Submitted:
10 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Plant-Based Expression Platforms for Recombinant Vaccine Antigen Production
2.1. Plant-Based Vaccines: Plants as Biofactories and Cellular Platforms for Antigen Production
2.1.1. Genetic Transformation and Vector Systems
2.1.2. Transcriptional and Translational Control
2.1.3. Glycoengineering and Humanized Glycosylation in Plant Systems
2.1.4. Subcellular Targeting and Protein Folding
2.1.5. Virus-like Particles (VLPs)
2.1.6. Downstream Processing and Purification Strategies
2.2. Microalgae-Derived Vaccines
2.2.1. Nuclear vs Chloroplast Expression in Microalgae
2.2.2. Transient/Viral-Vector and Episomal Systems
2.2.3. Promoters, UTRs, and Selectable Markers
2.2.4. Glycosylation and PTMs in Microalgae
2.2.5. Downstream Processing
3. Plant-Derived Compounds as Vaccine Adjuvants
3.1. Introduction and Immunological Rationale
3.2. Major Classes of Phytochemical Adjuvants
3.2.1. Saponins, Membrane-Active Enhancers
3.2.2. Polysaccharides, Depot-Forming Scaffolds and PRR Agonists
3.2.3. Flavonoids and Phenolics, Immunometabolic Modulators
3.2.4. Terpenoids, Emulsion-Based Adjuvant Systems
3.2.5. Alkaloids, Cytosolic Delivery and Modulators
3.2.6. Lectins, Mucosal Targeting Ligands
3.2.7. Plant Viruses as Vaccine Components and Adjuvants
3.3. Safety, Toxicity, and Translational Challenges
4. Plant-Based Vaccine Delivery Systems: Focus on Oral and Edible Platforms
4.1. Plant Matrices, Bioencapsulation, and Antigen Stability
4.2. Examples and Progress in Plant-Based Vaccine Delivery
4.3. Advantages, Challenges, and Limitations
5. Regulatory Considerations and Translational Challenges
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CaMV | Cauliflower Mosaic Virus |
| CRISPR | Clustered Regularly Interspaced Short Palindromic Repeats |
| Cas9 | CRISPR-associated protein 9 |
| RNAi | RNA interference |
| UTR | Untranslated Region |
| ER | Endoplasmic Reticulum |
| KDEL | Lys-Asp-Glu-Leu (ER retention signal) |
| VLP | Virus-Like Particle |
| HPV | Human Papillomavirus |
| HBcAg | Hepatitis B Core Antigen |
| CPMV | Cowpea Mosaic Virus |
| HA | Hemagglutinin |
| SARS-CoV- | Severe Acute Respiratory Syndrome Coronavirus 2 |
| APC | Antigen-Presenting Cell |
| MHC | Major Histocompatibility Complex |
| DSP | Downstream Processing |
| HCP | Host Cell Protein |
| SEC | Size-Exclusion Chromatography |
| AEC | Anion-Exchange Chromatography |
| SXC | Steric Exclusion Chromatography |
| HPH | High-Pressure Homogenization |
| CO2 | Carbon Dioxide |
| PTM | Post-Translational Modification |
| RBD | Receptor-Binding Domain |
| ACE2 | Angiotensin-Converting Enzyme 2 |
| CTB | Cholera Toxin B Subunit |
| LTB | Heat-Labile Enterotoxin B Subunit |
| qPCR | Quantitative Polymerase Chain Reaction |
| VP1 | Viral Protein 1 |
| VP2 | Viral Protein 2 |
| VP28 | Viral Protein 28 |
| GnTI | N-Acetylglucosaminyltransferase I |
| Neu5Ac | N-Acetylneuraminic Acid |
| CMP | Cytidine Monophosphate |
| Ti plasmid | Tumor-Inducing Plasmid |
| PRRs | Pattern Recognition Receptors |
| TLRs | Toll-Like Receptors |
| CLRs | C-Type Lectin Receptors |
| NLRs | NOD-Like Receptors |
| MPL | Monophosphoryl Lipid A |
| Tfh | T Follicular Helper Cells |
| GC | Germinal Center |
| APS | Astragalus Polysaccharides |
| EGCG | Epigallocatechin Gallate |
| LNP | Lipid Nanoparticle |
| sIgA | Secretory Immunoglobulin A |
| UEA-1 | Ulex europaeus Agglutinin-1 |
| GMTs | Geometric Mean Titers |
| GALT | Gut-Associated Lymphoid Tissue |
| NVCP | Norwalk Virus Capsid Protein |
| TGEV | Transmissible Gastroenteritis Virus |
| GMP | Good Manufacturing Practice |
| GMOs | Genetically Modified Organisms |
References
- Mao, H.H.; Chao, S. Advances in vaccines. In Current Applications of Pharmaceutical Biotechnology; 2019; pp. 155–88. [Google Scholar]
- Kis, Z.; Shattock, R.; Shah, N.; Kontoravdi, C. Emerging technologies for low-cost, rapid vaccine manufacture. Biotechnology journal 2019, 14, 1800376. [Google Scholar] [CrossRef] [PubMed]
- Boilesen, D.R.; Nielsen, K.N.; Holst, P.J. Novel antigenic targets of HPV therapeutic vaccines. Vaccines 2021, 9, 1262. [Google Scholar] [CrossRef] [PubMed]
- Giudice, E.L.; Campbell, J.D. Needle-free vaccine delivery. Advanced drug delivery reviews 2006, 58, 68–89. [Google Scholar] [CrossRef] [PubMed]
- Kwong, K.W.-Y.; Xin, Y.; Lai, N.C.-Y.; Sung, J.C.-C.; Wu, K.-C.; Hamied, Y.K.; et al. Oral vaccines: a better future of immunization. Vaccines 2023, 11, 1232. [Google Scholar] [CrossRef]
- Bandyopadhyay, A.S.; Zipursky, S. A novel tool to eradicate an ancient scourge: the novel oral polio vaccine type 2 story. The Lancet Infectious Diseases 2023, 23, e67–e71. [Google Scholar] [CrossRef]
- Boccalini, S.; Del Riccio, M.; Crescioli, G.; Salvati, C.; Sadotti, A.; Cacini, C.; et al. Safety of co-administration of injectable vaccines in individuals under 18 years of age: A systematic literature review. Human Vaccines & Immunotherapeutics 2025, 21, 2592425. [Google Scholar] [CrossRef]
- Carrier, M.J. Protein subunit vaccines and recombinant DNA technology; Routledge: Vaccination Strategies of Tropical Diseases, 2017; pp. 11–29. [Google Scholar]
- Lasky, L.A. From virus to vaccine: recombinant mammalian cell lines as substrates for the production of herpes simplex virus vaccines. Journal of medical virology 1990, 31, 59–61. [Google Scholar] [CrossRef]
- Chen, H.-L.; Huang, J.-Y.; Chu, T.-W.; Tsai, T.-C.; Hung, C.-M.; Lin, C.-C.; et al. Expression of VP1 protein in the milk of transgenic mice: a potential oral vaccine protects against enterovirus 71 infection. Vaccine 2008, 26, 2882–9. [Google Scholar] [CrossRef]
- Fischer, R.; Stoger, E.; Schillberg, S.; Christou, P.; Twyman, R.M. Plant-based production of biopharmaceuticals. Current opinion in plant biology 2004, 7, 152–8. [Google Scholar] [CrossRef]
- Streatfield, S.J.; Howard, J.A. Plant-based vaccines. International Journal for Parasitology 2003, 33, 479–93. [Google Scholar] [CrossRef]
- Scognamiglio, P.L.; Tesauro, D.; Roviello, G.N. Metallogels as Supramolecular Platforms for Biomedical Applications: A Review. Processes 2025, 13, 3671. [Google Scholar] [CrossRef]
- Sargsyan, T.; Simonyan, H.M.; Stepanyan, L.; Tsaturyan, A.; Vicidomini, C.; Pastore, R.; et al. Neuroprotective Properties of Clove (Syzygium aromaticum): State of the Art and Future Pharmaceutical Applications for Alzheimer’s Disease. Biomolecules 2025, 15, 452. [Google Scholar] [CrossRef]
- Sargsyan, T.; Stepanyan, L.; Tsaturyan, A.; Palumbo, R.; Vicidomini, C.; Roviello, G.N. Intracellular Parasitic Infections Caused by Plasmodium falciparum, Leishmania spp., Toxoplasma gondii, Echinococcus multilocularis, Among Key Pathogens: Global Burden, Transmission Dynamics, and Vaccine Advances—A Narrative Review with Contextual Insights from Armenia. Vaccines 2025, 13, 1082. [Google Scholar] [CrossRef]
- Najafi, S.; Jojani, M.; Najafi, K.; Costanzo, V.; Vicidomini, C.; Roviello, G.N. West Nile Virus: Epidemiology, Surveillance, and Prophylaxis with a Comparative Insight from Italy and Iran. Vaccines 2026, 14, 57. [Google Scholar] [CrossRef]
- Costanzo, M.; Roviello, G.N. Precision Therapeutics Through Bioactive Compounds: Metabolic Reprogramming, Omics Integration, and Drug Repurposing Strategies. International Journal of Molecular Sciences 2025, 26, 10047. [Google Scholar] [CrossRef]
- Vicidomini, C.; Roviello, G.N. Therapeutic Convergence in Neurodegeneration: Natural Products, Drug Repurposing, and Biomolecular Targets. Biomolecules 2025, 15, 1333. [Google Scholar] [CrossRef]
- Sargsyan, T.; Stepanyan, L.; Panosyan, H.; Hakobyan, H.; Israyelyan, M.; Tsaturyan, A.; et al. Synthesis and antifungal activity of Fmoc-protected 1, 2, 4-triazolyl-α-amino acids and their dipeptides against Aspergillus species. Biomolecules 2025, 15, 61. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, B.T.; Thompson, E.P.; Roviello, G.N.; Gale, T.F. C-Terminal Analogues of Camostat Retain TMPRSS2 Protease Inhibition: New Synthetic Directions for Antiviral Repurposing of Guanidinium-Based Drugs in Respiratory Infections. International Journal of Molecular Sciences 2025, 26, 6761. [Google Scholar] [CrossRef] [PubMed]
- Stepanyan, L.; Israyelyan, M.; Gori, A.; Tsaturyan, A.; Saribekyan, Z.; Hovsepyan, K.; et al. Natural and Synthetic Peptides as Alternatives to Antibiotics in Intestinal Infections—A Review. Antibiotics 2026, 15, 68. [Google Scholar] [CrossRef]
- Sargsyan, T.; Hakobyan, H.; Simonyan, H.; Soghomonyan, T.; Tsaturyan, A.; Hovhannisyan, A.; et al. Biomacromolecular interactions and antioxidant properties of novel synthetic amino acids targeting DNA and serum albumin. Journal of Molecular Liquids 2025, 128700. [Google Scholar] [CrossRef]
- Stepanyan, L.; Sargsyan, T.; Mittova, V.; Tsetskhladze, Z.R.; Motsonelidze, N.; Gorgoshidze, E.; et al. The Synthesis, characterization, and biological evaluation of a fluorenyl-methoxycarbonyl-containing thioxo-triazole-bearing dipeptide: Antioxidant, antimicrobial, and BSA/DNA binding studies for potential therapeutic applications in ROS scavenging and drug transport. Biomolecules 2025, 15, 933. [Google Scholar]
- Simonyan, H.; Palumbo, R.; Vicidomini, C.; Scognamiglio, P.L.; Petrosyan, S.; Sahakyan, L.; et al. Binding of G-quadruplex DNA and serum albumins by synthetic non-proteinogenic amino acids: Implications for c-Myc-related anticancer activity and drug delivery. Molecular Therapy Nucleic Acids 2025, 36. [Google Scholar] [CrossRef]
- Hayriyan, L.; Grigoryan, A.; Gevorgyan, H.; Tsaturyan, A.; Sargsyan, A.; Langer, P.; et al. A 3-Mannich coupling reaction via chiral propargylglycine Ni (ii) complex: an approach for synthesizing enantiomerically enriched unnatural α-amino acids. RSC advances 2025, 15, 35379–87. [Google Scholar] [CrossRef] [PubMed]
- Tovmasyan, A.S.; Mkrtchyan, A.F.; Tsaturyan, A.H.; Langer, P.; Malkov, A.V.; Saghyan, A.S. Strategy for synthesizing O-protected (S)-α-substituted serine analogs via sequential Ni (ii)-complex-mediated cross-coupling and cycloaddition reactions. New Journal of Chemistry 2025, 49, 11640–5. [Google Scholar] [CrossRef]
- Dadayan, A.S.; Mkrtchyan, A.F.; Poghosyan, A.S.; Dadayan, S.A.; Stepanyan, L.A.; Israyelyan, M.H.; et al. Unnatural Phosphorus-Containing α-Amino Acids and Their N-FMOC Derivatives: Synthesis and In Vitro Investigation of Anticholinesterase Activity. ChemistrySelect 2024, 9, e202303249. [Google Scholar] [CrossRef]
- Tovmasyan, A.S.; Mkrtchyan, A.F.; Khachatryan, H.N.; Hayrapetyan, M.V.; Hakobyan, R.M.; Poghosyan, A.S.; et al. Synthesis, characterization, and study of catalytic activity of chiral Cu (II) and Ni (II) salen complexes in the α-Amino Acid C-α alkylation reaction. Molecules 2023, 28, 1180. [Google Scholar] [CrossRef]
- Mkrtchyan, A.F.; Hayriyan, L.A.; Karapetyan, A.J.; Tovmasyan, A.S.; Tsaturyan, A.H.; Khrustalev, V.N.; et al. Using the Ni-[(benzylprolyl) amino] benzophenone complex in the Glaser reaction for the synthesis of bis α-amino acids. New Journal of Chemistry 2020, 44, 11927–32. [Google Scholar] [CrossRef]
- Mkrtchyan, A.F.; Saghyan, A.S.; Hayriyan, L.A.; Sargsyan, A.S.; Karapetyan, A.J.; Tovmasyan, A.S.; et al. Asymmetric synthesis, biological activity and molecular docking studies of some unsaturated α-amino acids, derivatives of glycine, allylglycine and propargylglycine. Journal of Molecular Structure 2020, 1208, 127850. [Google Scholar] [CrossRef]
- Parpart, S.; Petrosyan, A.; Shah, S.J.A.; Adewale, R.A.; Ehlers, P.; Grigoryan, T.; et al. Synthesis of optically pure (S)-2-amino-5-arylpent-4-ynoic acids by Sonogashira reactions and their potential use as highly selective potent inhibitors of aldose reductase. RSC advances 2015, 5, 107400–12. [Google Scholar] [CrossRef]
- Vicidomini, C.; Fontanella, F.; D’Alessandro, T.; Roviello, G.N.; De Stefano, C.; Stocchi, F.; et al. Resting-state functional MRI metrics to detect freezing of gait in Parkinson’s disease: a machine learning approach. Computers in Biology and Medicine 2025, 192, 110244. [Google Scholar] [CrossRef]
- Fontanella, F.; D’Alessandro, T.; Nardone, E.; De Stefano, C.; Vicidomini, C.; Roviello, G.N. Artificial Intelligence for Natural Products Drug Discovery in Neurodegeneration Therapies: A Review. Biomolecules 2026, 16, 129. [Google Scholar] [CrossRef] [PubMed]
- Roviello, G.N. Nature-Inspired Pathogen and Cancer Protein Covalent Inhibitors: From Plants and Other Natural Sources to Drug Development. Pathogens 2025, 14, 1153. [Google Scholar] [CrossRef]
- Mittova, V.; Tsetskhladze, Z.R.; Motsonelidze, N.; Palumbo, R.; Roviello, G.N. Georgian Grapes and Wines as a Source of Phenolic Compounds: Composition, Antioxidant Activity, and Traditional Winemaking. Molecules 2026, 31, 303. [Google Scholar] [CrossRef]
- Bagiyan, V.; Zakoyan, A.; Verdyan, A.; Ghazanchyan, N.; Kinosyan, M.; Davidyan, T.; et al. Yeast whey-enriched bread: nutritional profile and potential functional relevance. Functional Foods in Health and Disease-Online ISSN: 2160-3855 Print ISSN: 2378-7007. 2025. 15, 854–66. [CrossRef]
- Aghajanyan, A.; Avagyan, G.; Tsaturyan, A.; Hovhannisyan, G.; Yeghiyan, K.; Gasparyan, N.; et al. Valorization of grape pomace through melanin extraction and its biostimulant effects on yield and development of winter barley. Chilean journal of agricultural research 2025, 85, 635–49. [Google Scholar] [CrossRef]
- Hovsepyan, A.; Petrosyan, T.; Saghatelyan, L.; Tsaturyan, A.; Paronyan, M.; Koloyan, H.; et al. Safety evaluation of bacterial melanin as a plant growth stimulant for agricultural and food industry applications. Functional Food Science-Online ISSN: 2767-3146 2025, 5, 450–61. [Google Scholar] [CrossRef]
- Aghajanyan, A.; Minasyan, E.; Soghomonyan, T.; Melyan, G.; Hovhannisyan, G.; Yeghiyan, K.; et al. Isolation, purification, identification of melanin from grape pomace extracts, and its application areas. Bioactive Compounds in Health and Disease-Online ISSN: 2574-0334 2025, 8, 204–16. [Google Scholar] [CrossRef]
- Minasyan, E.; Aghajanyan, A.; Karapetyan, K.; Khachaturyan, N.; Hovhannisyan, G.; Yeghyan, K.; et al. Antimicrobial activity of melanin isolated from wine waste. Indian Journal of Microbiology 2024, 64, 1528–34. [Google Scholar] [CrossRef] [PubMed]
- Aghajanyan, A.E.; Hambardzumyan, A.A.; Minasyan, E.V.; Hovhannisyan, G.J.; Yeghiyan, K.I.; Soghomonyan, T.M.; et al. Efficient isolation and characterization of functional melanin from various plant sources. International Journal of Food Science and Technology 2024, 59, 3545–55. [Google Scholar] [CrossRef]
- Bagiyan, V.; Chitchyan, K.; Goginyan, V.; Tsaturyan, A. Baker’s yeast of the ttkhmor with high α-glucosidase activity for cultivation on whey. Food and Humanity 2024, 2, 100209. [Google Scholar] [CrossRef]
- Poladyan, A.; Trchounian, K.; Paloyan, A.; Minasyan, E.; Aghekyan, H.; Iskandaryan, M.; et al. Valorization of whey-based side streams for microbial biomass, molecular hydrogen, and hydrogenase production. Applied microbiology and biotechnology 2023, 107, 4683–96. [Google Scholar] [CrossRef] [PubMed]
- Stepanyan, L.; Sargsyan, T.; Israyelyan, M.; Karapetyan, K.; Tsaturyan, A. Study of the chemical composition, antibacterial and antioxidant activity of various extracts of the aerial part of Leonurus cardiaca. Farmacia 2023, 71, 1064–71. [Google Scholar] [CrossRef]
- Tsaturyan, A.; Arstamyan, L.; Sargsyan, A.; Saribekyan, J.; Voskanyan, A.; Minasyan, E.; et al. Development of an efficient method for obtaining lactose and lactulose from whey. Pharmacia 2023, 70, 1039–46. [Google Scholar] [CrossRef]
- Aghajanyan, A.E.; Hambardzumyan, A.A.; Minasyan, E.V.; Tsaturyan, A.H.; Paloyan, A.M.; Avetisyan, S.V.; et al. Development of the technology for producing water-soluble melanin from waste of vinary production and the study of its physicochemical properties. European Food Research and Technology 2022, 248, 485–95. [Google Scholar] [CrossRef]
- Tsaturyan, A.; Sahakyan, L.; Hayrapetyan, L.; Minasyan, E.; Chakhoyan, A.; Hayrapetyan, S.; et al. Ion-chromatographic determination of common anions in drinking water in some regions of the Republic of Armenia. Pharmacia 2024, 71, 1–9. [Google Scholar] [CrossRef]
- Kirakosyan, V.G.; Tsaturyan, A.H.; Poghosyan, L.E.; Minasyan, E.V.; Petrosyan, H.R.; Sahakyan, L.Y.; et al. Detection and development of a quantitation method for undeclared compounds in antidiabetic biologically active additives and its validation by high performance liquid chromatography. Pharmacia 2022, 69, 45–50. [Google Scholar] [CrossRef]
- Meyers, B.; Zaltsman, A.; Lacroix, B.; Kozlovsky, S.V.; Krichevsky, A. Nuclear and plastid genetic engineering of plants: comparison of opportunities and challenges. Biotechnology advances 2010, 28, 747–56. [Google Scholar]
- Sala, F.; Rigano, M.M.; Barbante, A.; Basso, B.; Walmsley, A.M.; Castiglione, S. Vaccine antigen production in transgenic plants: strategies, gene constructs and perspectives. Vaccine 2003, 21, 803–8. [Google Scholar] [CrossRef]
- Morley, S.A.; Nielsen, B.L. Chloroplast DNA Copy Number Changes during Plant Development in Organelle DNA Polymerase Mutants. Front Plant Sci. 2016, 7, 57. [Google Scholar] [CrossRef] [PubMed]
- Oey, M.; Lohse, M.; Kreikemeyer, B.; Bock, R. Exhaustion of the chloroplast protein synthesis capacity by massive expression of a highly stable protein antibiotic. The plant journal. 2009, 57, 436–45. [Google Scholar]
- Ruf, S.; Karcher, D.; Bock, R. Determining the transgene containment level provided by chloroplast transformation. Proc Natl Acad Sci U S A 2007, 104, 6998–7002. [Google Scholar] [CrossRef]
- Rigano, M.M.; Scotti, N.; Cardi, T. Unsolved problems in plastid transformation. Bioengineered 2012, 3, 329–33. [Google Scholar] [CrossRef] [PubMed]
- Komarova, T.V.; Baschieri, S.; Donini, M.; Marusic, C.; Benvenuto, E.; Dorokhov, Y.L. Transient expression systems for plant-derived biopharmaceuticals. Expert review of vaccines 2010, 9, 859–76. [Google Scholar] [PubMed]
- Rybicki, E.P. Plant-made vaccines and reagents for the One Health initiative. Hum Vaccin Immunother. 2017, 13, 2912–7. [Google Scholar] [CrossRef]
- Norkunas, K.; Harding, R.; Dale, J.; Dugdale, B. Improving agroinfiltration-based transient gene expression in Nicotiana benthamiana. Plant methods 2018, 14, 71. [Google Scholar] [CrossRef]
- Bevan, M. Binary Agrobacterium vectors for plant transformation. Nucleic Acids Res. 1984, 12, 8711–21. [Google Scholar]
- Kaur, M.; Manchanda, P.; Kalia, A.; Ahmed, F.K.; Nepovimova, E.; Kuca, K.; et al. Agroinfiltration mediated scalable transient gene expression in genome edited crop plants. International journal of molecular sciences 2021, 22, 10882. [Google Scholar] [CrossRef]
- Gelvin, S.B. Agrobacterium-mediated plant transformation: the biology behind the “gene-jockeying” tool. Microbiol Mol Biol Rev. table of contents. 2003, 67, 16–37. [Google Scholar] [CrossRef]
- Hefferon, K.L. Plant virus expression vectors set the stage as production platforms for biopharmaceutical proteins. Virology 2012, 433, 1–6. [Google Scholar] [CrossRef]
- Gleba, Y.; Klimyuk, V.; Marillonnet, S. Viral vectors for the expression of proteins in plants. Curr Opin Biotechnol. 2007, 18, 134–41. [Google Scholar] [CrossRef]
- Feng, Z.; Li, X.; Fan, B.; Zhu, C.; Chen, Z. Maximizing the Production of Recombinant Proteins in Plants: From Transcription to Protein Stability. International Journal of Molecular Sciences 2022, 23, 13516. [Google Scholar] [CrossRef] [PubMed]
- Nochi, T.; Takagi, H.; Yuki, Y.; Yang, L.; Masumura, T.; Mejima, M.; et al. Rice-based mucosal vaccine as a global strategy for cold-chain-and needle-free vaccination. Proceedings of the National Academy of Sciences 2007, 104, 10986–91. [Google Scholar] [CrossRef]
- Sandhu, J.S.; Krasnyanski, S.F.; Domier, L.L.; Korban, S.S.; Osadjan, M.D.; Buetow, D.E. Oral immunization of mice with transgenic tomato fruit expressing respiratory syncytial virus-F protein induces a systemic immune response. Transgenic research 2000, 9, 127–35. [Google Scholar]
- Ali, S.; Kim, W.-C. A fruitful decade using synthetic promoters in the improvement of transgenic plants. Frontiers in plant science 2019, 10, 1433. [Google Scholar] [CrossRef]
- Mushegian, A.R.; Shepherd, R.J. Genetic elements of plant viruses as tools for genetic engineering. Microbiological reviews 1995, 59, 548–78. [Google Scholar]
- Paremskaia, A.I.; Kogan, A.A.; Murashkina, A.; Naumova, D.A.; Satish, A.; Abramov, I.S.; et al. Codon-optimization in gene therapy: promises, prospects and challenges. Front Bioeng Biotechnol. 2024, 12, 1371596. [Google Scholar] [CrossRef]
- Liu, H.; Timko, M.P. Improving Protein Quantity and Quality-The Next Level of Plant Molecular Farming. Int J Mol Sci. 2022, 23. [Google Scholar] [CrossRef]
- Venkataraman, S.; Khan, I.; Habibi, P.; Le, M.; Lippert, R.; Hefferon, K. Recent advances in expression and purification strategies for plant made vaccines. Frontiers in Plant Science 2023, 14, 1273958. [Google Scholar] [CrossRef] [PubMed]
- Kanagarajan, S.; Tolf, C.; Lundgren, A.; Waldenström, J.; Brodelius, P.E. Transient Expression of Hemagglutinin Antigen from Low Pathogenic Avian Influenza A (H7N7) in Nicotiana benthamiana. PLOS ONE 2012, 7, e33010. [Google Scholar] [CrossRef] [PubMed]
- Mett, V.; Musiychuk, K.; Bi, H.; Farrance, C.E.; Horsey, A.; Ugulava, N.; et al. A plant-produced influenza subunit vaccine protects ferrets against virus challenge. Influenza and Other Respiratory Viruses 2008, 2, 33–40. [Google Scholar] [CrossRef]
- Altmann, F. The role of protein glycosylation in allergy. Int Arch Allergy Immunol. 2007, 142, 99–115. [Google Scholar] [CrossRef]
- Mercx, S.; Smargiasso, N.; Chaumont, F.; De Pauw, E.; Boutry, M.; Navarre, C. Inactivation of the β (1, 2)-xylosyltransferase and the α (1, 3)-fucosyltransferase genes in Nicotiana tabacum BY-2 cells by a multiplex CRISPR/Cas9 strategy results in glycoproteins without plant-specific glycans. Frontiers in plant science 2017, 8, 403. [Google Scholar] [CrossRef]
- Jansing, J.; Sack, M.; Augustine, S.M.; Fischer, R.; Bortesi, L. CRISPR/Cas9-mediated knockout of six glycosyltransferase genes in Nicotiana benthamiana for the production of recombinant proteins lacking β-1, 2-xylose and core α-1, 3-fucose. Plant biotechnology journal 2019, 17, 350–61. [Google Scholar] [CrossRef]
- Pantazica, A.M.; van Eerde, A.; Dobrica, M.O.; Caras, I.; Ionescu, I.; Costache, A.; et al. The “humanized” N-glycosylation pathway in CRISPR/Cas9-edited Nicotiana benthamiana significantly enhances the immunogenicity of a S/preS1 Hepatitis B Virus antigen and the virus-neutralizing antibody response in vaccinated mice. Plant biotechnology journal 2023, 21, 1176–90. [Google Scholar] [CrossRef] [PubMed]
- Palacpac, N.Q.; Yoshida, S.; Sakai, H.; Kimura, Y.; Fujiyama, K.; Yoshida, T.; et al. Stable expression of human β1, 4-galactosyltransferase in plant cells modifies N-linked glycosylation patterns. Proceedings of the National Academy of Sciences 1999, 96, 4692–7. [Google Scholar] [CrossRef] [PubMed]
- Kallolimath, S.; Castilho, A.; Strasser, R.; Grünwald-Gruber, C.; Altmann, F.; Strubl, S.; et al. Engineering of complex protein sialylation in plants. Proceedings of the National Academy of Sciences 2016, 113, 9498–503. [Google Scholar] [CrossRef]
- Moore, C.M.; Grandits, M.; Grünwald-Gruber, C.; Altmann, F.; Kotouckova, M.; Teh, A.Y.H.; et al. Characterisation of a highly potent and near pan-neutralising anti-HIV monoclonal antibody expressed in tobacco plants. Retrovirology 2021, 18, 17. [Google Scholar] [CrossRef]
- Castilho, A.; Neumann, L.; Gattinger, P.; Strasser, R.; Vorauer-Uhl, K.; Sterovsky, T.; et al. Generation of Biologically Active Multi-Sialylated Recombinant Human EPOFc in Plants. PLOS ONE 2013, 8, e54836. [Google Scholar] [CrossRef] [PubMed]
- Vitale, A.; Boston, R.S. Endoplasmic reticulum quality control and the unfolded protein response: insights from plants. Traffic 2008, 9, 1581–8. [Google Scholar] [CrossRef]
- Gomord, V.; Fitchette, A.C.; Menu-Bouaouiche, L.; Saint-Jore-Dupas, C.; Plasson, C.; Michaud, D.; et al. Plant-specific glycosylation patterns in the context of therapeutic protein production. Plant Biotechnol J. 2010, 8, 564–87. [Google Scholar] [CrossRef]
- Gomord, V.; Denmat, L.A.; Fitchette-Lainé, A.C.; Satiat-Jeunemaitre, B.; Hawes, C.; Faye, L. The C-terminal HDEL sequence is sufficient for retention of secretory proteins in the endoplasmic reticulum (ER) but promotes vacuolar targeting of proteins that escape the ER. The Plant Journal. 1997, 11, 313–25. [Google Scholar] [CrossRef] [PubMed]
- Rozov, S.M.; Deineko, E.V. Increasing the efficiency of the accumulation of recombinant proteins in plant cells: the role of transport signal peptides. Plants 2022, 11, 2561. [Google Scholar] [CrossRef] [PubMed]
- Shaaltiel, Y.; Bartfeld, D.; Hashmueli, S.; Baum, G.; Brill-Almon, E.; Galili, G.; et al. Production of glucocerebrosidase with terminal mannose glycans for enzyme replacement therapy of Gaucher’s disease using a plant cell system. Plant biotechnology journal. 2007, 5, 579–90. [Google Scholar] [CrossRef]
- Singh, N.D.; Ding, Y.; Daniell, H. Chloroplast-derived vaccine antigens and biopharmaceuticals: protocols for expression, purification, or oral delivery and functional evaluation; Recombinant Proteins From Plants: Methods and Protocols: Springer, 2009; pp. 163–92. [Google Scholar]
- Nooraei, S.; Bahrulolum, H.; Hoseini, Z.S.; Katalani, C.; Hajizade, A.; Easton, A.J.; et al. Virus-like particles: preparation, immunogenicity and their roles as nanovaccines and drug nanocarriers. Journal of nanobiotechnology 2021, 19, 59. [Google Scholar] [CrossRef]
- Marsian, J.; Lomonossoff, G.P. Molecular pharming—VLPs made in plants. Current opinion in biotechnology 2016, 37, 201–6. [Google Scholar] [CrossRef]
- Chen, Q.; Lai, H. Plant-derived virus-like particles as vaccines. Human vaccines & immunotherapeutics 2013, 9, 26–49. [Google Scholar]
- O’Rourke, J.P.; Peabody, D.S.; Chackerian, B. Affinity selection of epitope-based vaccines using a bacteriophage virus-like particle platform. Current opinion in virology 2015, 11, 76–82. [Google Scholar] [CrossRef]
- Ruedl, C.; Schwarz, K.; Jegerlehner, A.; Storni, T.; Manolova, V.; Bachmann, M.F. Virus-like particles as carriers for T-cell epitopes: limited inhibition of T-cell priming by carrier-specific antibodies. J Virol. 2005, 79, 717–24. [Google Scholar] [CrossRef]
- Ward, B.J.; Gobeil, P.; Séguin, A.; Atkins, J.; Boulay, I.; Charbonneau, P.-Y.; et al. Phase 1 randomized trial of a plant-derived virus-like particle vaccine for COVID-19. Nature Medicine 2021, 27, 1071–8. [Google Scholar] [CrossRef]
- Su, H.; van Eerde, A.; Rimstad, E.; Bock, R.; Branza-Nichita, N.; Yakovlev, I.A.; et al. Plant-made vaccines against viral diseases in humans and farm animals. In Frontiers in Plant Science; 2023; pp. 14–2023. [Google Scholar]
- Hodgins, B.; Pillet, S.; Landry, N.; Ward, B.J. A plant-derived VLP influenza vaccine elicits a balanced immune response even in very old mice with co-morbidities. PLOS ONE 2019, 14, e0210009. [Google Scholar] [CrossRef]
- Madeira, L.M.; Szeto, T.H.; Ma, J.K.-C.; Drake, P.M.W. Rhizosecretion improves the production of Cyanovirin-N in Nicotiana tabacum through simplified downstream processing. Biotechnology Journal. 2016, 11, 910–9. [Google Scholar] [CrossRef]
- Zahmanova, G.; Aljabali, A.A.A.; Takova, K.; Minkov, G.; Tambuwala, M.M.; Minkov, I.; et al. Green Biologics: Harnessing the Power of Plants to Produce Pharmaceuticals. International Journal of Molecular Sciences 2023, 24, 17575. [Google Scholar] [CrossRef]
- Buyel, J.F. Product safety aspects of plant molecular farming. In Frontiers in Bioengineering and Biotechnology; 2023; pp. 11–2023. [Google Scholar]
- Verdú-Navarro, F.; Moreno-Cid, J.A.; Weiss, J.; Egea-Cortines, M. The advent of plant cells in bioreactors. In Frontiers in Plant Science; 2023; pp. 14–2023. [Google Scholar]
- Wilson, S.A.; Roberts, S.C. Recent advances towards development and commercialization of plant cell culture processes for the synthesis of biomolecules. Plant Biotechnol J. 2012, 10, 249–68. [Google Scholar] [CrossRef] [PubMed]
- Nandi, S.; Kwong, A.T.; Holtz, B.R.; Erwin, R.L.; Marcel, S.; McDonald, K.A. Techno-economic analysis of a transient plant-based platform for monoclonal antibody production. mAbs 2016, 8, 1456–66. [Google Scholar] [CrossRef]
- Wolf, M.W.; Reichl, U. Downstream processing of cell culture-derived virus particles. Expert Rev Vaccines 2011, 10, 1451–75. [Google Scholar] [CrossRef] [PubMed]
- Schillberg, S.; Raven, N.; Spiegel, H.; Rasche, S.; Buntru, M. Critical Analysis of the Commercial Potential of Plants for the Production of Recombinant Proteins. In Frontiers in Plant Science; 2019; pp. 10–2019. [Google Scholar]
- Hemmati, F.; Hemmati-Dinarvand, M.; Karimzade, M.; Rutkowska, D.; Eskandari, M.H.; Khanizadeh, S.; et al. Plant-derived VLP: a worthy platform to produce vaccine against SARS-CoV-2. Biotechnology Letters 2022, 44, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Pillet, S.; Couillard, J.; Trépanier, S.; Poulin, J.-F.; Yassine-Diab, B.; Guy, B.; et al. Immunogenicity and safety of a quadrivalent plant-derived virus like particle influenza vaccine candidate—Two randomized Phase II clinical trials in 18 to 49 and ≥50 years old adults. PLOS ONE 2019, 14, e0216533. [Google Scholar] [CrossRef]
- Ward, B.J.; Makarkov, A.; Séguin, A.; Pillet, S.; Trépanier, S.; Dhaliwall, J.; et al. Efficacy, immunogenicity, and safety of a plant-derived, quadrivalent, virus-like particle influenza vaccine in adults (18–64 years) and older adults (≥65 years): two multicentre, randomised phase 3 trials. The Lancet 2020, 396, 1491–503. [Google Scholar] [CrossRef]
- Muthamilselvan, T.; Khan, M.R.I.; Hwang, I. Assembly of human papillomavirus 16 L1 protein in Nicotiana benthamiana chloroplasts into highly immunogenic virus-like particles. Journal of Plant Biology 2023, 66, 331–40. [Google Scholar] [CrossRef]
- Tacket, C.O.; Mason, H.S.; Losonsky, G.; Estes, M.K.; Levine, M.M.; Arntzen, C.J. Human Immune Responses to a Novel Norwalk Virus Vaccine Delivered in Transgenic Potatoes. The Journal of Infectious Diseases 2000, 182, 302–5. [Google Scholar] [CrossRef]
- Thanavala, Y.; Mahoney, M.; Pal, S.; Scott, A.; Richter, L.; Natarajan, N.; et al. Immunogenicity in humans of an edible vaccine for hepatitis B. Proc Natl Acad Sci U S A 2005, 102, 3378–82. [Google Scholar] [CrossRef]
- Yuki, Y.; Nojima, M.; Kashima, K.; Sugiura, K.; Maruyama, S.; Kurokawa, S.; et al. Oral MucoRice-CTB vaccine is safe and immunogenic in healthy US adults. Vaccine 2022, 40, 3372–9. [Google Scholar] [CrossRef]
- Pêra, F.F.P.G.; Mutepfa, D.L.R.; Khan, A.M.; Els, J.H.; Mbewana, S.; van Dijk, A.A.A.; et al. Engineering and expression of a human rotavirus candidate vaccine in Nicotiana benthamiana. Virology Journal 2015, 12, 205. [Google Scholar] [CrossRef]
- Kurokawa, N.; Lavoie, P.-O.; D’Aoust, M.-A.; Couture, M.M.J.; Dargis, M.; Trépanier, S.; et al. Development and characterization of a plant-derived rotavirus-like particle vaccine. Vaccine 2021, 39, 4979–87. [Google Scholar] [CrossRef]
- Modelska, A.; Dietzschold, B.; Sleysh, N.; Fu, Z.F.; Steplewski, K.; Hooper, D.C.; et al. Immunization against rabies with plant-derived antigen. Proceedings of the National Academy of Sciences 1998, 95, 2481–5. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, S.; Singh, P.K.; Yadav, D.K.; Shahnawaz, M.; Mishra, S.; Sawant, S.V.; et al. High level expression of surface glycoprotein of rabies virus in tobacco leaves and its immunoprotective activity in mice. Journal of Biotechnology 2005, 119, 1–14. [Google Scholar] [CrossRef]
- Park, Y.; Kang, H.; Min, K.; Kim, N.; Park, M.; Ouh, I.-O.; et al. Rabies virus glycoprotein produced in Nicotiana benthamiana is an immunogenic antigen in mice. Czech Journal of Genetics and Plant Breeding 2021, 57, 26–35. [Google Scholar] [CrossRef]
- Phoolcharoen, W.; Bhoo, S.H.; Lai, H.; Ma, J.; Arntzen, C.J.; Chen, Q.; et al. Expression of an immunogenic Ebola immune complex in Nicotiana benthamiana. Plant Biotechnology Journal 2011, 9, 807–16. [Google Scholar] [CrossRef]
- Pang, E.L.; Peyret, H.; Ramirez, A.; Loh, H.-S.; Lai, K.-S.; Fang, C.-M.; et al. Epitope Presentation of Dengue Viral Envelope Glycoprotein Domain III on Hepatitis B Core Protein Virus-Like Particles Produced in Nicotiana benthamiana. In Frontiers in Plant Science; 2019; pp. 10–2019. [Google Scholar]
- Tacket, C.O.; Mason, H.S.; Losonsky, G.; Clements, J.D.; Levine, M.M.; Arntzen, C.J. Immunogenicity in humans of a recombinant bacterial antigen delivered in a transgenic potato. Nature Medicine 1998, 4, 607–9. [Google Scholar] [CrossRef] [PubMed]
- Margolin, E.; Chapman, R.; Meyers, A.E.; van Diepen, M.T.; Ximba, P.; Hermanus, T.; et al. Production and Immunogenicity of Soluble Plant-Produced HIV-1 Subtype C Envelope gp140 Immunogens. In Frontiers in Plant Science; 2019; pp. 10–2019. [Google Scholar]
- Lamphear, B.J.; Streatfield, S.J.; Jilka, J.M.; Brooks, C.A.; Barker, D.K.; Turner, D.D.; et al. Delivery of subunit vaccines in maize seed. Journal of Controlled Release 2002, 85, 169–80. [Google Scholar] [CrossRef] [PubMed]
- Dreesen, I.A.; Charpin-El Hamri, G.; Fussenegger, M. Heat-stable oral alga-based vaccine protects mice from Staphylococcus aureus infection. Journal of biotechnology 2010, 145, 273–80. [Google Scholar] [CrossRef] [PubMed]
- Berndt, A.J.; Smalley, T.N.; Ren, B.; Simkovsky, R.; Badary, A.; Sproles, A.E.; et al. Recombinant production of a functional SARS-CoV-2 spike receptor binding domain in the green algae Chlamydomonas reinhardtii. PLOS ONE 2021, 16, e0257089. [Google Scholar] [CrossRef]
- Govea-Alonso, D.O.; Malla, A.; Bolaños-Martínez, O.C.; Vimolmangkang, S.; Rosales-Mendoza, S. An Algae-Made RBD from SARS-CoV-2 Is Immunogenic in Mice. Pharmaceuticals 2022, 15, 1298. [Google Scholar] [CrossRef]
- Lucas, P.L.; Mathieu-Rivet, E.; Song, P.C.; Oltmanns, A.; Loutelier-Bourhis, C.; Plasson, C.; et al. Multiple xylosyltransferases heterogeneously xylosylate protein N-linked glycans in Chlamydomonas reinhardtii. The Plant Journal 2020, 102, 230–45. [Google Scholar] [CrossRef]
- Sun, M.; Qian, K.; Su, N.; Chang, H.; Liu, J.; Shen, G. Foot-and-mouth disease virus VP1 protein fused with cholera toxin B subunit expressed in Chlamydomonas reinhardtii chloroplast. Biotechnology letters 2003, 25, 1087–92. [Google Scholar] [CrossRef]
- Rout, S.S.; de Grahl, I.; Yu, X.; Reumann, S. Production of a viral surface protein in Nannochloropsis oceanica for fish vaccination against infectious pancreatic necrosis virus. Applied Microbiology and Biotechnology 2022, 106, 6535–49. [Google Scholar] [CrossRef]
- Bañuelos-Hernández, B.; Monreal-Escalante, E.; González-Ortega, O.; Angulo, C.; Rosales-Mendoza, S. Algevir: an expression system for microalgae based on viral vectors. Frontiers in microbiology 2017, 8, 1100. [Google Scholar] [CrossRef] [PubMed]
- Poliner, E.; Takeuchi, T.; Du, Z.-Y.; Benning, C.; Farré, E.M. Nontransgenic Marker-Free Gene Disruption by an Episomal CRISPR System in the Oleaginous Microalga, Nannochloropsis oceanica CCMP1779. ACS Synthetic Biology 2018, 7, 962–8. [Google Scholar] [CrossRef] [PubMed]
- Moroi, K.; Yamamoto, T.; Kurita, T. Double-strand break-free and transgene-free genome editing in the microalga Nannochloropsis oceanica using removable vectors containing the CRISPR base editing system. Scientific Reports 2025, 15, 42431. [Google Scholar] [CrossRef]
- Specht, E.A.; Mayfield, S.P. Algae-based oral recombinant vaccines. In Frontiers in Microbiology; 2014; pp. 5–2014. [Google Scholar]
- Esland, L.; Larrea-Alvarez, M.; Purton, S. Selectable Markers and Reporter Genes for Engineering the Chloroplast of Chlamydomonas reinhardtii. Biology 2018, 7, 46. [Google Scholar] [CrossRef]
- Mathieu-Rivet, E.; Scholz, M.; Arias, C.; Dardelle, F.; Schulze, S.; Le Mauff, F.; et al. Exploring the N-glycosylation Pathway in Chlamydomonas reinhardtii Unravels Novel Complex Structures*. Molecular & Cellular Proteomics 2013, 12, 3160–83. [Google Scholar] [CrossRef]
- Oltmanns, A.; Hoepfner, L.; Scholz, M.; Zinzius, K.; Schulze, S.; Hippler, M. Novel Insights Into N-Glycan Fucosylation and Core Xylosylation in C. reinhardtii. Front Plant Sci. 2019, 10, 1686. [Google Scholar]
- Vanier, G.; Lucas, P.-L.; Loutelier-Bourhis, C.; Vanier, J.; Plasson, C.; Walet-Balieu, M.-L.; et al. Heterologous expression of the N-acetylglucosaminyltransferase I dictates a reinvestigation of the N-glycosylation pathway in Chlamydomonas reinhardtii. Scientific Reports 2017, 7, 10156. [Google Scholar] [CrossRef]
- Waheed, M.T.; Ismail, H.; Gottschamel, J.; Mirza, B.; Lössl, A.G. Plastids: the green frontiers for vaccine production. Frontiers in Plant Science 2015, 6, 1005. [Google Scholar] [CrossRef]
- Pataro, G.; Eslami, E.; Pignataro, F.; Procentese, A. Downstream Processes in a Microalgae Biorefinery: Cascaded Enzymatic Hydrolysis and Pulsed Electric Field as Green Solution. Processes 2025, 13, 1629. [Google Scholar] [CrossRef]
- Wijffels, R.H.; Barbosa, M.J. An outlook on microalgal biofuels. science 2010, 329, 796–9. [Google Scholar] [CrossRef] [PubMed]
- Choi, S. Powering point-of-care diagnostic devices. Biotechnology advances 2016, 34, 321–30. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Wang, Z.; Huang, H. Physical Cell Disruption Technologies for Intracellular Compound Extraction from Microorganisms. Processes 2024, 12, 2059. [Google Scholar] [CrossRef]
- Halim, R.; Rupasinghe, T.W.; Tull, D.L.; Webley, P.A. Mechanical cell disruption for lipid extraction from microalgal biomass. Bioresour Technol. 2013, 140, 53–63. [Google Scholar] [CrossRef]
- Sun, H.; Wang, Y.; He, Y.; Liu, B.; Mou, H.; Chen, F.; et al. Microalgae-derived pigments for the food industry. Marine Drugs 2023, 21, 82. [Google Scholar] [CrossRef]
- Carvalho, S.B.; Freire, J.M.; Moleirinho, M.G.; Monteiro, F.; Gaspar, D.; Castanho, M.A.R.B.; et al. Bioorthogonal Strategy for Bioprocessing of Specific-Site-Functionalized Enveloped Influenza-Virus-Like Particles. Bioconjugate Chemistry 2016, 27, 2386–99. [Google Scholar] [CrossRef]
- Labisch, J.J.; Wiese, G.P.; Pflanz, K. Steric Exclusion Chromatography for Purification of Biomolecules—A Review. Separations 2023, 10, 183. [Google Scholar] [CrossRef]
- Hillebrandt, N.; Hubbuch, J. Size-selective downstream processing of virus particles and non-enveloped virus-like particles. In Frontiers in Bioengineering and Biotechnology; 2023; pp. 11–2023. [Google Scholar]
- Khan, M.F.H.; Baudin, F.; Sudalaiyadum Perumal, A.; Kamen, A.A. Freeze-Drying of mRNA-LNPs Vaccines: A Review. Vaccines 2025, 13, 853. [Google Scholar] [CrossRef]
- Arpagaus, C.; Collenberg, A.; Rütti, D.; Assadpour, E.; Jafari, S.M. Nano spray drying for encapsulation of pharmaceuticals. International Journal of Pharmaceutics 2018, 546, 194–214. [Google Scholar] [CrossRef]
- Kasper, J.C.; Friess, W. The freezing step in lyophilization: physico-chemical fundamentals, freezing methods and consequences on process performance and quality attributes of biopharmaceuticals. Eur J Pharm Biopharm. 2011, 78, 248–63. [Google Scholar] [CrossRef] [PubMed]
- Zou, M.; Lei, C.; Huang, D.; Liu, L.; Han, Y. Application of plant-derived products as adjuvants for immune activation and vaccine development. Vaccine 2024, 42, 126115. [Google Scholar] [CrossRef]
- Lavelle, E.C.; McEntee, C.P. Vaccine adjuvants: Tailoring innate recognition to send the right message. Immunity 2024, 57, 772–89. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Cai, Y.; Jiang, Y.; He, X.; Wei, Y.; Yu, Y.; et al. Vaccine adjuvants: mechanisms and platforms. Signal transduction and targeted therapy 2023, 8, 283. [Google Scholar] [CrossRef]
- Ben-Akiva, E.; Chapman, A.; Mao, T.; Irvine, D.J. Linking vaccine adjuvant mechanisms of action to function. Science Immunology 2025, 10, eado5937. [Google Scholar] [CrossRef] [PubMed]
- Fu, X. Current landscape and challenges in adjuvant and antigen delivery systems for vaccine. Vaccine: X 2025, 100735. [Google Scholar] [CrossRef]
- Chen, G.; Jiang, N.; Zheng, J.; Hu, H.; Yang, H.; Lin, A.; et al. Structural characterization and anti-inflammatory activity of polysaccharides from Astragalus membranaceus. International Journal of Biological Macromolecules 2023, 241, 124386. [Google Scholar] [CrossRef] [PubMed]
- Morais, V.; Suarez, N.; Silveira, F. Methods of saponin purification from Quillaja sp. for vaccine adjuvant production. Frontiers in Natural Products 2025, 3, 1524624. [Google Scholar] [CrossRef]
- Marty-Roix, R.; Vladimer, G.I.; Pouliot, K.; Weng, D.; Buglione-Corbett, R.; West, K.; et al. Identification of QS-21 as an inflammasome-activating molecular component of saponin adjuvants. Journal of Biological Chemistry 2016, 291, 1123–36. [Google Scholar] [CrossRef]
- Coccia, M.; Collignon, C.; Hervé, C.; Chalon, A.; Welsby, I.; Detienne, S.; et al. Cellular and molecular synergy in AS01-adjuvanted vaccines results in an early IFNγ response promoting vaccine immunogenicity. npj Vaccines 2017, 2, 25. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Wang, N.; Zhang, X.; Wang, M.; Liu, Y.; Shi, Y. Potentials of saponins-based adjuvants for nasal vaccines. Frontiers in immunology 2023, 14, 1153042. [Google Scholar] [CrossRef]
- Vn, Mata-Haro; Cekic, C.; Martin, M.; Chilton, P.M.; Casella, C.R.; Mitchell, T.C. The vaccine adjuvant monophosphoryl lipid A as a TRIF-biased agonist of TLR4. Science 2007, 316, 1628–32. [Google Scholar] [CrossRef]
- Joyce, M.G.; King, H.A.; Elakhal-Naouar, I.; Ahmed, A.; Peachman, K.K.; Macedo Cincotta, C.; et al. A SARS-CoV-2 ferritin nanoparticle vaccine elicits protective immune responses in nonhuman primates. Science translational medicine 2021, 14, eabi5735. [Google Scholar] [CrossRef] [PubMed]
- Welsby, I.; Detienne, S.; N’Kuli, F.; Thomas, S.; Wouters, S.; Bechtold, V.; et al. Lysosome-dependent activation of human dendritic cells by the vaccine adjuvant QS-21. Frontiers in immunology 2017, 7, 663. [Google Scholar] [CrossRef]
- Datoo, M.S.; Natama, M.H.; Somé, A.; Traoré, O.; Rouamba, T.; Bellamy, D.; et al. Efficacy of a low-dose candidate malaria vaccine, R21 in adjuvant Matrix-M, with seasonal administration to children in Burkina Faso: a randomised controlled trial. The Lancet 2021, 397, 1809–18. [Google Scholar] [CrossRef]
- Luo, X.; Song, Z.; Zeng, X.; Ye, Y.; Zheng, H.; Cai, D.; et al. A promising self-nanoemulsifying adjuvant with plant-derived saponin D boosts immune response and exerts an anti-tumor effect. Frontiers in Immunology 2023, 14, 1154836. [Google Scholar] [CrossRef]
- Maraskovsky, E.; Schnurr, M.; Wilson, N.S.; Robson, N.C.; Boyle, J.; Drane, D. Development of prophylactic and therapeutic vaccines using the ISCOMATRIX adjuvant. Immunology and cell biology 2009, 87, 371–6. [Google Scholar] [CrossRef]
- Neeland, M.R.; Shi, W.; Collignon, C.; Taubenheim, N.; Meeusen, E.N.; Didierlaurent, A.M.; et al. The lymphatic immune response induced by the adjuvant AS01: a comparison of intramuscular and subcutaneous immunization routes. The Journal of Immunology 2016, 197, 2704–14. [Google Scholar] [CrossRef]
- Cleland, J.L.; Kensil, C.R.; Lim, A.; Jacobsen, N.E.; Basa, L.; Spellman, M.; et al. Isomerization and formulation stability of the vaccine adjuvant QS-21. Journal of pharmaceutical sciences 1996, 85, 22–8. [Google Scholar] [CrossRef] [PubMed]
- Brewer, J.M.; Conacher, M.; Hunter, C.A.; Mohrs, M.; Brombacher, F.; Alexander, J. Aluminium hydroxide adjuvant initiates strong antigen-specific Th2 responses in the absence of IL-4-or IL-13-mediated signaling. The Journal of Immunology 1999, 163, 6448–54. [Google Scholar] [CrossRef] [PubMed]
- HogenEsch, H. Mechanism of immunopotentiation and safety of aluminum adjuvants. Frontiers in immunology 2013, 3, 406. [Google Scholar] [CrossRef]
- Stylianou, V.V.; Bertram, K.M.; Dunn, E.B.; Baharlou, H.; Terre, D.J.; Elhindi, J.; et al. Innate immune cell activation by adjuvant AS01 in human lymph node explants is age independent. The Journal of Clinical Investigation 2024, 134. [Google Scholar] [CrossRef] [PubMed]
- Del Giudice, G.; Rappuoli, R.; Didierlaurent, A.M. Correlates of adjuvanticity: A review on adjuvants in licensed vaccines; Elsevier: Seminars in immunology, 2018; pp. 14–21. [Google Scholar]
- Adam, A.; Lee, C.; Jones, M.C.; Harrington, B.R.; Zou, J.; Peng, B.-H.; et al. VSA-2-, a novel plant-derived adjuvant for SARS-CoV-2 subunit vaccine. Vaccine 2026, 75, 128255. [Google Scholar] [CrossRef]
- Wang, D.; Liu, Y.; Zhao, W. The adjuvant effects on vaccine and the immunomodulatory mechanisms of polysaccharides from traditional Chinese medicine. Frontiers in Molecular Biosciences 2021, 8, 655570. [Google Scholar] [CrossRef]
- Hayashi, M.; Aoshi, T.; Haseda, Y.; Kobiyama, K.; Wijaya, E.; Nakatsu, N.; et al. Advax, a delta inulin microparticle, potentiates in-built adjuvant property of co-administered vaccines. EBioMedicine 2017, 15, 127–36. [Google Scholar] [CrossRef]
- Gordon, D.; Kelley, P.; Heinzel, S.; Cooper, P.; Petrovsky, N. Immunogenicity and safety of Advax™, a novel polysaccharide adjuvant based on delta inulin, when formulated with hepatitis B surface antigen: a randomized controlled Phase 1 study. Vaccine 2014, 32, 6469–77. [Google Scholar] [CrossRef]
- Honda-Okubo, Y.; Vaghasiya, U.; Petrovsky, N. Immunogenicity in mice and non-human primates of an Advax-CpG55. 2-adjuvanted recombinant hemagglutinin seasonal quadrivalent influenza vaccine. Vaccine 2025, 47, 126707. [Google Scholar] [CrossRef]
- Zhao, D.; Chen, X.; Wang, L.; Zhang, J.; Zhao, Z.; Yue, N.; et al. Bidirectional and persistent immunomodulation of Astragalus polysaccharide as an adjuvant of influenza and recombinant SARS-CoV-2 vaccine. International Journal of Biological Macromolecules 2023, 234, 123635. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Wu, B.; Ma, Y.; Liu, X.; Tao, L.; Jia, L.; et al. Astragalus polysaccharides: structure-immunomodulation relationships, multi-target pharmacological activities, and cutting-edge applications in immune modulation. Frontiers in Immunology 2025, 16–2025. [Google Scholar] [CrossRef] [PubMed]
- Leroux-Roels, G.; Marchant, A.; Levy, J.; Van Damme, P.; Schwarz, T.F.; Horsmans, Y.; et al. Impact of adjuvants on CD4+ T cell and B cell responses to a protein antigen vaccine: Results from a phase II, randomized, multicenter trial. Clinical Immunology 2016, 169, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Chen, J.; Wang, L.; Wu, Y.; Kong, W.; Chen, Y.; et al. Astragalus polysaccharide and aluminum adjuvant synergize to amplify immune responses induced by a recombinant COVID-19 vaccine. Human Vaccines & Immunotherapeutics 2025, 21, 2559504. [Google Scholar] [CrossRef]
- Thilakarathna, S.H.; Rupasinghe, H.V. Flavonoid bioavailability and attempts for bioavailability enhancement. Nutrients 2013, 5, 3367–87. [Google Scholar] [CrossRef]
- Wu, J.; Ou, Y.; Yao, M.; Liu, J.; Ran, H.; Wu, Z.; et al. The immunostimulatory activity of Epimedium flavonoids involves toll-like receptor 7/8. Frontiers in Pharmacology 2025, 16, 1514284. [Google Scholar] [CrossRef]
- Mokra, D.; Adamcakova, J.; Mokry, J. Green tea polyphenol (-)-epigallocatechin-3-gallate (EGCG): a time for a new player in the treatment of respiratory diseases? Antioxidants 2022, 11, 1566. [Google Scholar] [CrossRef]
- Javed, Z.; Sadia, H.; Iqbal, M.J.; Shamas, S.; Malik, K.; Ahmed, R.; et al. Apigenin role as cell-signaling pathways modulator: implications in cancer prevention and treatment. Cancer Cell International 2021, 21, 189. [Google Scholar] [CrossRef]
- Prigent, S.V.; Gruppen, H.; Visser, A.J.; Van Koningsveld, G.A.; De Jong, G.A.; Voragen, A.G. Effects of non-covalent interactions with 5-O-caffeoylquinic acid (chlorogenic acid) on the heat denaturation and solubility of globular proteins. Journal of Agricultural and Food Chemistry 2003, 51, 5088–95. [Google Scholar] [CrossRef]
- Hoskin, D.W.; Coombs, M.R.P. Immune modulation by flavonoids; Frontiers Media SA, 2022; p. 899577. [Google Scholar]
- O’Hagan, D.T.; van der Most, R.; Lodaya, R.N.; Coccia, M.; Lofano, G. “World in motion”–emulsion adjuvants rising to meet the pandemic challenges. npj Vaccines 2021, 6, 158. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, A.L.; Kazmin, D.; Napolitani, G.; Clutterbuck, E.A.; Pulendran, B.; Siegrist, C.-A.; et al. AS03-and MF59-adjuvanted influenza vaccines in children. Frontiers in immunology 2017, 8, 1760. [Google Scholar] [CrossRef] [PubMed]
- O’Hagan, D.T.; Ott, G.S.; De Gregorio, E.; Seubert, A. The mechanism of action of MF59 – An innately attractive adjuvant formulation. Vaccine 2012, 30, 4341–8. [Google Scholar] [CrossRef]
- Ko, E.-J.; Kang, S.-M. Immunology and efficacy of MF59-adjuvanted vaccines. Human Vaccines & Immunotherapeutics 2018, 14, 3041–5. [Google Scholar] [CrossRef]
- Morel, S.; Didierlaurent, A.; Bourguignon, P.; Delhaye, S.; Baras, B.; Jacob, V.; et al. Adjuvant System AS03 containing α-tocopherol modulates innate immune response and leads to improved adaptive immunity. Vaccine 2011, 29, 2461–73. [Google Scholar] [CrossRef]
- Pulendran, B.; S. Arunachalam, P.; O’Hagan, D.T. Emerging concepts in the science of vaccine adjuvants. Nature Reviews Drug Discovery 2021, 20, 454–75. [Google Scholar] [CrossRef]
- Mendes, A.; Azevedo-Silva, J.; Fernandes, J.C. From Sharks to Yeasts: Squalene in the Development of Vaccine Adjuvants. Pharmaceuticals 2022, 15, 265. [Google Scholar] [CrossRef]
- Gohil, N.; Bhattacharjee, G.; Khambhati, K.; Braddick, D.; Singh, V. Engineering Strategies in Microorganisms for the Enhanced Production of Squalene: Advances, Challenges and Opportunities. In Frontiers in Bioengineering and Biotechnology; 2019; pp. 7–2019. [Google Scholar]
- O’Hagan, D.T.; Ott, G.S.; Nest, G.V.; Rappuoli, R.; Giudice, G.D. The history of MF59® adjuvant: a phoenix that arose from the ashes. Expert review of vaccines 2013, 12, 13–30. [Google Scholar] [CrossRef] [PubMed]
- Garçon, N.; Vaughn, D.W.; Didierlaurent, A.M. Development and evaluation of AS03, an Adjuvant System containing α-tocopherol and squalene in an oil-in-water emulsion. Expert review of vaccines 2012, 11, 349–66. [Google Scholar] [CrossRef]
- Xiao, W.; Qin, S.; Li, X.; Jin, X.; Zhu, A.; He, X.; et al. Tomatine as a versatile adjuvant boosts mRNA vaccine responses. Materials Today Bio 2025, 35, 102360. [Google Scholar] [CrossRef]
- Wang, K.; Yin, J.; Chen, J.; Ma, J.; Si, H.; Xia, D. Inhibition of inflammation by berberine: Molecular mechanism and network pharmacology analysis. Phytomedicine 2024, 128, 155258. [Google Scholar] [CrossRef] [PubMed]
- Stojanović-Radić, Z.; Pejčić, M.; Dimitrijević, M.; Aleksić, A.; V. Anil Kumar, N.; Salehi, B.; et al. Piperine-A Major Principle of Black Pepper: A Review of Its Bioactivity and Studies. Applied Sciences 2019, 9, 4270. [Google Scholar] [CrossRef]
- Reyna-Margarita, H.-R.; Irais, C.-M.; Mario-Alberto, R.-G.; Agustina, R.-M.; Luis-Benjamín, S.-G.; David, P.-E. Plant Phenolics and Lectins as Vaccine Adjuvants. Current Pharmaceutical Biotechnology 2019, 20, 1236–43. [Google Scholar] [CrossRef]
- da Silva, T.A.; Mariano, V.S.; Sardinha-Silva, A.; de Souza, M.A.; Mineo, T.W.P.; Roque-Barreira, M.C. IL-17 Induction by ArtinM is Due to Stimulation of IL-23 and IL-1 Release and/or Interaction with CD3 in CD4+ T Cells. PLOS ONE 2016, 11, e0149721. [Google Scholar] [CrossRef]
- Diesner, S.C.; Wang, X.-Y.; Jensen-Jarolim, E.; Untersmayr, E.; Gabor, F. Use of lectin-functionalized Particles for Oral Immunotherapy. Therapeutic Delivery 2012, 3, 277–90. [Google Scholar] [CrossRef] [PubMed]
- Rybicki, E.P. Plant-based vaccines against viruses. Virology journal 2014, 11, 205. [Google Scholar] [CrossRef]
- Lebel M-È; Chartrand, K.; Leclerc, D.; Lamarre, A. Plant viruses as nanoparticle-based vaccines and adjuvants. Vaccines 2015, 3, 620–37. [Google Scholar] [CrossRef]
- Lico, C.; Schoubben, A.; Baschieri, S.; Blasi, P.; Santi, L. Nanoparticles in biomedicine: new insights from plant viruses. Current medicinal chemistry 2013, 20, 3471–87. [Google Scholar] [CrossRef]
- Röder, J.; Dickmeis, C.; Commandeur, U. Small, smaller, nano: New applications for potato virus X in nanotechnology. Frontiers in Plant Science 2019, 10, 158. [Google Scholar] [CrossRef]
- Nikitin, N.; Vasiliev, Y.; Kovalenko, A.; Ryabchevskaya, E.; Kondakova, O.; Evtushenko, E.; et al. Plant viruses as adjuvants for next-generation vaccines and immunotherapy. Vaccines 2023, 11, 1372. [Google Scholar] [CrossRef] [PubMed]
- Pifferi, G.; Restani, P. The safety of pharmaceutical excipients. Il Farmaco 2003, 58, 541–50. [Google Scholar] [CrossRef] [PubMed]
- Zafar, S.; Akhtar, A.; Sayed, E.; Onaiwu, E.; Arshad, M.S.; Ahmad, Z. Vaccine formulation design: challenges and opportunities. RSC Pharmaceutics 2025, 2, 490–516. [Google Scholar] [CrossRef]
- Petrovsky, N.; Cooper, P.D. Advax™, a novel microcrystalline polysaccharide particle engineered from delta inulin, provides robust adjuvant potency together with tolerability and safety. Vaccine 2015, 33, 5920–6. [Google Scholar] [CrossRef]
- Zhou, L.; Liu, Z.; Wang, Z.; Yu, S.; Long, T.; Zhou, X.; et al. Astragalus polysaccharides exerts immunomodulatory effects via TLR4-mediated MyD88-dependent signaling pathway in vitro and in vivo. Scientific reports 2017, 7, 44822. [Google Scholar] [CrossRef]
- Zhong, X.; Wang, G.; Li, F.; Fang, S.; Zhou, S.; Ishiwata, A.; et al. Immunomodulatory effect and biological significance of β-glucans. Pharmaceutics 2023, 15, 1615. [Google Scholar] [CrossRef]
- Holland, C.; Ryden, P.; Edwards, C.H.; Grundy, M.M.-L. Plant cell walls: Impact on nutrient bioaccessibility and digestibility. Foods 2020, 9, 201. [Google Scholar] [CrossRef]
- Kwon, K.-C.; Verma, D.; Singh, N.D.; Herzog, R.; Daniell, H. Oral delivery of human biopharmaceuticals, autoantigens and vaccine antigens bioencapsulated in plant cells. Advanced drug delivery reviews 2013, 65, 782–99. [Google Scholar] [CrossRef] [PubMed]
- Cerovic, V.; Pabst, O.; Mowat, A.M. The renaissance of oral tolerance: merging tradition and new insights. Nature Reviews Immunology 2025, 25, 42–56. [Google Scholar] [CrossRef]
- Mörbe, U.M.; Jørgensen, P.B.; Fenton, T.M.; von Burg, N.; Riis, L.B.; Spencer, J.; et al. Human gut-associated lymphoid tissues (GALT); diversity, structure, and function. Mucosal immunology 2021, 14, 793–802. [Google Scholar] [CrossRef]
- Rezende, R.M.; Weiner, H.L. Oral tolerance: an updated review. Immunology Letters 2022, 245, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Rybicki, E.P. Plant-made vaccines for humans and animals. Plant biotechnology journal 2010, 8, 620–37. [Google Scholar] [CrossRef]
- Shahid, N.; Daniell, H. Plant-based oral vaccines against zoonotic and non-zoonotic diseases. Plant biotechnology journal. 2016, 14, 2079–99. [Google Scholar] [CrossRef]
- Singh, L.; Jain, H.; Jaswal, S.; Patel, R.; Banjare, P. Plant-Based Oral Vaccines: Molecular Biotechnology Approaches Toward Functional Food-Based Immunization. In Molecular Biotechnology; 2026. [Google Scholar]
- Mason, H.S. Plant-based vaccines: Expression and oral immunogenicity. In Vitro Cellular & Developmental Biology - Plant. 2002, 38, 237–40. [Google Scholar]
- Pudhuvai, B.; Koul, B.; Mishra, A.K. Insights into the world of edible vaccines: From lab to reality. Current Research in Biotechnology 2025, 9, 100290. [Google Scholar] [CrossRef]
- Lamphear, B.J.; Jilka, J.M.; Kesl, L.; Welter, M.; Howard, J.A.; Streatfield, S.J. A corn-based delivery system for animal vaccines: an oral transmissible gastroenteritis virus vaccine boosts lactogenic immunity in swine. Vaccine 2004, 22, 2420–4. [Google Scholar] [CrossRef]
- Kapusta, J.; Modelska, A.; Figlerowicz, M.; Pniewski, T.; Letellier, M.; Lisowa, O.; et al. A plant-derived edible vaccine against hepatitis B virus. Faseb j. 1999, 13, 1796–9. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.B.S.; Ganapathi, T.R.; Revathi, C.J.; Srinivas, L.; Bapat, V.A. Expression of hepatitis B surface antigen in transgenic banana plants. Planta 2005, 222, 484–93. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Qin, S.; Li, K.; Yin, X.; Sun, D.; Chang, J. Rotavirus Reverse Genetics Systems and Oral Vaccine Delivery Vectors for Mucosal Vaccination. Microorganisms 2025, 13, 1579. [Google Scholar] [CrossRef] [PubMed]
- Lavelle, E.C.; Ward, R.W. Mucosal vaccines—fortifying the frontiers. Nature Reviews Immunology 2022, 22, 236–50. [Google Scholar] [CrossRef]
- Sinha, D.; Gopalakrishna, P.K.; Paul, S.; Longet, S. A user’s guide to designing efficient and safe mucosal vaccines: Challenges & potentials. Oxford Open Immunology 2025, 6, iqaf007. [Google Scholar] [CrossRef]
- Hou, Y.; Li, J.; Wu, Y. Modulation of oral vaccine efficacy by the gut microbiota. npj Vaccines 2025, 10, 179. [Google Scholar] [CrossRef]
- Sahoo, A.; Mandal, A.K.; Dwivedi, K.; Kumar, V. A cross talk between the immunization and edible vaccine: Current challenges and future prospects. Life Sciences 2020, 261, 118343. [Google Scholar] [CrossRef]
- Mayer, L.; Shao, L. Therapeutic potential of oral tolerance. Nature Reviews Immunology 2004, 4, 407–19. [Google Scholar] [CrossRef]
- Mbongue, J.C.; Vanterpool, E.; Langridge, W.H.R. Exploring the Potential of Plant-Based CTB-INS Oral Vaccines in Treating Type 1 Diabetes. Immuno 2023, 3, 217–27. [Google Scholar] [CrossRef]
- Azizi, A.; Kumar, A.; Diaz-Mitoma, F.; Mestecky, J. Enhancing Oral Vaccine Potency by Targeting Intestinal M Cells. PLOS Pathogens 2010, 6, e1001147. [Google Scholar] [CrossRef]
- Kobayashi, N.; Takahashi, D.; Takano, S.; Kimura, S.; Hase, K. The Roles of Peyer’s Patches and Microfold Cells in the Gut Immune System: Relevance to Autoimmune Diseases. In Frontiers in Immunology; 2019; pp. 10–2019. [Google Scholar]
- Han, J.; Zhang, J.; Hu, L.; Wang, C.; Wang, S.; Miao, G. Chloroplast display of subunit vaccines and their efficacy via oral administration. International Journal of Biological Macromolecules 2024, 258, 129125. [Google Scholar] [CrossRef]
- Rosales-Mendoza, S.; Nieto-Gómez, R. Green Therapeutic Biocapsules: Using Plant Cells to Orally Deliver Biopharmaceuticals. Trends in Biotechnology 2018, 36, 1054–67. [Google Scholar] [CrossRef] [PubMed]
- Streatfield, S.J. Approaches to achieve high-level heterologous protein production in plants. Plant biotechnology journal 2007, 5, 2–15. [Google Scholar] [CrossRef]
- Tusé, D.; Tu, T.; McDonald, K.A. Manufacturing economics of plant-made biologics: case studies in therapeutic and industrial enzymes. BioMed research international 2014, 2014, 256135. [Google Scholar] [CrossRef] [PubMed]
- Kwon, K.-C.; Daniell, H. Low-cost oral delivery of protein drugs bioencapsulated in plant cells. Plant biotechnology journal 2015, 13, 1017. [Google Scholar] [CrossRef] [PubMed]
- Fischer, R.; Schillberg, S.; F Buyel, J.; M Twyman, R. Commercial aspects of pharmaceutical protein production in plants. Current pharmaceutical design 2013, 19, 5471–7. [Google Scholar] [CrossRef]
- Daniell, H.; Singh, N.D.; Mason, H.; Streatfield, S.J. Plant-made vaccine antigens and biopharmaceuticals. Trends in Plant Science 2009, 14, 669–79. [Google Scholar] [CrossRef] [PubMed]
- Mowat, A.M. Anatomical basis of tolerance and immunity to intestinal antigens. Nature Reviews Immunology 2003, 3, 331–41. [Google Scholar] [CrossRef] [PubMed]
- Diao, H.-P.; Guo, Y.-F.; Chen, Y.; Hwang, I.; Song, S.-J. Plant-derived vaccines: Advances in delivery approaches. Plant Communications 2026, 7, 101676. [Google Scholar] [CrossRef]
- Marciani, D.J. Elucidating the Mechanisms of Action of Saponin-Derived Adjuvants. Trends in Pharmacological Sciences 2018, 39, 573–85. [Google Scholar] [CrossRef]
- Eidenberger, L.; Kogelmann, B.; Steinkellner, H. Plant-based biopharmaceutical engineering. Nature Reviews Bioengineering 2023, 1, 426–39. [Google Scholar] [CrossRef]
- Taylor, L.E., II; Dai, Z.; Decker, S.R.; Brunecky, R.; Adney, W.S.; Ding, S.-Y.; et al. Heterologous expression of glycosyl hydrolases in planta: a new departure for biofuels. Trends in Biotechnology 2008, 26, 413–24. [Google Scholar] [CrossRef]
- Kopertekh, L.; Wilhelm, R. Biosafety, Risk Analysis, and Regulatory Framework for Molecular Farming in Europe. In Tools & Techniques of Plant Molecular Farming; Kole, C., Chaurasia, A., Hefferon, K.L., Panigrahi, J., Eds.; Springer Nature Singapore: Singapore, 2023; pp. 359–81. [Google Scholar]
- Buyel, J.F. Plant Molecular Farming – Integration and Exploitation of Side Streams to Achieve Sustainable Biomanufacturing. In Frontiers in Plant Science; 2019; pp. 9–2018. [Google Scholar]
- Sparrow, P.A.C. GM Risk Assessment. Molecular Biotechnology 2010, 44, 267–75. [Google Scholar] [CrossRef] [PubMed]
- Fausther-Bovendo, H.; Kobinger, G. Plant-made vaccines and therapeutics. Science 2021, 373, 740–1. [Google Scholar] [CrossRef]
| Pathogen/Vaccine Platform | Antigen/Product Description | Plant Host/Expression Organism | Expression Technology/Method | Platform type | Regulator y Status/Use in Humans | References |
|---|---|---|---|---|---|---|
| SARS-CoV-2 (COVID-19) | Spike (S) protein VLP (CoVLP/Covifenz®) | Nicotiana benthamiana | Transient expression (Agrobacterium-mediated agroinfiltration) | VLP | Authorized for human use in Canada (Covifenz®); authorization was later cancelled by the sponsor (March 31, 2023) | [103] |
| Influenza (Seasonal & Pandemic) | Hemagglutinin (HA)/Quadrivalent Virus-Like Particles (QVLP) | Nicotiana benthamiana | Transient expression (Agrobacterium-mediated) | VLP | Advanced Clinical Trials — Phase III completed (efficacy trials conducted) | [104,105] |
| Human Papillomavirus (HPV) | L1 capsid protein VLPs | Nicotiana benthamiana | Transient expression | VLP | Preclinical / Early clinical | [106] |
| Norovirus | Norwalk virus capsid protein (NVCP) | Potato (Solanum tuberosum) | Stable transformation (nuclear) | Edible/Oral | Early human trials (oral immunogenicity) | [107] |
| Hepatitis B | Hepatitis B surface antigen (HBsAg) expressed in plants | Potato (Solanum tuberosum), Lettuce (Lactuca sativa, edible); Nicotiana tabacum (non-edible research host) | Stable transformation (nuclear) | Edible/Oral | Early clinical studies (Phase I/II oral immunogenicity), but no licensed product | [108] |
| Cholera | Cholera toxin B subunit (CTB) expressed in MucoRice-CTB (rice) | Oryza sativa (rice) MucoRice-CTB | Stable transformation (seed-based) | Edible/Oral | Phase I clinical trial (oral delivery) completed | [109] |
| Rotavirus | VP6/VP7 antigens (plant-expressed) | Nicotiana spp. | Transient/stable expression | Subunit | Preclinical | [110,111] |
| Rabies | Rabies virus glycoprotein (G protein) (plant-produced antigen/chimeric constructs) | Spinach (Spinacia oleracea, chimeric plant virus vaccine), Nicotiana benthamiana, Nicotiana tabacum | Transient expression & stable/transgenic expression approaches | Subunit/Oral | Preclinical/Early human exploratory immunogenicity (Phase I-like oral feeding studies) — no licensed product | [112,113,114] |
| Ebola Virus | Ebola Immune Complex (EIC) based on Ebola glycoprotein GP1 fused to an antibody scaffold | Nicotiana benthamiana | Transient expression (geminiviral replicon vectors) | Immune complex | Preclinical — immunogenic in mice; plant system also used for ZMapp mAbs (therapeutic antibodies), but no licensed plant-derived vaccine | [115] |
| Dengue Virus | Envelope protein (E) / domain III constructs | Nicotiana benthamiana | Transient expression | Subunit | Preclinical | [116] |
| Enterotoxigenic E. coli (ETEC) | Heat-labile enterotoxin B subunit (LTB) | Transgenic maize (Zea mays), Transgenic potato (Solanum tuberosum) | Stable transformation (nuclear) | Edible / Oral | Early human oral immunogenicity studies (Phase I-like / exploratory) — no licensed product | [117] |
| HIV | gp120 / gp140 glycoproteins | Nicotiana benthamiana | Glycoengineered transient expression | Subunit | Preclinical | [118] |
|
Veterinary (TGEV) |
Coronavirus antigen (TGEV) | Transgenic maize | Stable expression (seed-based) | Edible (feed-based) | Veterinary use (preclinical/field studies) | [119] |
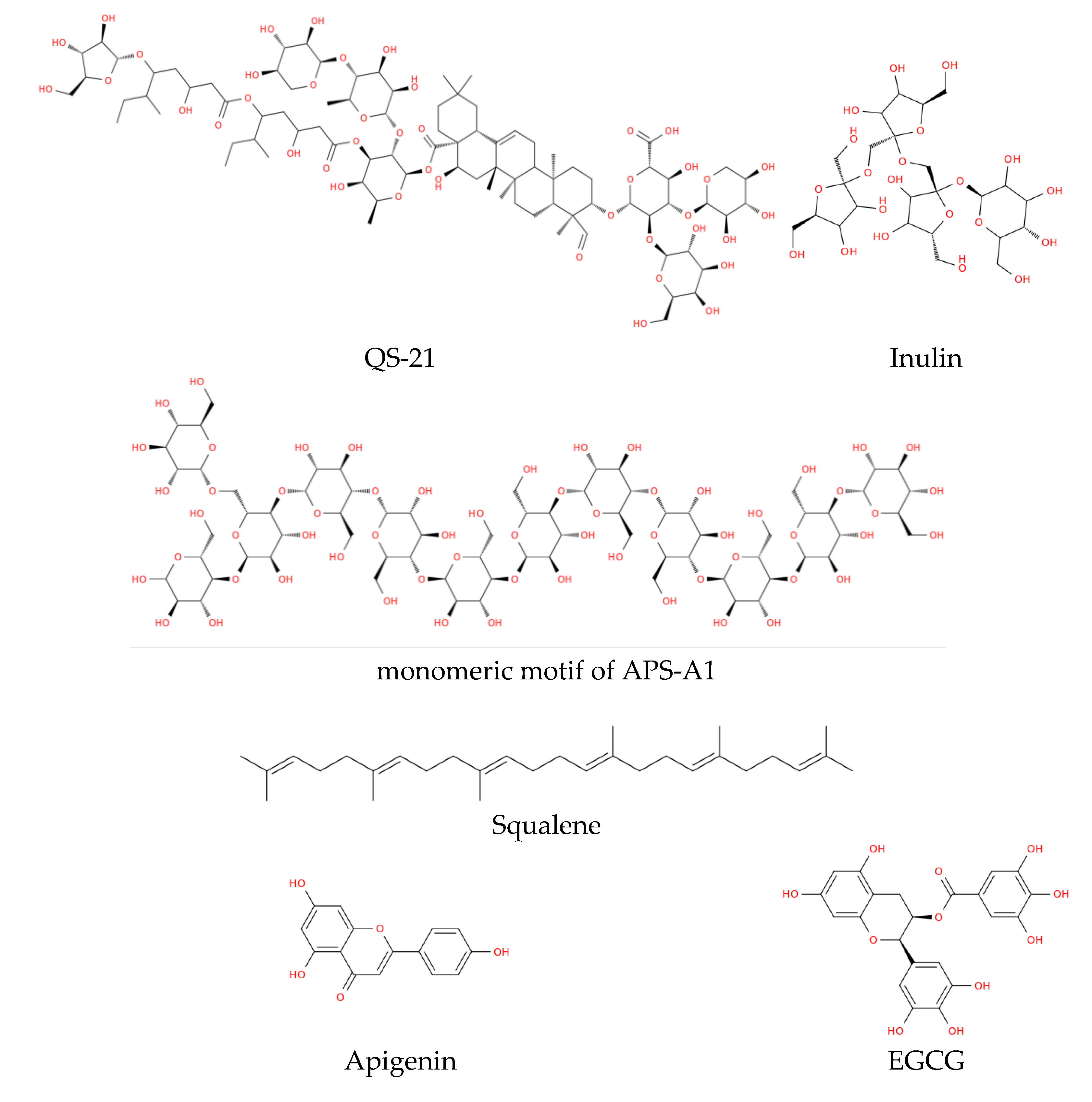
| Category | Primary Example | Botanical Source | Target Receptor/Mechanism | Clinical Status/Application | Ref. |
|---|---|---|---|---|---|
| Saponins | QS-21 | Quillaja saponaria | NLRP3 Inflammasome, Syk kinase | Licensed (Shingrix, Mosquirix, Arexvy) | [154] |
| Saponins | ISCOMs / ISCOMATRIX | Quillaja saponaria | Antigen co-delivery, MHC I/II presentation | Clinical / Advanced development | [162,163] |
| Saponins | Matrix-M™ | Quillaja saponaria | DC recruitment, Th1/Th2 balance | Licensed (Nuvaxovid, R21) |
[160] |
| Saponins | Quil-A | Quillaja saponaria | Membrane permeabilization, immune activation | Preclinical / Veterinary use | [153] |
| Saponins | VSA-2 | Quillaja saponaria | Enhanced Th1 response, improved stability | Preclinical (SARS-CoV-2 models) | [169] |
| Polysaccharides | Advax™ (delta inulin) | Dahlia variabilis | Alternative pathway (Non-inflammasome) | Clinical Trials (HBV, Influenza) | [207] |
| Polysaccharides | APS | Astragalus membranaceus | TLR4, MyD88- NF-κB pathway, Dendritic cell maturation | Preclinical (Synergy with Alum) | [208] |
| Polysaccharides | β-glucans | Various plants/fungi | Dectin-1, complement activation | Preclinical / Immunomodulation | [209] |
| Terpenoids | Squalene (MF59) |
Shark-derived, Amaranthus caudatus (Alternative) | Chemokine release (CCL2, CXCL8) | Licensed (MF59, Fluad) |
[184,185,186,187] |
| Terpenoids | Squalene (AS03) |
Plant-derived / mixed sources | Enhanced innate activation (α-tocopherol synergy) | Licensed (Pandemrix, Arepanrix) | [185,188] |
| Flavonoids | EGCG | Camellia sinensis | TLR7/8, PI3K/Akt/mTOR | Preclinical/ Cancer research | [180,181] |
| Flavonoids | Apigenin | Various plants | PI3K/Akt/mTOR, MAPK pathways | Preclinical | [181] |
| Flavonoids | Epimedium flavonoids | Epimedium spp. | TLR7/8 activation | Experimental | [179] |
| Alkaloids | Tomatine | Solanum lycopersicum | Membrane disruption, LNP stabilizer | Preclinical (mRNA platforms) | [194] |
| Alkaloids | Berberine | Berberis spp. | NF-κB, mTOR modulation | Preclinical / adjunct therapy | [195] |
| Alkaloids | Piperine | Piper nigrum | Bioavailability enhancement | Experimental | [196] |
| Lectins | ArtinM | Artocarpus integrifolia | IL-12 induction, Th1 polarization | Preclinical | [198] |
| Lectins | UEA-1 | Ulex europaeus | M-cell targeting, mucosal delivery | Experimental | [199] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




