Submitted:
12 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Case Presentation
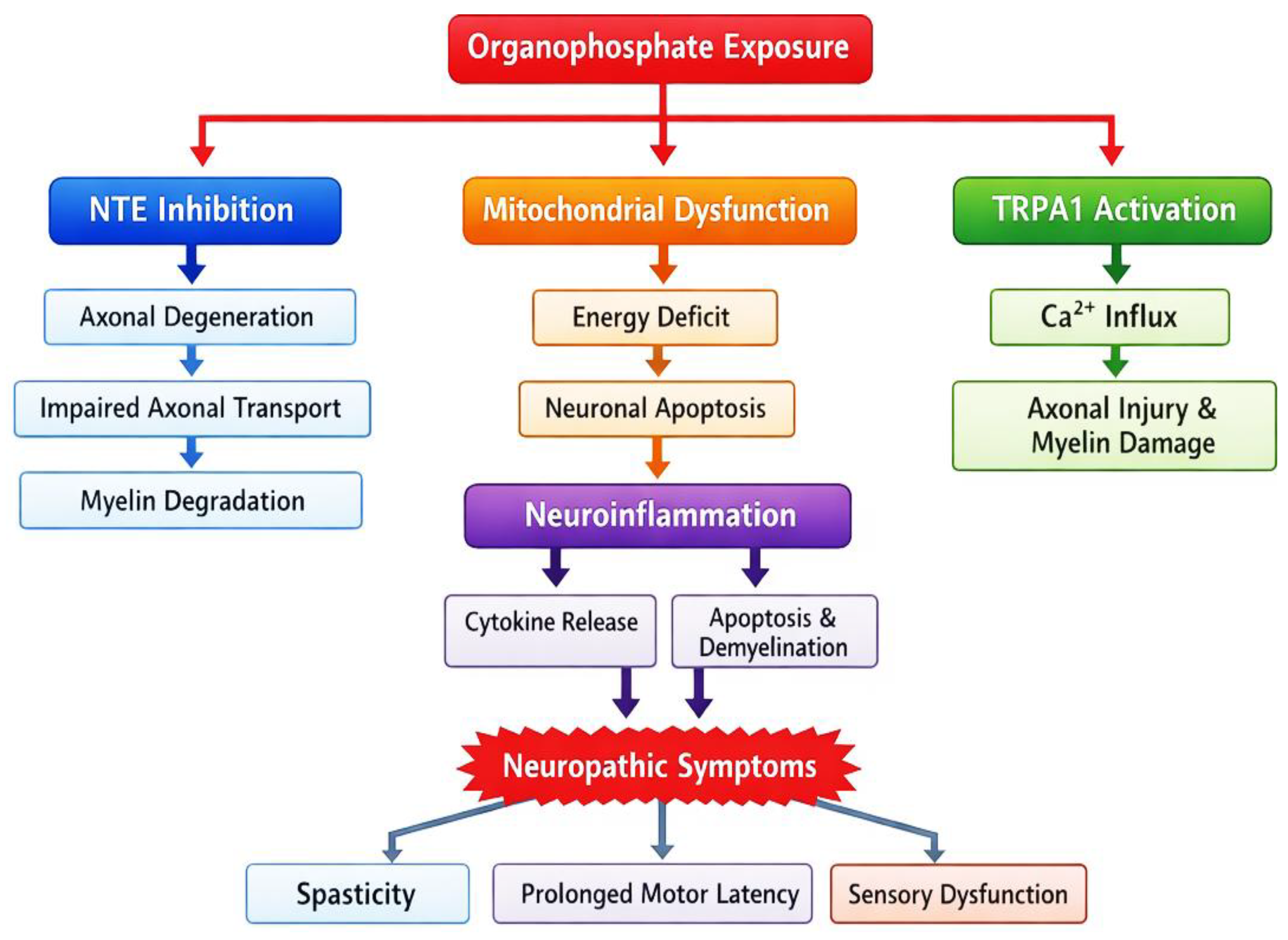
Discussion
Conclusion
- ▪
- Chronic low-level occupational exposure to organophosphates can cause OPIDN without a preceding acute cholinergic crisis.
- ▪
- Diagnosis requires a high index of suspicion, a detailed occupational history, and appropriate investigations (NCS/EMG and serum cholinesterase levels).
- ▪
- Management is primarily supportive, including physical therapy, pain management, and spasticity control.
- ▪
- Off-label therapies such as methylprednisolone may have a role but are not considered standard of care.
List of Abbreviations
Ethics Approval and Consent to Participate
Consent for Publication
Availability of Data and Materials
Competing Interests
Author Contributions
Funding
Acknowledgements
Author Information
References
- Ansal Mundu P, Kumar M, Satapathy RP, Mitra JK. Organophosphate-induced delayed neuropathy: a case report. Int J Contemp Med Res. 2016;3(8):2289–91.
- Rao BR, Mohanty L, Kampali H, et al. Organophosphate-induced delayed neuropathy: a rare case presentation. J Integr Med Res. 2024; 2:33–6.
- Viswanath A, Barman A, Sahoo J, et al. Organophosphorus poisoning induced delayed neurotoxicity: a report of two cases. Spinal Cord Ser Cases. 2023;9(1):54.
- Goncharov NV, Belinskaia DA, Avdonin PV. Organophosphate-induced pathology: mechanisms of development, principles of therapy, and features of experimental studies. J Evol Biochem Physiol. 2023;59(5):1756–96.
- Ganie SY, Javaid D, Hajam YA, et al. Mechanisms and treatment strategies of organophosphate pesticide-induced neurotoxicity in humans: a critical appraisal. Toxicol. 2022; 472:153181.
- Tessema RA, Nagy K, Ádám B. Occupational and environmental pesticide exposure and associated health risks among pesticide applicators and non-applicator residents in rural Ethiopia. Front Public Health. 2022; 10:1017189.
- Desye B, Tesfaye AH, Daba C, et al. Pesticide safe use practice and acute health symptoms, and associated factors among farmers in developing countries: a systematic review and meta-analysis of epidemiological evidence. BMC Public Health. 2024; 24:3313.
- Patel A, Chavan G, Nagpal AK. Navigating the neurological abyss: a comprehensive review of organophosphate poisoning complications. Cureus. 2024;16(2).
- Chhabra J, Oberoi A, Pandita N, et al. The enigma of delayed neurotoxicity in organophosphate poisoning: a case report of clinical presentation with normal MRI findings. Cureus. 2024;16(6): e62877.
- Emerick GL, DeOliveira GH, dos Santos AC, et al. Mechanisms for consideration for intervention in the development of organophosphorus-induced delayed neuropathy. Chem Biol Interact. 2012;199(3):177-84.
- Gautam S, Sapkota S, Ojha R, et al. Delayed myelopathy after organophosphate intoxication: a case report. SAGE Open Med Case Rep. 2022; 10:1–4.
- Kobayashi S, Okubo R, Ugawa Y. Delayed polyneuropathy induced by organophosphate poisoning. Intern Med. 2017; 56:1903–5.
- Acharya H, Kelaiya A, Singel V, et al. Organophosphorus agent-induced delayed neuropathy: a case report. Int J Res Med Sci. 2016; 4:674–6.
- Prasai P, Thakur J, Karki KT. Organophosphorus poisoning induced delayed poly-axonal motor neuropathy: a case report. Egneuro. 2022;4(1):27–9.
- Akçay Y, Yalbuzdağ Ş, İnce B, et al. Organophosphate-induced delayed neuropathy: a case report. Turk J Phys Med Rehabil. 2017; 63:88–91.
- Chowdhury MMH, Masum A, Kawnayn G. A case of organophosphate-induced delayed motor neuropathy without any sensory involvement. BIRDEM Med J. 2024;14(1):49–52.
- Perry J, Cotton J, Rahman MA, et al. Organophosphate exposure and the chronic effects on farmers: a narrative review. Rural Remote Health. 2020;20(1):206–22.
- Vasconcellos LF, Leite AC, Nascimento OJ. Organophosphate-induced delayed neuropathy: case report. Arq Neuropsiquiatr. 2002;60(4):1003–7.
- Bereda G. Organophosphate poisoning: insights from a case report of acute cholinergic syndrome. Clin Case Rep. 2025;13(10): e71183.
- Bereda G. Poisoning by organophosphate pesticides: a case report. Cureus. 2022; 14(10): e29842.
- Bereda G. Acute Cardiac Arrhythmias in Organophosphate Poisoning: From Bradycardia to Atrial Fibrillation. Indian J Clin Cardiol. 2025; 6(4):398-404.
| Witness observations of the symptoms and progression of the disease | ||
|---|---|---|
| Year 1–5 | Year 6–8 | Year 9 |
| ⁃Occasional headaches ⁃Dizziness ⁃Mild fatigue |
⁃Tingling & numbness in hands and feet ⁃Muscle cramps & occasional tremors ⁃Difficulty gripping objects & weakness in the legs |
⁃Progressive muscle weakness, worsened over months ⁃Gait disturbances (difficulty walking & frequent tripping) ⁃Burning pain in legs, worse at night ⁃Mild memory & concentration problems |
| Nerve conduction study (NCS) | ||||
|---|---|---|---|---|
| Right median | Right ulnar | Left tibial | ||
| Motor nerve conduction (MNC) | CMAP amplitude (mV) | 5.9 | 7.8 | 3.7 |
| Normal value | >4 | >4 | >4 | |
| Nerve conduction velocity (ms) | 57 | 54 | 46 | |
| Normal value | >50 | >50 | >40 | |
| Distal latency (ms) | 3.9 | 2.7 | 4.6 | |
| Normal value | <4.4 | <3.3 | <5.8 | |
| Sensory nerve conduction (SNC) | SNAP amplitude (µV) | 49.3 | 34 | 18.8 |
| Normal value | >20 | >17 | >6 | |
| Distal latency (ms) | 4.8 | 5.1 | 9.9 | |
| Normal Value | 2–3 | 2–3 | 2–3 | |
| Differential Diagnosis | Rationale for Consideration | Exclusion criteria |
|---|---|---|
| Nutritional Neuropathy | Diabetes & B12/folate deficiency can cause peripheral neuropathy | Normal fasting glucose, HbA1c, vitamin B12, & folate levels |
| Hereditary Neuropathies | Distal weakness, foot drop, & sensory deficits | No family history; adult-onset; & NCS pattern inconsistent with hereditary demyelinating neuropathy |
| CNS Demyelination | Weakness, spasticity, & sensory changes | MRI brain & spinal cord normal; symptoms predominantly peripheral |
| Stroke | Acute/subacute limb weakness | MRI spine showed only mild spinal cord atrophy |
| Motor Neuron Disease | Progressive weakness, & muscle atrophy | Sensory involvement present; reflexes largely preserved; pattern inconsistent with motor neuron disease |
| Chronic Inflammatory Demyelinating Polyneuropathy | Progressive motor & sensory deficits | NCS shows mixed axonal features but lacks a classic CIDP pattern; & no relapsing course |
| Toxin-Induced Neuropathy | Lead, arsenic, or other toxins can cause neuropathy | No history of relevant exposure; blood tests unremarkable; & symptom pattern aligns with OP exposure |
| Measure | Baseline | Week 4 | Week 8 | Week 12 | Week 16 | Color/Representation | Notes |
|---|---|---|---|---|---|---|---|
| Proximal muscle strength | 2/5 | 3/5 | 3/5 | 4/5 | 4/5 | Solid brown line | Objective clinical assessment |
| Distal muscle strength | 2–3/5 | 3/5 | 3–4/5 | 4/5 | 4/5 | Solid blue line | Objective clinical assessment |
| Spasticity | 2–3 | 2–3 | 2 | 1–2 | 1 | Broken brown line | Clinician-assessed; partly subjective |
| Neuropathic pain (VAS) | 7 | 6 | 5 | 4 | 3 | Broken blue line | Patient-reported; subjective measure |
| Overall functional recovery | Low | Moderate | Moderate | Moderate | High | Shaded area (combined gains) | Represents combined improvement in strength, spasticity, and pain |
| Findings from earlier published studies | ||||||||
|---|---|---|---|---|---|---|---|---|
| References | Age/gender | OPs type | History of ACS | History of long-term exposure | Time frame of OPIDN occurrence | Clinical Features | Clinical interventions | |
| Kobayashi, et al 2017 [12] | 89-year-old male farmer | 200 mL of a 50% Diptelex solution (trichlorfon) | Yes | No | Unknown | Rapidly progressive distal weakness and sensory disturbance | Gastric lavage, intravenous atropine, and pralidoxime | |
| Akçay, 2017 [15] | 27-year-old-male | 40 mL chlorpyrifos | Yes | No | 1 month | Motor axonal neuropathy involves the long axons of the peripheral nervous system. | A cold pack, Baclofen, and Botulinum toxin | |
| Rao et al., 2024 [2] | 36-year-old female | 50% chlorpyrifos | Yes | No | 20 days | Severe axonal motor neuropathy affecting both upper and lower limbs | Atropine infusion, pralidoxime, and antibiotics | |
| Gautam et al., 2022 [11] | 16-year-old male | 50% chlorpyrifos and 5% cypermethrin | Yes | No | 6 weeks | Normal motor and sensory amplitudes, no muscle atrophy, but spasticity was present in both the lower limbs. | IV methylprednisolone, calcium, and vitamin B1 supplements, and regular extensive physiotherapy. | |
| Acharya et al., 2016 [13] | 40-year-old male | High suspicion of poisoning by a food adulterant | No | No | 20 days | Difficulty in walking with inability to flex the foot and toes in both feet (“foot drop”) | Physiotherapy | |
| Chhabra et al., 2024 [9] | A 28-year-old man | 80 ml of OP (chlorpyrifos) | Yes | No | 5 weeks | Weakness in bilateral lower limbs, difficulty walking, and an abnormal gait | Gabapentin and nortriptyline, physiotherapy, and a foot splint. | |
| Viswanath, 2023 [3] | 20-year-old male | Organophosphorus pesticides | Yes | No | 3 weeks | Weakness and tightness in both lower limbs, and difficulty in walking, with distal weakness in both lower limbs | Cold packs, Baclofen, and Botulinum toxin A | |
| 34-year-old male | Organophosphorus pesticides | Yes | No | 2 weeks | Tightness in both lower extremities, difficulty walking, and painful spasms in sleep | Galvanic intermittent electrical stimulation, cold packs, Baclofen, and Botulinum toxin A injection | ||
| Chowdhury MMH et al, 2024 [16] | 29-year-old male | Chlorpyrifos | Yes | No | 4 weeks | Progressive lower-limb weakness and motor axonal neuropathy | Supplemental oxygen, hydrocortisone, nebulization, atropine pralidoxime, IV fluid, thiamine, and physiotherapy | |
| Ansal Mundu et al., 2016 [1] | 22-year-old male | Chlorpyriphos 200 ml | Yes | No | 3 weeks | Tingling sensation in the right foot, weakness in the right lower limb, and difficulty in walking | Gastric lavage, inj Atropine, pralidoxime, and oxygen therapy | |
| Prasai Pet al., 2022 [14] | 23-year-old lady | Organophosphorus compound | Yes | No | 4 weeks | Distal weakness in the lower limbs and motor neuropathy | Steroids and physiotherapy | |
| Vasconcellos et al., 2002 [18] | 39-year-old female | Dichlorvos-based insecticide | Yes | No | 2 weeks | Cramping calf pain, hyperesthesia in the plantar area, and distal weakness in the lower and upper limbs | Amitriptyline, carbamazepine, capsaicin, thiamin, and physiotherapy | |
| Finding of this manuscript | ||||||||
| Bereda. 2025 | 44-year-old male pesticide applicator | Many (e.g., chlorpyrifos, malathion, & diazinon) | No | Yes | 9 years | Axonal degeneration with demyelination in the lower limbs, as evidenced by prolonged distal motor latencies, reduced conduction velocity, and diminished sensory nerve action potentials. | Gabapentin, baclofen, antioxidant supplementation with vitamin E, vitamin C, methylprednisolone, and Physiotherapy | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




