Submitted:
12 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
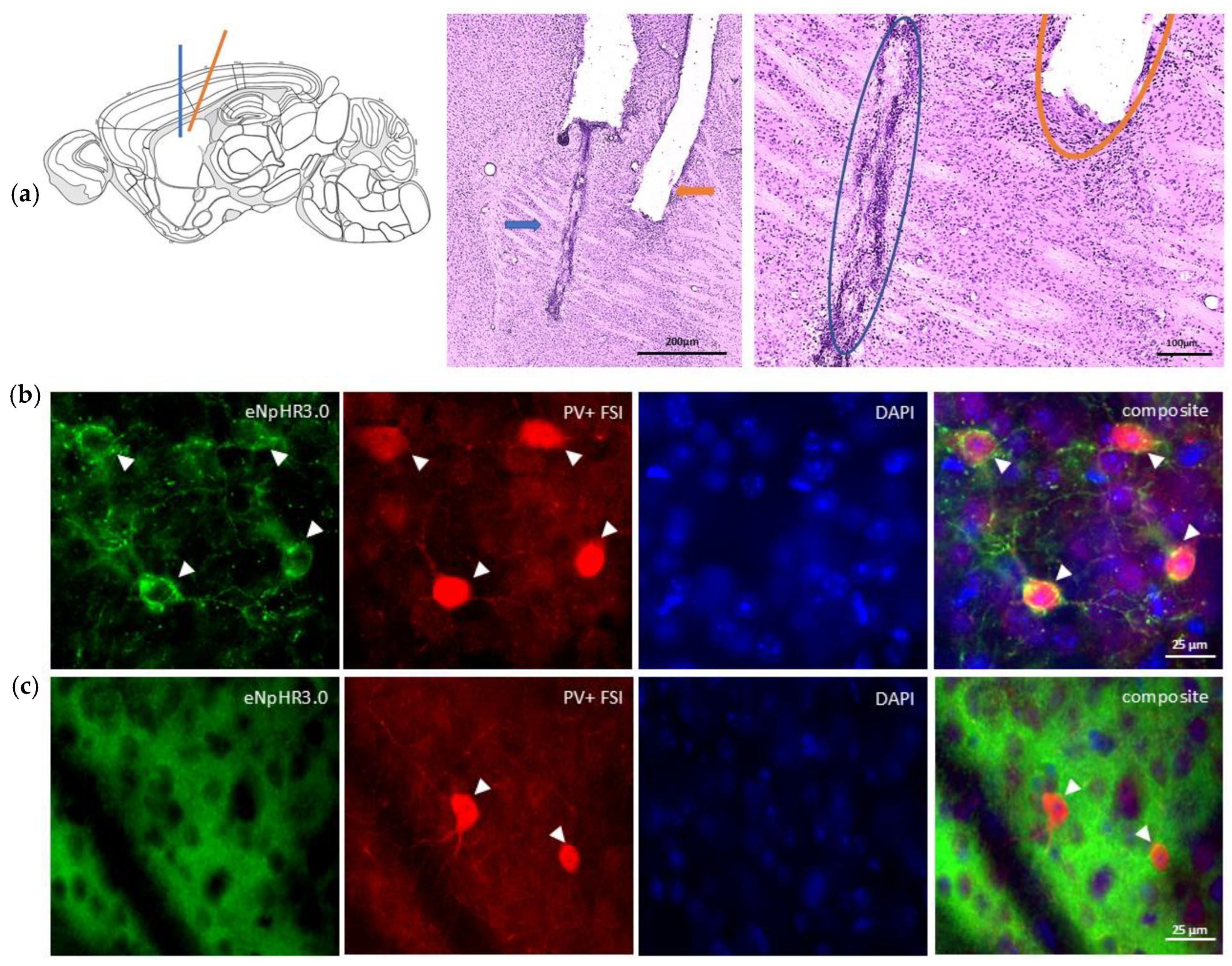
2.1. Verifying Localization and Expression
2.2. Extracellular Neurotransmitter and Neuromodulator Levels
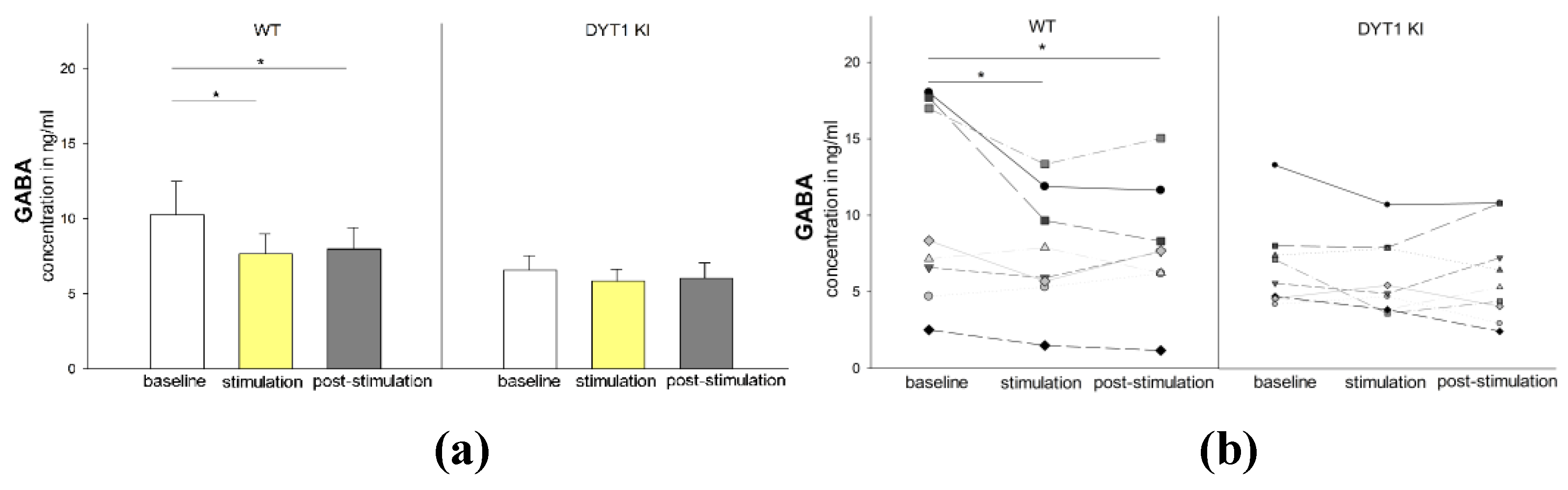
2.2.1. GABA Levels Decreased Upon Optogenetic Inhibition of PV+ FSI in WT but not in DYT1 KI
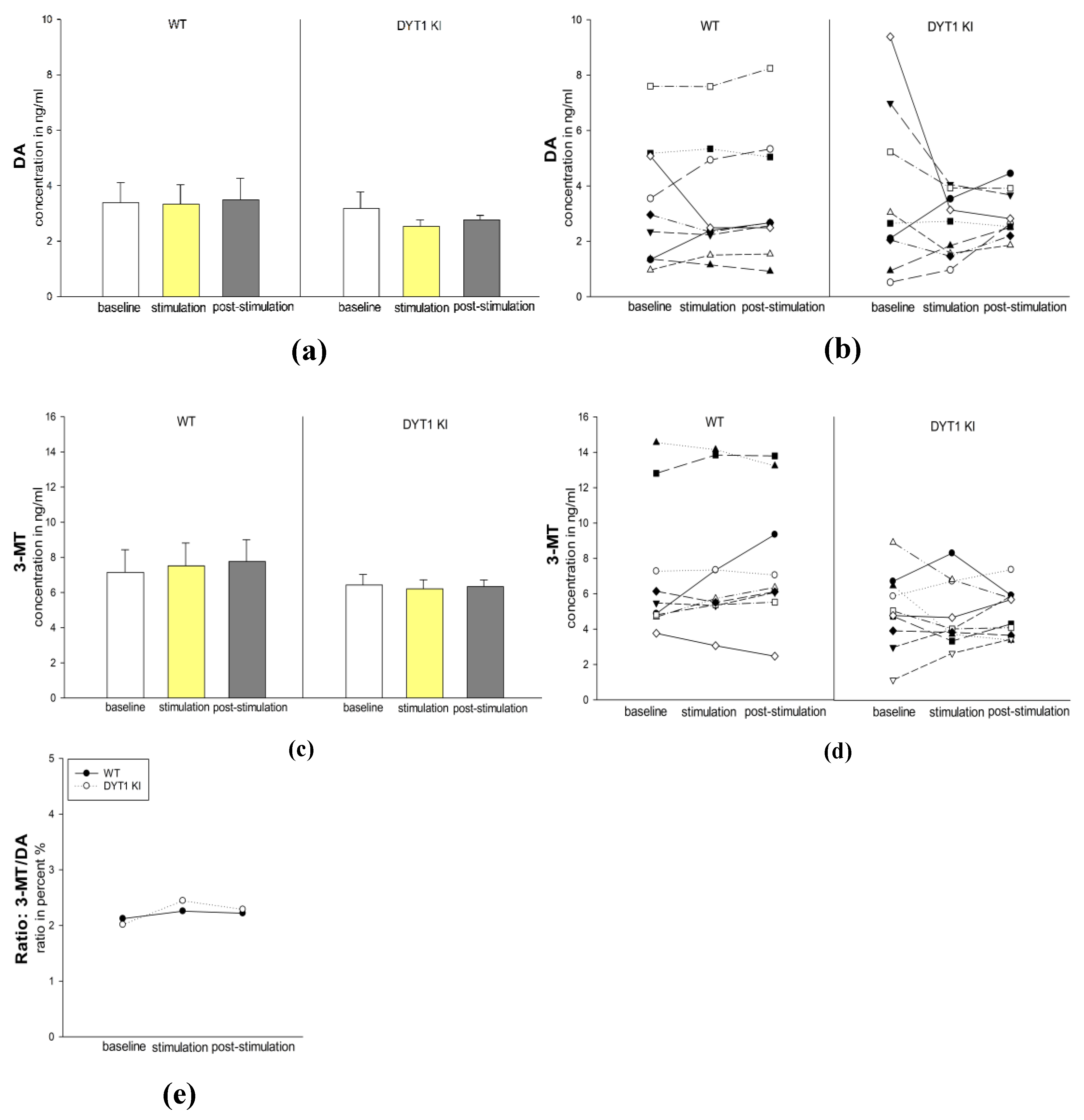
2.2.2. DA and 3-MT Levels Remained Stable Across Stimulation
2.2.3. Extracellular Adenosine (ADE) Levels Decreased with a Delay During Inhibition of PV+ FSI in DYT1 KI
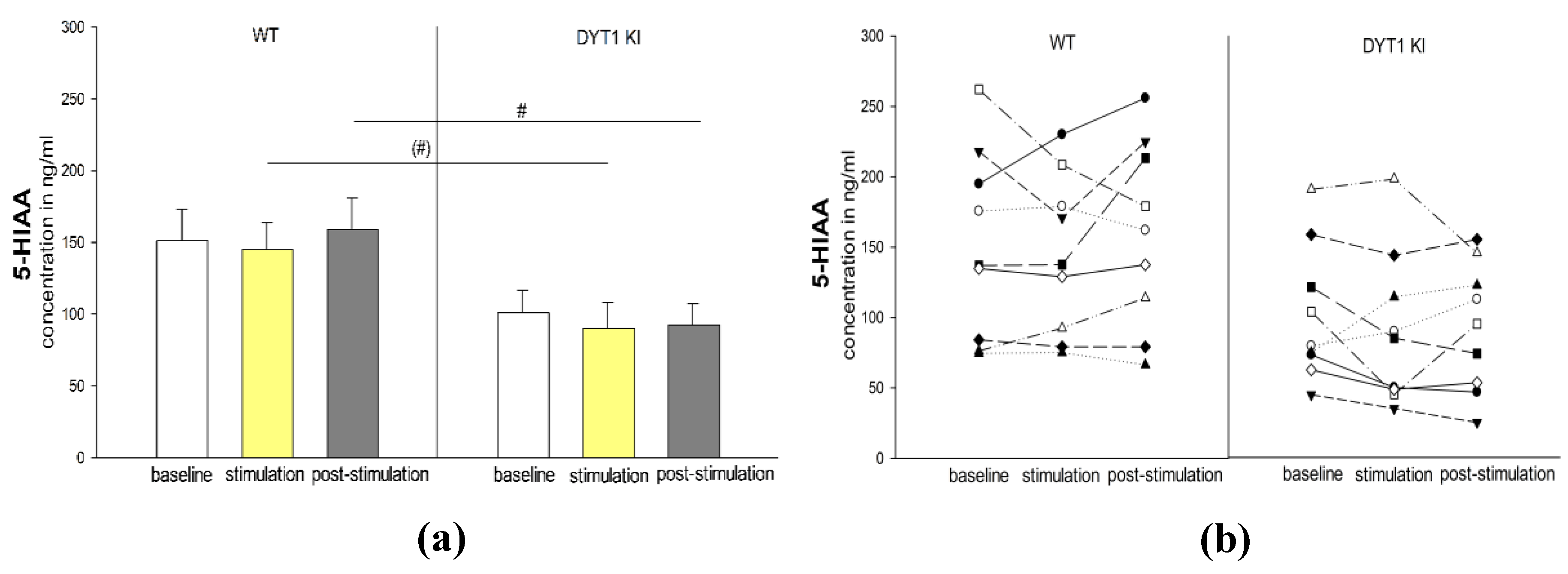
2.2.4. 5-HIAA Levels Were Selectively Reduced in DYT1 KI Mice During and After Optogenetic Inhibition of PF+ FSI
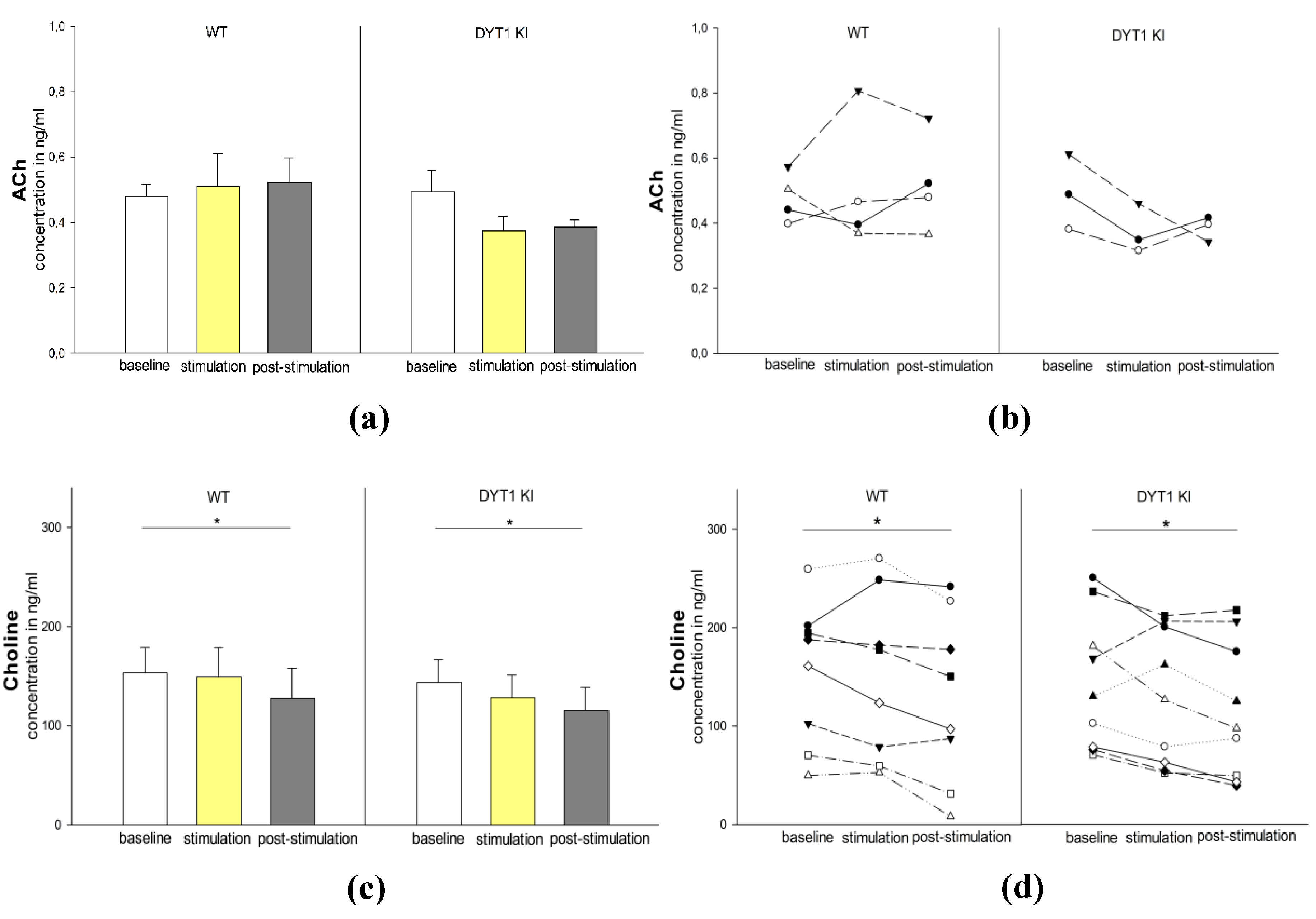
2.2.5. ACh Levels Were Unaffected by Optogenetic Inhibition, Choline Decreased Post-Stimulation in Both Genotypes
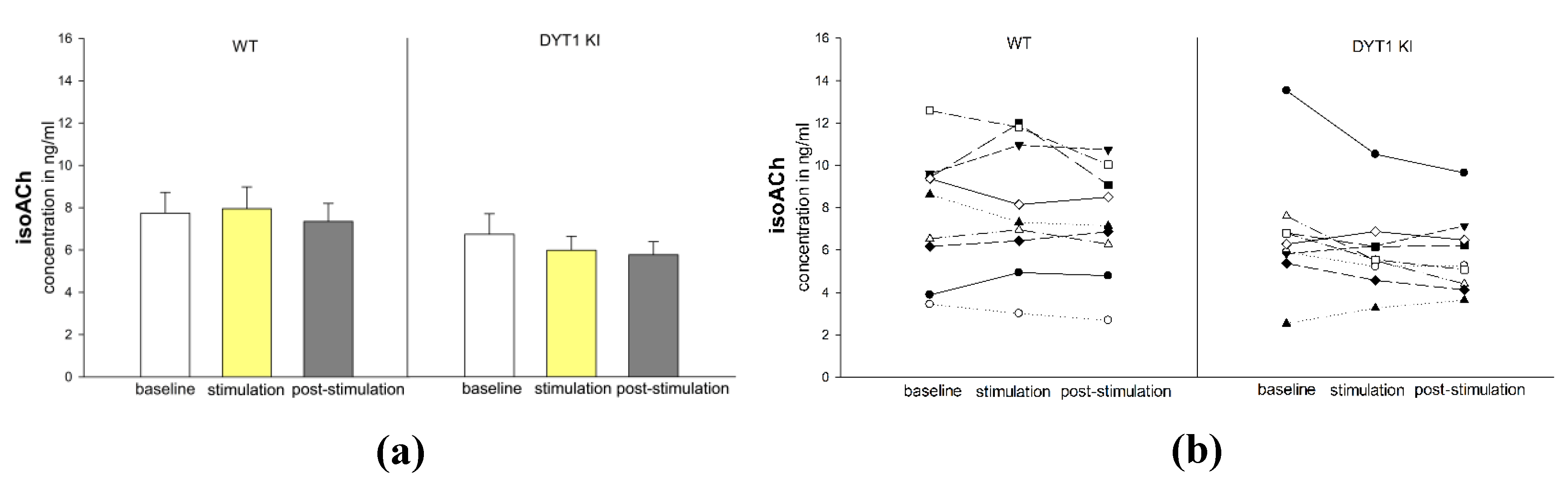
2.2.6. Iso-Acetylcholine (isoACh) Tended to be Lower in DYT1 KI

3. Discussion
4. Materials and Methods
4.1. Animals
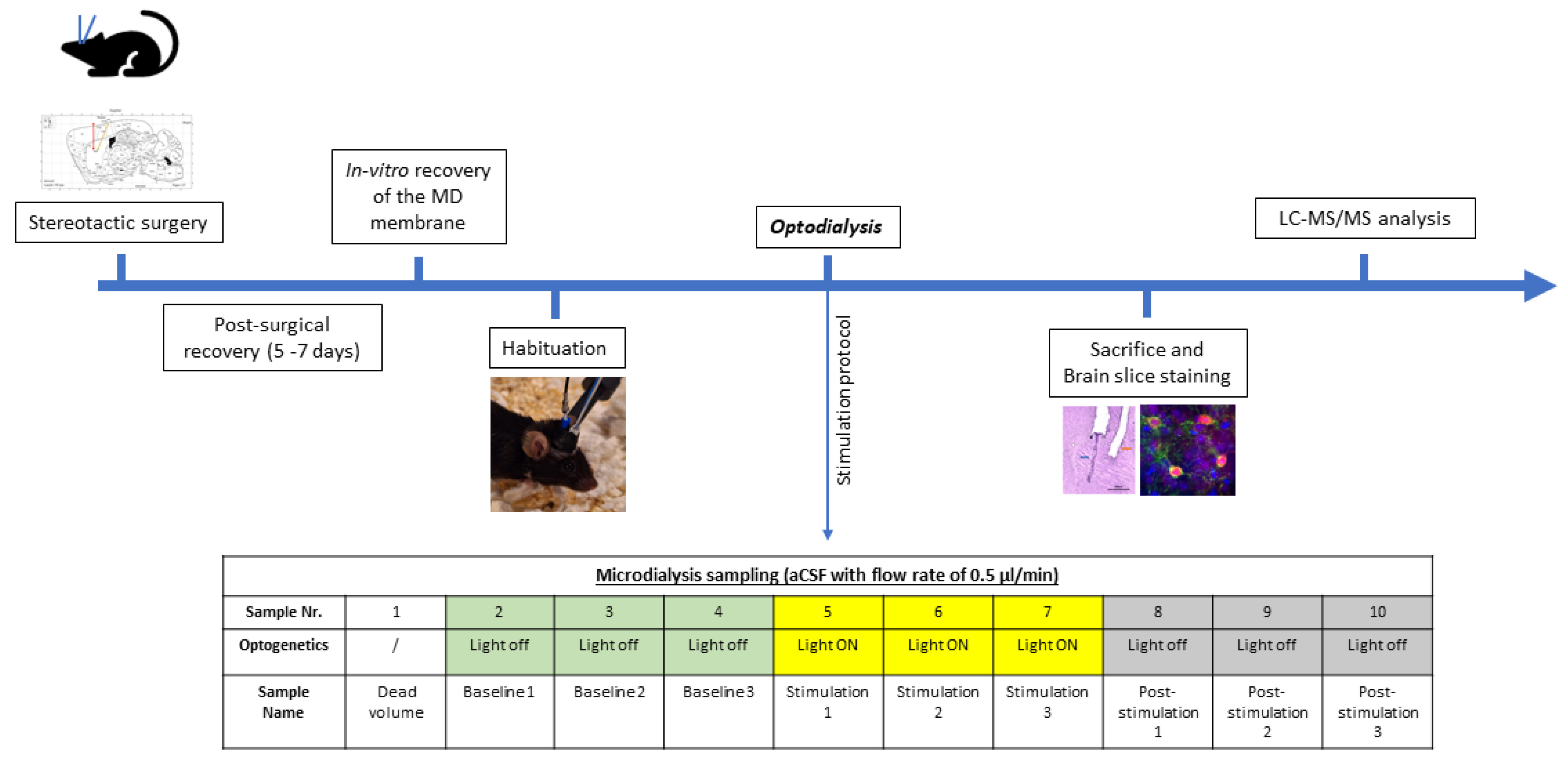
4.2. Stereotaxic Surgery
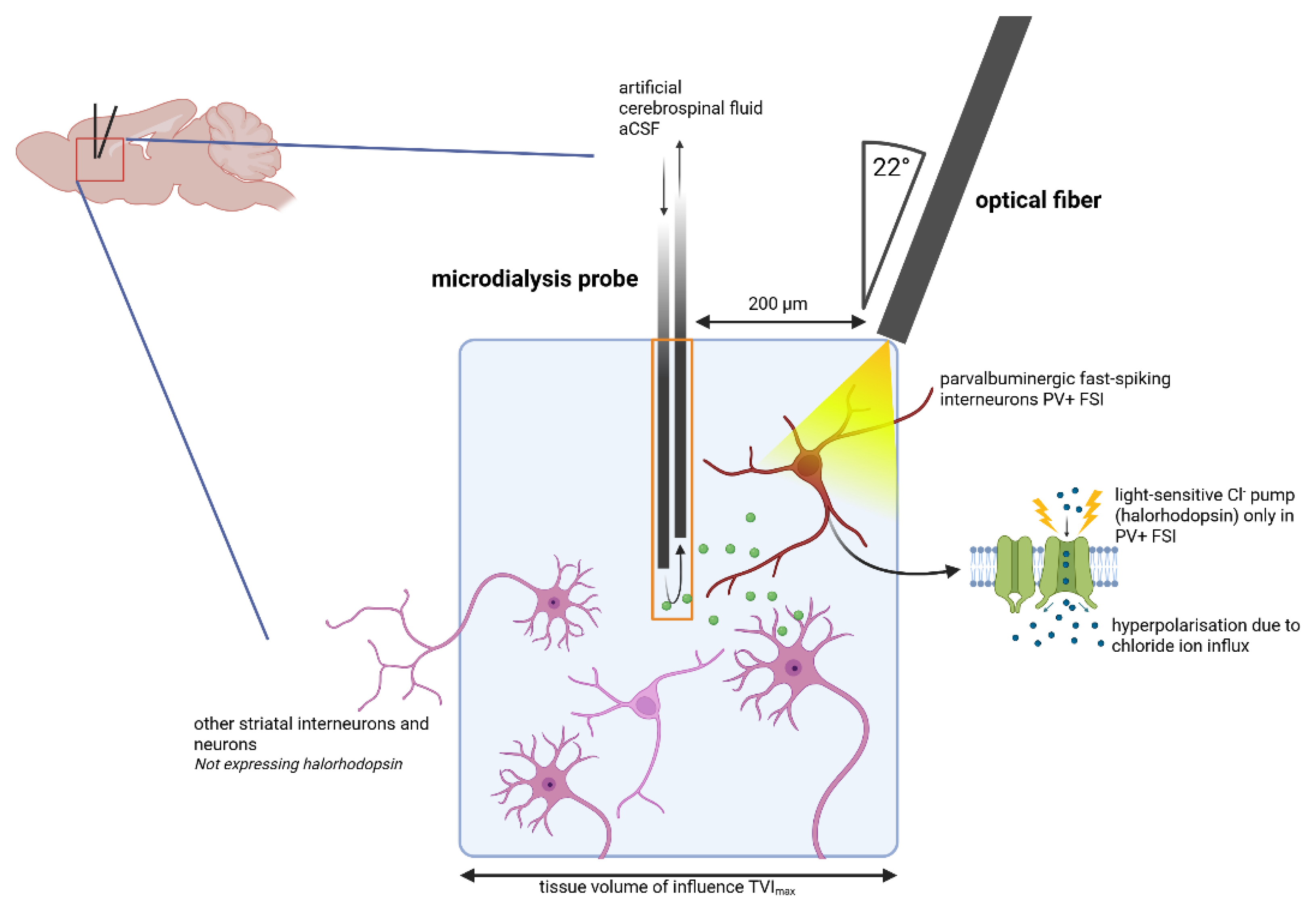
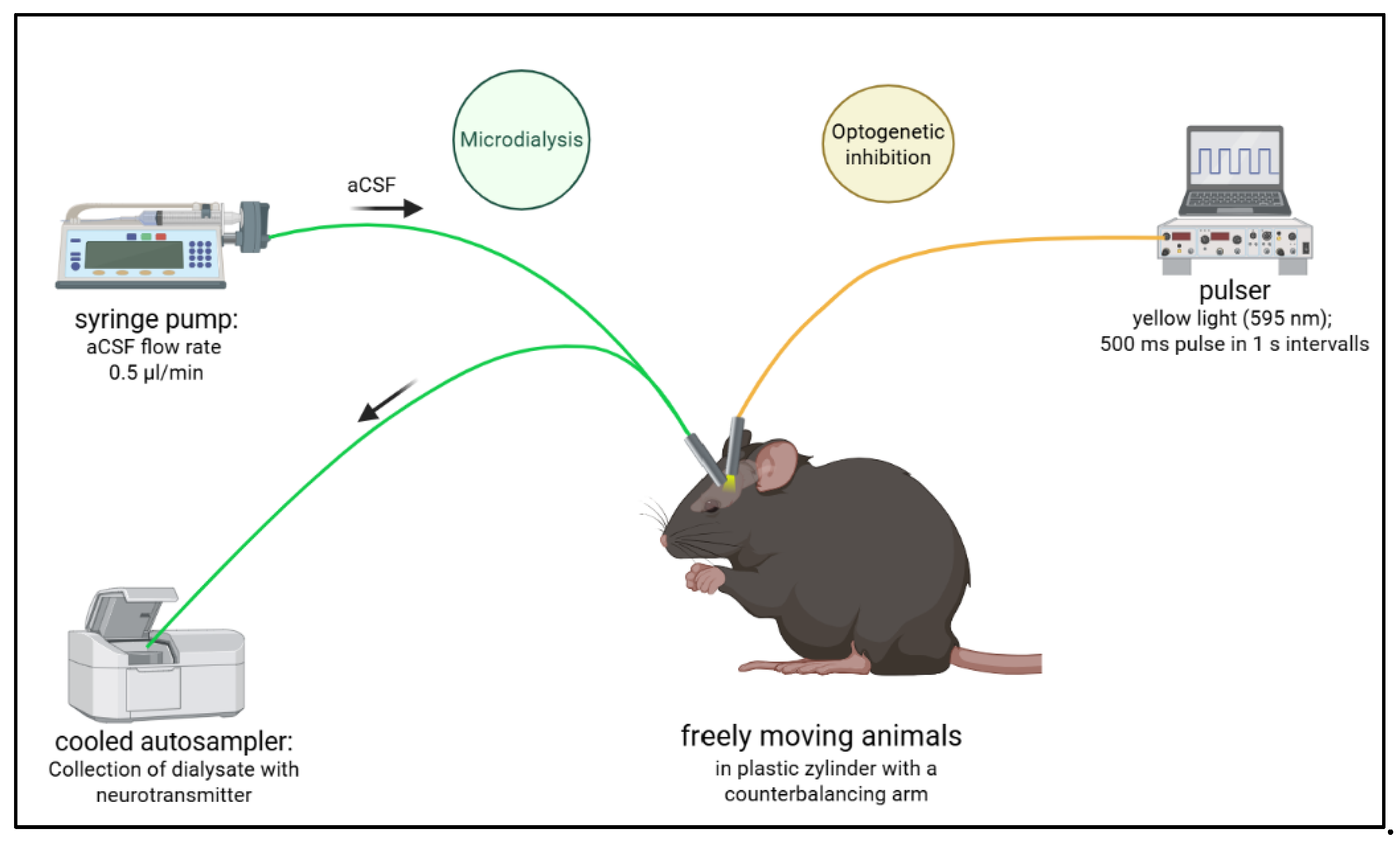
4.3. Optodialysis Procedure
4.4. LC-MS/MS Analysis
4.5. Brain Slice Staining and Image Acquisition
4.6. Statistics
4.6.1. Power Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 3-MT 5-HIAA |
3-methoxytyramine 5-hydroxyindoleacetic acid |
| ACh | acetylcholine |
| ADE | adenosine |
| DA | dopamine |
| DYT1 KI | knock-in mouse model for DYT1 dystonia |
| GABA | γ-aminobutyric acid |
| isoACh | γ-butyrobetaine (former iso-acetylcholine), isobaric compound of ACh |
| MD | microdialysis |
| PV+ FSI | Parvalbumin-positive fast-spiking interneurons |
References
- Albanese, A.; Bhatia, K.; Bressman, S.B.; Delong, M.R.; Fahn, S.; Fung, V.S.C.; Hallett, M.; Jankovic, J.; Jinnah, H.A.; Klein, C.; et al. Phenomenology and classification of dystonia: a consensus update. Mov. Disord. 2013, 28, 863–873. [Google Scholar] [CrossRef]
- Domingo, A.; Yadav, R.; Ozelius, L.J. Isolated dystonia: clinical and genetic updates. J Neural Transm 2021, 128, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Si, Z.; Wang, L.; Zhang, L. DYT-TOR1A dystonia: an update on pathogenesis and treatment. Front. Neurosci. 2023, 17, 1216929. [Google Scholar] [CrossRef] [PubMed]
- Goodchild, R.E.; Kim, C.E.; Dauer, W.T. Loss of the dystonia-associated protein torsinA selectively disrupts the neuronal nuclear envelope. Neuron 2005, 48, 923–932. [Google Scholar] [CrossRef] [PubMed]
- Scarduzio, M.; Hess, E.J.; Standaert, D.G.; Eskow Jaunarajs, K.L. Striatal Synaptic Dysfunction in Dystonia and Levodopa-Induced Dyskinesia. Neurobiol. Dis. 2022, 166, 105650. [Google Scholar] [CrossRef]
- Goodchild, R.E.; Dauer, W.T. The AAA+ protein torsinA interacts with a conserved domain present in LAP1 and a novel ER protein. J. Cell Biol. 2005, 168, 855–862. [Google Scholar] [CrossRef]
- Song, C.-H.; Fan, X.; Exeter, C.J.; Hess, E.J.; Jinnah, H.A. Functional analysis of dopaminergic systems in a DYT1 knock-in mouse model of dystonia. Neurobiol. Dis. 2012, 48, 66–78. [Google Scholar] [CrossRef]
- Richter, F.; Gerstenberger, J.; Bauer, A.; Liang, C.-C.; Richter, A. Sensorimotor tests unmask a phenotype in the DYT1 knock-in mouse model of dystonia. Behav. Brain Res. 2017, 317, 536–541. [Google Scholar] [CrossRef]
- Maltese, M.; Stanic, J.; Tassone, A.; Sciamanna, G.; Ponterio, G.; Vanni, V.; Martella, G.; Imbriani, P.; Bonsi, P.; Mercuri, N.B.; et al. Early structural and functional plasticity alterations in a susceptibility period of DYT1 dystonia mouse striatum. Elife 2018, 7. [Google Scholar] [CrossRef]
- Downs, A.M.; Fan, X.; Donsante, C.; Jinnah, H.A.; Hess, E.J. Trihexyphenidyl rescues the deficit in dopamine neurotransmission in a mouse model of DYT1 dystonia. Neurobiol. Dis. 2019, 125, 115–122. [Google Scholar] [CrossRef]
- Downs, A.M.; Fan, X.; Kadakia, R.F.; Donsante, Y.; Jinnah, H.A.; Hess, E.J. Cell-intrinsic effects of TorsinA(ΔE) disrupt dopamine release in a mouse model of TOR1A dystonia. Neurobiol. Dis. 2021, 155, 105369. [Google Scholar] [CrossRef]
- Scarduzio, M.; Zimmerman, C.N.; Jaunarajs, K.L.; Wang, Q.; Standaert, D.G.; McMahon, L.L. Strength of cholinergic tone dictates the polarity of dopamine D2 receptor modulation of striatal cholinergic interneuron excitability in DYT1 dystonia. Exp. Neurol. 2017, 295, 162–175. [Google Scholar] [CrossRef]
- Tepper, J.M.; Tecuapetla, F.; Koós, T.; Ibáñez-Sandoval, O. Heterogeneity and diversity of striatal GABAergic interneurons. Front. Neuroanat. 2010, 4, 150. [Google Scholar] [CrossRef]
- Wegman, E.; Wosiski-Kuhn, M.; Luo, Y. The dual role of striatal interneurons: circuit modulation and trophic support for the basal ganglia. Neural Regen. Res. 2024, 19, 1277–1283. [Google Scholar] [CrossRef]
- Gernert, M.; Hamann, M.; Bennay, M.; Löscher, W.; Richter, A. Deficit of striatal parvalbumin-reactive GABAergic interneurons and decreased basal ganglia output in a genetic rodent model of idiopathic paroxysmal dystonia. J. Neurosci. 2000, 20, 7052–7058. [Google Scholar] [CrossRef]
- Gittis, A.H.; Leventhal, D.K.; Fensterheim, B.A.; Pettibone, J.R.; Berke, J.D.; Kreitzer, A.C. Selective inhibition of striatal fast-spiking interneurons causes dyskinesias. J. Neurosci. 2011, 31, 15727–15731. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Sun, J.; Moss, J.; Wen, Z.; Sun, G.J.; Hsu, D.; Zhong, C.; Davoudi, H.; Christian, K.M.; Toni, N.; et al. Parvalbumin interneurons mediate neuronal circuitry-neurogenesis coupling in the adult hippocampus. Nat Neurosci 2013, 16, 1728–1730. [Google Scholar] [CrossRef]
- Bode, C.; Richter, F.; Spröte, C.; Brigadski, T.; Bauer, A.; Fietz, S.; Fritschy, J.-M.; Richter, A. Altered postnatal maturation of striatal GABAergic interneurons in a phenotypic animal model of dystonia. Exp. Neurol. 2017, 287, 44–53. [Google Scholar] [CrossRef]
- Schulz, A.; Richter, F.; Richter, A. In vivo optogenetic inhibition of striatal parvalbumin-reactive interneurons induced genotype-specific changes in neuronal activity without dystonic signs in male DYT1 knock-in mice. J. Neurosci. Res. 2023, 101, 448–463. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, T.; Morigaki, R.; Hayasawa, H.; Koyama, H.; Oda, T.; Miyake, K.; Takagi, Y. Striatal parvalbumin interneurons are activated in a mouse model of cerebellar dystonia. Dis. Model. Mech. 2024, 17. [Google Scholar] [CrossRef] [PubMed]
- Hamann, M.; Richter, F.; Richter, A. Acute effects of neurosteroids in a rodent model of primary paroxysmal dystonia. Horm. Behav. 2007, 52, 220–227. [Google Scholar] [CrossRef]
- Deubner, J.; Coulon, P.; Diester, I. Optogenetic approaches to study the mammalian brain. Curr. Opin. Struct. Biol. 2019, 57, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Anderzhanova, E.; Wotjak, C.T. Brain microdialysis and its applications in experimental neurochemistry. Cell Tissue Res. 2013, 354, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Helmschrodt, C.; Becker, S.; Perl, S.; Schulz, A.; Richter, A. Development of a fast liquid chromatography-tandem mass spectrometry method for simultaneous quantification of neurotransmitters in murine microdialysate. Anal. Bioanal. Chem. 2020, 412, 7777–7787. [Google Scholar] [CrossRef]
- Becker, S.; Schulz, A.; Kreyer, S.; Dreßler, J.; Richter, A.; Helmschrodt, C. Sensitive and simultaneous quantification of 16 neurotransmitters and metabolites in murine microdialysate by fast liquid chromatography-tandem mass spectrometry. Talanta 2023, 253, 123965. [Google Scholar] [CrossRef] [PubMed]
- Tepper, J.M. GABAergic Interneurons of the Striatum. In Handbook of Basal Ganglia Structure and Function; Elsevier, 2010, 2016; pp. pp 151–166. ISBN 9780123747679. [Google Scholar]
- Sciamanna, G.; Bonsi, P.; Tassone, A.; Cuomo, D.; Tscherter, A.; Viscomi, M.T.; Martella, G.; Sharma, N.; Bernardi, G.; Standaert, D.G.; et al. Impaired striatal D2 receptor function leads to enhanced GABA transmission in a mouse model of DYT1 dystonia. Neurobiol. Dis. 2009, 34, 133–145. [Google Scholar] [CrossRef]
- Fino, E.; Vandecasteele, M.; Perez, S.; Saudou, F.; Venance, L. Region-specific and state-dependent action of striatal GABAergic interneurons. Nat. Commun. 2018, 9, 3339. [Google Scholar] [CrossRef]
- Holly, E.N.; Davatolhagh, M.F.; España, R.A.; Fuccillo, M.V. Striatal low-threshold spiking interneurons locally gate dopamine. Curr. Biol. 2021, 31, 4139–4147.e6. [Google Scholar] [CrossRef]
- Roberts, B.M.; Lopes, E.F.; Cragg, S.J. Axonal Modulation of Striatal Dopamine Release by Local γ-Aminobutyric Acid (GABA) Signalling. Cells 2021, 10. [Google Scholar] [CrossRef]
- Granata, A.; Schiavo, G.; Warner, T.T. TorsinA and dystonia: from nuclear envelope to synapse. J. Neurochem. 2009, 109, 1596–1609. [Google Scholar] [CrossRef]
- Martella, G.; Maltese, M.; Nisticò, R.; Schirinzi, T.; Madeo, G.; Sciamanna, G.; Ponterio, G.; Tassone, A.; Mandolesi, G.; Vanni, V.; et al. Regional specificity of synaptic plasticity deficits in a knock-in mouse model of DYT1 dystonia. Neurobiol. Dis. 2014, 65, 124–132. [Google Scholar] [CrossRef]
- Tepper, J.M.; Koós, T.; Ibanez-Sandoval, O.; Tecuapetla, F.; Faust, T.W.; Assous, M. Heterogeneity and Diversity of Striatal GABAergic Interneurons: Update 2018. Front. Neuroanat. 2018, 12, 91. [Google Scholar] [CrossRef]
- Taverna, S.; Ilijic, E.; Surmeier, D.J. Recurrent collateral connections of striatal medium spiny neurons are disrupted in models of Parkinson’s disease. J. Neurosci. 2008, 28, 5504–5512. [Google Scholar] [CrossRef]
- Dang, M.T.; Yokoi, F.; McNaught, K.S.P.; Jengelley, T.-A.; Jackson, T.; Li, J.; Li, Y. Generation and characterization of Dyt1 DeltaGAG knock-in mouse as a model for early-onset dystonia. Exp. Neurol. 2005, 196, 452–463. [Google Scholar] [CrossRef] [PubMed]
- Eskow Jaunarajs, K.L.; Bonsi, P.; Chesselet, M.F.; Standaert, D.G.; Pisani, A. Striatal cholinergic dysfunction as a unifying theme in the pathophysiology of dystonia. Prog. Neurobiol. 2015, 127-128, 91–107. [Google Scholar] [CrossRef] [PubMed]
- Martella, G.; Tassone, A.; Sciamanna, G.; Platania, P.; Cuomo, D.; Viscomi, M.T.; Bonsi, P.; Cacci, E.; Biagioni, S.; Usiello, A.; et al. Impairment of bidirectional synaptic plasticity in the striatum of a mouse model of DYT1 dystonia: role of endogenous acetylcholine. Brain 2009, 132, 2336–2349. [Google Scholar] [CrossRef]
- Sebastião, A.M.; Ribeiro, J.A. Adjusting the brakes to adjust neuronal activity: Adenosinergic modulation of GABAergic transmission. Neuropharmacology 2023, 236, 109600. [Google Scholar] [CrossRef]
- Mori, A. How do adenosine A2A receptors regulate motor function? Parkinsonism Relat. Disord. 2020, 80 Suppl 1, S13–S20. [Google Scholar] [CrossRef]
- Cunha, R.A. Adenosine as a neuromodulator and as a homeostatic regulator in the nervous system: different roles, different sources and different receptors. Neurochem. Int. 2001, 38, 107–125. [Google Scholar] [CrossRef] [PubMed]
- Schiffmann, S.N.; Fisone, G.; Moresco, R.; Cunha, R.A.; Ferré, S. Adenosine A2A receptors and basal ganglia physiology. Prog. Neurobiol. 2007, 83, 277–292. [Google Scholar] [CrossRef]
- Yang, C.; Franciosi, S.; Brown, R.E. Adenosine inhibits the excitatory synaptic inputs to Basal forebrain cholinergic, GABAergic, and parvalbumin neurons in mice. Front. Neurol. 2013, 4, 77. [Google Scholar] [CrossRef]
- Wu, Z.; Cui, Y.; Wang, H.; Wu, H.; Wan, Y.; Li, B.; Wang, L.; Pan, S.; Peng, W.; Dong, A.; et al. Neuronal activity-induced, equilibrative nucleoside transporter-dependent, somatodendritic adenosine release revealed by a GRAB sensor. Proc. Natl. Acad. Sci. U. S. A. 2023, 120, e2212387120. [Google Scholar] [CrossRef]
- Boison, D. Adenosine as a neuromodulator in neurological diseases. Curr. Opin. Pharmacol. 2008, 8, 2–7. [Google Scholar] [CrossRef]
- Chen, J.-F.; Lee, C.; Chern, Y. Adenosine receptor neurobiology: overview. Int. Rev. Neurobiol. 2014, 119, 1–49. [Google Scholar] [CrossRef]
- Taiwo, F.T.; Adebayo, P.B. Neuroimaging findings in DYT1 dystonia and the pathophysiological implication: A systematic review. Brain Behav. 2023, 13, e3023. [Google Scholar] [CrossRef]
- D’Angelo, V.; Giorgi, M.; Paldino, E.; Cardarelli, S.; Fusco, F.R.; Saverioni, I.; Sorge, R.; Martella, G.; Biagioni, S.; Mercuri, N.B.; et al. A2A Receptor Dysregulation in Dystonia DYT1 Knock-Out Mice. Int. J. Mol. Sci. 2021, 22. [Google Scholar] [CrossRef] [PubMed]
- Richter, A.; Hamann, M. Effects of adenosine receptor agonists and antagonists in a genetic animal model of primary paroxysmal dystonia. Br. J. Pharmacol. 2001, 134, 343–352. [Google Scholar] [CrossRef]
- Balcioglu, A.; Kim, M.-O.; Sharma, N.; Cha, J.-H.; Breakefield, X.O.; Standaert, D.G. Dopamine release is impaired in a mouse model of DYT1 dystonia. J. Neurochem. 2007, 102, 783–788. [Google Scholar] [CrossRef] [PubMed]
- Page, M.E.; Bao, L.; Andre, P.; Pelta-Heller, J.; Sluzas, E.; Gonzalez-Alegre, P.; Bogush, A.; Khan, L.E.; Iacovitti, L.; Rice, M.E.; et al. Cell-autonomous alteration of dopaminergic transmission by wild type and mutant (DeltaE) TorsinA in transgenic mice. Neurobiol. Dis. 2010, 39, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Bert, L.; Favale, D.; Jego, G.; Greve, P.; Guilloux, J.-P.; Guiard, B.P.; Gardier, A.M.; Suaud-Chagny, M.F.; Lestage, P. Rapid and precise method to locate microdialysis probe implantation in the rodent brain. J. Neurosci. Methods 2004, 140, 53–57. [Google Scholar] [CrossRef]
- Richter, F.; Richter, A. Genetic animal models of dystonia: common features and diversities. Prog. Neurobiol. 2014, 121, 91–113. [Google Scholar] [CrossRef] [PubMed]
- Lopes, E.F.; Roberts, B.M.; Siddorn, R.E.; Clements, M.A.; Cragg, S.J. Inhibition of Nigrostriatal Dopamine Release by Striatal GABAA and GABAB Receptors. J. Neurosci. 2019, 39, 1058–1065. [Google Scholar] [CrossRef]
- Mathur, B.N.; Lovinger, D.M. Serotonergic action on dorsal striatal function. Parkinsonism Relat. Disord. 2012, 18 Suppl 1, S129–31. [Google Scholar] [CrossRef] [PubMed]
- Blomeley, C.P.; Bracci, E. Serotonin excites fast-spiking interneurons in the striatum. Eur. J. Neurosci. 2009, 29, 1604–1614. [Google Scholar] [CrossRef]
- Stenfors, C.; Ross, S.B. Changes in extracellular 5-HIAA concentrations as measured by in vivo microdialysis technique in relation to changes in 5-HT release. Psychopharmacology (Berl) 2004, 172, 119–128. [Google Scholar] [CrossRef]
- Smith, J.B.; Klug, J.R.; Ross, D.L.; Howard, C.D.; Hollon, N.G.; Ko, V.I.; Hoffman, H.; Callaway, E.M.; Gerfen, C.R.; Jin, X. Genetic-Based Dissection Unveils the Inputs and Outputs of Striatal Patch and Matrix Compartments. Neuron 2016, 91, 1069–1084. [Google Scholar] [CrossRef]
- Smit, M.; Bartels, A.L.; van Faassen, M.; Kuiper, A.; Niezen-Koning, K.E.; Kema, I.P.; Dierckx, R.A.; de Koning, T.J.; Tijssen, M.A. Serotonergic perturbations in dystonia disorders-a systematic review. Neurosci. Biobehav. Rev. 2016, 65, 264–275. [Google Scholar] [CrossRef]
- Ferguson, S.M.; Savchenko, V.; Apparsundaram, S.; Zwick, M.; Wright, J.; Heilman, C.J.; Yi, H.; Levey, A.I.; Blakely, R.D. Vesicular localization and activity-dependent trafficking of presynaptic choline transporters. J. Neurosci. 2003, 23, 9697–9709. [Google Scholar] [CrossRef]
- Szydlowski, S.N.; Pollak Dorocic, I.; Planert, H.; Carlén, M.; Meletis, K.; Silberberg, G. Target selectivity of feedforward inhibition by striatal fast-spiking interneurons. J. Neurosci. 2013, 33, 1678–1683. [Google Scholar] [CrossRef]
- Burke, D.A.; Rotstein, H.G.; Alvarez, V.A. Striatal Local Circuitry: A New Framework for Lateral Inhibition. Neuron 2017, 96, 267–284. [Google Scholar] [CrossRef] [PubMed]
- Assous, M.; Tepper, J.M. Excitatory extrinsic afferents to striatal interneurons and interactions with striatal microcircuitry. Eur. J. Neurosci. 2019, 49, 593–603. [Google Scholar] [CrossRef]
- Kocaturk, S.; Shah, F.; Guven, B.; Tepper, J.M.; Assous, M. Cholinergic control of striatal GABAergic microcircuits; Cold Spring Harbor Laboratory, 2021. [Google Scholar]
- Richter, F.; Bauer, A.; Perl, S.; Schulz, A.; Richter, A. Optogenetic augmentation of the hypercholinergic endophenotype in DYT1 knock-in mice induced erratic hyperactive movements but not dystonia. EBioMedicine 2019, 41, 649–658. [Google Scholar] [CrossRef]
- Tassone, A.; Martella, G.; Meringolo, M.; Vanni, V.; Sciamanna, G.; Ponterio, G.; Imbriani, P.; Bonsi, P.; Pisani, A. Vesicular Acetylcholine Transporter Alters Cholinergic Tone and Synaptic Plasticity in DYT1 Dystonia. Movement Disorders 2021, 36, 2768–2779. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, T.; Fukuyama, Y.; Fujita, M.; Shirasaka, Y.; Tamai, I. Carnitine precursor γ-butyrobetaine is a novel substrate of the Na(+)- and Cl(-)-dependent GABA transporter Gat2. Drug Metab. Pharmacokinet. 2011, 26, 632–636. [Google Scholar] [CrossRef] [PubMed]
- Dambrova, M.; Chlopicki, S.; Liepinsh, E.; Kirjanova, O.; Gorshkova, O.; Kozlovski, V.I.; Uhlen, S.; Liepina, I.; Petrovska, R.; Kalvinsh, I. The methylester of gamma-butyrobetaine, but not gamma-butyrobetaine itself, induces muscarinic receptor-dependent vasodilatation. Naunyn Schmiedebergs. Arch. Pharmacol. 2004, 369, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Lamy, E.; Pilyser, L.; Paquet, C.; Bouaziz-Amar, E.; Grassin-Delyle, S. High-sensitivity quantification of acetylcholine and choline in human cerebrospinal fluid with a validated LC-MS/MS method. Talanta 2021, 224, 121881. [Google Scholar] [CrossRef]
- Chefer, V.I.; Thompson, A.C.; Zapata, A.; Shippenberg, T.S. Overview of brain microdialysis. Curr. Protoc. Neurosci. 2009, Chapter 7, Unit7.1. [Google Scholar] [CrossRef]
- Zhu, M.; Liu, G.; Chen, H.; Ma, W.; Cui, L.; Nimmu, N.V.; Hou, H.; Hu, Q.; Zhang, Y. Analytical strategies in neurotransmitter measurements: A mini literature review. Biomed. Chromatogr. 2023, 37, e5521. [Google Scholar] [CrossRef]
- Hascup, E.R.; Bjerkén, S. af; Hascup, K.N.; Pomerleau, F.; Huettl, P.; Strömberg, I.; Gerhardt, G.A. Histological studies of the effects of chronic implantation of ceramic-based microelectrode arrays and microdialysis probes in rat prefrontal cortex. Brain Res. 2009, 1291, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Richner, T.J.; Baumgartner, R.; Brodnick, S.K.; Azimipour, M.; Krugner-Higby, L.A.; Eliceiri, K.W.; Williams, J.C.; Pashaie, R. Patterned optogenetic modulation of neurovascular and metabolic signals. J. Cereb. Blood Flow Metab. 2015, 35, 140–147. [Google Scholar] [CrossRef]
- Emiliani, V.; Entcheva, E.; Hedrich, R.; Hegemann, P.; Konrad, K.R.; Lüscher, C.; Mahn, M.; Pan, Z.-H.; Sims, R.R.; Vierock, J.; et al. Optogenetics for light control of biological systems. Nat. Rev. Methods Primers 2022, 2. [Google Scholar] [CrossRef] [PubMed]
- Paxinos, G.; Franklin, K.B.J. Paxinos and Franklin’s The mouse brain in stereotaxic coordinates, Fifth edition; Academic Press an imprint of Elsevier: London, 2019; ISBN 0128161582. [Google Scholar]
- Wiegert, J.S.; Mahn, M.; Prigge, M.; Printz, Y.; Yizhar, O. Silencing Neurons: Tools, Applications, and Experimental Constraints. Neuron 2017, 95, 504–529. [Google Scholar] [CrossRef] [PubMed]
- Bansal, H.; Gupta, N.; Roy, S. Comparison of low-power, high-frequency and temporally precise optogenetic inhibition of spiking in NpHR, eNpHR3.0 and Jaws-expressing neurons. Biomed. Phys. Eng. Express 2020, 6, 45011. [Google Scholar] [CrossRef] [PubMed]
| GABA 1), 2) | ACh | isoACh | Cholin 1), 3) | DA | 3-MT | 5-HIAA | ADE 1), 2) | |
|---|---|---|---|---|---|---|---|---|
| WT (n = 8) DYT1 (n = 9) |
WT (n = 4) DYT1 (n = 3) |
WT (n = 9) DYT1 (n = 9) |
WT (n = 8) DYT1 (n = 9) |
WT (n = 9) DYT1 (n = 9) |
WT (n = 9) DYT1 (n = 10) |
WT (n = 9) DYT1 (n = 9) |
WT (n = 9) DYT1 (n = 9) |
|
| Baseline | ||||||||
| WT | 10.25 ± 2.23* | 0.48 ± 0.04 | 7.74 ± 0.99 | 153.36 ± 25.64 | 3.37 ± 0.74 | 7.15 ± 1.29 | 150.84 ± 22.28 | 26.58 ± 7.30 * |
| DYT1 | 6.57 ± 0.97 | 0.49 ± 0.07 | 6.73 ± 0.97 | 143.68 ± 23.01 | 3.19 ± 0.59 | 6.43 ± 0.59 | 100.74 ± 15.97 | 31.96 ± 9.85 # |
| Stimulation | ||||||||
| WT | 7.64 ± 1.37 * | 0.51 ± 0.10 | 7.94 ± 1.04 | 149.08 ± 29.73 | 3.33 ± 0.71 | 7.52 ± 1.29 | 144.65 ± 18.79 | 14.90 ± 4.63 * |
| DYT1 | 5.82 ± 0.82 | 0.37 ± 0.04 | 5.98 ± 0.67 | 128.33 ± 22.85 | 2.54 ± 0.23 | 6.21 ± 0.51 | 89.70 ± 18.10 | 23.64 ± 7.84 |
| Post-Stim | ||||||||
| WT | 7.98 ± 1.44 * | 0.52 ± 0.07 | 7.34 ± 0.86 | 127.51 ± 30.52 | 6.50 ± 0.76 | 7.77 ± 1.24 | 159.15 ± 21.91 G | 11.23 ± 2.79 * |
| DYT1 | 6.01 ± 1.04 | 0.38 ± 0,02 | 5.77 ± 0.62 | 115.50 ± 23.28 | 2.77 ± 0.17 | 6.34 ± 0.37 | 91.99 ± 15.19 G | 18.54 ± 5.25# |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




