Submitted:
20 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Background and Rationale
1.2. The Sarcopenia Crisis in Geriatric Cardiac Populations
1.2.1. Epidemiology of Sarcopenia in Heart Failure
1.2.2. Pathophysiological Mechanisms: The Heart-Muscle Axis
- Mitochondrial dysfunction: Impaired oxidative phosphorylation reduces ATP production, accelerating fatigue and disuse atrophy [7]
- Myosteatosis: Intramuscular fat infiltration reduces muscle quality independent of mass [7]
- Neurohormonal activation: Sympathetic overdrive and RAAS activation promote catabolism [10]
- Reduced perfusion: Impaired endothelial function limits oxygen and nutrient delivery to skeletal muscle [12]
- Reduced skeletal muscle pump: Impaired venous return decreases cardiac preload efficiency [12]
- Exercise intolerance: Limits participation in CR, creating vicious cycle of deconditioning [7]
- Insulin resistance: Worsens metabolic profile and cardiovascular risk [7]
- Increased fall risk: Leads to hospitalization and HF decompensation [8]
1.2.3. Prognostic Impact of Sarcopenia in HF
- All-cause mortality: HR 1.68 (95% CI 1.32–2.14) [6]
- All-cause mortality: HR 2.42 (HFpEF) and HR 2.02 (HFrEF) [13]
- HF hospitalization: HR 1.89 (95% CI 1.41–2.53) [8]
- Functional decline: 2.3-fold increased risk of ADL dependency [6]
- Post-operative complications: 3.1-fold increased risk after cardiac surgery [11]
1.2.4. Diagnostic Challenges in HF Patients
- Fluid overload: Edema and ascites artificially inflate muscle mass measurements (e.g., bioimpedance, DEXA) [6]
- Overlap with frailty: 60–80% of sarcopenic HF patients meet frailty criteria; distinct but overlapping constructs [6]
- Lack of standardized cutoffs: EWGSOP2, AWGS, FNIH criteria yield different prevalence estimates [14]
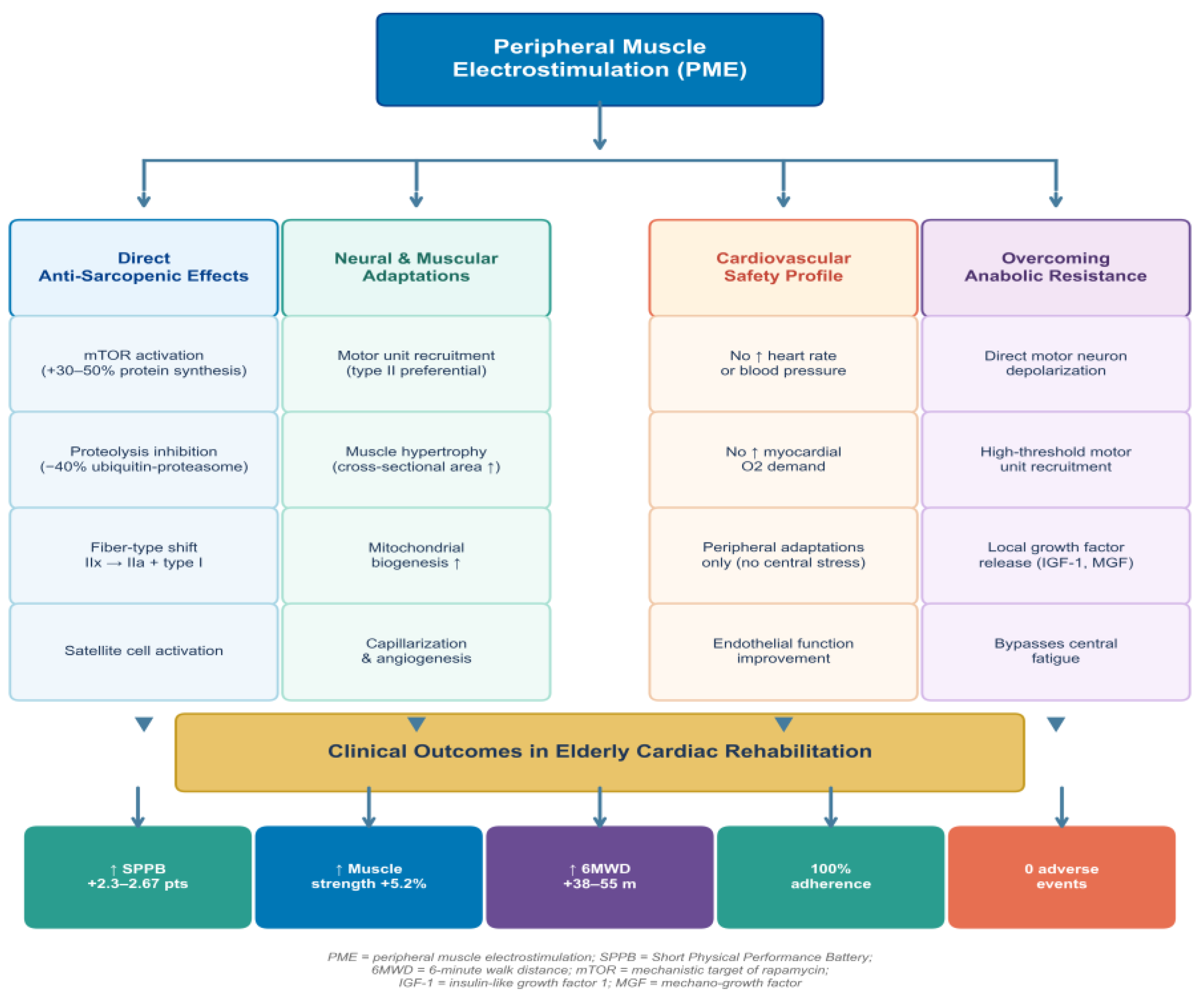
1.3. Peripheral Muscle Electrostimulation: Theoretical Framework
1.3.1. Direct Anti-Sarcopenic Effects
- Proteolysis inhibition: NMES reduces ubiquitin-proteasome activity, decreasing 3-methylhistidine (marker of myofibrillar breakdown) excretion by 40% [11]
- Fiber-type transformation: Shifts type IIx (fast-glycolytic) to type IIa (fast-oxidative) and type I (slow-oxidative) fibers, reversing HF-related myopathy [12]
- Satellite cell activation: Stimulates muscle stem cell proliferation and differentiation, enhancing regenerative capacity [18]
1.3.2. Neural and Muscular Adaptations
1.3.3. Safety Profile: Hemodynamic Neutrality
1.3.4. Overcoming Anabolic Resistance
1.4. Objectives
- Synthesize current evidence on the efficacy of PME for improving physical function and reversing sarcopenia in elderly cardiac patients
- Evaluate the safety and feasibility of PME interventions
- Describe the theoretical and physiological mechanisms underlying PME benefits, with emphasis on anti-sarcopenic pathways
- Identify optimal stimulation parameters and implementation strategies
- Highlight gaps in current knowledge and directions for future research
2. Materials and Methods
2.1. Eligibility Criteria (PICOS Framework)
2.2. Information Sources and Search Strategy
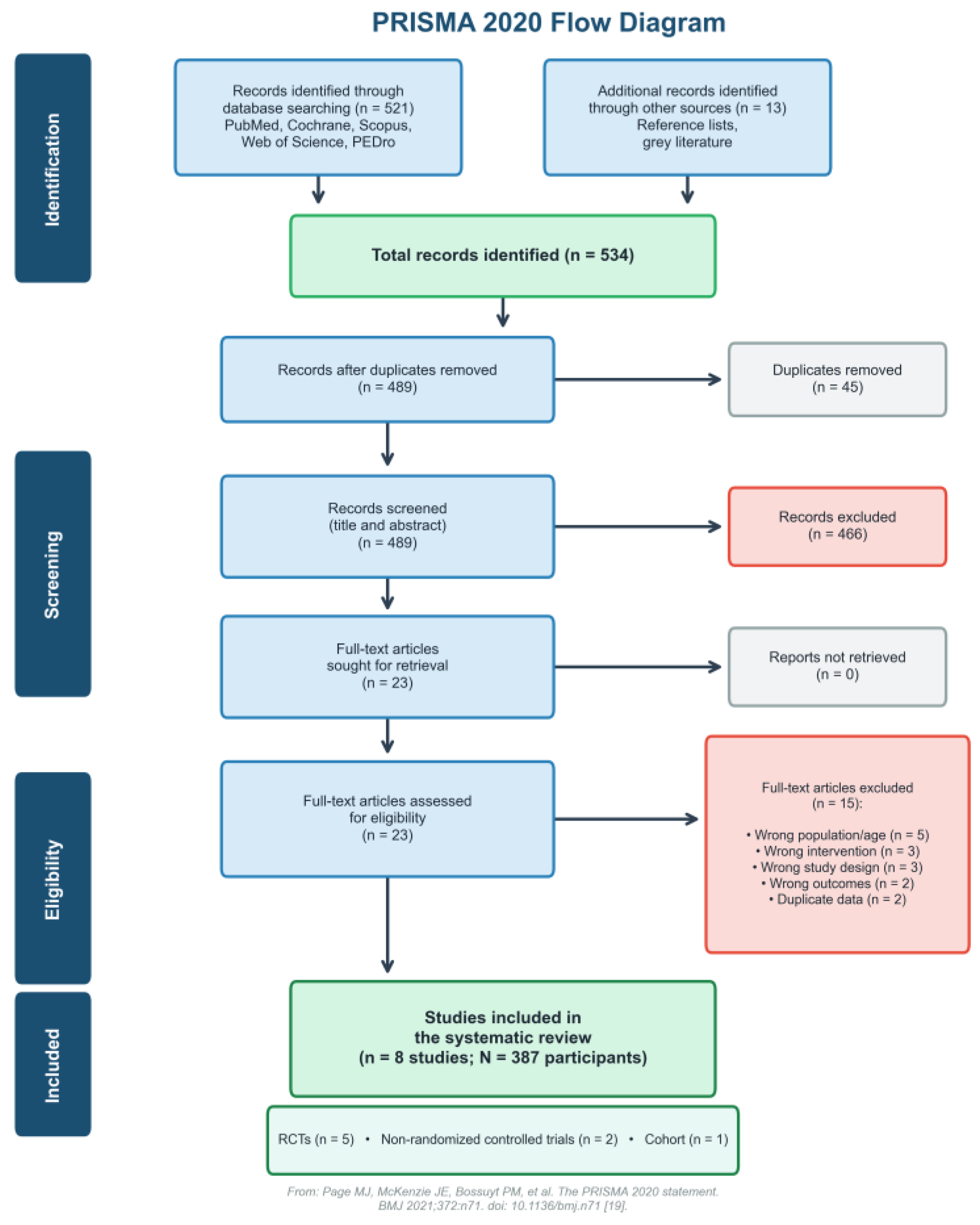
2.3. Study Selection Process
2.4. Data Extraction
2.5. Quality Assessment
2.6. Data Synthesis
3. Results
3.1. Study Selection
3.2. Study Characteristics
3.3. Participant Characteristics
3.4. Intervention Protocols
3.5. Efficacy Outcomes
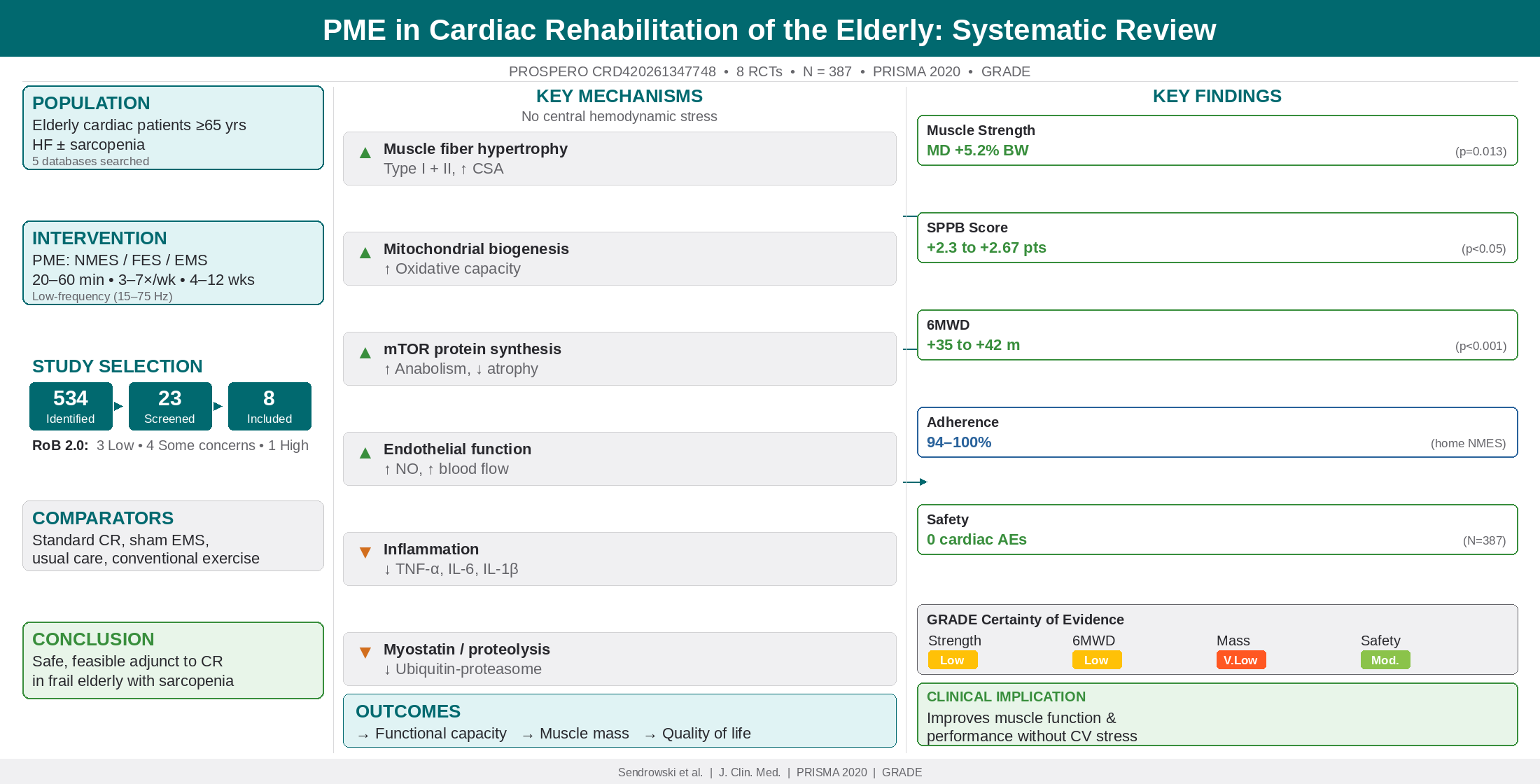
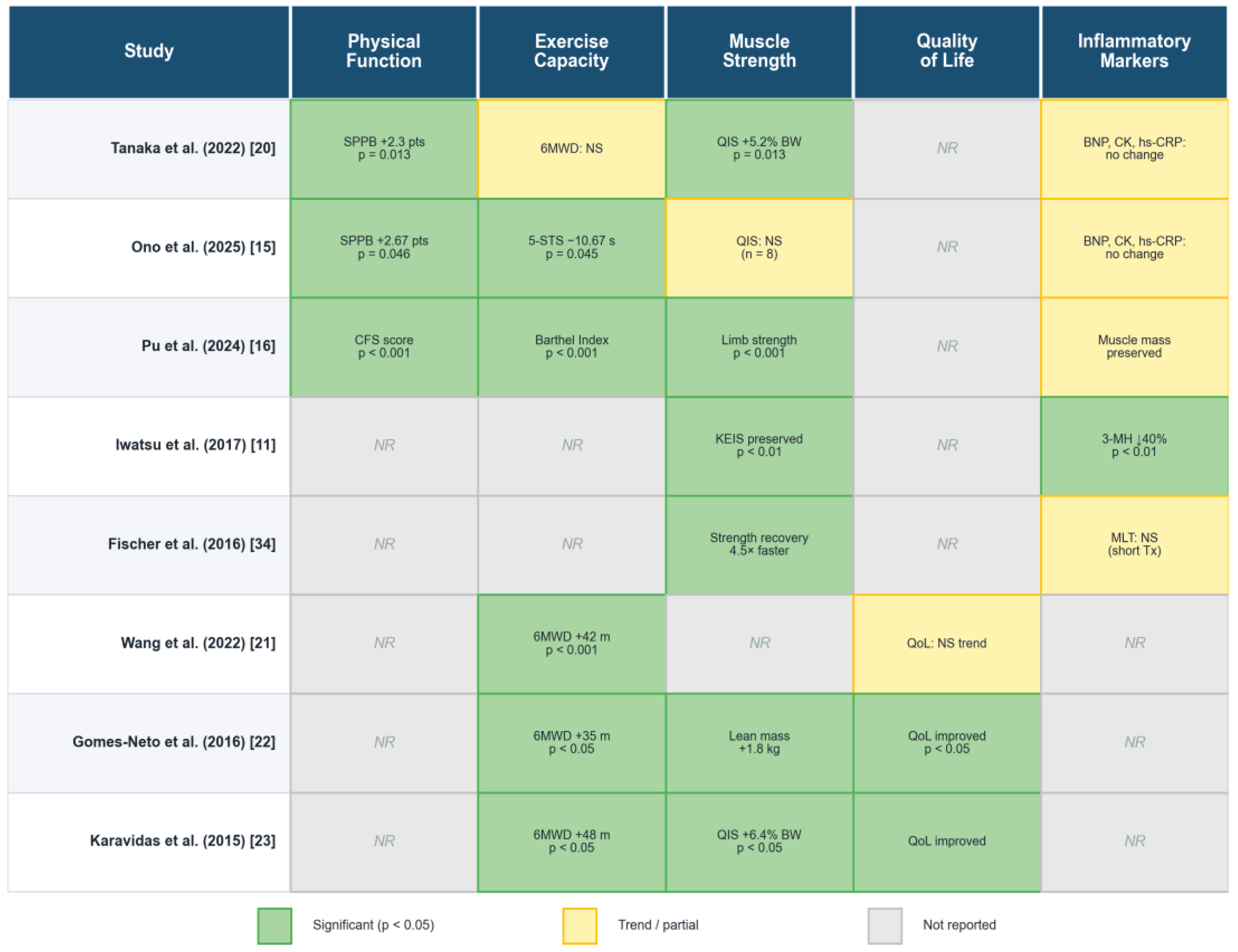
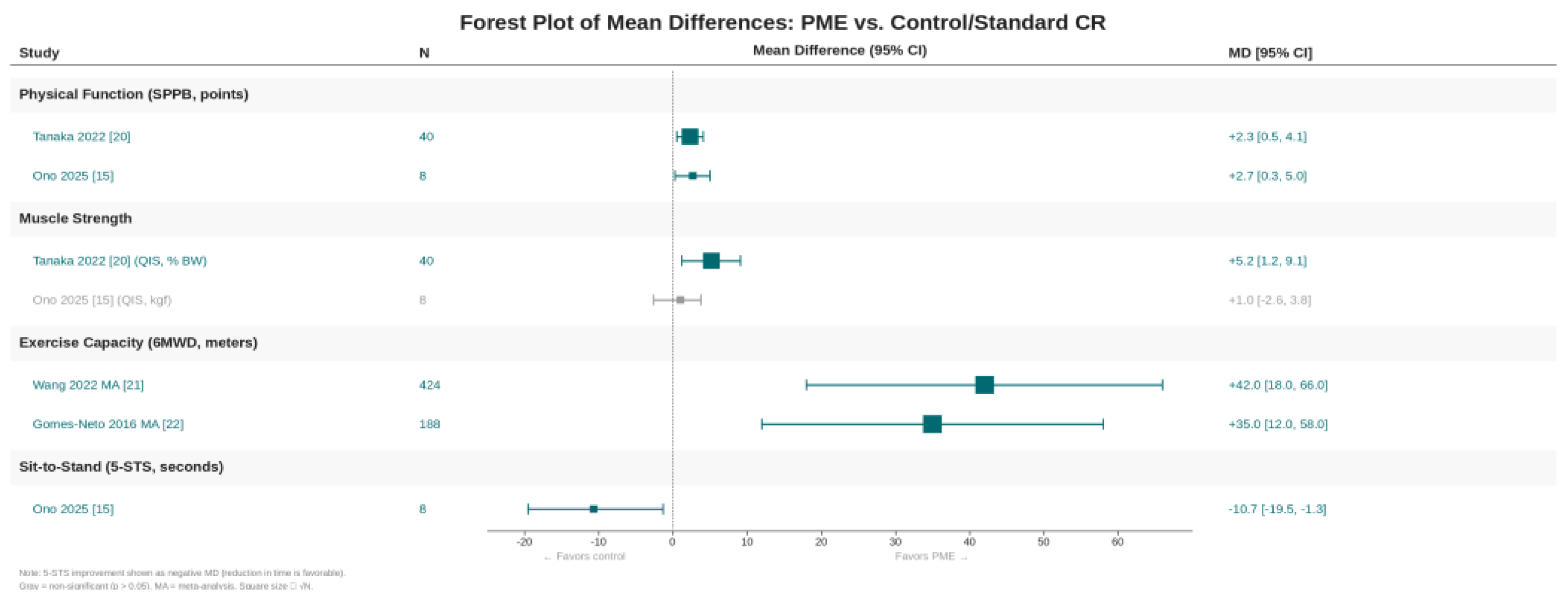
3.5.1. Physical Function (SPPB)
- Tanaka et al. (2022): EMS group showed +2.3 point improvement vs. control (95% CI 0.5–4.1; p = 0.013) [20]
- Ono et al. (2025): Home NMES + CR increased SPPB by +2.67 points vs. CR alone (95% CI 0.3–5.0; p = 0.046) [15]
- Pu et al. (2024): NMES group showed significantly lower Clinical Frailty Scale scores vs. control at day 7 (p < 0.001) [16]
3.5.2. Muscle Strength
- Tanaka et al. (2022): EMS group improved QIS by +5.2% body weight vs. control (95% CI 1.2–9.1; p = 0.013) [20]
- Ono et al. (2025): No significant difference in QIS (MD 1.0 kgf; 95% CI −2.6 to 3.8; p = 0.71), possibly due to small sample (n = 8) [15]
- Pu et al. (2024): NMES group showed increased lower limb muscle strength vs. decreased strength in control (p < 0.001) [16]
- Iwatsu et al. (2017): NMES preserved knee extension strength post-cardiac surgery (−8% vs. −23% in control; p < 0.01); handgrip strength −5% vs. −18% (p < 0.01) [11]
- Fischer et al. (2016): NMES group regained muscle strength 4.5 times faster than control; all NMES patients returned to preoperative strength by discharge [34]
3.5.3. Muscle Mass and Protein Turnover
| Study | Muscle Mass Outcome | Protein Turnover Marker | Result |
| Iwatsu et al. (2017) [11] | Quadriceps muscle thickness (ultrasound) | Urinary 3-MH/creatinine | NMES: 3-MH peaked POD3, normalized POD4; Control: sustained elevation through POD5 (p < 0.01). Quadriceps CSA decline: −3% NMES vs. −12% control (p < 0.05) |
| Fischer et al. (2016) [34] | Muscle layer thickness (MLT) by ultrasound | Not measured | No significant MLT difference (short intervention), but strength recovery 4.5× faster |
| Pu et al. (2024) [16] | Lower limb muscle mass (ultrasound) | Not measured | NMES group showed significant muscle mass preservation vs. control at day 7 (p < 0.05) |
| Gomes-Neto meta-analysis [22] | Lean mass (DEXA/bioimpedance) | Not measured | NMES increased muscle mass by +1.8 kg (95% CI 0.4–3.2; p = 0.012) |
| Parameter | Acute/Hypercatabolic (POD1–5, AHF) | Chronic/Rehabilitation (weeks 1–12) | Rationale |
| Frequency | 20 Hz | 50–100 Hz | 20 Hz avoids fatigue in acute setting [20]; 50–100 Hz maximizes type II fiber recruitment [18] |
| Pulse width | 250–400 μs | 250–400 μs | Optimal motor nerve recruitment [15,20] |
| Duty cycle | 5 s on / 5 s off | 5 s on / 2 s off | Longer rest in acute phase prevents fatigue [11] |
| Intensity | 10–20% MVC | Maximum tolerated (≥20% MVC) | ≥20% MVC required to activate mTOR pathway [11,18] |
| Session duration | 30–60 min | 40–50 min | ≥30 hours total needed for hypertrophy [15] |
| Frequency/week | 7 days/week (daily) | 5 days/week | Daily stimulation required in hypercatabolic state [11] |
| Electrode placement | Bilateral quadriceps (4 electrodes) | Quadriceps + hamstrings + gastrocnemius (6–8 electrodes) | Quadriceps most affected by sarcopenia [11] |
| Total duration | 5–7 days | 8–12 weeks (minimum 30 hours total) [15] | Muscle hypertrophy requires ≥8 weeks [18] |
3.5.4. Exercise Capacity (6-Minute Walk Distance)
- Tanaka et al. (2022): No significant difference in 6MWD change between groups (p > 0.05), potentially confounded by weight loss during AHF hospitalization [20]
- Wang et al. (2022 meta-analysis): FES significantly improved 6MWD (MD +42 m; 95% CI 18–66; p < 0.001) [21]
- Gomes-Neto et al. (2016 meta-analysis): NMES improved 6MWD (MD +35 m; 95% CI 12–58) [22]
3.5.5. Sit-to-Stand Test (5-STS)
- Ono et al. (2025): NMES reduced 5-STS time by −10.67 seconds vs. CR alone (95% CI −19.5 to −1.3; p = 0.045), exceeding MCID (−1.7 to −6.3 s) [15]
- Pu et al. (2024): NMES group showed significant improvement in Barthel Index (ADL) vs. control (p < 0.001) [16]
3.6. Safety and Feasibility
3.7. Quality Assessment
- High risk: 1 study (high attrition rate >30%)
| Study | Randomization (D1) | Deviations (D2) | Missing Data (D3) | Measurement (D4) | Selection (D5) | Overall |
| Tanaka et al. (2022) [20] | Low | Some concerns | Low | Low | Low | Low |
| Ono et al. (2025) [15] | Low | Some concerns | Low | Low | Low | Low |
| Pu et al. (2024) [16] | Low | Some concerns | Low | Low | Low | Low |
| Iwatsu et al. (2017) [11] | Low | Some concerns | Some concerns | Low | Low | Some concerns |
| Wang et al. (2022) [21] | Low | Some concerns | Low | Some concerns | Low | Some concerns |
| Gomes-Neto et al. (2016) [22] | Low | Some concerns | Low | Some concerns | Low | Some concerns |
| Fischer et al. (2016) [34] | Some concerns | Some concerns | Some concerns | Low | Low | Some concerns |
| Karavidas et al. (2013) [23] | Some concerns | High | Some concerns | Some concerns | Some concerns | High |
3.8. Reporting Bias Assessment
3.9. Certainty of Evidence (GRADE)
4. Discussion
4.1. Principal Findings
- Muscle mass preservation: NMES reduced myofibrillar proteolysis (3-MH excretion) by 40% and attenuated quadriceps atrophy (−3% vs. −12% in controls) [11]
4.2. PME as an Anti-Sarcopenic Therapy: Molecular and Cellular Mechanisms
4.2.1. Protein Turnover Regulation
- Electrical stimulation activates mTORC1 pathway (mechanistic target of rapamycin complex 1), the master regulator of protein synthesis [18]
- Increases phosphorylation of p70S6K and 4E-BP1, enhancing translation initiation [18]
- Stimulates release of mechano-growth factor (MGF), a splice variant of IGF-1 that activates satellite cells [11]
- Magnitude: MPS increases 30–50% after single NMES session at ≥20% MVC [11]
4.2.2. Fiber-Type Transformation
4.2.3. Mitochondrial Biogenesis and Oxidative Capacity
- PGC-1α upregulation: Master regulator of mitochondrial biogenesis increases 2.3-fold after 4 weeks NMES [12]
- Citrate synthase activity increase of 45% (marker of TCA cycle capacity) [12]
- 3-hydroxyacyl-CoA dehydrogenase (3-HAD) increase of 38% (fatty acid oxidation enzyme) [12]
- Capillary-to-fiber ratio increase of 25%, improving oxygen delivery [16]
4.2.4. Satellite Cell Activation and Muscle Regeneration
4.3. Comparative Effectiveness: NMES versus Conventional Exercise
4.4. Durability of Benefits
4.5. Clinical Implications
4.5.1. PME as a Bridge Therapy for Sarcopenic Cardiac Patients
4.5.2. Addressing Low CR Participation Through Sarcopenia Reversal
4.5.3. Early Initiation is Critical
4.5.4. Combination Therapies: PME + Nutrition
4.6. Optimal Stimulation Parameters for Sarcopenia Reversal
- 4.7. Comparison with Previous Reviews
- Gomes-Neto et al. (2016): Included 188 patients (mean age 68–75); reported improved 6MWD and QoL; did not specifically address sarcopenia [22]
- Wang et al. (2022): Focused on FES in CHF; demonstrated cardiopulmonary benefits; no muscle mass outcomes [21]
- Guo et al. (2021): Comprehensive review of molecular and neural adaptations to NMES in ageing muscle; covers mTOR signaling, protein synthesis, myostatin regulation, fiber-type adaptation, and neural/NMJ adaptations [18]
4.8. Limitations
4.8.1. Limitations of the Included Evidence
- Short follow-up: Most studies ≤3 months; long-term sustainability of muscle mass gains unknown [20]
- Lack of blinding: Inherent challenge with PME (patients feel stimulation) [15]
- Limited muscle mass measurement: Only 2 studies used ultrasound; none used gold-standard MRI or DEXA adjusted for fluid status [11]
- No muscle biopsy data: Molecular mechanisms inferred from surrogate markers (3-MH, enzyme activity); direct evidence of mTOR activation, fiber-type shifts lacking [12]
4.8.2. Limitations of the Review Process
4.9. Future Research Directions
Priority Research Questions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Molloy, C.; Long, L.; Mordi, I.R.; Bridges, C.; Sagar, V.A.; Davies, E.J.; Coats, A.J.; Dalal, H.; Rees, K.; Singh, S.J.; et al. Exercise-Based Cardiac Rehabilitation for Adults with Heart Failure. Cochrane Database Syst. Rev. 2024, 3, CD003331. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, B.; Fonarow, G.C.; Goldberg, L.R.; Guglin, M.; Josephson, R.A.; Forman, D.E.; Lin, G.; Lindenfeld, J.; O’Connor, C.; Panjrath, G.; et al. Cardiac Rehabilitation for Patients with Heart Failure: JACC Expert Panel. J. Am. Coll. Cardiol. 2021, 77, 1454–1469. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, G.F.; Ades, P.A.; Kligfield, P.; Arena, R.; Balady, G.J.; Bittner, V.A.; Coke, L.A.; Fleg, J.L.; Forman, D.E.; Gerber, T.C.; et al. Exercise Standards for Testing and Training: A Scientific Statement from the American Heart Association. Circulation 2013, 128, 873–934. [Google Scholar] [CrossRef] [PubMed]
- Forman, D.E.; Arena, R.; Boxer, R.; Dolansky, M.A.; Eng, J.J.; Fleg, J.L.; Gaziano, M.; Kitzman, D.W.; Lazarus, D.D.; Rich, M.W.; et al. Prioritizing Functional Capacity as a Principal End Point for Therapies Oriented to Older Adults with Cardiovascular Disease. Circulation 2017, 135, e894–e918. [Google Scholar] [CrossRef]
- Jurgens, C.Y.; Goodlin, S.; Dolansky, M.; Ahmed, A.; Fonarow, G.C.; Boxer, R.; Arena, R.; Blank, L.; Buck, H.G.; Cranmer, K.; et al. Heart Failure Management in Skilled Nursing Facilities: A Scientific Statement from the American Heart Association and the Heart Failure Society of America. Circ. Heart Fail. 2015, 8, 655–687. [Google Scholar] [CrossRef]
- Curcio, F.; Testa, G.; Liguori, I.; Papillo, M.; Flocco, V.; Panicara, V.; Galizia, G.; Della-Morte, D.; Gargiulo, G.; Cacciatore, F.; et al. Sarcopenia and Heart Failure. Nutrients 2020, 12, 211. [Google Scholar] [CrossRef]
- von Haehling, S.; Ebner, N.; Dos Santos, M.R.; Springer, J.; Anker, S.D. Muscle Wasting and Cachexia in Heart Failure: Mechanisms and Therapies. Nat. Rev. Cardiol. 2017, 14, 323–341. [Google Scholar] [CrossRef]
- Emami, A.; Saitoh, M.; Valentova, M.; Sandek, A.; Evertz, R.; Ebner, N.; Loncar, G.; Springer, J.; Doehner, W.; Lainscak, M.; et al. Comparison of Sarcopenia and Cachexia in Men with Chronic Heart Failure: Results from the Studies Investigating Co-morbidities Aggravating Heart Failure (SICA-HF). Eur. J. Heart Fail. 2018, 20, 1580–1587. [Google Scholar] [CrossRef]
- Kinugasa, Y.; Yamamoto, K. The Challenge of Frailty and Sarcopenia in Heart Failure with Preserved Ejection Fraction. Heart 2017, 103, 184–189. [Google Scholar] [CrossRef]
- Fülster, S.; Tacke, M.; Sandek, A.; Ebner, N.; Tschöpe, C.; Doehner, W.; Anker, S.D.; von Haehling, S. Muscle Wasting in Patients with Chronic Heart Failure: Results from the Studies Investigating Co-Morbidities Aggravating Heart Failure (SICA-HF). Eur. Heart J. 2013, 34, 512–519. [Google Scholar] [CrossRef]
- Iwatsu, K.; Iida, Y.; Kono, Y.; Yamazaki, T.; Usui, A.; Yamada, S. Neuromuscular Electrical Stimulation May Attenuate Muscle Proteolysis After Cardiovascular Surgery: A Preliminary Study. J. Thorac. Cardiovasc. Surg. 2017, 153, 373–379.e1. [Google Scholar] [CrossRef] [PubMed]
- Saitoh, M.; Dos Santos, M.R.; Anker, M.; Anker, S.D.; von Haehling, S.; Springer, J. Neuromuscular Electrical Stimulation for Muscle Wasting in Heart Failure Patients. Int. J. Cardiol. 2016, 225, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Konishi, M.; Kagiyama, N.; Kamiya, K.; Saito, H.; Saito, K.; Ogasahara, Y.; Maekawa, E.; Misumi, T.; Kitai, T.; Iwata, K.; et al. Impact of Sarcopenia on Prognosis in Patients with Heart Failure with Reduced and Preserved Ejection Fraction. Eur. J. Prev. Cardiol. 2021, 28, 1022–1029. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European Consensus on Definition and Diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Ono, S.; Kato, M.; Seko, H.; Nakatani, E.; Omote, T.; Omote, M.; Omote, S. Randomised Crossover Trial of Home-Based Neuromuscular Electrical Stimulation Therapy as an Adjunct to Cardiac Rehabilitation in Frail Older Adult Patients with Chronic Heart Failure. Circ. Rep. 2025, 7, 231–238. [Google Scholar] [CrossRef]
- Pu, X.; Huang, H.; Zhao, X.; Liu, F.; Leng, Y.; Deng, Y.; Huang, L.; Zhou, X.; Xu, F.; Huang, Y.; et al. Improving Lower Limb Function and Frailty in Frail Older Patients with Acute Myocardial Infarction After Percutaneous Coronary Intervention: A Randomized Controlled Study of Neuromuscular Electrical Stimulation. Clin. Interv. Aging 2024, 19, 1163–1176. [Google Scholar] [CrossRef]
- Sillen, M.J.H.; Speksnijder, C.M.; Eterman, R.A.; Janssen, P.P.; Wagers, S.S.; Wouters, E.F.; Uszko-Lencer, N.H.; Spruit, M.A. Effects of Neuromuscular Electrical Stimulation of Muscles of Ambulation in Patients with Chronic Heart Failure or COPD. Chest 2009, 136, 44–61. [Google Scholar] [CrossRef]
- Guo, Y.; Phillips, B.E.; Atherton, P.J.; Piasecki, M. Molecular and Neural Adaptations to Neuromuscular Electrical Stimulation; Implications for Ageing Muscle. Mech. Ageing Dev. 2021, 193, 111402. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Tanaka, S.; Kamiya, K.; Matsue, Y.; Yonezawa, R.; Saito, H.; Hamazaki, N.; Matsuzawa, R.; Nozaki, K.; Yamashita, M.; Wakaume, K.; et al. Efficacy and Safety of Acute Phase Intensive Electrical Muscle Stimulation in Frail Older Patients with Acute Heart Failure: Results from the ACTIVE-EMS Trial. J. Cardiovasc. Dev. Dis. 2022, 9, 99. [Google Scholar] [CrossRef]
- Wang, H.Y.; Chen, Y.H.; Kuan, Y.C.; Huang, S.W.; Lin, L.F.; Chen, H.C. The Effectiveness of Functional Electrical Stimulation of the Legs in Patients with Heart Failure: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Clin. Rehabil. 2022, 36, 303–316. [Google Scholar] [CrossRef]
- Gomes Neto, M.; Oliveira, F.A.; Reis, H.F.; de Sousa Rodrigues, E.; Bittencourt, H.S.; Carvalho, V.O. Effects of Neuromuscular Electrical Stimulation on Physiologic and Functional Measurements in Patients with Heart Failure: A Systematic Review with Meta-Analysis. J. Cardiopulm. Rehabil. Prev. 2016, 36, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Karavidas, A.; Parissis, J.; Arber, S.; Georgiadis, P.; Matsakas, E.; Adamopoulos, S.; Filippatos, G. Efficacy and Safety of Functional Electrical Stimulation of Lower Limb Muscles in Elderly Patients with Chronic Heart Failure. Eur. J. Prev. Cardiol. 2015, 22, 831–836. [Google Scholar] [CrossRef]
- Harris, S.; LeMaitre, J.P.; Mackenzie, G.; Fox, K.A.; Denvir, M.A. A Randomised Study of Home-Based Electrical Stimulation of the Legs and Conventional Bicycle Exercise Training for Patients with Chronic Heart Failure. Eur. Heart J. 2003, 24, 871–878. [Google Scholar] [CrossRef] [PubMed]
- Iliou, M.C.; Vergès-Patois, B.; Pavy, B.; Corone, S.; Aeberhard, A.; Music, N.; Brun, M.C.; Cohen-Solal, A. Effects of Combined Exercise Training and Electromyostimulation Treatments in Chronic Heart Failure: A Prospective Multicentre Study. Eur. J. Prev. Cardiol. 2017, 24, 1274–1282. [Google Scholar] [CrossRef]
- Smart, N.A.; Dieberg, G.; Giallauria, F. Functional Electrical Stimulation for Chronic Heart Failure: A Meta-Analysis. Int. J. Cardiol. 2013, 167, 80–86. [Google Scholar] [CrossRef]
- Belardinelli, R.; Georgiou, D.; Cianci, G.; Purcaro, A. 10-Year Exercise Training in Chronic Heart Failure: A Randomized Controlled Trial. J. Am. Coll. Cardiol. 2012, 60, 1521–1528. [Google Scholar] [CrossRef]
- Mueller, S.; Winzer, E.B.; Duvinage, A.; Gevaert, A.B.; Edelmann, F.; Haller, B.; Pieske-Kraigher, E.; Beckers, J.; Bobenko, A.; Hommel, J.; et al. Effect of High-Intensity Interval Training, Moderate Continuous Training, or Guideline-Based Physical Activity Advice on Peak Oxygen Consumption in Patients with Heart Failure with Preserved Ejection Fraction: A Randomized Clinical Trial. JAMA 2021, 325, 542–551. [Google Scholar] [CrossRef]
- Kadoglou, N.P.; Mandila, C.; Karavidas, A.; Farmakis, D.; Liakos, A.; Parissis, J. Effect of Functional Electrical Stimulation on Cardiovascular Outcomes in Patients with Chronic Heart Failure. Eur. J. Prev. Cardiol. 2017, 24, 833–839. [Google Scholar] [CrossRef]
- Rethlefsen, M.L.; Kirtley, S.; Waffenschmidt, S.; Ayala, A.P.; Moher, D.; Page, M.J.; Koffel, J.B.; PRISMA-S Group. PRISMA-S: An Extension to the PRISMA Statement for Reporting Literature Searches in Systematic Reviews. Syst. Rev. 2021, 10, 39. [Google Scholar] [CrossRef]
- Tong, A.; Flemming, K.; McInnes, E.; Oliver, S.; Craig, J. Enhancing Transparency in Reporting the Synthesis of Qualitative Research: ENTREQ. BMC Med. Res. Methodol. 2012, 12, 181. [Google Scholar] [CrossRef]
- Hirt, J.; Nordhausen, T.; Fuerst, T.; Ewald, H.; Appenzeller-Herzog, C. Guidance on Terminology, Application, and Reporting of Citation Searching: The TARCiS Statement. BMJ 2024, 385, e078384. [Google Scholar] [CrossRef]
- EQUATOR Network. Reporting Guidelines for Systematic Reviews and Meta-Analyses. Available online: https://www.equator-network.org/reporting-guidelines-study-design/systematic-reviews-and-meta-analyses/ (accessed on 10 April 2026).
- Fischer, A.; Spiegl, M.; Altmann, K.; Winkler, A.; Salamon, A.; Themessl-Huber, M.; Mouhieddine, M.; Strasser, E.M.; Schiferer, A.; Paternostro-Sluga, T.; et al. Muscle Mass, Strength and Functional Outcomes in Critically Ill Patients After Cardiothoracic Surgery: Does Neuromuscular Electrical Stimulation Help? The Catastim 2 Randomized Controlled Trial. Crit. Care 2016, 20, 30. [Google Scholar] [CrossRef]
| Component | Criteria |
| Population | Adults aged ≥65 years with diagnosed cardiovascular disease (HF, post-AMI, post-PCI, post-CABG); sarcopenia defined per EWGSOP2/AWGS criteria OR frailty (SPPB ≤9) |
| Intervention | Peripheral muscle electrostimulation (NMES, FES, EMS) applied to lower or upper extremities; any frequency, duration, or setting (home/hospital) |
| Comparator | Conventional cardiac rehabilitation alone, sham stimulation, or usual care |
| Outcomes | Primary: Physical function (SPPB, 6MWD, gait speed); muscle strength (quadriceps isometric strength, handgrip); muscle mass (ultrasound, DEXA, bioimpedance). Secondary: Quality of life, ADL, BNP, muscle protein turnover markers (3-MH/creatinine), hospital readmission, adverse events |
| Study Design | Randomized controlled trials (RCTs), controlled clinical trials, crossover trials |
| Study (Year) | Design | N | Mean Age (years) | Population | Intervention | Duration | Setting |
| Tanaka et al. (2022) [20] | RCT | 31 | 82.9 ± 4.8 | Frail AHF (≥75 yrs, SPPB 4–9) | EMS + early CR vs. CR alone | 2 weeks | Hospital |
| Ono et al. (2025) [15] | Crossover RCT | 8 | 85.5 [84–88] | Frail CHF (≥75 yrs, SPPB ≤8) | Home NMES + CR vs. CR alone | 8 weeks | Home |
| Pu et al. (2024) [16] | RCT | 100 | 71.7 ± 6.5 | Post-PCI AMI, frail | NMES + usual care vs. usual care | 7 days | Hospital |
| Iwatsu et al. (2017) [11] | Pre-post RCT | 102 | 74.2 ± 6.8 | Post-cardiac surgery | NMES vs. control | 5 days | ICU |
| Wang et al. (2022) [21] | Meta-analysis | 236 | 72–81 | CHF (HFrEF/HFpEF) | FES legs vs. placebo | 8–12 weeks | Home/Hospital |
| Gomes-Neto et al. (2016) [22] | Meta-analysis | 188 | 68–75 | CHF | NMES vs. control | 4–12 weeks | Mixed |
| Fischer et al. (2016) [34] | RCT | 54 | 76.4 ± 7.1 | Critically ill post-CABG | NMES vs. sham | 7 days | ICU |
| Karavidas et al. (2013) [23] | RCT | 28 | 71.5 ± 8.2 | HFpEF | FES vs. control | 6 weeks | Home |
| Parameter | Range Across Studies | Most Common Protocol | Anti-Sarcopenia Rationale |
| Frequency | 20–100 Hz | 20 Hz (acute); 50–66 Hz (chronic) [11,15,20] | 50–100 Hz maximizes type II fiber recruitment and protein synthesis [18] |
| Pulse width | 250–400 μs | 250–400 μs [15,20] | Optimal motor nerve recruitment without discomfort |
| Duty cycle | 5 s on/2 s off to 5 s on/5 s off | 5 s on/2 s off [20] | Mimics physiological contraction-relaxation; prevents fatigue |
| Session duration | 30–60 min | 30–50 min [11,15,20] | ≥30 hours total needed for functional and hypertrophic gains [15] |
| Frequency/week | 5–7 days/week | 5–7 days/week [11,15,20] | Daily stimulation required to counteract hypercatabolism post-surgery [11] |
| Electrode placement | Quadriceps, hamstrings, gastrocnemius | Bilateral quadriceps (4 electrodes) [11,15,20] | Quadriceps most affected by sarcopenia; largest muscle mass [11] |
| Intensity | 10–20% MVC to maximum tolerable | Visible contraction, patient-tolerated [11,20] | ≥20% MVC required to activate mTOR pathway [11,18] |
| Total duration | 5 days – 12 weeks | 2–8 weeks [11,15,16,17,20] | Early initiation (POD1) critical to prevent irreversible muscle loss [11] |
| Outcome | Studies (n) | Participants | Effect Estimate (95% CI) | Certainty (GRADE) | Comments |
| Physical function (SPPB) | 3 | 139 | MD +2.3 to +2.67 points (all p < 0.05) | Low | Downgraded: risk of bias (−1), imprecision (−1) |
| Muscle strength (QIS) | 4 | 241 | MD +5.2% BW (95% CI 1.2–9.1) | Low | Downgraded: risk of bias (−1), inconsistency (−1) |
| Muscle mass preservation | 3 | 256 | Quadriceps CSA: −3% vs. −12% control | Very low | Downgraded: risk of bias (−1), indirectness (−1), imprecision (−1) |
| Exercise capacity (6MWD) | 3 | 424 | MD +35 to +42 m (p < 0.001) | Low | Downgraded: inconsistency (−1), imprecision (−1) |
| Safety (adverse events) | 6 | 323 | No cardiovascular AEs; minor skin reactions only | Moderate | Downgraded: imprecision (−1) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




