Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
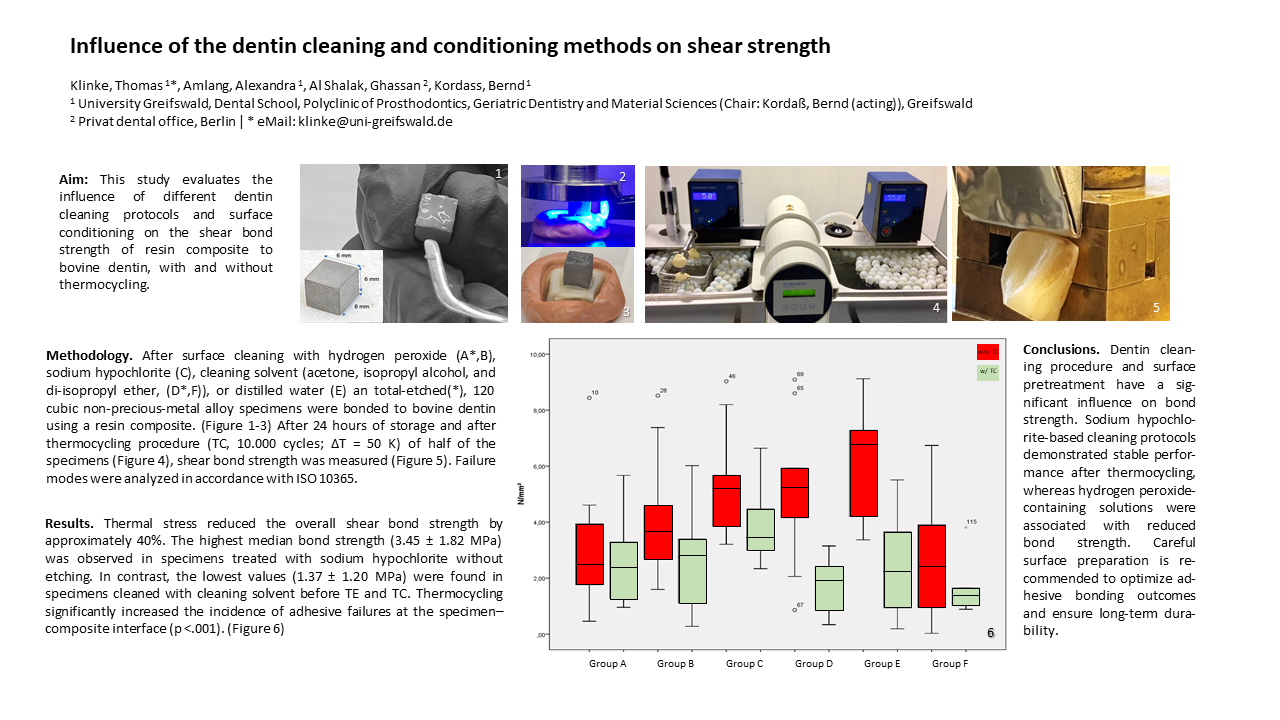
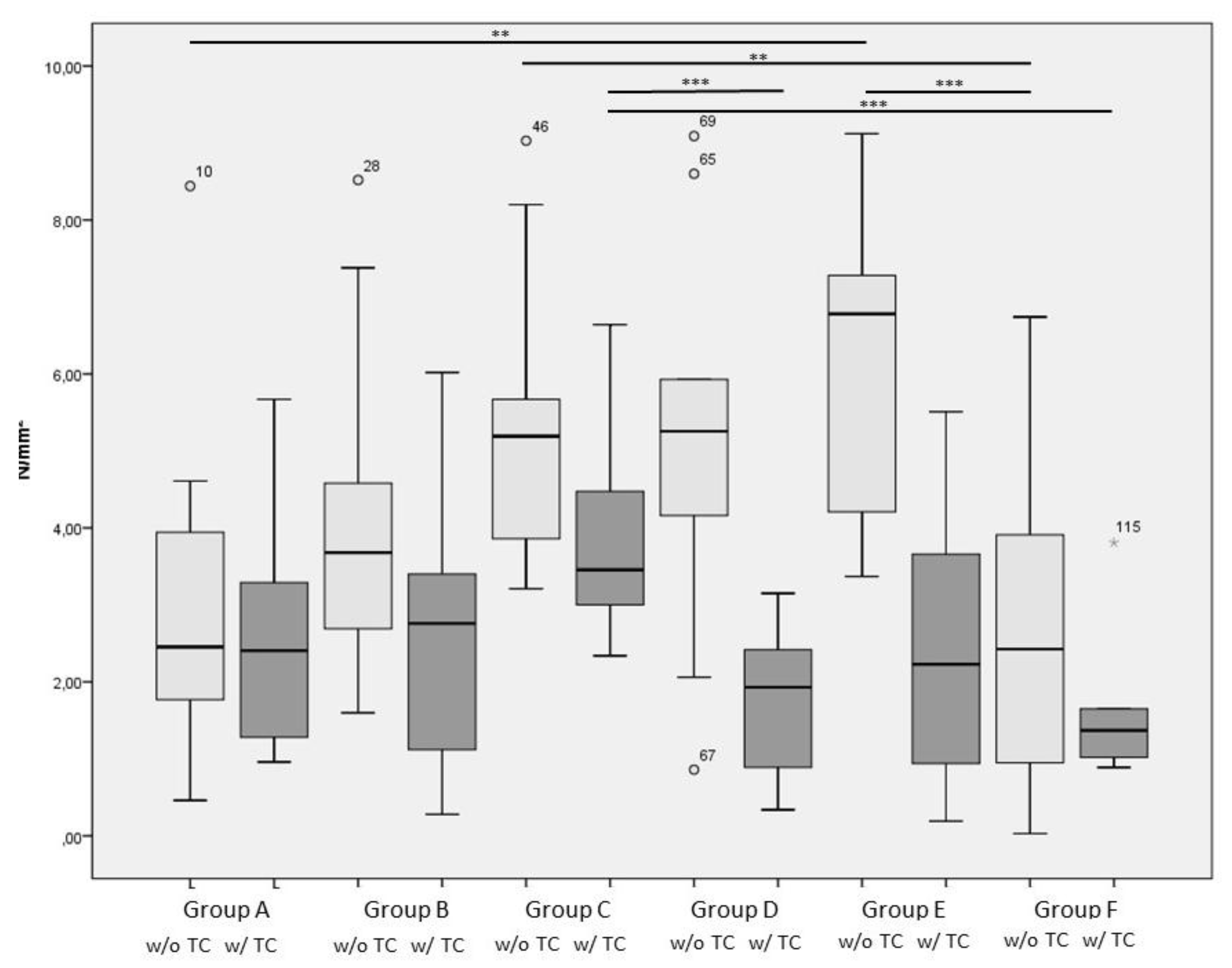
3. Results
3.1. Fracture Analysis
3.2. Failure Mechanisms and Shear Bond Strength
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| K | Kelvin |
| (CH₃)₂CHOH | Iso-propanol |
| ANOVA | Analysis if Variance |
| CH₃CH₂OH | Ethanol |
| CHX | Chlorhexidine |
| FDP | Fixed-partial denture |
| H₂O₂ | Hydro peroxide |
| ISO | International Standard Organisazion |
| MMP | matrix metalloproteinase |
| MPa | Megapascal, N/mm² |
| MW | Mean |
| SD | Standard deviation |
| TC | Thermocycling |
| TE | Total-etch |
References
- Van Landuyt, K.L., et al., Bonding to Dentin: Smear Layer and the Process of Hybridization, in Dental Hard Tissues and Bonding, D.C.W. G. Eliades, T. Eliades, Editor. 2005, Heidelberg: Springer. p. 89-116.
- Cardoso, M.V. , et al., Current aspects on bonding effectiveness and stability in adhesive dentistry. Aust Dent J 2011, 56, 31–44. [Google Scholar] [CrossRef]
- Aminoroaya, A. , et al., A review of dental composites: Challenges, chemistry aspects, filler influences, and future insights. Composites Part B: Engineering 2021, 216, 108852. [Google Scholar] [CrossRef]
- Wysokińska-Miszczuk, J. , et al., Composite Materials Used for Dental Fillings. Materials 2024, 17(19), 4936. [Google Scholar] [CrossRef] [PubMed]
- Tani, C.; Finger, W.J. Effect of smear layer thickness on bond strength mediated by three all-in-one self-etching priming adhesives. J Adhes Dent 2002, 4(4), 283–289. [Google Scholar]
- Ding, P.G.F. , et al., Relationship between microtensile bond strength and nanoleakage at the composite-dentin interface. Dent Mater 2009, 25(1), 135–41. [Google Scholar] [CrossRef] [PubMed]
- Ding, P.G.F. , et al., Relationship between microtensile bond strength and submicron hiatus at the composite–dentin interface using CLSM visualization technique. Dental Materials 2010, 26(3), 257–263. [Google Scholar] [CrossRef]
- Nakabayashi, N. Bonding of restorative materials to dentine: the present status in Japan. Int Dent J 1985, 35(2), 145–54. [Google Scholar]
- Kenshima, S. , et al., Effect of smear layer thickness and pH of self-etching adhesive systems on the bond strength and gap formation to dentin. J Adhes Dent 2005, 7(2), 117–26. [Google Scholar] [PubMed]
- Saikaew, P. , et al., Effect of smear layer and surface roughness on resin-dentin bond strength of self-etching adhesives. Dental materials journal 2018, 37(6), 973–980. [Google Scholar] [CrossRef]
- Giachetti, L.; Bertini, F.; Scaminaci Russo, D. Investigation into the nature of dentin resin tags: a scanning electron microscopic morphological analysis of demineralized bonded dentin. J Prosthet Dent 2004, 92(3), 233–8. [Google Scholar] [CrossRef]
- Gwinnett, A.J. Quantitative contribution of resin infiltration/hybridization to dentin bonding. Am J Dent 1993, 6(1), 7–9. [Google Scholar] [PubMed]
- Tao, L.; Pashley, D.H. Shear bond strengths to dentin: effects of surface treatments, depth and position. Dental Materials 1988, 4(6), 371–378. [Google Scholar] [CrossRef] [PubMed]
- Albaladejo, A. , et al., Hybrid layers of etch-and-rinse versus self-etching adhesive systems. Med Oral Patol Oral Cir Bucal 2010, 15(1), e112–8. [Google Scholar] [CrossRef]
- Van Meerbeek, B. , et al., Morphological aspects of the resin-dentin interdiffusion zone with different dentin adhesive systems. J Dent Res 1992, 71(8), 1530–40. [Google Scholar] [CrossRef] [PubMed]
- Dagostin, A.; Ferrari, M. In vivo bonding mechanism of an experimental dual-cure enamel-dentin bonding system. Am J Dent 2001, 14(2), 105–8. [Google Scholar]
- Pucci, C.R. , et al., Effects of Contamination by Hemostatic Agents and Use of Cleaning Agent on Etch-and-Rinse Dentin Bond Strength. Braz Dent J 2016, 27(6), 688–692. [Google Scholar] [CrossRef]
- Almahdy, A. , et al., Effects of MMP inhibitors incorporated within dental adhesives. J Dent Res 2012, 91(6), 605–11. [Google Scholar] [CrossRef]
- Dietrich, T.; Kraemer, M.L.J.; Roulet, J.-F. Blood contamination and dentin bonding—effect of anticoagulant in laboratory studies. Dental Materials 2002, 18(2), 159–162. [Google Scholar] [CrossRef]
- Ricci, H.A. , et al., Chlorhexidine increases the longevity of in vivo resin-dentin bonds. Eur J Oral Sci 2010, 118(4), 411–6. [Google Scholar] [CrossRef]
- Zhang, S.C.; Kern, M. The role of host-derived dentinal matrix metalloproteinases in reducing dentin bonding of resin adhesives. Int J Oral Sci 2009, 1(4), 163–76. [Google Scholar] [CrossRef] [PubMed]
- Pioch, T. , et al., Influence of different etching times on hybrid layer formation and tensile bond strength. Am J Dent. 1998, 11, 202 ̶ 206. [Google Scholar]
- Perdigão, J.; Lopes, M. The effect of etching time on dentin demineralization. Quintessence Int. 2001, 32(1), 19 ̶ 26. [Google Scholar]
- Keul, C. , et al., The effect of ceramic primer on shear bond strength of resin composite cement to zirconia: a function of water storage and thermal cycling. J Am Dent Assoc. 2013, 144(11), 1261–71. [Google Scholar] [CrossRef]
- Schwickerath, H.; Coca, I. Einzelkronen aus Glaskeramik. Phillip Journal fur Restaurative Zahnmedizin 1987, 4(6), 336–338. [Google Scholar] [PubMed]
- ISO. 2022. Adhesives — Designation of main failure patterns.
- Inoue, T.; Miyazaki, T.; Nishimura, F. Tensile strength and durability of bovine dentin. Dent Mater J 2007, 26(3), 348–54. [Google Scholar] [CrossRef] [PubMed]
- Cattani-Lorente, M. , et al., Effect of different surface treatments on the hydrothermal degradation of a 3Y-TZP ceramic for dental implants. Dent Mater 2014, 30(10), 1136–46. [Google Scholar] [CrossRef] [PubMed]
- Coelho, A. , et al., Effect of Cavity Disinfectants on Dentin Bond Strength and Clinical Success of Composite Restorations—A Systematic Review of In Vitro, In Situ and Clinical Studies. International Journal of Molecular Sciences 2021, 22(1), 353. [Google Scholar] [CrossRef] [PubMed]
- Coelho, A. , et al., Effect of Different Cavity Disinfectants on Adhesion to Dentin of Permanent Teeth. Journal of Functional Biomaterials 2022, 13(4), 209. [Google Scholar] [CrossRef]
- Ercan, E. , et al., Effect of different cavity disinfectants on shear bond strength of composite resin to dentin. J Adhes Dent 2009, 11(5), 343–6. [Google Scholar]
- Nakamura, T. , et al., Mechanical properties of new self-adhesive resin-based cement. J Prosthodont Res 2010, 54(2), 59–64. [Google Scholar] [CrossRef]
- Prati, C.; Chersoni, S.; Pashley, D.H. Effect of removal of surface collagen fibrils on resin–dentin bonding. Dental Materials 1999, 15(5), 323–331. [Google Scholar] [CrossRef] [PubMed]
| Group | Surface cleaning and conditioning method | Number | Fracture analysis | ||
|---|---|---|---|---|---|
| Type 1 | Type 2 | Type 3 | |||
| A** | Hydrogen peroxide (H2O2) w/ surface conditioning (TE) | 20 | 18 | 1 | 1 |
| B | Hydrogen peroxide (H2O2) w/o TE | 20 | 14 | 1 | 5 |
| C | Sodium hypochlorite (NaClO, 3 %) w/o TE | 20 | 20 | 0 | 0 |
| D | Acetone, isopropyl alcohol and di-isopropyl ether solvent (Fokaldry®) w/ TE | 20 | 19 | 0 | 1 |
| E* | Distilled water w/o TE | 20 | 19 | 0 | 1 |
| F | Acetone, isopropyl alcohol and di-isopropyl ether solvent (Fokaldry®) w/ TE | 20 | 18 | 1 | 1 |
| Fracture type | Fractur localization |
|---|---|
| 1 | Adhesive fracture > 75% within the bonded zone between the test specimen and the luting composite |
| 2 | Adhesive fracture > 75% within the bonded zone between the luting composite and the dentin |
| 3 | Cohesive fracture with < 75% of the surface covered, no clear classification possible. |
| Surface conditioning (total-etch, TE) |
w/ TE (N=38) | w/o TE (N=73) | ||
|---|---|---|---|---|
| Thermo cycling (TC) | w/o TC (N=20) | w/ TC (N=18) |
w/o TC (N=39) |
w/ EC (N=34) |
| Mean ± SD | 4.50 ± 2.43 | 2.14 ± 1.23 | 4.60 ± 2.35 | 2.69 ± 1.66 |
| Confidence interval (95% CI) | 2.97 – 5.59 | 1.57 – 2.72 | 3.83 – 5.37 | 2.11 – 3.27 |
| Median | 4.38 | 2.10 | 4.21 | 2.76 |
| Interquartile distance | 3.84 | 1.57 | 3.53 | 2.60 |
| Standard error (SE) | 0.62 | 0.27 | 0.38 | 0.28 |
| Cleaning method | Group A | Group B | Group C | |||
| w/o TC | w/ TC | w/oi TC | w/ TC | w/o TC | w/ TC | |
| Mean ± SD | 3.15 ± 2.46 | 2.50 ± 1.41 | 4.14 ± 2.25 | 2.57 ± 1.72 | 5.43 ± 2.00 | 3.85 ± 1.40 |
| Confidence interval (95% CI) |
1.09 – 5.21 | 1.49 – 3.52 | 2.53 – 5.76 | 1.34 – 3.81 | 3.89 – 6,97 | 2.68 – 5.02 |
| Median | 2.45 | 2.40 | 3.68 | 2.76 | 5.,19 | 3.45 |
| Interquartile distance | 2.51 | 2.10 | 2.84 | 2.49 | 3.27 | 1.82 |
| Standard error | 0.87 | 0.44 | 0.71 | 0.54 | 0.66 | 0.49 |
| Cleaning method | Group D | Group E | Group F | |||
| w/o TC | w/ TC | w/o TC | w/ TC | w/o TC | w/ TC | |
| Mean ± SD | 5.17 ± 2.52 | 1.79 ± 0.93 | 6.14 ± 1.87 | 2.48 ± 1.72 | 2.76 ± 1.99 | 1.68 ± 1.08 |
| Confidence interval (95% CI) |
3.36 – 6.98 | 1.12 – 2.45 | 4.80 -7.49 | 1.24 – 3.71 | 1.34 – 4.19 | 0.54 – 2.82 |
| Median | 5.25 | 1.93 | 6.78 | 2.23 | 2.42 | 1.37 |
| Interquartile distance | 2.96 | 1.66 | 3.16 | 2.88 | 3.12 | 1.20 |
| Standard error | 0.79 | 0.29 | 0.59 | 0.54 | 0.63 | 0.44 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




