Submitted:
14 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
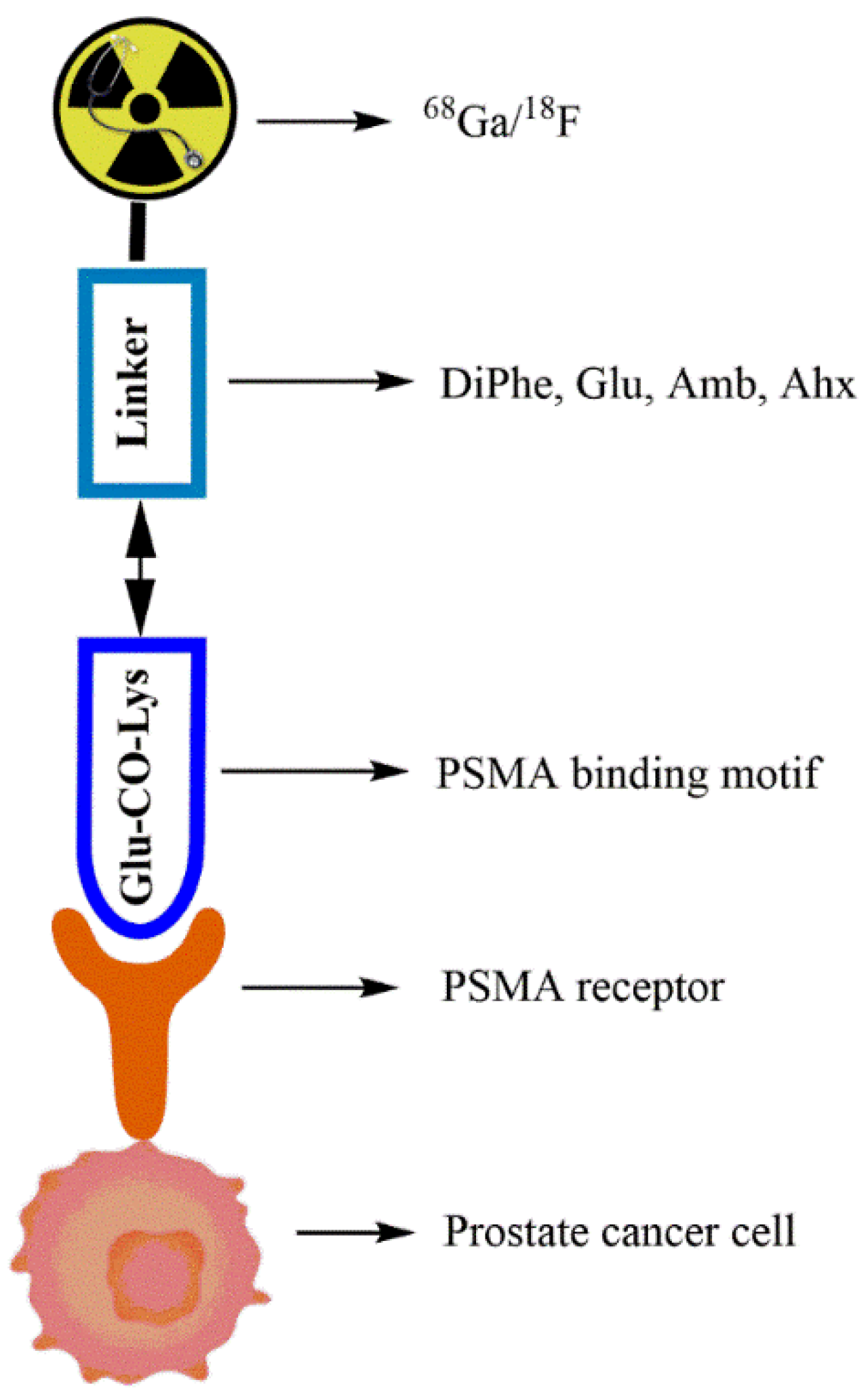
1. Introduction
2. Materials and Methods
General
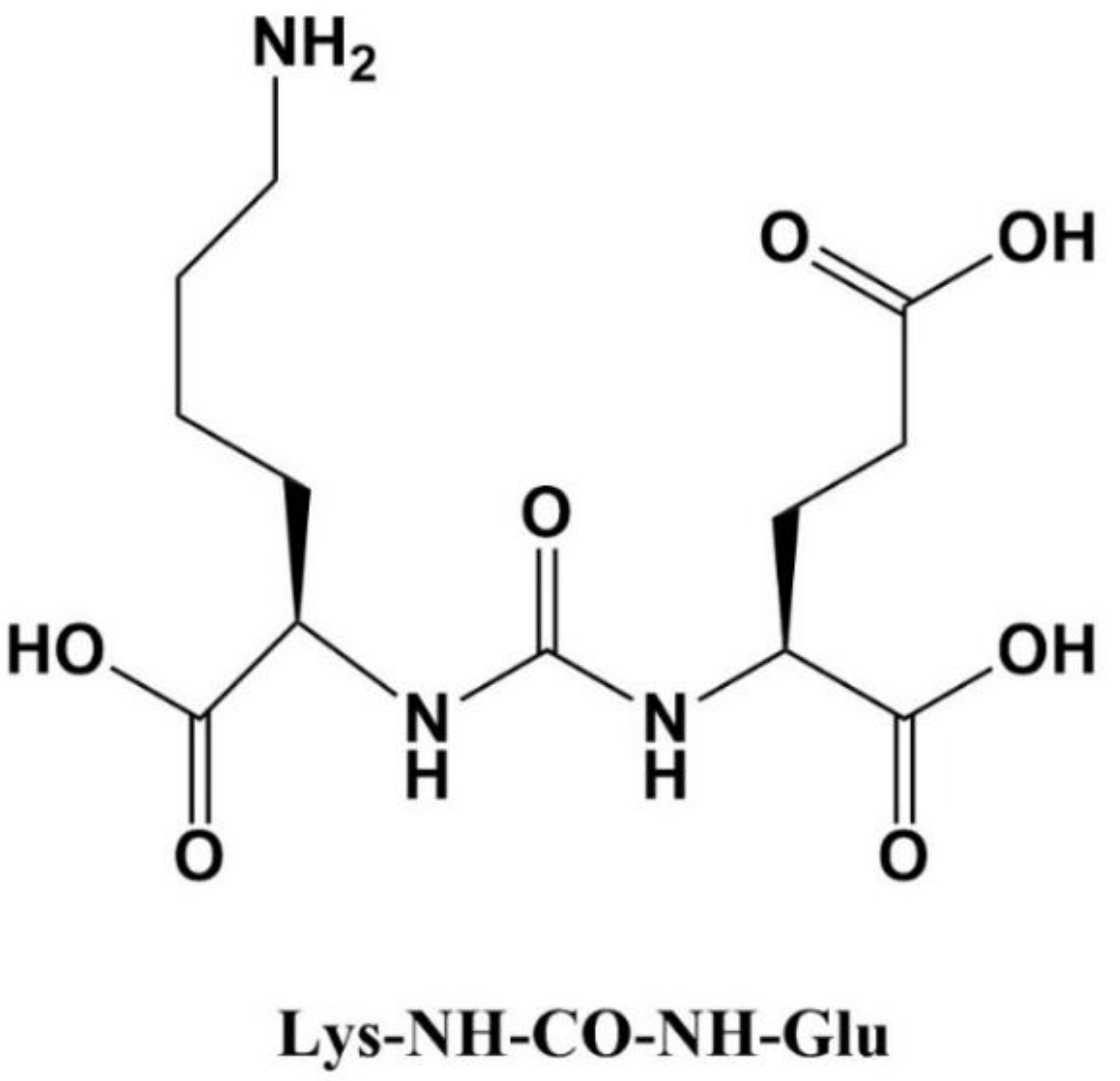
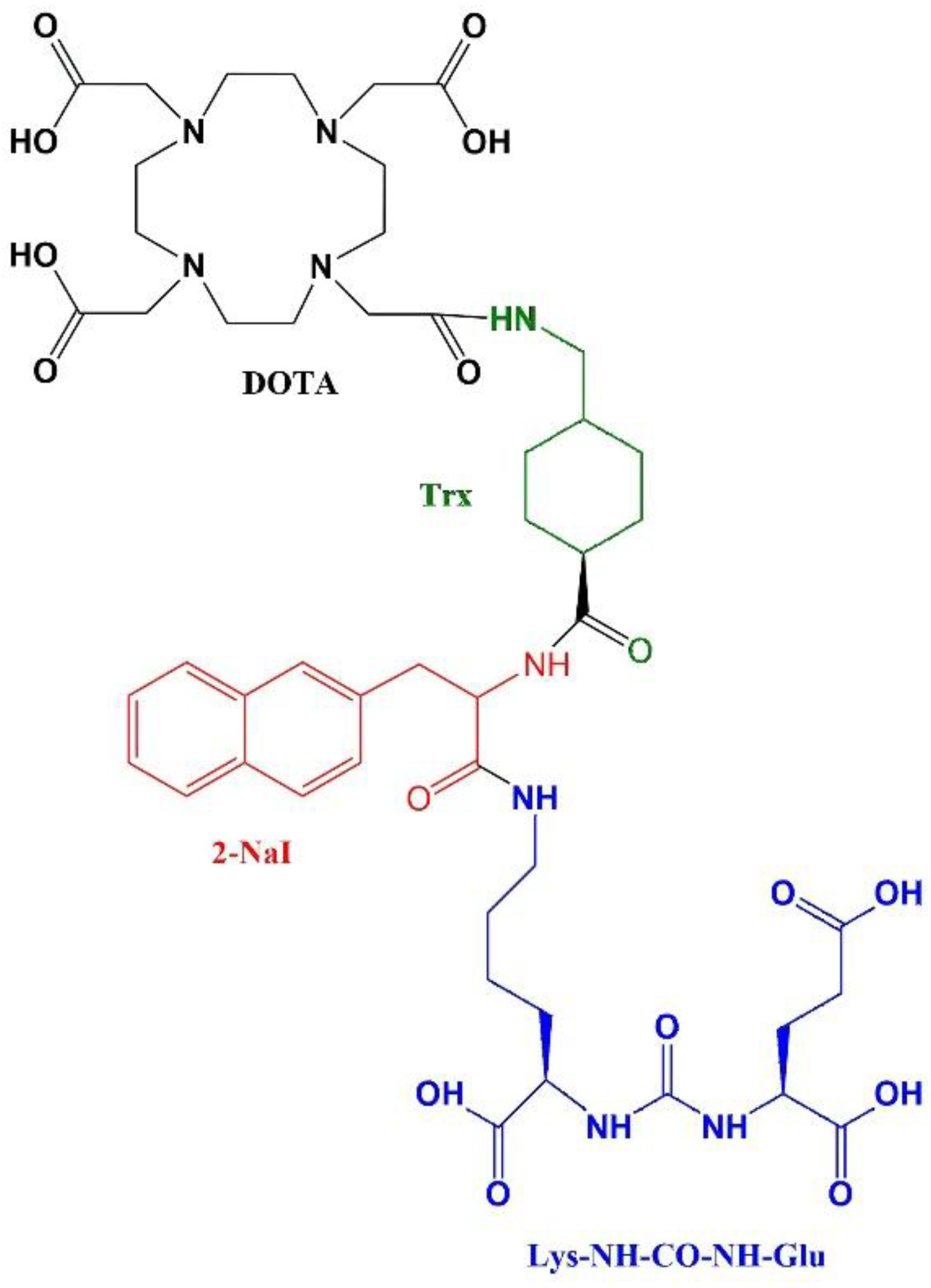
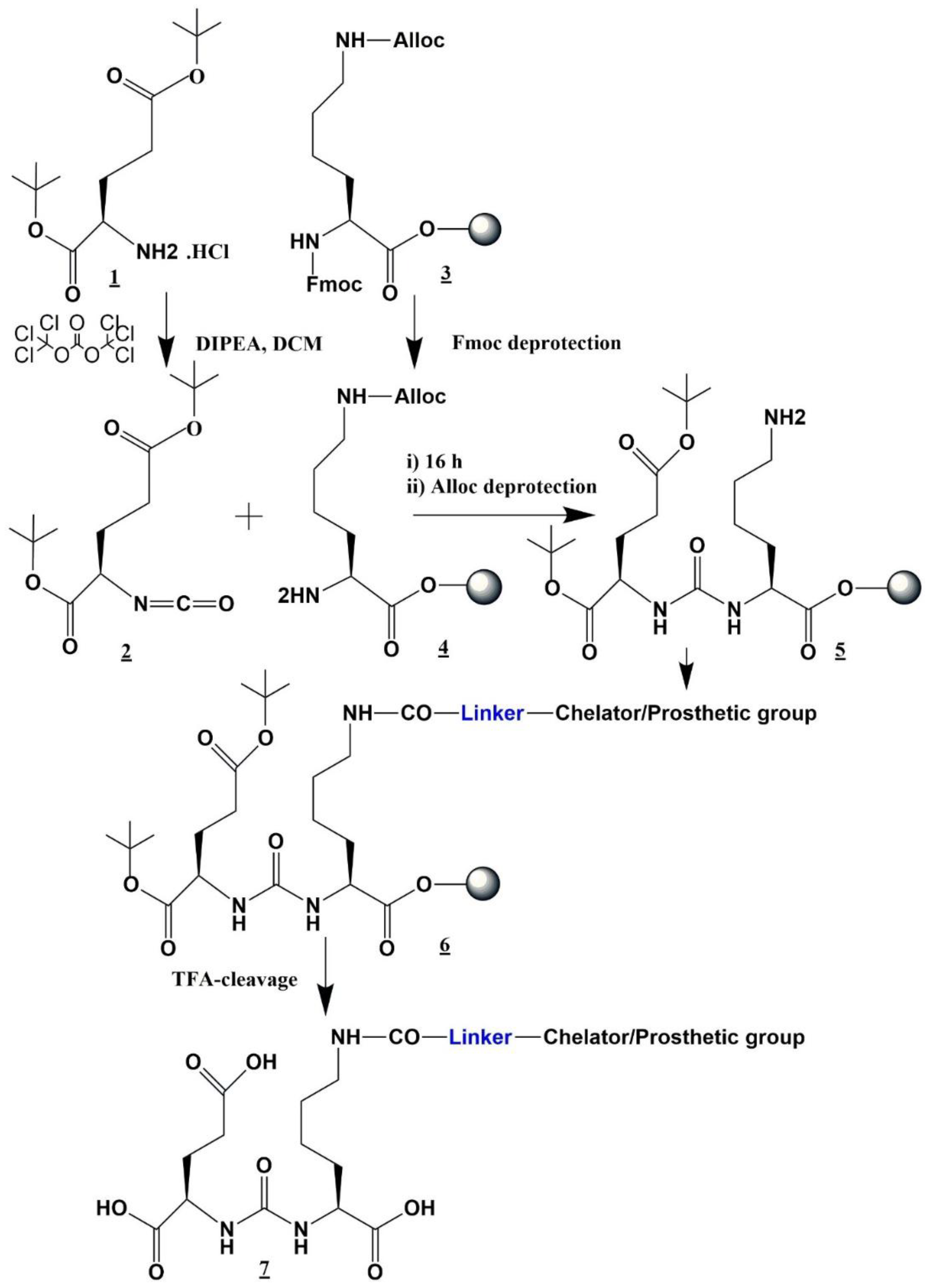
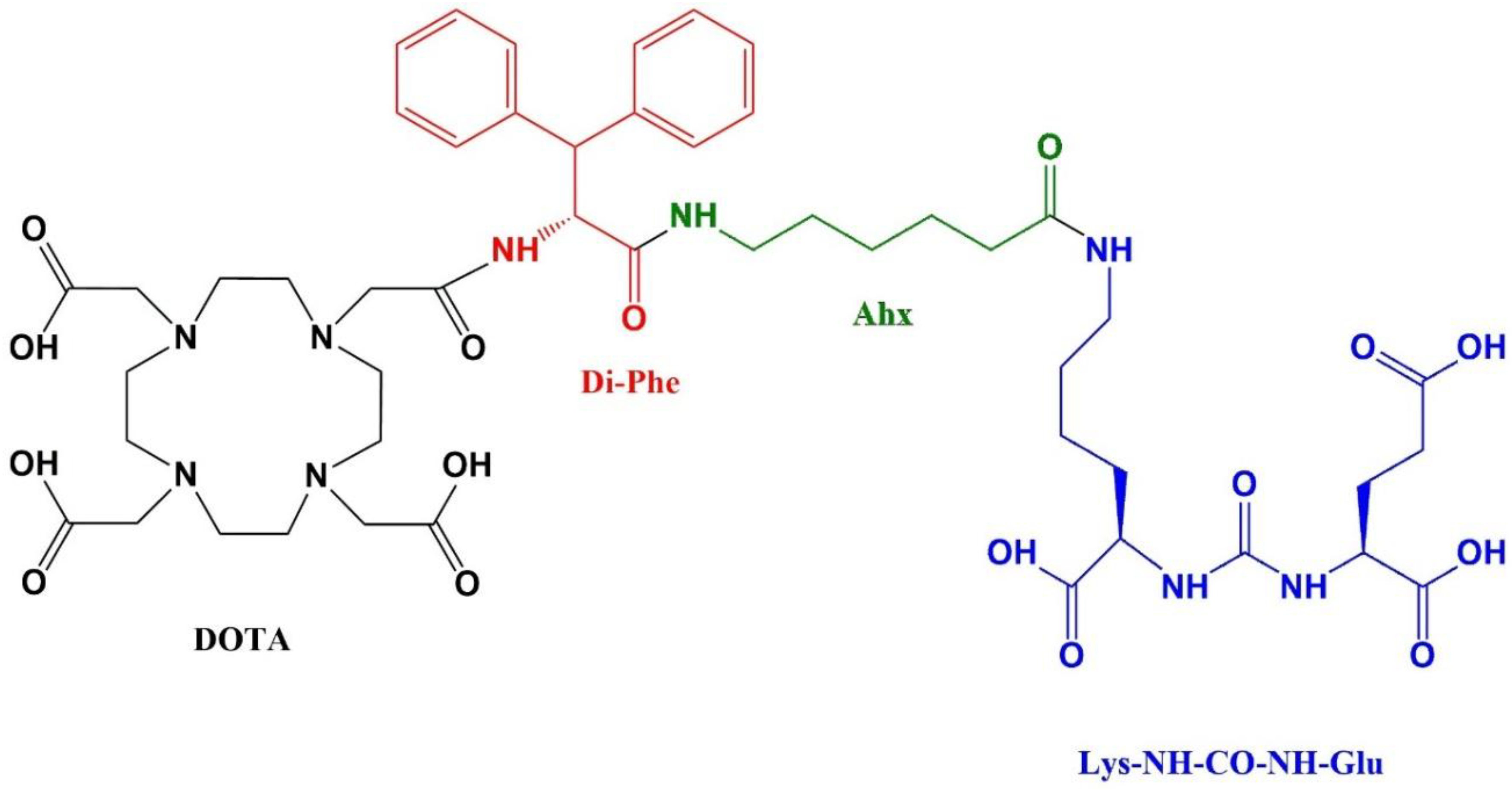
Preparation of Glu-NH-CO-NH-Lys-Ahx-DiPhe-DOTA
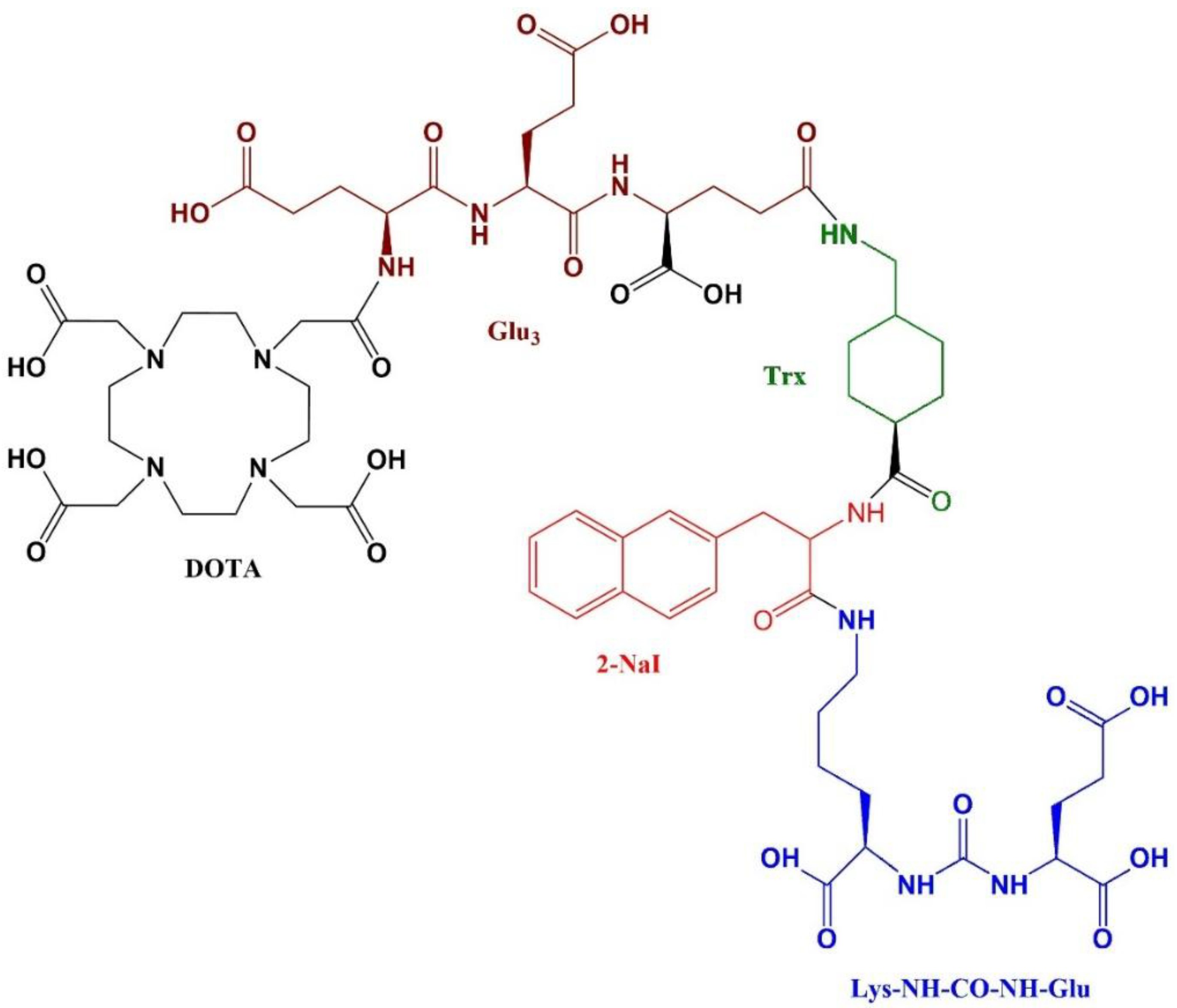
Preparation of Glu-NH-CO-NH-Lys-2-NaI-Trx-Glu-Glu-Glu-DOTA
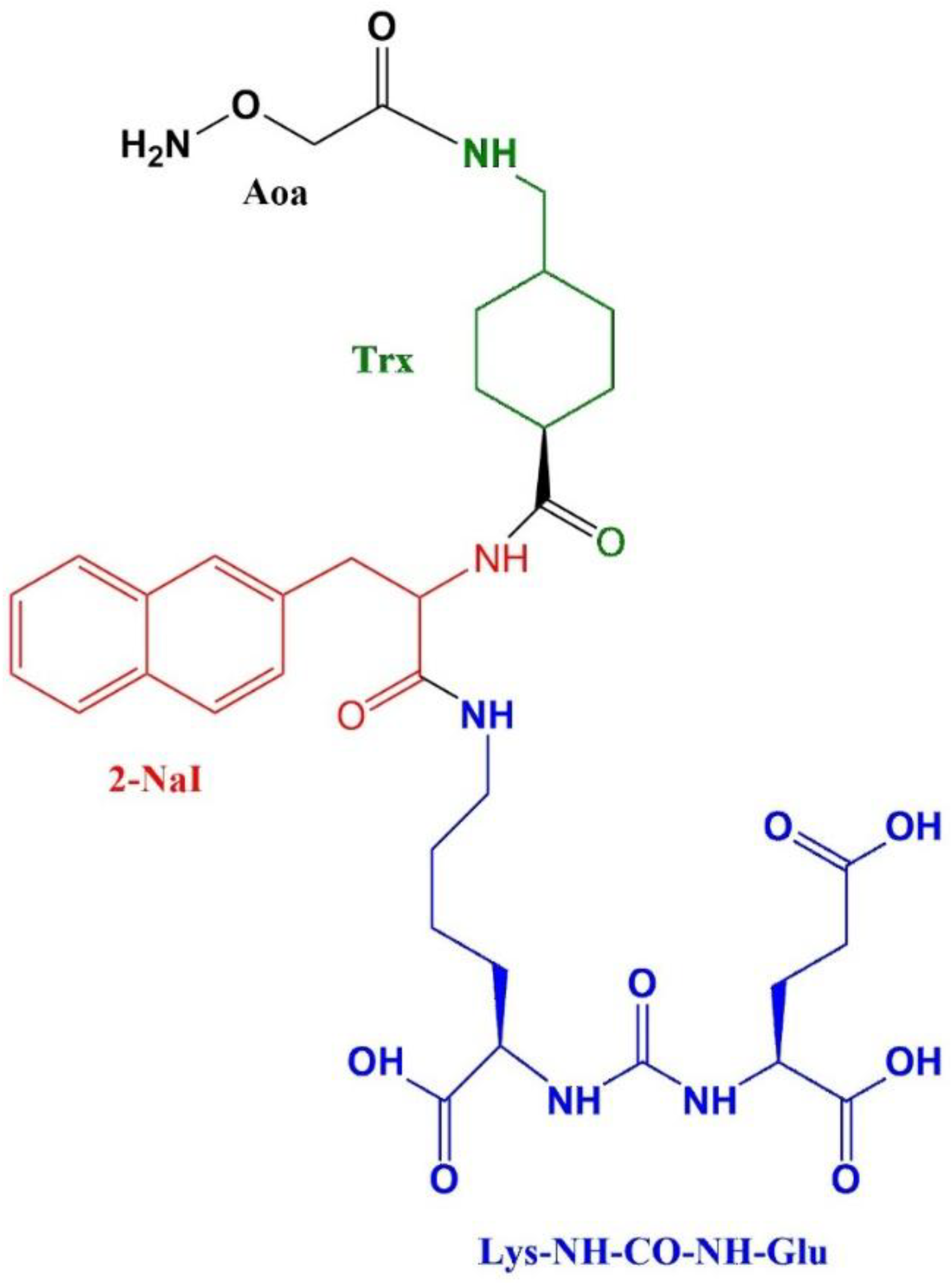
Preparation of Glu-NH-CO-NH-Lys-2-NaI-Trx-Aoa
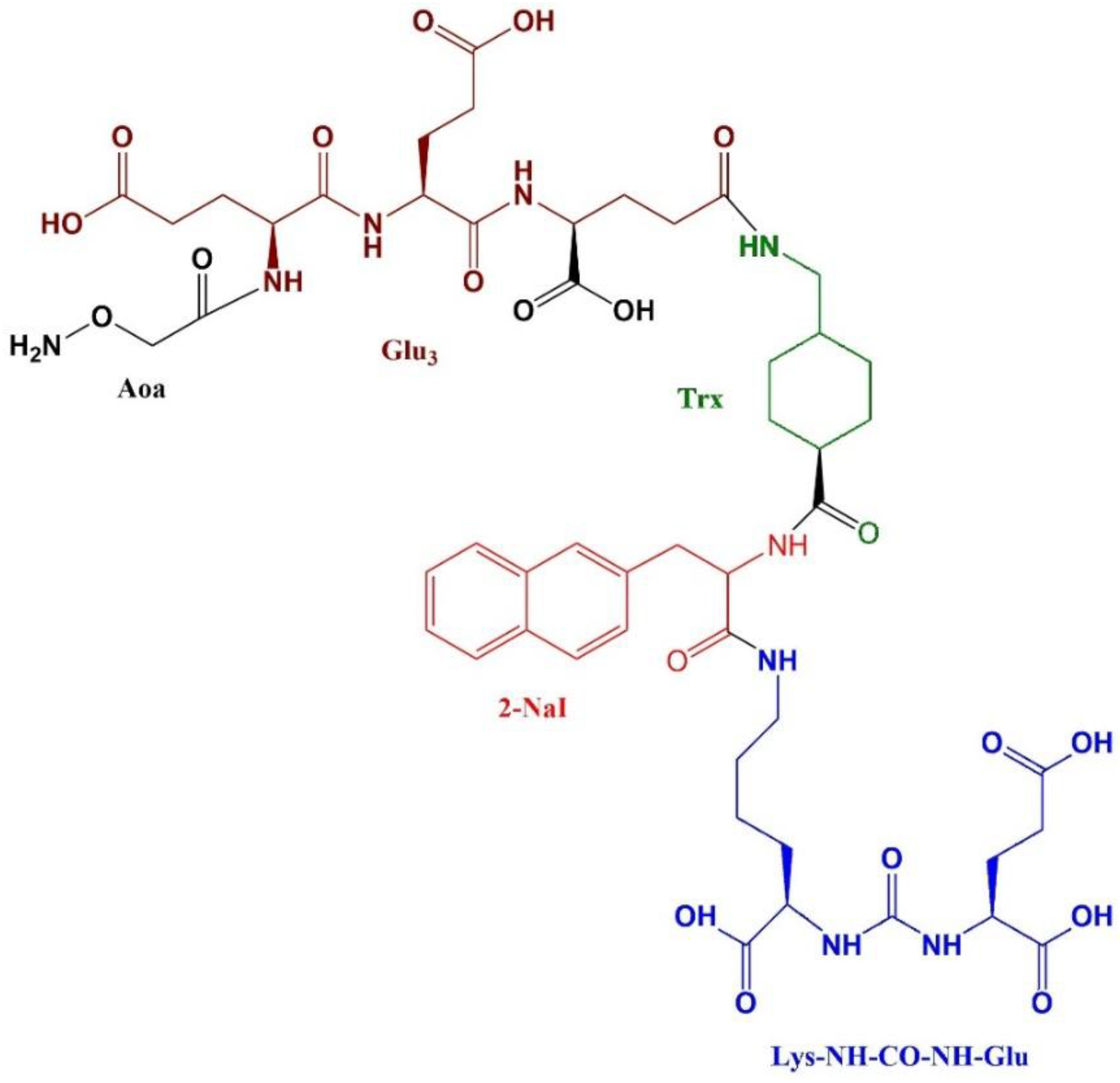
Preparation of Glu-NH-CO-NH-Lys-2-NaI-Trx-Glu-Glu-Glu-Aoa
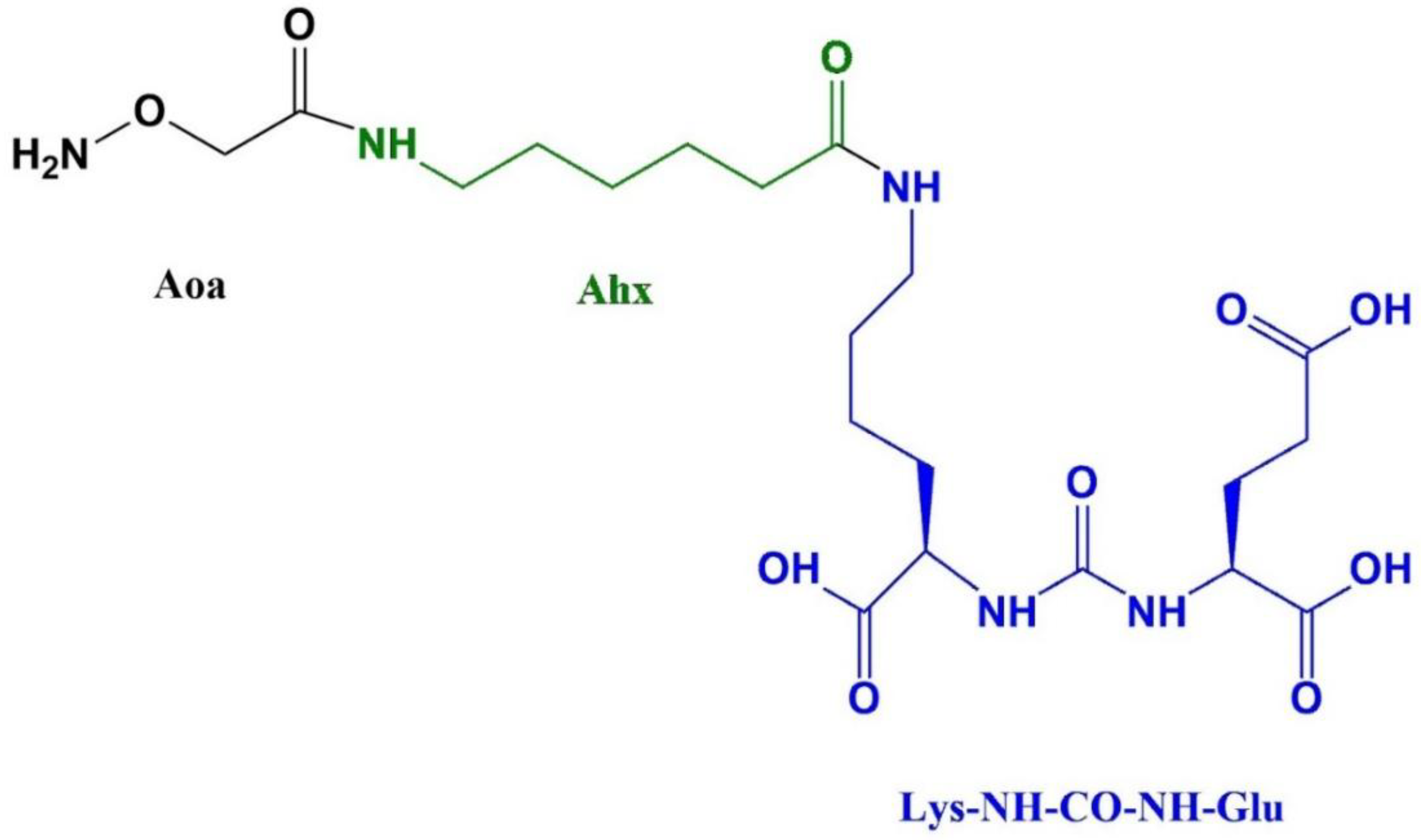
Preparation of Glu-NH-CO-NH-Lys-Ahx-Aoa
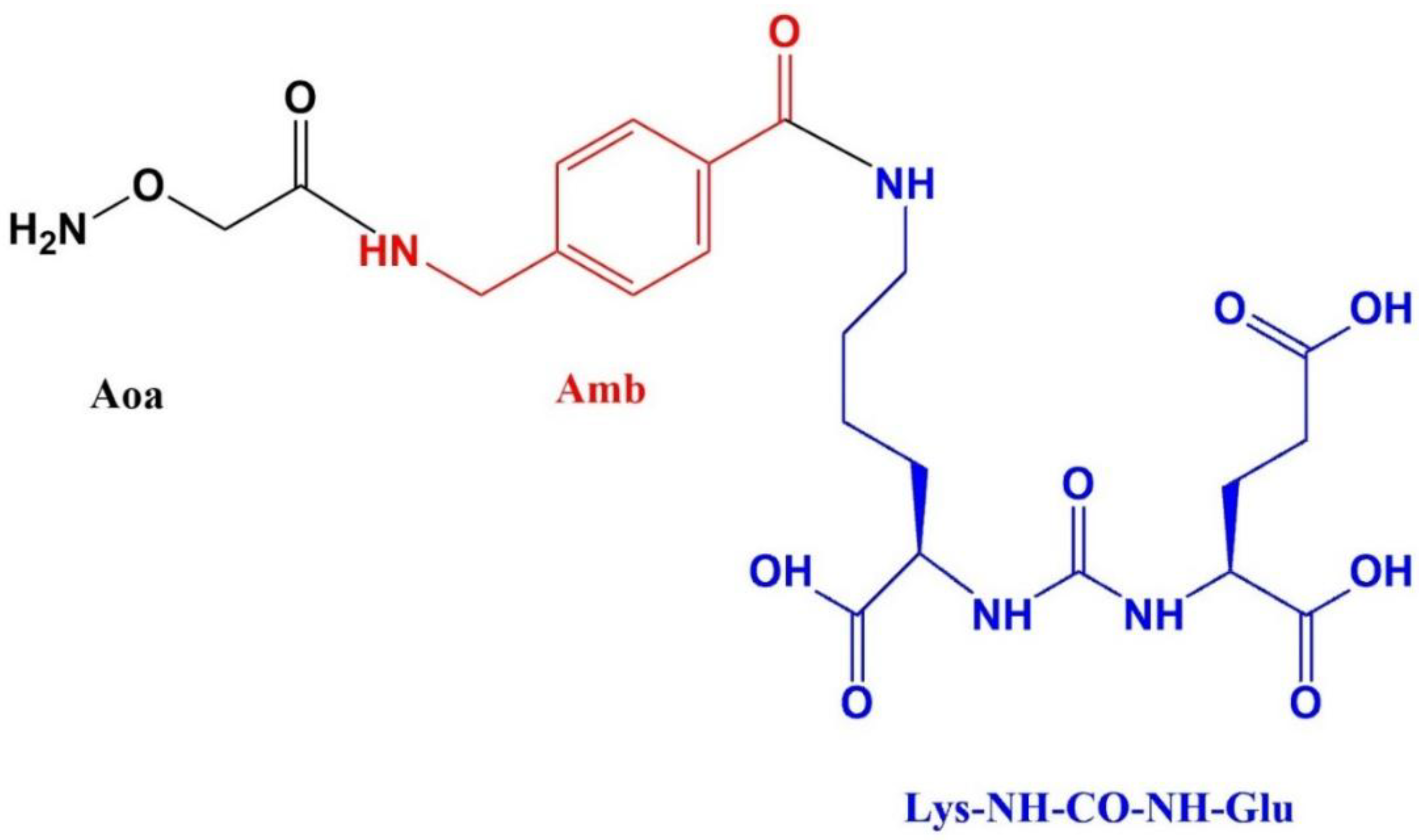
Preparation of Glu-NH-CO-NH-Lys-4-Amb-Aoa
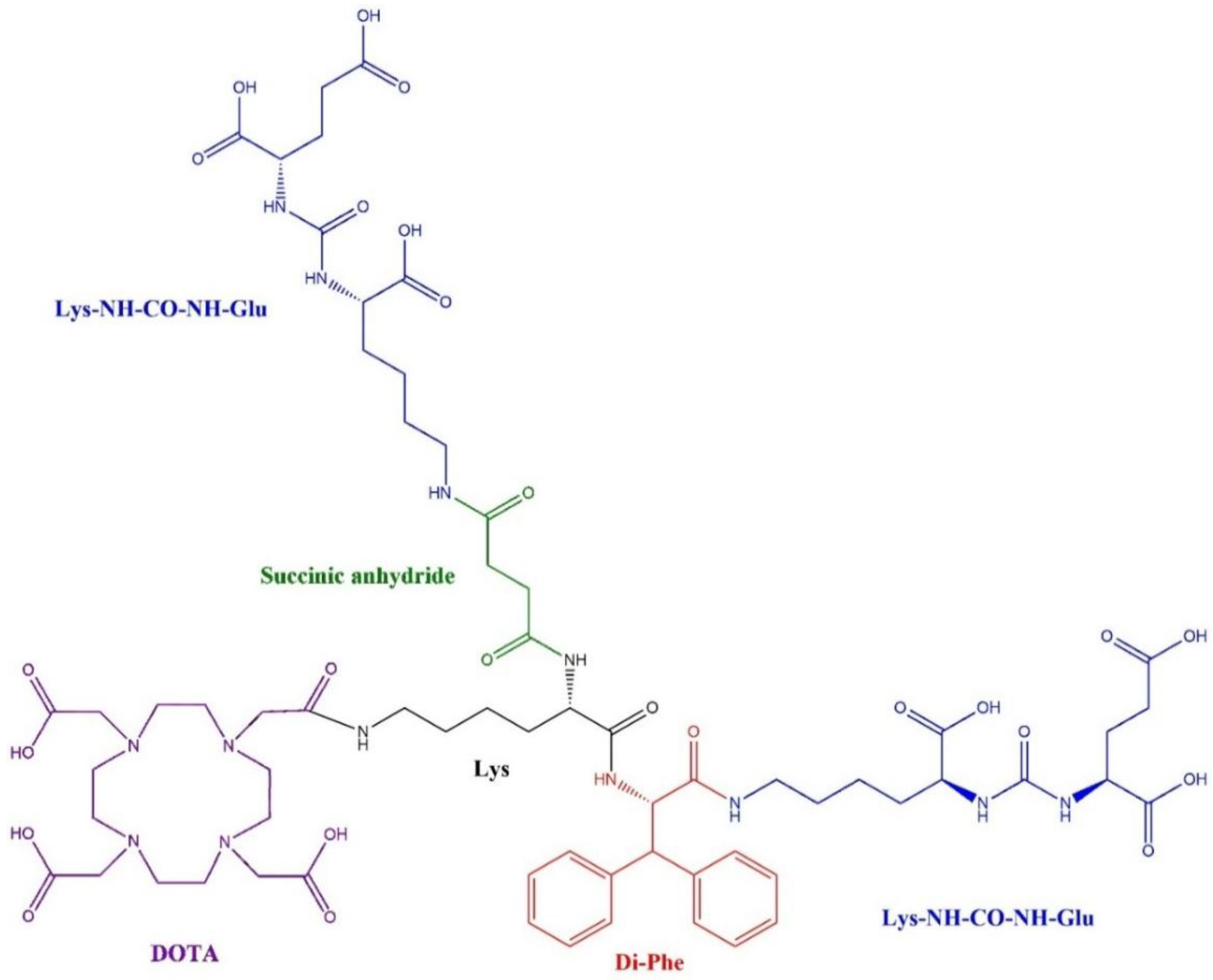
2. Synthesis of DOTA-Coupled Bis-PSMA-Binding Motif
Radiolabeling with 68Ga
Radiolabeling with 18F
Sep-Pak C18 Purification
3. Results
Synthesis
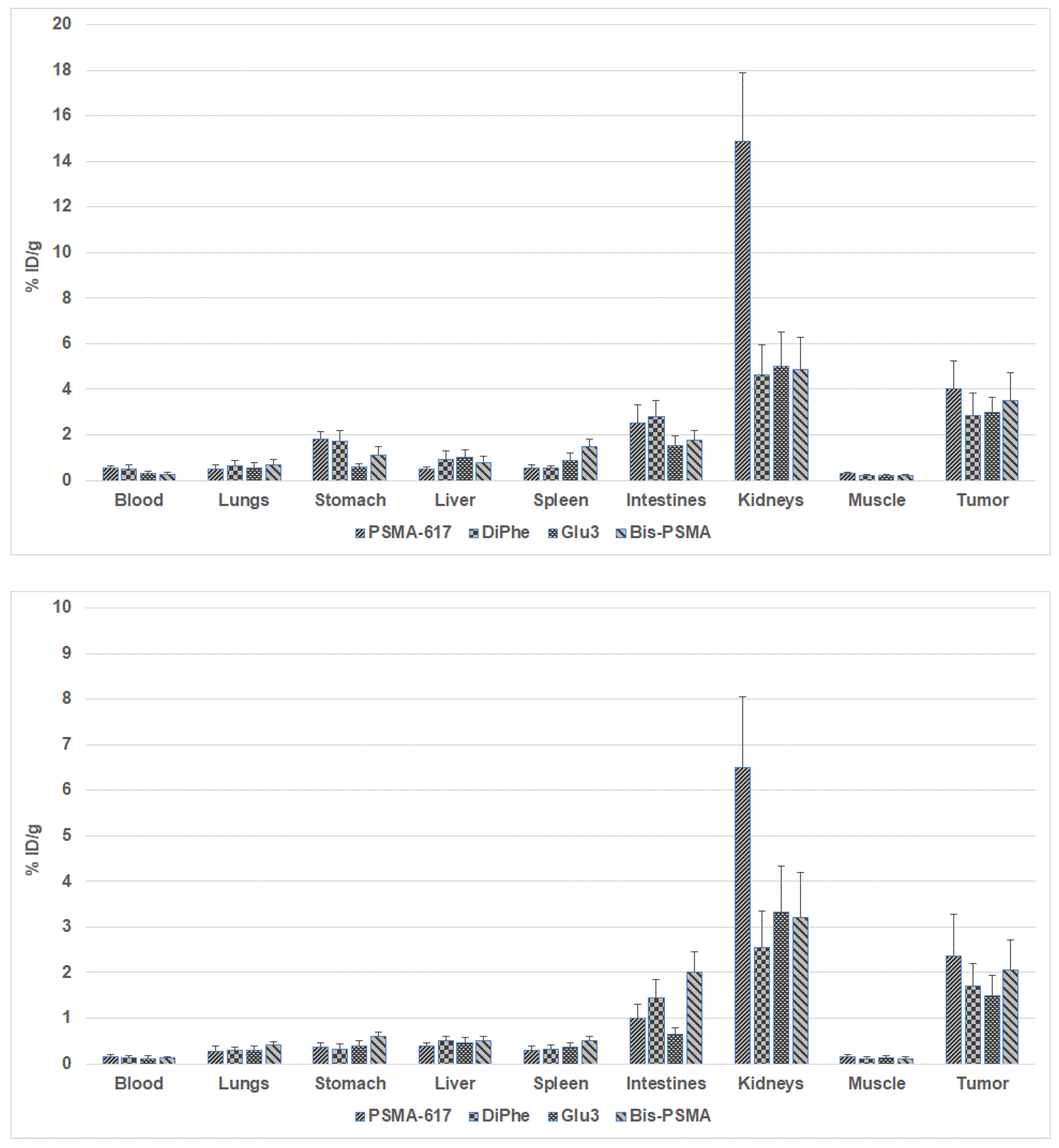
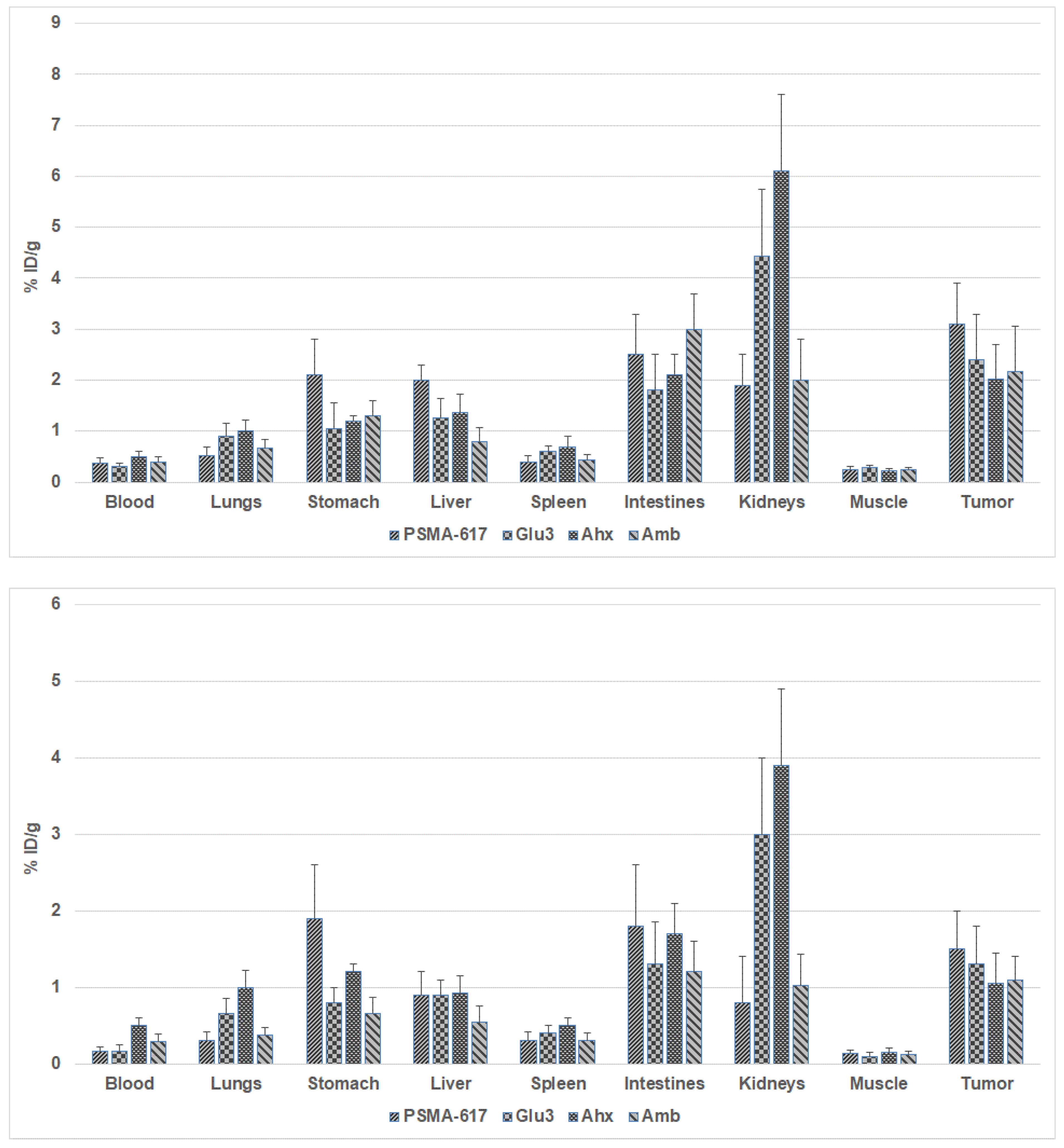
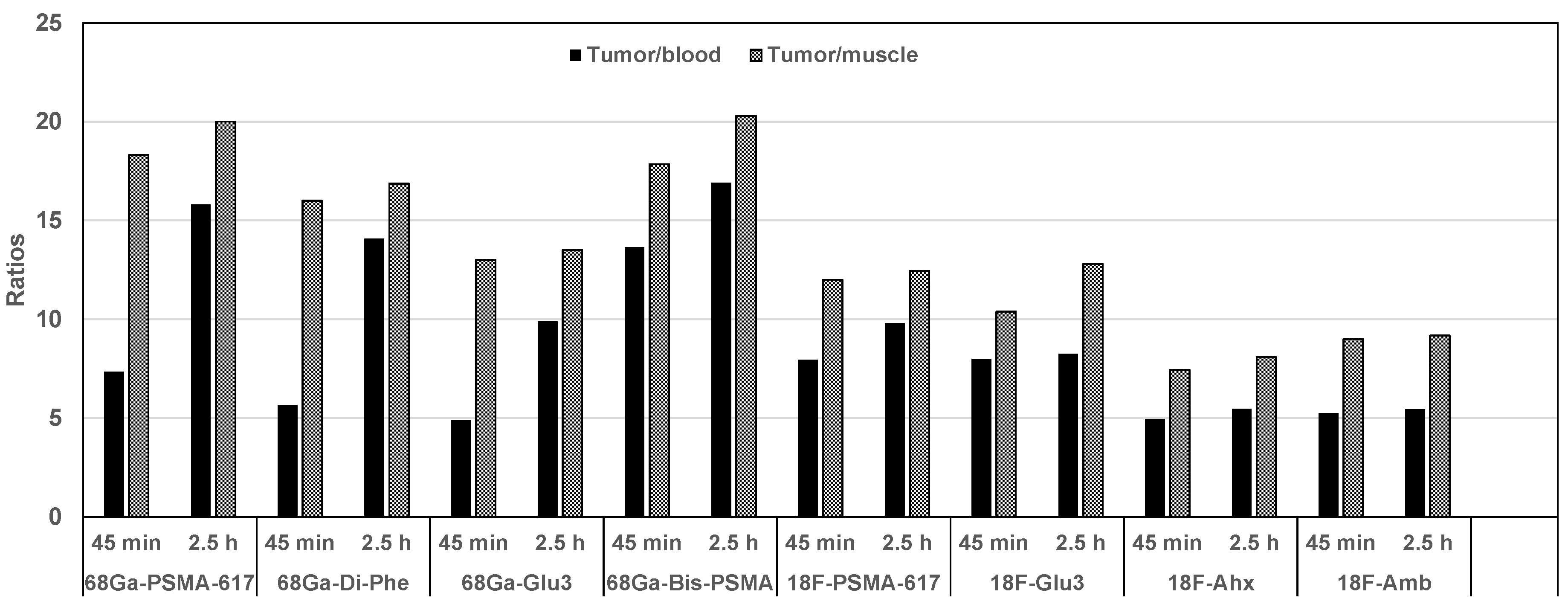
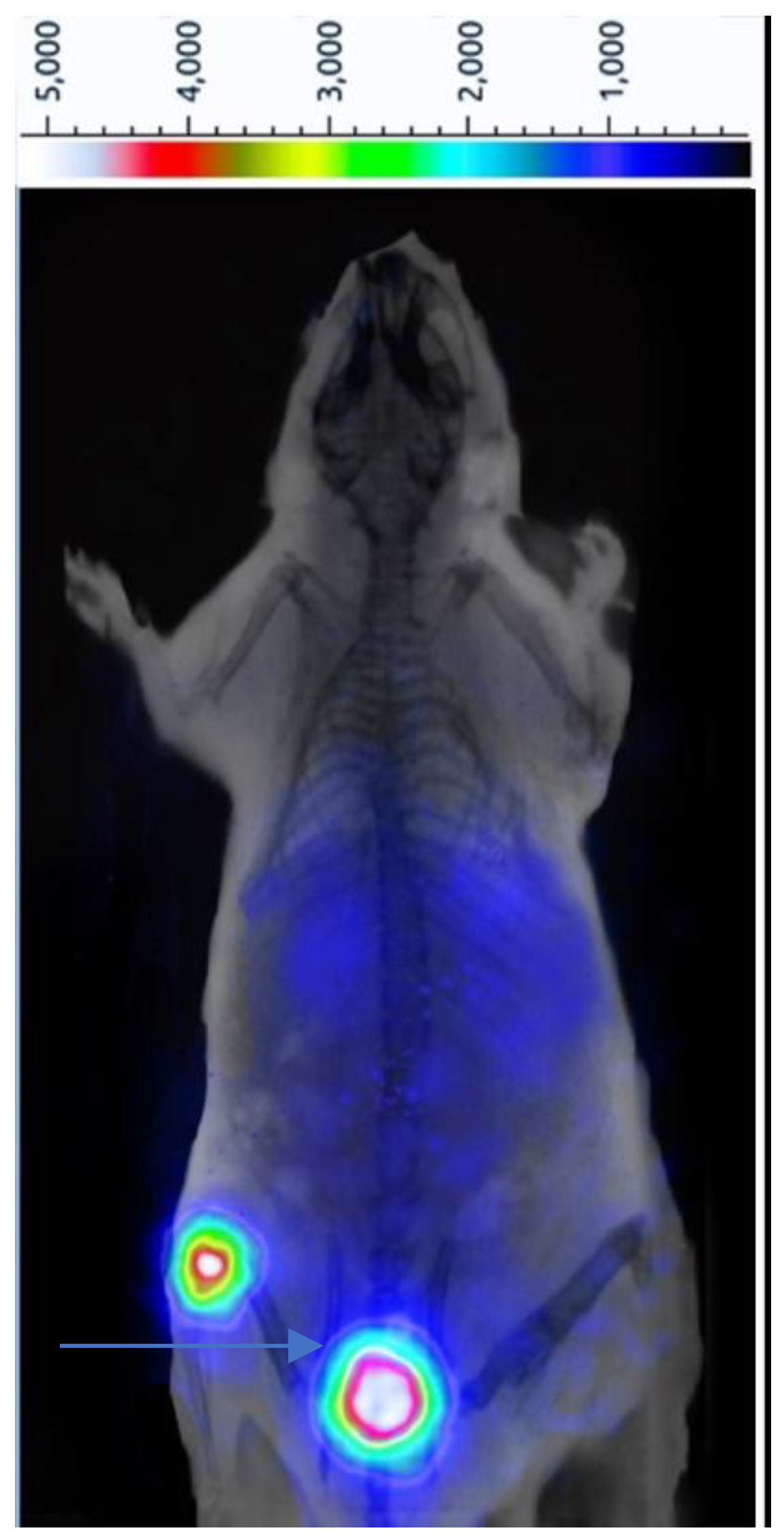
Small Animal PET Imaging
4. Discussion
5. Conclusion
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Leslie, S.W.; Soon-Sutton, T.L.; Skelton, W.P. Prostate Cancer. 2024 Oct 4. In StatPearls [Internet]; StatPearls Publishing: Treasure Island (FL), 2026. [Google Scholar] [PubMed]
- Okarvi, S.M. Recent developments of prostate-specific membrane antigen (PSMA)-specific radiopharmaceuticals for precise imaging and therapy of prostate cancer: an overview. Clin Transl Imaging 2019, 7, 189–208. [Google Scholar] [CrossRef]
- Debnath, S.; Zhou, N.; McLaughlin, M.; Rice, S.; Pillai, A.K.; Hao, G.; Sun, X. PSMA-Targeting Imaging and Theranostic Agents-Current Status and Future Perspective. Int J Mol Sci. 2022, 23(3), 1158. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wüstemann, T.; Bauder-Wüst, U.; Schäfer, M.; Eder, M.; Benesova, M.; Leotta, K.; Kratochwil, C.; Haberkorn, U.; Kopka, K.; Mier, W. Design of Internalizing PSMA-specific Glu-ureido-based Radiotherapeuticals. Theranostics 2016, 6(8), 1085–95. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rogers, O.C.; Rosen, D.M.; Antony, L.; et al. Targeted delivery of cytotoxic proteins to prostate cancer via conjugation to small-molecule urea-based PSMA inhibitors. Sci Rep 2021, 11, 14925. [Google Scholar] [CrossRef]
- Murce, E.; Beekman, S.; Spaan, E.; Handula, M.; Stuurman, D.; de Ridder, C.; Seimbille, Y. Preclinical Evaluation of a PSMA-Targeting Homodimer with an Optimized Linker for Imaging of Prostate Cancer. Molecules 2023, 28, 4022. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.; Yang, J.; Li, J.; Qin, J.; Tang, R.; Zhao, J.; Peng, Y.; Qiu, L.; Lin, J. Preclinical Evaluation Study of 68Ga-Labeled PSMA-Targeted Dimer Probe. Chem Biomed Imaging 2025, 4(1), 54–63. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hennrich, U.; Eder, M. [177Lu]Lu-PSMA-617 (PluvictoTM): The First FDA-Approved Radiotherapeutical for Treatment of Prostate Cancer. Pharmaceuticals 2022, 15(10), 1292. [Google Scholar] [CrossRef]
- Geis, N.M.; Braunwarth, Y.; Meyer, P.T.; Eder, M.; Eder, A.C. Optimizing PSMA-617-based inhibitors through charged linker modifications: Insights into structure-activity relationships. Theranostics 2026, 16(6), 2798–2810. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lundmark, F.; Olanders, G.; Rinne, S.S.; Abouzayed, A.; Orlova, A.; Rosenström, U. Design, Synthesis, and Evaluation of Linker-Optimised PSMA-Targeting Radioligands. Pharmaceutics 2022, 14(5), 1098. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Salih, S.; Elliyanti, A.; Alkatheeri, A.; AlYafei, F.; Almarri, B.; Khan, H. The Role of Molecular Imaging in Personalized Medicine. J Pers Med. 2023, 13(2), 369. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Singh, D.; Dhiman, V.K.; Pandey, M.; Sharma, A.; Pandey, H.; Verma, S.K.; Pandey, R. Personalized medicine: An alternative for cancer treatment. Cancer Treatment and Research Communications, Volume 2024, Volume 42, 100860. [Google Scholar] [CrossRef]
- Benešová, M.; Bauder-Wüst, U.; Schäfer, M.; Klika, K.D.; Mier, W.; Haberkorn, U.; Kopka, K.; Eder, M. Linker Modification Strategies To Control the Prostate-Specific Membrane Antigen (PSMA)-Targeting and Pharmacokinetic Properties of DOTA-Conjugated PSMA Inhibitors. J Med Chem. 2016, 59(5), 1761–75. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.S.; Wang, X.; Zhang, Y.; Doke, A.; DiFilippo, F.P.; Heston, W.D. Improving the biodistribution of PSMA-targeting tracers with a highly negatively charged linker. Prostate 2014, 74(7), 702–13. [Google Scholar] [CrossRef] [PubMed]
- Benešová, M.; Schäfer, M.; Bauder-Wüst, U.; Afshar-Oromieh, A.; Kratochwil, C.; Mier, W.; Haberkorn, U.; Kopka; K. Eder, M. Preclinical Evaluation of a Tailor-Made DOTA-Conjugated PSMA Inhibitor with Optimized Linker Moiety for Imaging and Endoradiotherapy of Prostate Cancer. J Nucl Med. 2015, 56(6), 914–20. [Google Scholar] [CrossRef] [PubMed]
- Zia, N.A.; Cullinane, C.; Van Zuylekom, J.K.; Waldeck, K.; McInnes, L.E.; G. Buncic, G.; Haskali, M.B.; Roselt, P.D.; Hicks, R.J.; Donnelly, P.S. A Bivalent Inhibitor of Prostate Specific Membrane Antigen Radiolabeled with Copper-64 with High Tumor Uptake and Retention. Angew. Chem. Int. Ed. 2019, 58, 14991–14994. [Google Scholar] [CrossRef] [PubMed]
- Schäfer, M.; Bauder-Wüst, U.; Leotta, K.; Zoller, F.; Mier, W.; Haberkorn, U.; Eisenhut, M.; Eder, M. A dimerized urea-based inhibitor of the prostate-specific membrane antigen for 68Ga-PET imaging of prostate cancer. EJNMMI Res 2012, 2(1), 23. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Martin, S.; Schreck, M.V.; Stemler, T.; Maus, S.; Rosar, F.; Burgard, C.; Schaefer-Schuler, A.; Ezziddin, S.; Bartholomä, M.D. Development of a homotrimeric PSMA radioligand based on the NOTI chelating platform. EJNMMI Radiopharm Chem. 2024, 9(1), 84. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nordquist, L.; Lengyelova, E.; Saltzstein, D.; Josephson, D.; Franklin, G.; Morrish, G.; Gervasio, O.; Parker, M.; Miller, R.; Shore, N. COBRA: Assessment of safety and efficacy of 64Cu-SAR-bisPSMA in patients with biochemical recurrence of prostate cancer following definitive therapy. Journal of Nuclear Medicine 2024, 65 (supplement 2), 242291. [Google Scholar] [CrossRef]
- Gorin, M.A.; Lengyelova, E.L.; Morrish, G.; Gervasio, O.; Miller, R.M.; Shore, N.D. CLARIFY: Positron emission tomography using 64Cu-SAR-bisPSMA in patients with high-risk prostate cancer prior to radical prostatectomy—A phase 3 diagnostic performance study. J Clin Oncol 2025, 43, TPS429 No 5_suppl. [Google Scholar] [CrossRef]
- Chan, W.; White, P. Fmoc solid phase peptide synthesis: A practical approach; Oxford Academic, 1999. [Google Scholar]
- Okarvi, S.M.; Maliki, Y. Preparation and preclinical evaluation of 18F-labeled folate-RGD peptide conjugate for PET imaging of triple-negative breast carcinoma. EJNMMI Radiopharm Chem 2025, 10(1), 25. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Okarvi, S.M. A novel solid phase synthesis of an estradiol derivative and preclinical evaluation for targeting estrogen receptor positive breast cancer. Scientific Reports | 2025, 15, 30143. [Google Scholar] [CrossRef] [PubMed]
- Wendeler, M.; Grinberg, L.; Wang, X.; Dawson, P.E.; Baca, M. Enhanced catalysis of oxime-based bioconjugations by substituted anilines. Bioconjug Chem. 2014, 25, 93–101. [Google Scholar] [PubMed]
- Vegt, E.; de Jong, M.; Wetzels, J.F.; Masereeuw, R.; Melis, M.; Oyen, W.J.; Gotthardt, M.; Boerman, O.C. Renal toxicity of radiolabeled peptides and antibody fragments: mechanisms, impact on radionuclide therapy, and strategies for prevention. J Nucl Med. Epub. 2010, 51(7), 1049–58. [Google Scholar] [CrossRef] [PubMed]
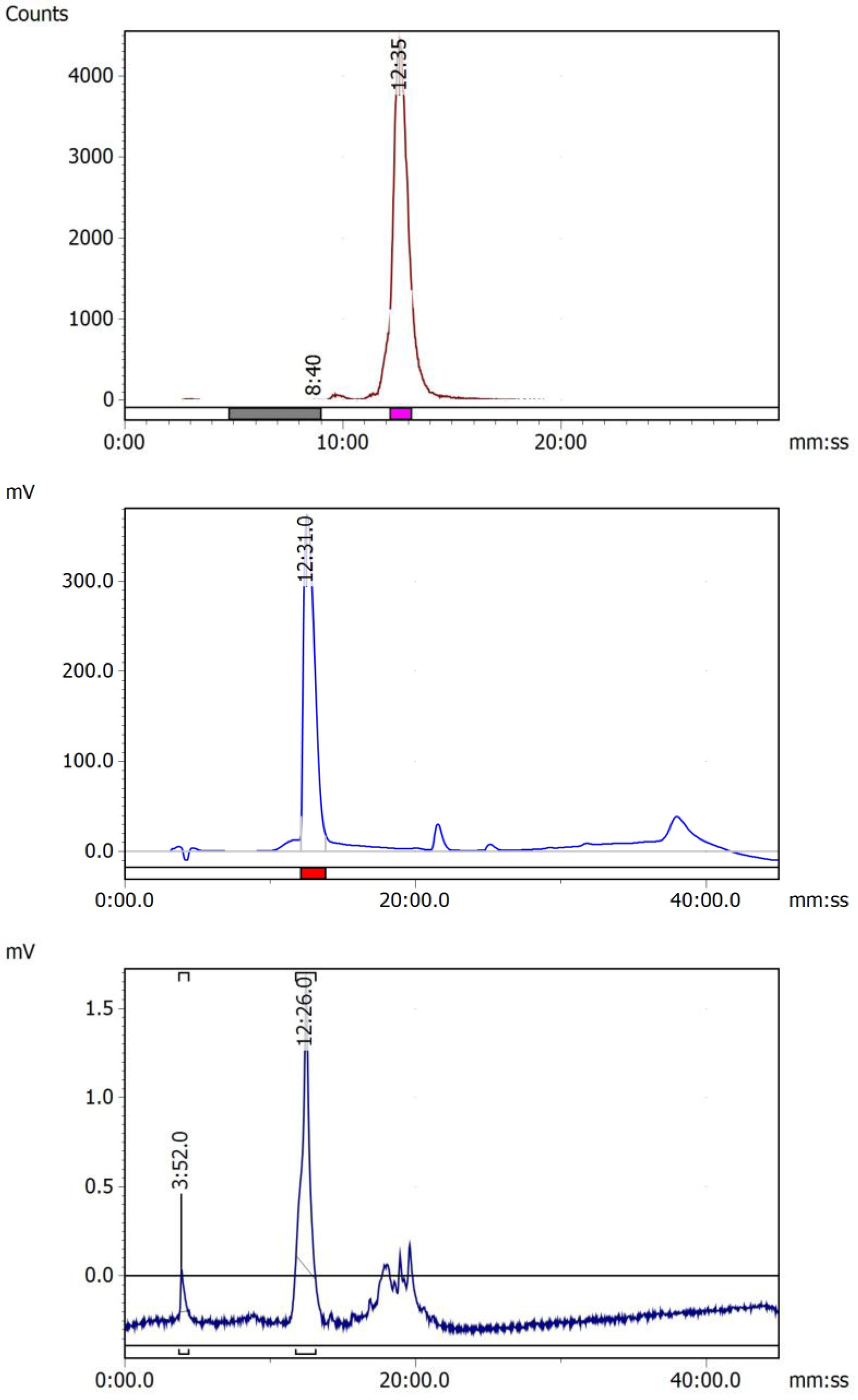
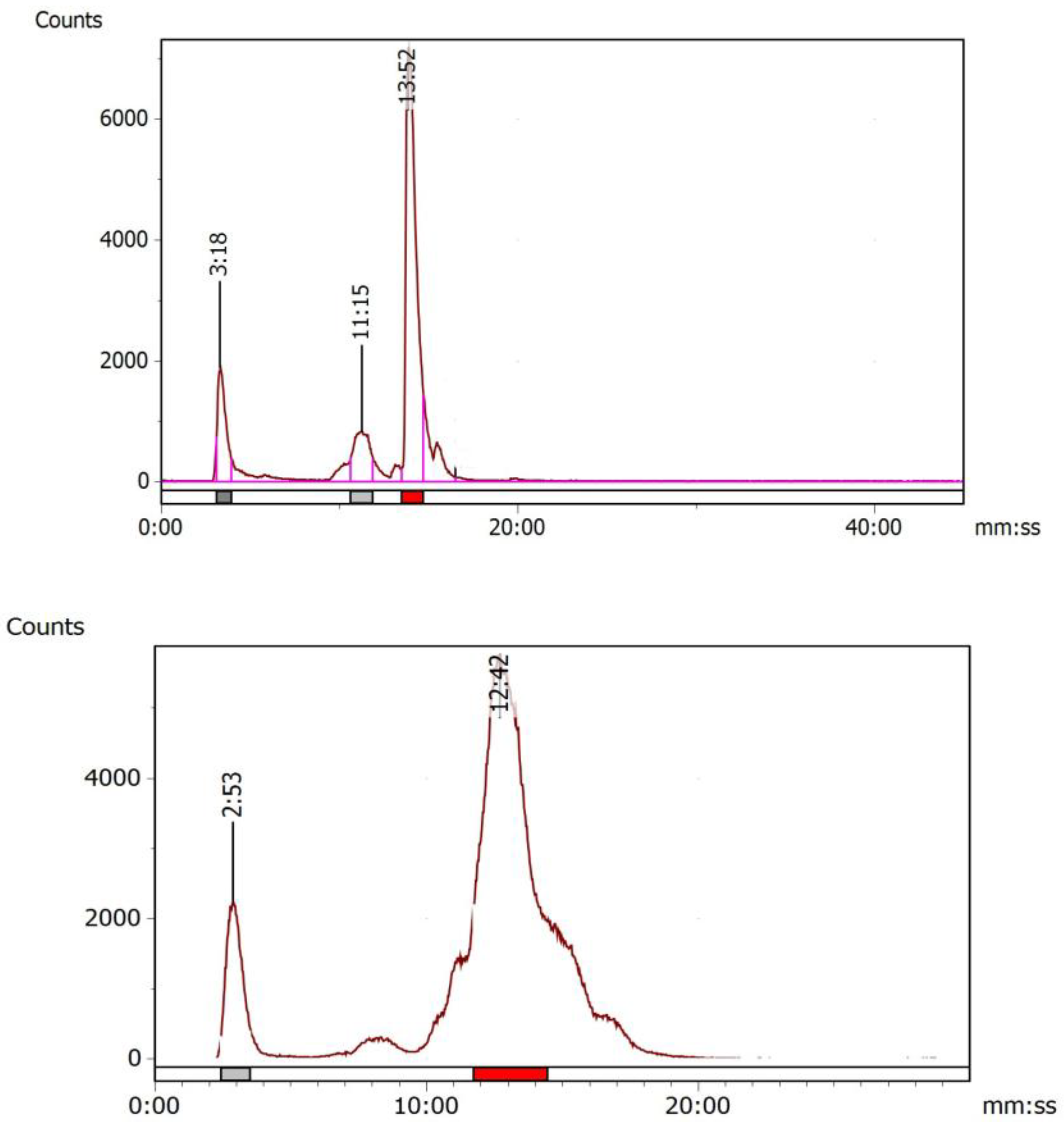
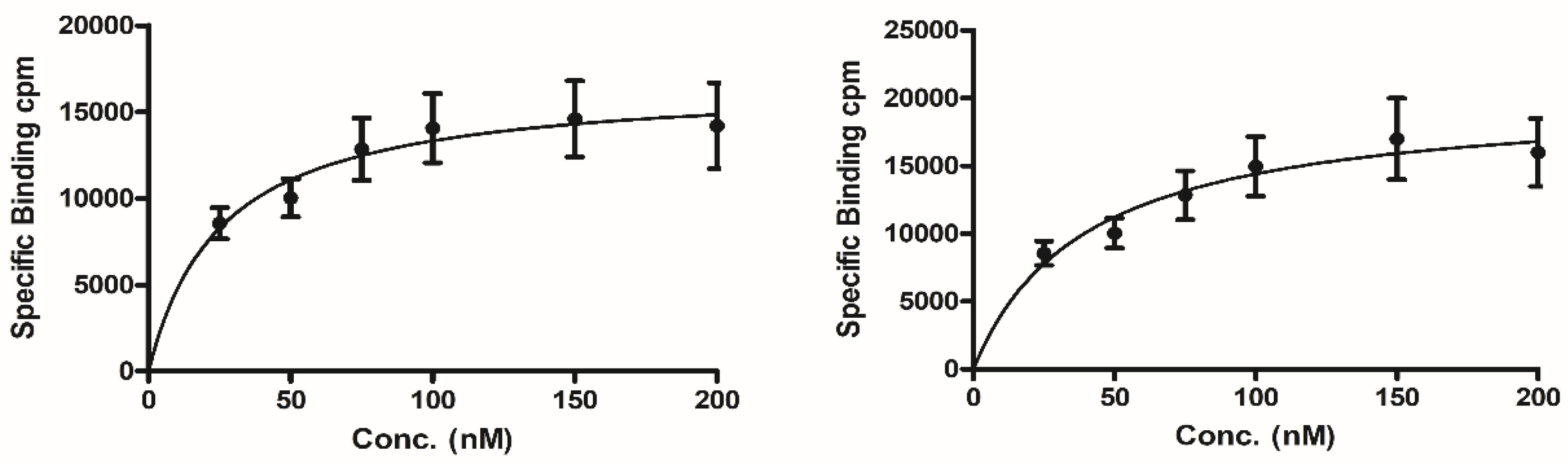
| Compound | Molecular weight | HPLC retention time (min) | Log P | Binding affinity (nM) |
|---|---|---|---|---|
| [68Ga]-DOTA-PSMA 617 [68Ga]-PSMA-617 |
1042.14 | 13.15 | -2.63±0.12 | 25.72 ± 4.84 |
| [68Ga]-DOTA-Di-Phe-Lys-CO-Glu-OH [68Ga]-Di-Phe |
1042.15 | 13.06 | -2.86±0.13 | 66.43 ± 12.35 |
| [68Ga]-DOTA-Glu3-Lys-CO-Glu-OH [68Ga]-Glu3 |
1429.4 | 12.45 | -3.09±0.11 | 59.12 ± 10.13 |
| [68Ga]-DOTA-Bis-PSMA-(Lys-CO-Glu)2 [68Ga]-Bis-PSMA |
1458.5 | 12.35 | -3.18±0.15 | 39.92 ± 9.89 |
| [18F]-Aoa-2-NaI-Trx-Lys-CO-Glu-OH PSMA-617 [18F]-PSMA-617 |
729 | 13.0 | -1.51±0.11 | 40.09 ± 8.76 |
| [18F]-Aoa-Glu3 Lys-CO-Glu-OH [18F]-Glu3 |
1503.5 | 12.39 | -2.40±0.13 | 72 ± 14.50 |
| [18F]-Aoa-Ahx Lys-CO-Glu-OH [18F]-Ahx |
506 | 13.3 | -1.11±0.10 | 94.10 ± 13.78 |
| [18F]-Aoa-Amb-Lys-CO-Glu-OH [18F]-Amb |
526 | 12.49 | -2.16±0.12 | 93.0 ± 15.04 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




