual hearing post cochlear implant surgery. Acta oto-laryngologica 2016, 136, 420-424. [CrossRef]
- Zhang, J.; Wang, H.; Yan, C.; Guan, J.; Yin, L.; Lan, L.; Li, J.; Zhao, L.; Wang, Q. The Frequency of Common Deafness-Associated Variants Among 3,555,336 Newborns in China and 141,456 Individuals Across Seven Populations Worldwide. Ear and hearing 2023, 44, 232-241. [CrossRef]
- Liu, Y.; Wang, L.; Yuan, L.; Li, Y.; Chen, Z.; Yang, B.; Wang, D.; Sun, Y. Hereditary deafness carrier screening in 9,993 Chinese individuals. Frontiers in genetics 2023, 14, 1327258. [CrossRef]
- Zhao, S.; Xiang, J.; Fan, C.; Asan; Shang, X.; Zhang, X.; Chen, Y.; Zhu, B.; Cai, W.; Chen, S.; et al. Pilot study of expanded carrier screening for 11 recessive diseases in China: results from 10,476 ethnically diverse couples. European journal of human genetics : EJHG 2019, 27, 254-262. [CrossRef]
- Gregg, A.R.; Aarabi, M.; Klugman, S.; Leach, N.T.; Bashford, M.T.; Goldwaser, T.; Chen, E.; Sparks, T.N.; Reddi, H.V.; Rajkovic, A.; et al. Screening for autosomal recessive and X-linked conditions during pregnancy and preconception: a practice resource of the American College of Medical Genetics and Genomics (ACMG). Genetics in medicine : official journal of the American College of Medical Genetics 2021, 23, 1793-1806. [CrossRef]
- Sagaser, K.G.; Malinowski, J.; Westerfield, L.; Proffitt, J.; Hicks, M.A.; Toler, T.L.; Blakemore, K.J.; Stevens, B.K.; Oakes, L.M. Expanded carrier screening for reproductive risk assessment: An evidence-based practice guideline from the National Society of Genetic Counselors. Journal of genetic counseling 2023, 32, 540-557. [CrossRef]
- Dai, P.; Huang, L.H.; Wang, G.J.; Gao, X.; Qu, C.Y.; Chen, X.W.; Ma, F.R.; Zhang, J.; Xing, W.L.; Xi, S.Y.; et al. Concurrent Hearing and Genetic Screening of 180,469 Neonates with Follow-up in Beijing, China. American journal of human genetics 2019, 105, 803-812. [CrossRef]
- Schimmenti, L.A.; Warman, B.; Schleiss, M.R.; Daly, K.A.; Ross, J.A.; McCann, M.; Jurek, A.M.; Berry, S.A. Evaluation of newborn screening bloodspot-based genetic testing as second tier screen for bedside newborn hearing screening. Genetics in medicine : official journal of the American College of Medical Genetics 2011, 13, 1006-1010. [CrossRef]
- Zhu, Q.W.; Li, M.T.; Zhuang, X.; Chen, K.; Xu, W.Q.; Jiang, Y.H.; Qin, G. Assessment of Hearing Screening Combined With Limited and Expanded Genetic Screening for Newborns in Nantong, China. JAMA network open 2021, 4, e2125544. [CrossRef]
- Hoefsloot, L.H.; Roux, A.F.; Bitner-Glindzicz, M. EMQN Best Practice guidelines for diagnostic testing of mutations causing non-syndromic hearing impairment at the DFNB1 locus. European journal of human genetics : EJHG 2013, 21, 1325-1329. [CrossRef]
- Wilch, E.; Azaiez, H.; Fisher, R.A.; Elfenbein, J.; Murgia, A.; Birkenhäger, R.; Bolz, H.; Da Silva-Costa, S.M.; Del Castillo, I.; Haaf, T.; et al. A novel DFNB1 deletion allele supports the existence of a distant cis-regulatory region that controls GJB2 and GJB6 expression. Clinical genetics 2010, 78, 267-274. [CrossRef]
- Alford, R.L.; Arnos, K.S.; Fox, M.; Lin, J.W.; Palmer, C.G.; Pandya, A.; Rehm, H.L.; Robin, N.H.; Scott, D.A.; Yoshinaga-Itano, C. American College of Medical Genetics and Genomics guideline for the clinical evaluation and etiologic diagnosis of hearing loss. Genetics in medicine : official journal of the American College of Medical Genetics 2014, 16, 347-355. [CrossRef]
- Li, M.M.; Tayoun, A.A.; DiStefano, M.; Pandya, A.; Rehm, H.L.; Robin, N.H.; Schaefer, A.M.; Yoshinaga-Itano, C. Clinical evaluation and etiologic diagnosis of hearing loss: A clinical practice resource of the American College of Medical Genetics and Genomics (ACMG). Genetics in medicine : official journal of the American College of Medical Genetics 2022, 24, 1392-1406. [CrossRef]
- Antoniadi, T.; Pampanos, A.; Petersen, M.B. Prenatal diagnosis of prelingual deafness: carrier testing and prenatal diagnosis of the common GJB2 35delG mutation. Prenatal diagnosis 2001, 21, 10-13. [CrossRef]
- Chen, H.L.; Lin, P.H.; Chiang, Y.T.; Huang, W.J.; Lin, C.F.; Ma, G.C.; Chang, S.P.; Fan, J.Y.; Lin, S.Y.; Wu, C.C.; et al. Preimplantation Genetic Diagnosis in Hereditary Hearing Impairment. Diagnostics (Basel, Switzerland) 2021, 11. [CrossRef]
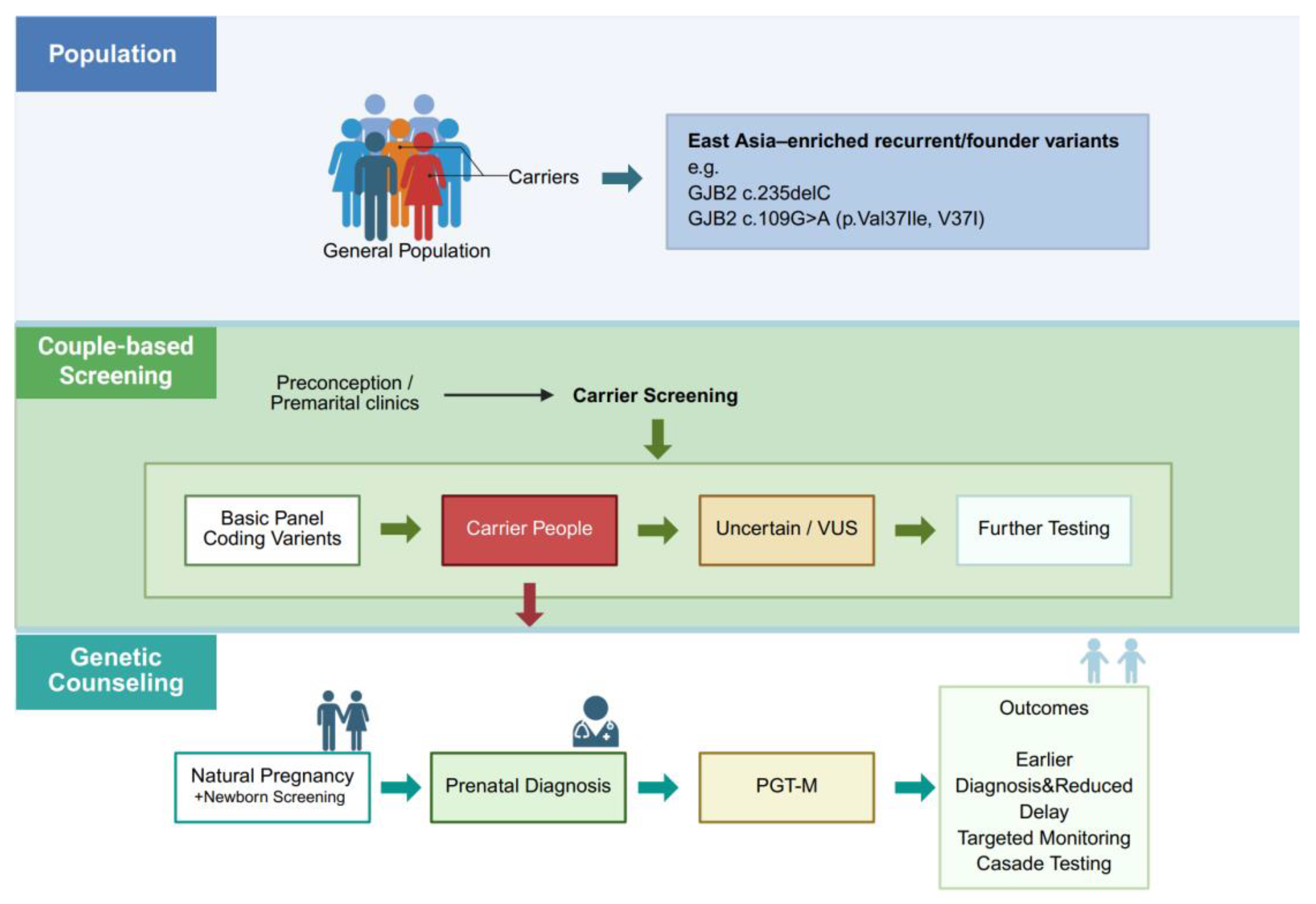
| Population/Region | Major recurrent/founder alleles | Notes |
| East Asia [50,51,52,53] | c.235delC c.109G>A(p.Val37Ile) c.299_300delAT c.-23+1G>A (region-specific) |
A limited, population-matched panel can capture a substantial proportion of DFNB1 cases; p.Val37Ile is often linked to milder/variable or later-onset hearing loss. |
| Europe [54] | c.35delG | High-yield first-tier variant in many cohorts; consider reflex testing for DFNB1 structural variants (e.g., GJB6 deletions) when only one pathogenic GJB2 allele is detected. |
| Yakut / Native Siberian groups [55] | c.-23+1G>A (IVS1+1G>A) | Classic founder architecture in Yakut and related groups; emphasizes ancestry-matched panels. |
| Ashkenazi Jewish / parts of the Middle East [56] | c.167delT | Frequent founder allele; inclusion improves first-tier yield in relevant populations. |
| South Asia & Romani [57,58] | c.71G>A (p.Trp24Ter; “W24X”) | High carrier frequency reported in some Romani and South Asian subpopulations; useful for targeted screening. |
| West Africa (e.g., Ghana) [59,60] | c.427C>T (p.Arg143Trp) | A regionally prevalent recurrent allele, highlighting the need for population-tailored screening panels |
| Syndrome | cardinal symptom(s) | Hearing phenotype | Representative GJB2 variants | inheritance mechanism | Translational relevance |
|---|---|---|---|---|---|
|
Palmoplantar keratoderma with deafness (PPK + deafness) [79,80,81] |
Diffuse palmoplantar keratoderma, sometimes knuckle involvement | Usually congenital or early-onset SNHL; severity variable | R75W, R75Q, H73R, G59A, S183F | Usually AD; often dominant-negative / trans-dominant effects on Cx26/Cx30 | A useful bridge phenotype linking skin disease and cochlear dysfunction; relevant to dominant-variant editing strategies |
|
Vohwinkel syndrome [82,83] |
Honeycomb PPK, starfish-like keratoses, pseudoainhum/constriction bands | Usually progressive SNHL, often mild to moderate but variable | Y65H, D66H; some reports also include G130V | Usually AD; impaired gap-junction function with variant-specific dominant effects | Highlights the overlap between keratoderma and auditory phenotypes; useful for discussing pleiotropy rather than immediate therapy |
|
Bart–Pumphrey syndrome [84,85] |
PPK, knuckle pads, leukonychia | Sensorineural HL with variable severity | N54K | AD; defective trafficking and dominant / trans-dominant effects have been reported | Mechanistically informative because it sits between milder keratoderma phenotypes and more severe KID-spectrum disease |
|
Keratitis–ichthyosis–deafness (KID) syndrome [86,87] |
Keratitis, ichthyosis/erythrokeratoderma, severe skin barrier disease, infection/cancer risk in some patients | Usually severe congenital SNHL; some variants associated with lethal early disease | D50N, G45E, A88V, G12R, N14K, N14Y, I30N | Usually AD/de novo; gain-of-function hyperactive hemichannels, plus additional dominant effects in some variants | Best current example for mechanism-based therapy: hemichannel blockade (Figure 1), anti-hemichannel mAb delivery, and dominant-variant editing all have preclinical support |
|
Hystrix-like ichthyosis with deafness (HID) / overlap phenotypes [88] |
Severe ichthyotic or spiky hyperkeratotic skin changes | Congenital or early-onset SNHL | Often overlaps with D50N-related spectrum | Overlap disorder within the Cx26 syndromic continuum rather than a wholly separate mechanism | Better treated as a spectrum/extreme phenotype than a standalone major entity in a hearing-focused review |
| Year | Study | Model | Treatment window | Delivery route | Principal target cells | Key findings |
| 2011 [99] |
Crispino et al. | Cx26 conditional deletion, organ of Corti explant culture | Early postnatal tissue, ex vivo | Ex vivo viral transduction in cochlear explants | Non sensory epithelium and supporting cell network | Restored Cx26 expression and improved intercellular coupling, providing proof of concept that GJB2 supplementation can re-engage epithelial gap-junction function. |
| 2014 [100] |
Yu et al. | Conditional Gjb2 loss in supporting cell lineages | Neonatal, around birth | Cochlear local injection targeting scala media | Supporting cells and adjacent epithelial cells | Re-established the gap-junction network and reduced epithelial injury, but hearing recovery remained limited or inconsistent. |
| 2015 [109] |
Iizuka et al. | Cx26 conditional loss model | Perinatal versus adult comparison | Round window based local delivery | Supporting cell enriched patterns in the cochlea | Perinatal delivery improved ABR thresholds and preserved cochlear architecture, whereas adult-stage treatment showed little benefit, supporting a narrow developmental rescue window. |
| 2021 [110] |
Guo et al. | Inducible Gjb2 deficiency model | More mature stage, postnatal weeks | Round window related local cochlear delivery | Supporting cells with unintended inner hair cell transduction | Restored Cx26 signal in supporting cells but failed to improve hearing; ectopic inner hair cell expression was associated with hair-cell loss, highlighting the need for strict cell specificity. |
| 2025 [111] |
Wang et al. | Conditional Cx26 deficiency model | Neonatal | Local cochlear delivery | Supporting cell targeted expression | Vector delivery triggered marked immune activation and could compromise hearing in wild-type ears; adjunct anti-inflammatory treatment improved tolerability and functional outcome. |
| 2025 [112] |
Ivanchenko et al. | DFNB1 mouse models with non-human primate validation | Early postnatal | Round window local delivery | Expression constrained to physiologic cochlear cell populations | Regulatory-element-constrained expression improved cochlear pathology and hearing in DFNB1 models, while showing appropriate localization and minimal threshold disturbance in non-human primate cochlea. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




