Submitted:
13 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Evidence Base and Variable Selection
- i.
- Guideline-based indicators drawn from the WHO IMCI algorithm and chart booklet (World Health Organization, 2014; Gera et al., 2016), which define the cardinal signs of “some” and “severe” dehydration.
- ii.
- Validated clinical scales, specifically the Gorelick and CDS instruments, that provide structured symptom scoring for pediatric dehydration (Jauregui et al., 2014).
- iii.
- Meta-analytic findings from Ogbolu et al. (2025) that quantified pooled diagnostic accuracy for each sign across pediatrics, adult, and elderly populations, complemented by studies addressing geriatric hydration screening and thirst/urine indices (Alsanie et al., 2022; Elliott et al., 2024; Parkinson et al., 2023).
- i.
- Appeared in at least two validated scales or guidelines, or
- ii.
- Demonstrated sensitivity ≥ 80 % and specificity ≥ 65 % in pooled or individual-study data.
2.3. Weight Assignment and Scoring Scheme
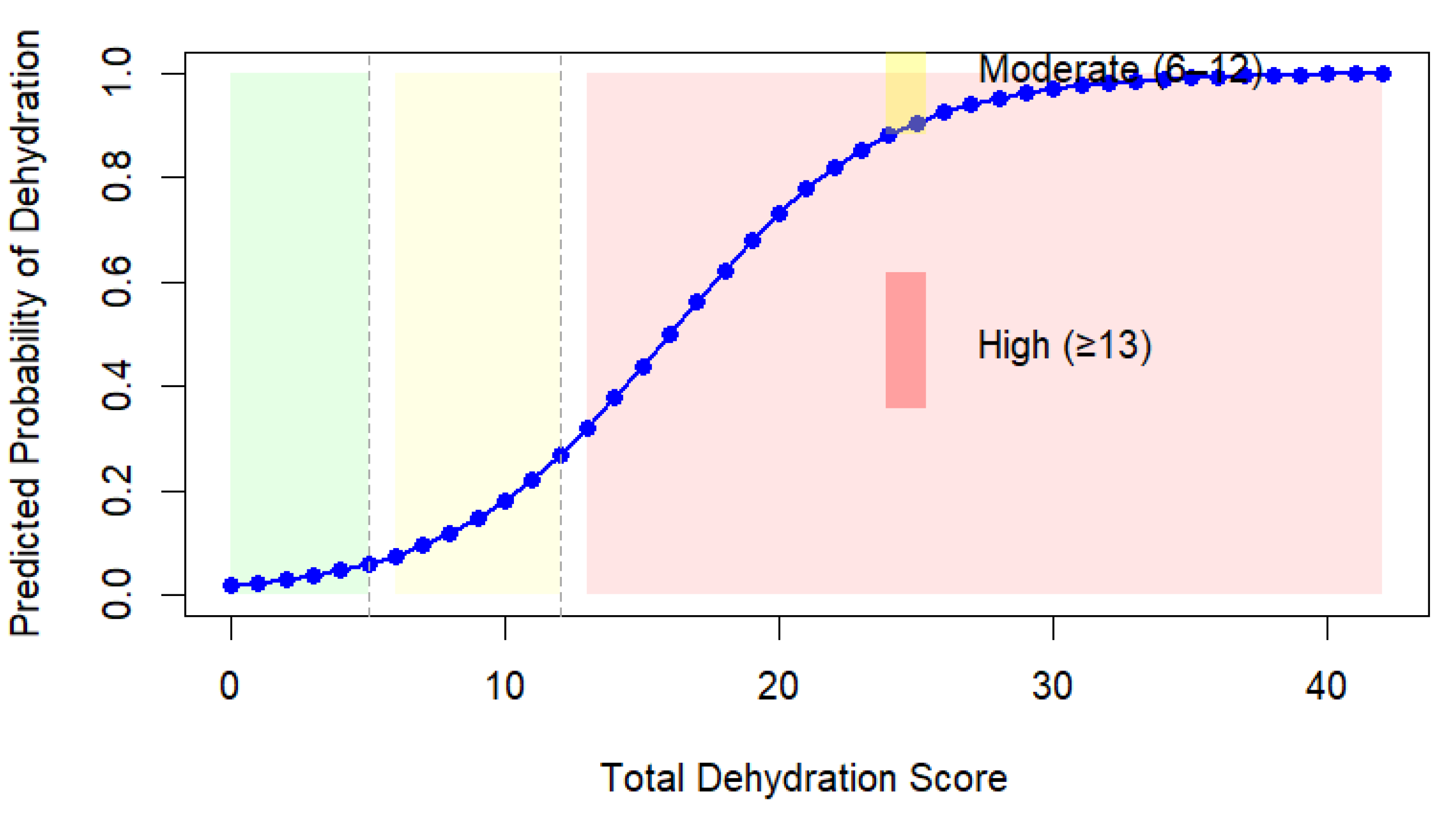
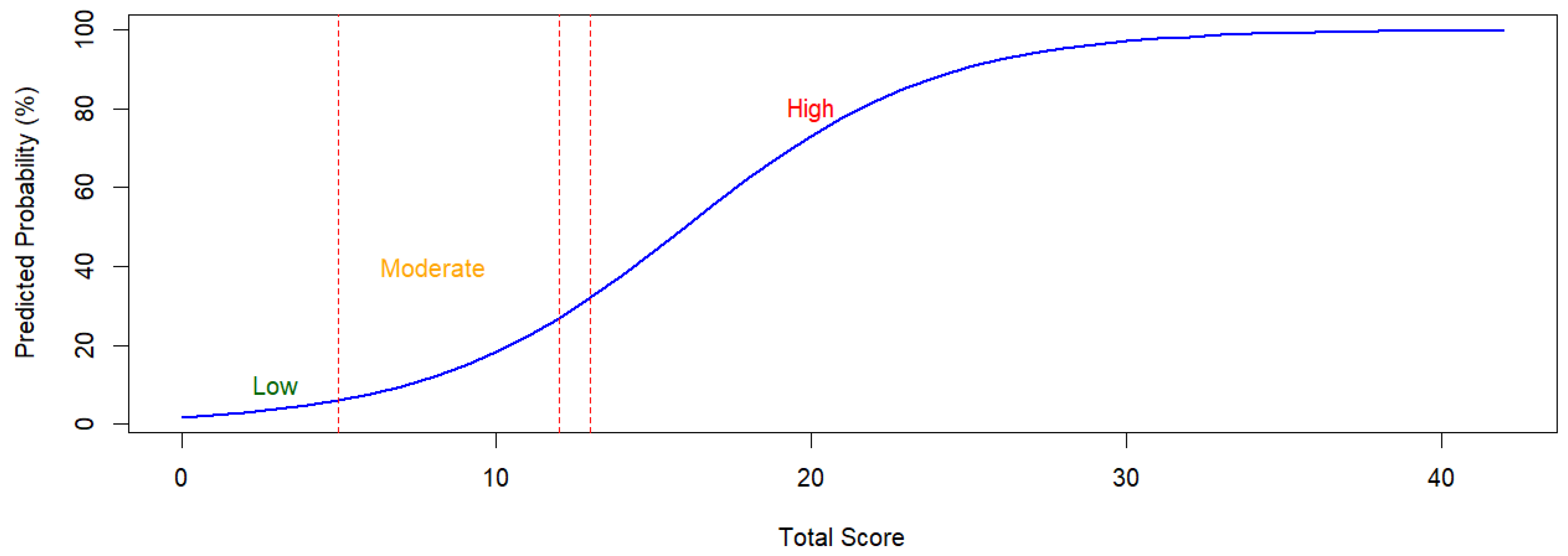
2.4. Risk Categorization
- i.
- Low risk: points
- ii.
- Moderate risk: points
- iii.
- High risk: ≥ 13 points
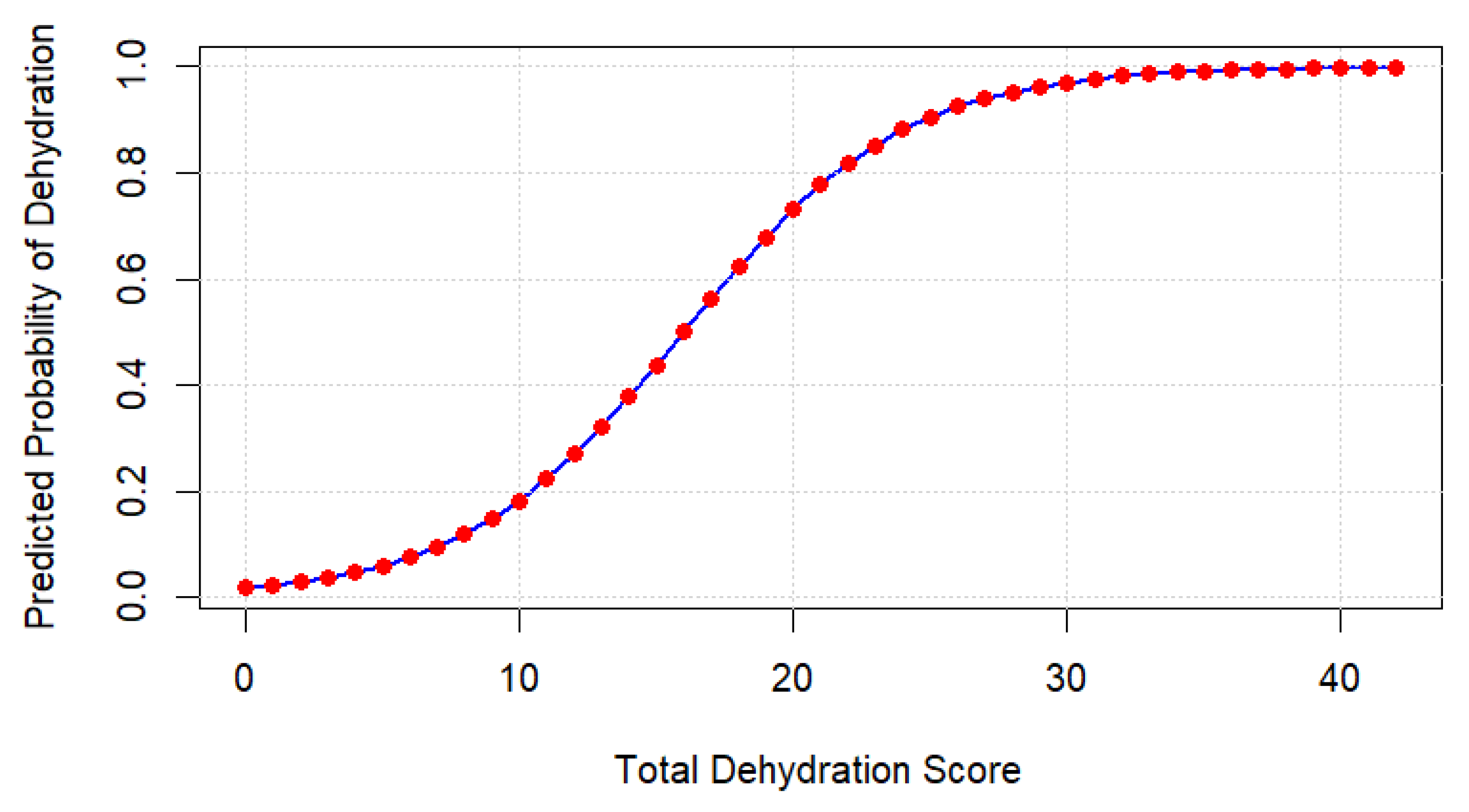
2.5. Probability Mapping
- i.
- A score of 5 corresponds to an estimated probability of
- ii.
- A score of
- iii.
- A score of and
- iv.
- A score approaches predicted likelihood of dehydration.
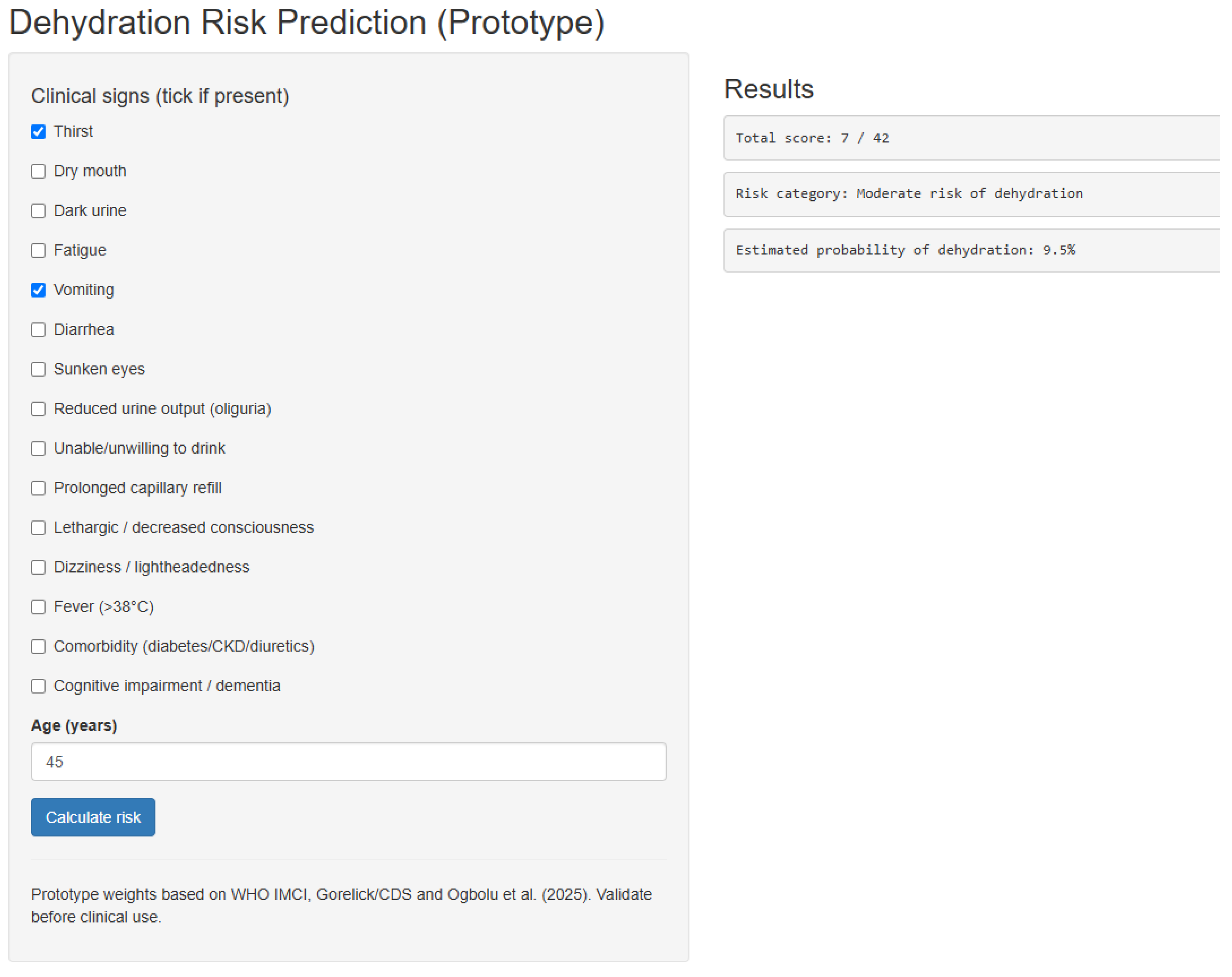
2.6. The Implementation of the MetaDehydrate Application
- i.
- Input panel: 16 predictor fields (checkboxes or drop-downs) allowing users to indicate the presence/absence of each sign or modifier (e.g., thirst, dark urine, age ≥ 65 years).
- ii.
- Computation module: executes internal functions to encode binary inputs, calculate the total weighted score, and apply the logistic equation defined in Section 2.5.
- iii.
- Output panel displays the resulting total score, predicted probability (%), and categorical risk level (low, moderate, high), accompanied by a color-coded progress bar for quick interpretation.
| Step | Input | Process | Output | Description |
| 1 | User-selected predictors (16 variables) | Binary encoding of presence = 1 / absence = 0 and application of assigned weights | Weighted total score | Captures clinical and demographic information from interface inputs |
| 2 | Total weighted score (0–42) | Logistic transformation using | Predicted probability (%) | Converts the discrete score to a continuous risk estimate |
| 3 | Probability (%) + score range | Risk-tier classification logic | Risk category (Low, Moderate, High) + visual gauge | Outputs text summary and color-coded indicator for clinical interpretation |
2.7. Prototype Testing and Usability Evaluation
| Domain | Metric / Test | Method | Result | Interpretation |
| Functional verification | Score calculation correctness | Comparison of system-generated scores with independent hand calculations across 120 simulated profiles | 120 / 120 exact matches (100%) | Confirms correct implementation of weighting and summation logic |
| Functional verification | Probability transformation correctness | Manual verification of logistic transformation outputs using predefined equation | 120 / 120 exact matches (100%) | Confirms correct execution of probability mapping formula |
| Functional verification | Risk-tier classification | Verification of categorical assignment against predefined score thresholds | 100% agreement | Confirms correct rule-based classification |
| Computational performance | Mean response time per session | Timestamp difference between input submission and output rendering | 0.18 s (SD ± 0.05 s) | Immediate feedback during user interaction |
| System stability | Error incidence | Monitoring of computational and interface errors over repeated runs | 0 errors across >500 runs | Indicates stable software behavior |
| Concurrency handling | Multi-user access | Simultaneous access via shinyapps.io cloud deployment | No crashes or delays observed | Demonstrates robustness under concurrent use |
| Cross-platform rendering | Device compatibility | Testing on desktop and mobile browsers | Stable rendering across devices | Confirms interface portability |
| Usability verification | Data entry and interpretation time | Timed pilot testing with users (n = 10) | < 1 minute per profile | Indicates efficient interaction flow |
| Usability verification | Interface clarity | Structured feedback from pilot users | Positive qualitative feedback | Supports interpretability of outputs |
3. Results
3.1. Model Structure and Scoring Behavior
3.2. Example Scenarios
3.3. Probability Mapping Results
3.4. Application Testing Outcomes – The MetaDehydrate
3.5. Model Performance and Internal Validation
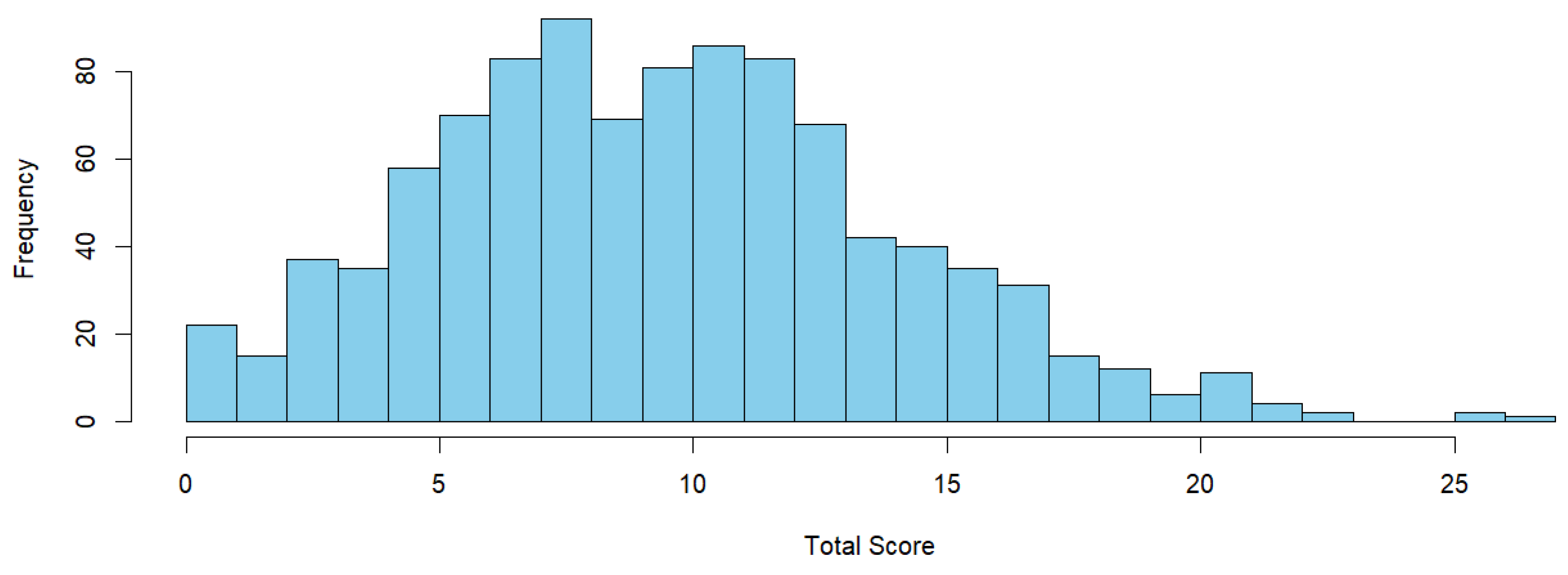
3.5.1. Overall Model Behaviour
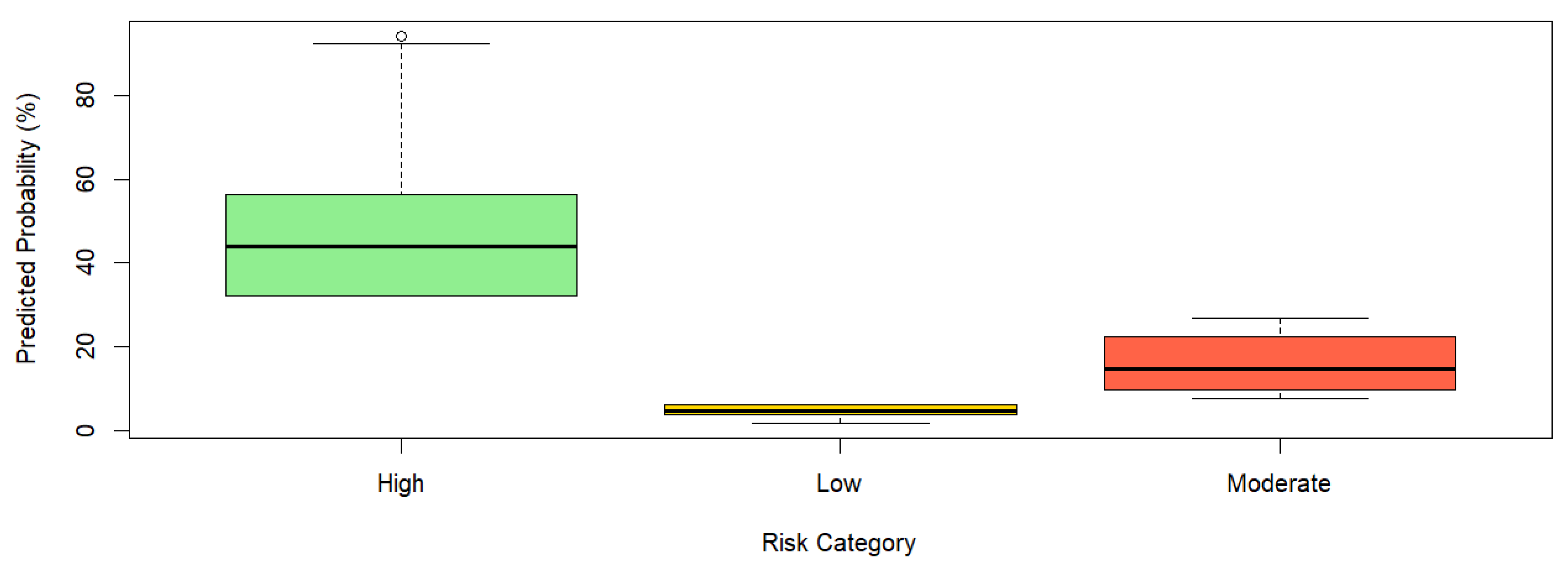
3.5.2. Descriptive Statistics by Risk Category
3.5.3. Variable-Level Trends and Probability Mapping
4. Discussion
4.1. Summary of Findings
4.2. Comparison with Existing Tools
- i.
- Evidence integration: It synthesizes pooled sensitivity, and specificity estimates from multi-age studies (Ogbolu et al., 2025) to assign transparent heuristic weights, rather than treating all symptoms equally.
- ii.
- Expanded scope: It also includes adult-related predictors, including dark urine, dizziness, fatigue, and cognitive impairments with geriatric hydration research underpinning the use of the tool (Mentes, 2006; Rosi et al., 2022).
- iii.
- Digital implementation: The R Shiny (the MetaDehydrate) application calculates the total score, estimated probability, and risk tier automatically, unlike paper-based scale, which reduces the inter-observer variation and can be integrated with mobile or clinical information systems.
4.3. Strengths
4.4. Limitations
4.5. Future Directions
5. Conclusions
Acknowledgments
References
- Alsanie, S., S. Lim, and S. A. Wootton. 2022. Detecting low-intake dehydration using bioelectrical impedance analysis in older adults in acute care settings: A systematic review. BMC Geriatrics 22, 954. [Google Scholar] [CrossRef] [PubMed]
- Gravel, J., S. Manzano, C. Guimont, L. Lacroix, A. Gervaix, and B. Bailey. 2010. Multicenter validation of the clinical dehydration scale for children. Archives de pediatrie: organe officiel de la Societe francaise de pediatrie 17, 12: 1645–1651. [Google Scholar] [CrossRef] [PubMed]
- Elliott, K. B., M. S. Keefe, J. J. S. Rolloque, N. C. Jiwan, R. A. Dunn, H. Y. Luk, and Y. Sekiguchi. 2024. Relationships between morning thirst and later hydration status and total water intake. Nutrients 16, 18: 3212. [Google Scholar] [CrossRef] [PubMed]
- Falszewska, A., H. Szajewska, and P. Dziechciarz. 2018. Diagnostic accuracy of three clinical dehydration scales: A systematic review. Archives of Disease in Childhood 103, 4: 383–388. [Google Scholar] [CrossRef] [PubMed]
- Marzuillo, P., G. Rivetti, A. Galeone, G. Capasso, P. Tirelli, A. Di Sessa, and F. Nunziata. 2024. Heart rate to identify non-febrile children with dehydration and acute kidney injury in emergency department: a prospective validation study. European Journal of Pediatrics 183, 11: 5043–5048. [Google Scholar] [CrossRef] [PubMed]
- Goldman, R. D., J. N. Friedman, and P. C. Parkin. 2008. Validation of the clinical dehydration scale for children with acute gastroenteritis. Pediatrics 122, 3: 545–549. [Google Scholar] [CrossRef] [PubMed]
- Jauregui, J., D. Nelson, E. Choo, M. Galicia, and K. J. S. Anand. 2014. External validation and comparison of three pediatric clinical dehydration scales. PLOS ONE 9, 5: e95739. [Google Scholar] [CrossRef] [PubMed]
- Bennett, B. L., T. Hew-Butler, M. H. Rosner, T. Myers, and G. S. Lipman. 2020. Wilderness medical society clinical practice guidelines for the management of exercise-associated hyponatremia: 2019 update. Wilderness & environmental medicine 31, 1: 50–62. [Google Scholar]
- Mentes, J. C. 2006. Oral hydration in older adults: Greater awareness is needed. American Journal of Nursing 106, 6: 40–49. [Google Scholar] [CrossRef] [PubMed]
- Ogbolu, M. O., O. D. Eniade, and M. Kozlovszky. 2025. Systematic review and meta-analysis of risk factors for dehydration and development of a predictive scoring system. Healthcare 13, 16: 1974. [Google Scholar] [CrossRef] [PubMed]
- Parkinson, E., L. Hooper, J. Fynn, S. H. Wilsher, T. Oladosu, F. Poland, and D. Bunn. 2023. Low-intake dehydration prevalence in non-hospitalised older adults: Systematic review and meta-analysis. Clinical Nutrition 42, 8: 1510–1520. [Google Scholar] [CrossRef] [PubMed]
- Rosi, I. M., R. Milos, I. Cortinovis, D. Laquintana, and L. Bonetti. 2022. Sensitivity and specificity of the new geriatric dehydration screening tool: An observational diagnostic study. Nutrition 101, 111695. [Google Scholar] [CrossRef] [PubMed]
- Sekiguchi, Y., C. L. Benjamin, C. R. Butler, M. C. Morrissey, E. M. Filep, R. L. Stearns, and D. J. Casa. 2022. Relationships between WUT (body weight, urine colour, and thirst level) criteria and urine indices of hydration status. Sports Health 14, 4: 566–574. [Google Scholar] [CrossRef] [PubMed]
- Keefe, M. S., H. Y. Luk, J. J. S. Rolloque, N. C. Jiwan, T. B. McCollum, and Y. Sekiguchi. 2024. The weight, urine colour and thirst Venn diagram is an accurate tool compared with urinary and blood markers for hydration assessment at morning and afternoon timepoints in euhydrated and free-living individuals. British Journal of Nutrition 131, 7: 1181–1188. [Google Scholar] [CrossRef] [PubMed]
- Keefe, M. S., H. Y. Luk, J. J. Rolloque, N. C. Jiwan, and Y. Sekiguchi. 2025. Hydration assessment in males and females using the WUT (weight, urine color, and thirst) Venn diagram compared to blood and urinary indices. Nutrients 17, 4: 689. [Google Scholar] [CrossRef] [PubMed]
- Stookey, J. D., S. A. Kavouras, H. Suh, and F. Lang. 2020. Underhydration is associated with obesity, chronic diseases, and death within 3 to 6 years in U.S. adults aged 51–70 years. Nutrients 12, 4: 905. [Google Scholar] [CrossRef] [PubMed]
- Gera, T., D. Shah, P. Garner, M. Richardson, and H. S. Sachdev. 2016. Integrated management of childhood illness (IMCI) strategy for children under five. Cochrane database of systematic reviews (6). [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. 2014. Integrated Management of Childhood Illness: Chart Booklet (In-service training) (WHO Library Cataloguing-in-Publication Data). Geneva, Switzerland: Author: Available online: https://cdn.who.int/media/docs/default-source/mca-documents/child/imci-integrated-management-of-childhood-illness/imci-in-service-training/imci-chart-booklet.pdf?sfvrsn=f63af425_1.
- Chang, W, J Cheng, J Allaire, C Sievert, B Schloerke, G Aden-Buie, Y Xie, J Allen, J McPherson, A Dipert, and B Borges. 2026. shiny: Web Application Framework for R. R package version 1.12.1.9000. Available online: https://shiny.posit.co/.
| Predictor | Clinical Definition / Description | Evidence Source(s) |
| Thirst | Patient reports desire to drink or requests fluids; early subjective sign of fluid deficit. | Ogbolu et al., 2025; Elliott et al., 2024 |
| Dry mouth / mucous membranes | Observable dryness of tongue, lips, or oral mucosa. | World Health Organization, 2014; Falszewska et al., 2018 |
| Dark urine | Concentrated urine (amber/brown) indicates reduced volume. | Ogbolu et al., 2025; World Health Organization, 2014 |
| Fatigue / weakness | Reduced energy or general tiredness associated with fluid deficit. | Ogbolu et al., 2025; Stookey et al., 2020 |
| Vomiting | ≥ 1 episode of emesis within 24 h; direct fluid loss. | World Health Organization, 2014; Gera et al., 2016 |
| Diarrhoea | ≥ 3 loose stools in 24 h or increased stool frequency. | World Health Organization, 2014 |
| Sunken eyes | Noticeable retraction of eyeballs within orbits. | World Health Organization, 2014 |
| Reduced urine output (oliguria) | Markedly decreased voiding frequency or < 0.5 mL/kg/h. | Ogbolu et al., 2025; Rosi et al., 2022 |
| Unable / unwilling to drink | Patients cannot or refuse to take fluids orally. | World Health Organization, 2014; Ogbolu et al., 2025 |
| Prolonged capillary refill time | > 2 s after nailbed or sternum pressure test. | Falszewska et al., 2018 |
| Lethargy / decreased consciousness | Drowsiness, slow responses, or unresponsiveness. | World Health Organization, 2014; Ogbolu et al., 2025 |
| Dizziness / light-headedness | Feeling of imbalance or presyncope on standing. | Ogbolu et al., 2025 |
| Fever (> 38 °C) | Axillary / oral temperature ≥ 38 °C causing increased fluid loss. | World Health Organization, 2014; Bennett et al., 2020 |
| Age ≥ 65 years | Older-adult modifier for diminished thirst perception. | Parkinson et al., 2023; Alsanie et al., 2022 |
| Comorbidity (e.g., diabetes, CKD) | Chronic conditions predispose dehydration. | Ogbolu et al., 2025; Stookey et al., 2020 |
| Cognitive impairment | Documented dementia or reduced mental status limiting fluid intake. | Mentes, 2006; Ogbolu et al., 2025 |
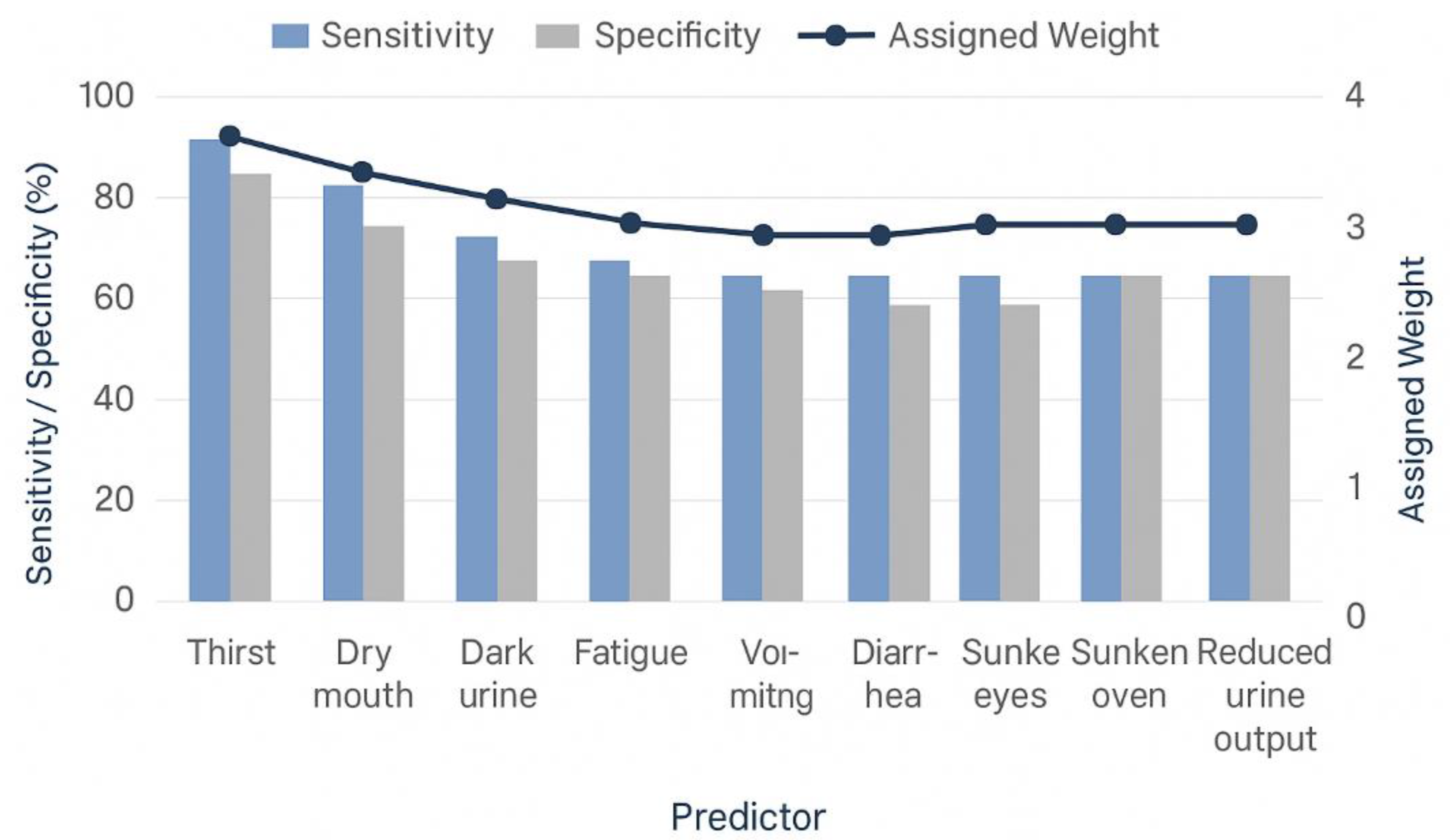
| Predictor | Assigned Weight (points) | Supporting Diagnostic Evidence (approx.) | Evidence Source(s) |
| Thirst | 4 | Sensitivity , Specificity | Ogbolu et al., 2025; Elliott et al., 2024; Keefe et al., 2025 |
| Dry mouth / mucous membranes | 3 | Sens. | Falszewska et al., 2018; World Health Organization, 2014. |
| Dark urine | 3 | Sens. | Ogbolu et al., 2025; Keefe et al., 2024 |
| Fatigue / weakness | 2 | Sens. | Ogbolu et al., 2025; Stookey et al., 2020 |
| Vomiting | 3 | Strong indicator of acute fluid loss | World Health Organization, 2014; Gera et al., 2016 |
| Diarrhoea | 3 | Primary cause of volume loss in IMCI criteria | World Health Organization, 2014. |
| Sunken eyes | 3 | Consistent sign in pediatric and elderly assessment | World Health Organization, 2014; Goldman et al., 2008 |
| Reduced urine output (oliguria) | 3 | Sens. ≈ 80 %, Spec. ≈ 70 % | Ogbolu et al., 2025; Rosi et al., 2022 |
| Unable / unwilling to drink | 4 | Critical IMCI “danger sign” | World Health Organization, 2014. |
| Prolonged capillary refill time | 3 | Sens. ≈ 75 %, Spec. ≈ 70 % | Falszewska et al., 2018 |
| Lethargy / decreased consciousness | 4 | Marker of severe volume depletion | World Health Organization, 2014; Ogbolu et al., 2025 |
| Dizziness / light-headedness | 2 | Symptom of orthostatic hypovolemia | Ogbolu et al., 2025; Mentes, 2006 |
| Fever (> 38 °C) | 2 | Contributor to insensible fluid loss | World Health Organization, 2014; Bennett et al., 2020 |
| Age ≥ 65 years | 1 | Risk modifier for reduced thirst drive | Parkinson et al., 2023; Alsanie et al., 2022 |
| Comorbidity (e.g., diabetes, CKD) | 1 | Chronic risk for fluid imbalance | Stookey et al., 2020 |
| Cognitive impairment | 1 | Limits self-hydration ability | Mentes, 2006 |
| Maximum Total Score | 42 |
| Case ID | Key Predictors Present | Total Score (/42) | Predicted Probability (%) | Risk Category | Interpretation |
| Case A – Low risk | Fatigue only (no major signs) | 4 | 15 % | Low | Likely well hydrated; monitor fluid intake and re-assess if symptoms persist. |
| Case B – Moderate risk | Thirst + vomiting + dark urine | 10 | 45 % | Moderate | Suggests mild-to-moderate dehydration; initiate oral rehydration and observe. |
| Case C – High risk | Inability to drink + lethargy + diarrhoea + sunken eyes | 25 | 89 % | High | Indicates severe dehydration; requires urgent clinical evaluation and possible IV rehydration. |
| Risk Category | Total Score (Mean ± SD) | Predicted Probability (Mean ± SD) |
| Low | 3.49 ± 1.51 | 4.44 ± 1.40 |
| Moderate | 9.06 ± 1.99 | 16.07 ± 6.49 |
| High | 15.69 ± 2.68 | 47.69 ± 14.87 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




