Submitted:
11 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
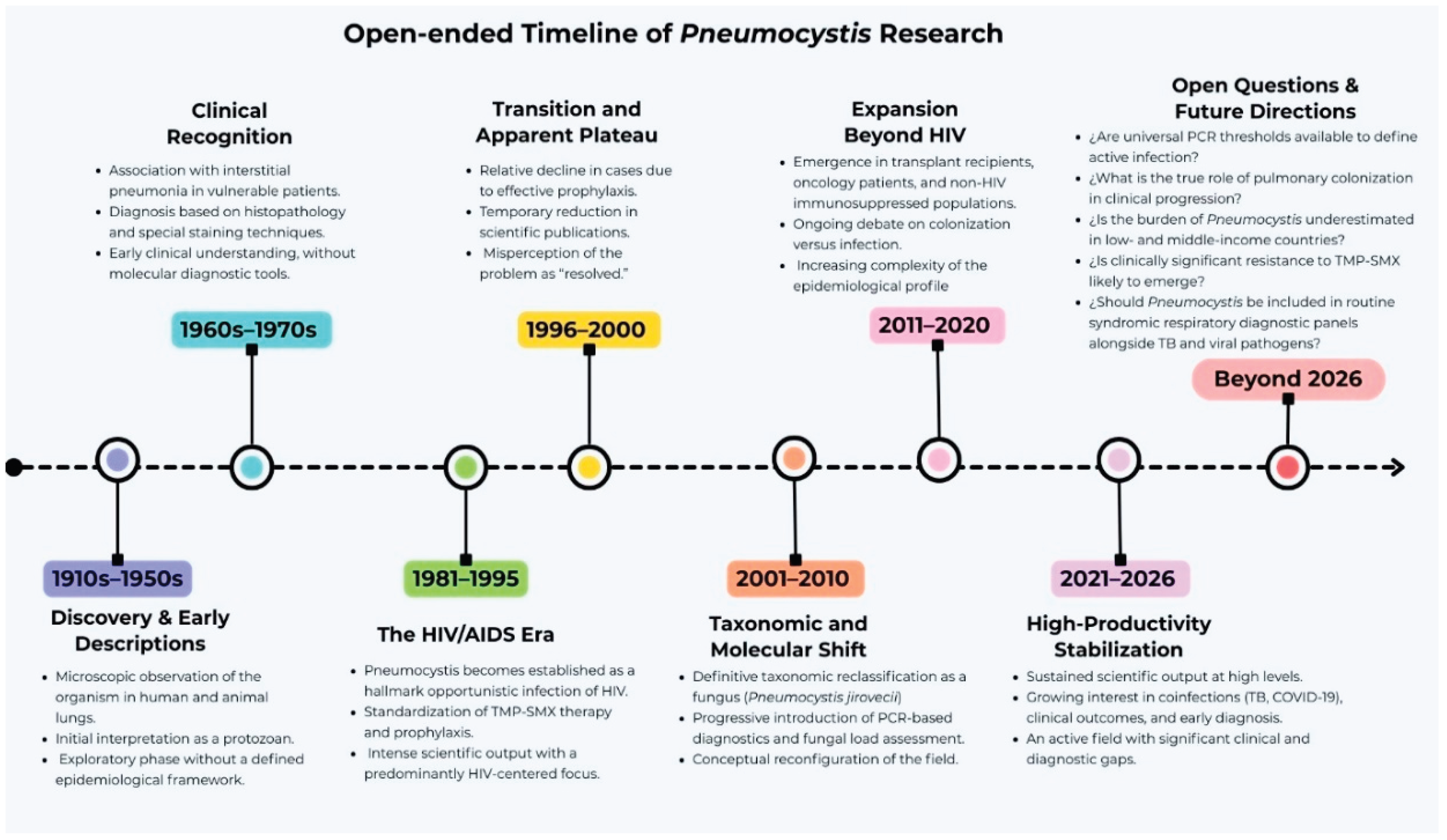
1. Introduction
2. Materials and Methods
2.1. Data Source and Bibliographic Time Window
2.2. Search Strategy
2.3. Bibliometric Data Extraction
- ▪ Productivity indicators: year of publication, document type, source title, and subject area.
- ▪ Collaboration indicators: authorship information, institutional affiliations, and countries/regions, used to assess co-authorship patterns.
- ▪ Thematic indicators: author keywords and indexed keywords (when available), used for co-occurrence analysis and thematic mapping.
- ▪ Source indicators: leading journals/sources based on publication volume and citation impact, according to citation counts provided by Scopus.
2.4. Bibliometric Data Analysis
- ▪ Retrieval of records from Scopus using the predefined query and filters.
- ▪ Export of the resulting dataset (9 February 2026).
- ▪ Descriptive analysis of publication and citation patterns.
- ▪ Network visualization through scientific mapping.
2.5. Comparative Bibliometric Analysis of Major Pneumonia-Associated Pathogens (2001–2025)
3. Results and Discussion
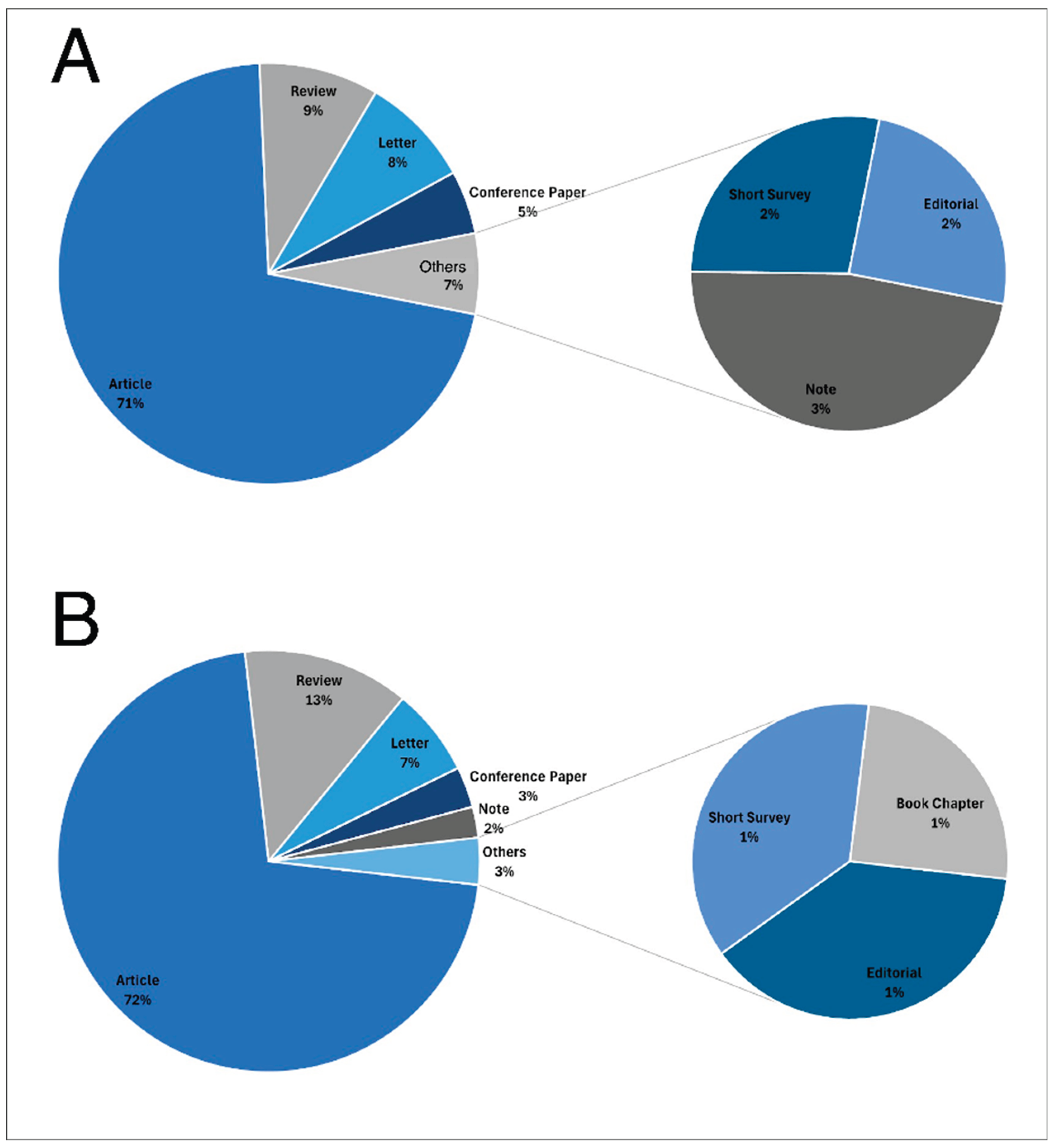
3.1. Publication Output and Document Types
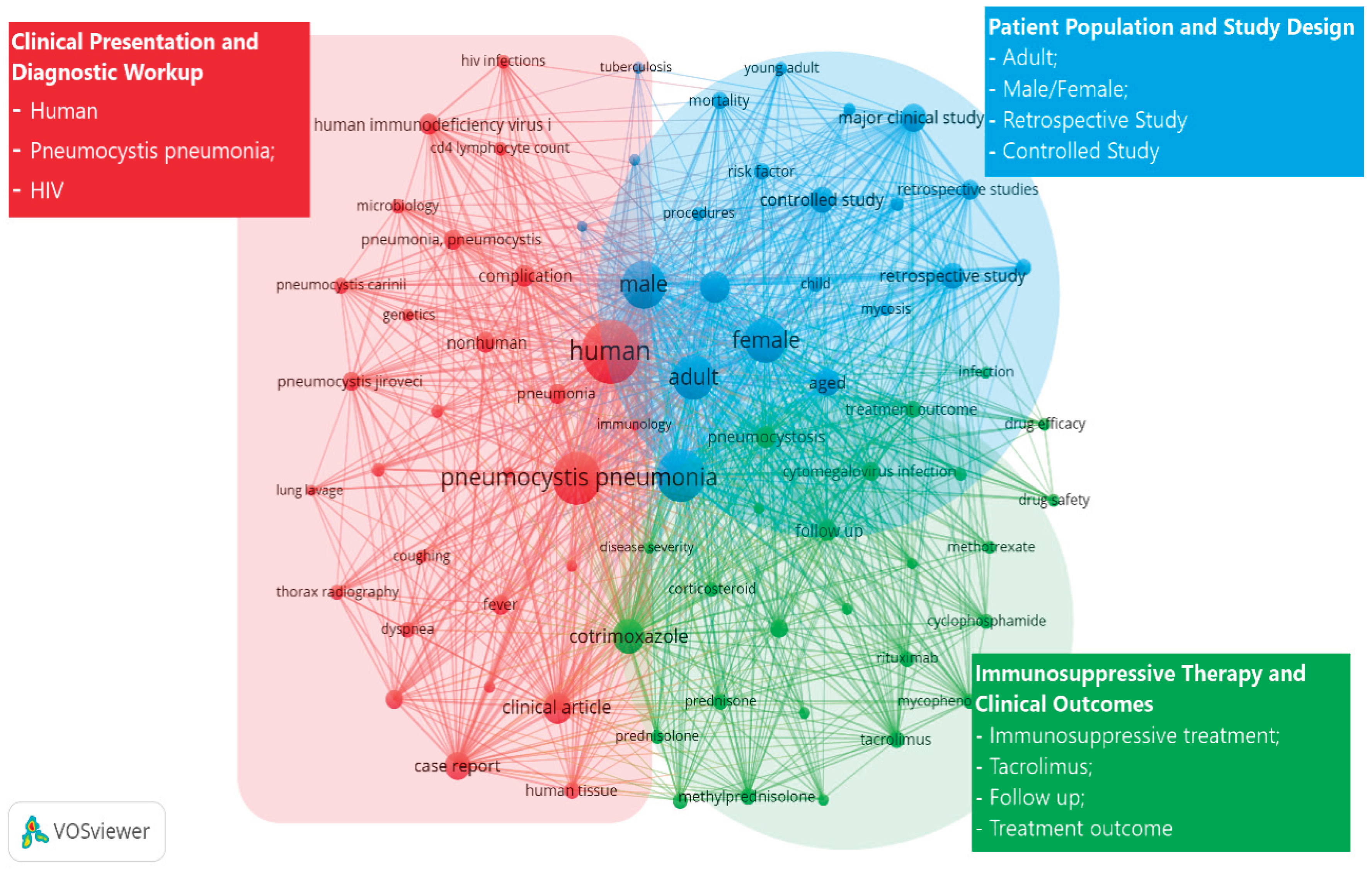
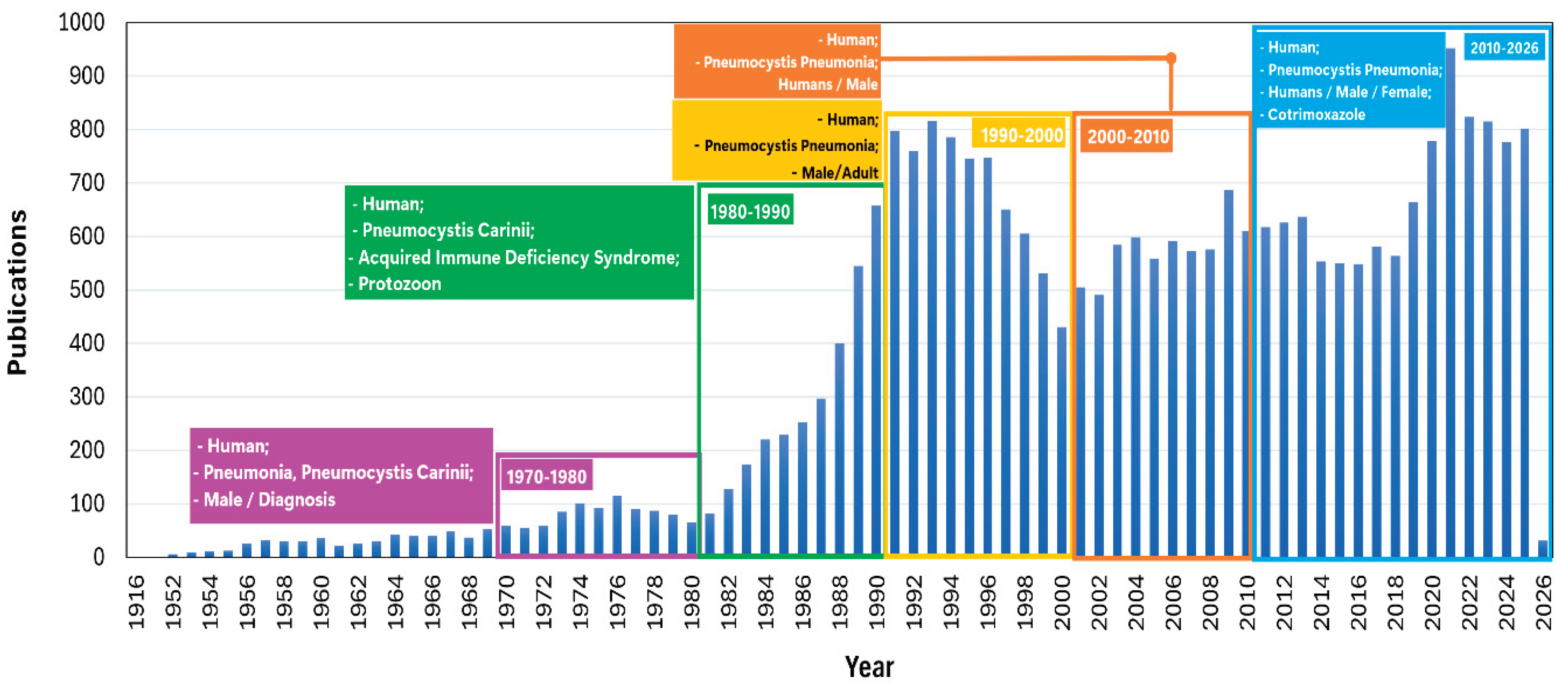
3.2. Publications by Year and the More Cited Keywords
3.3. Authors
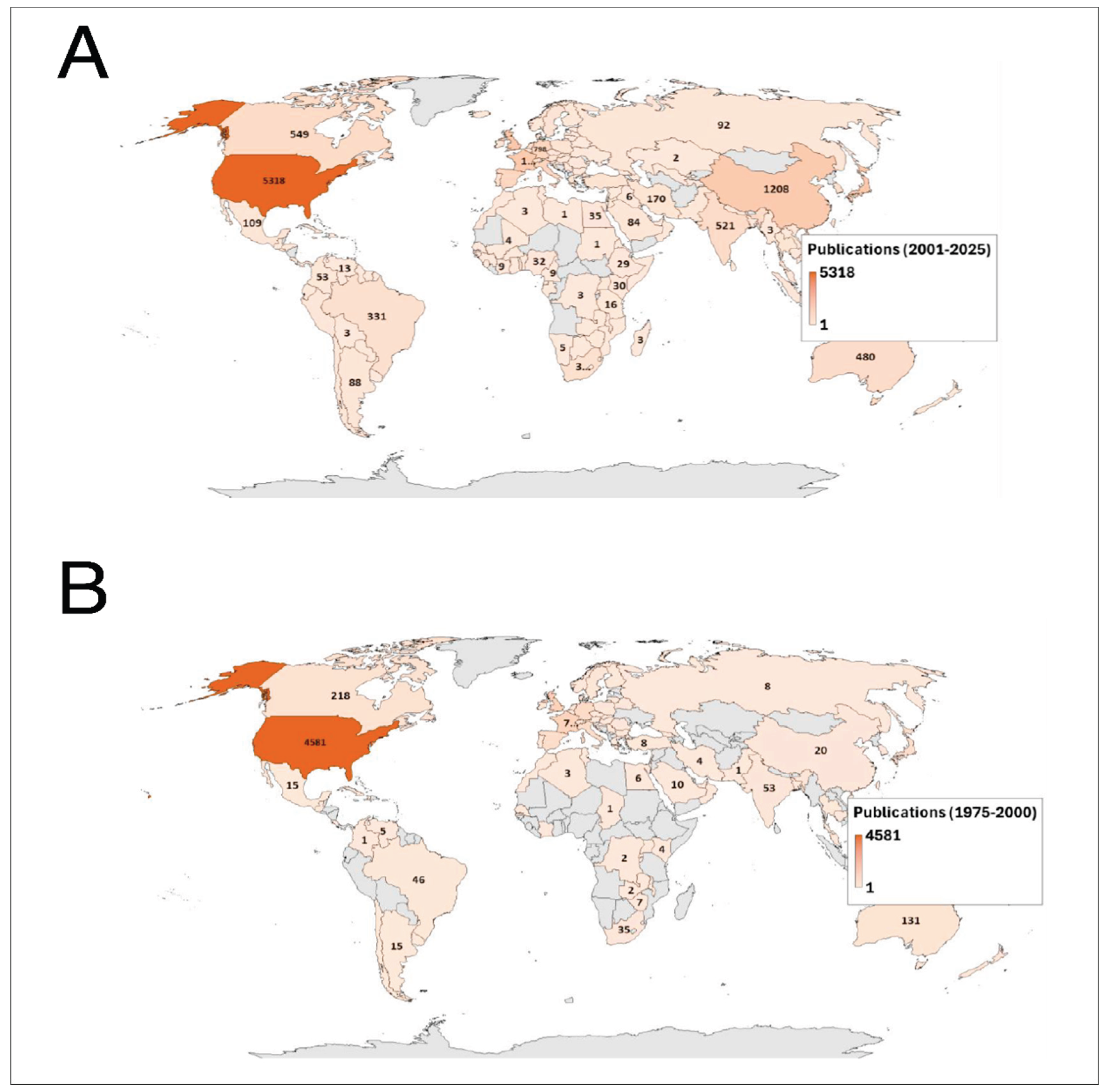
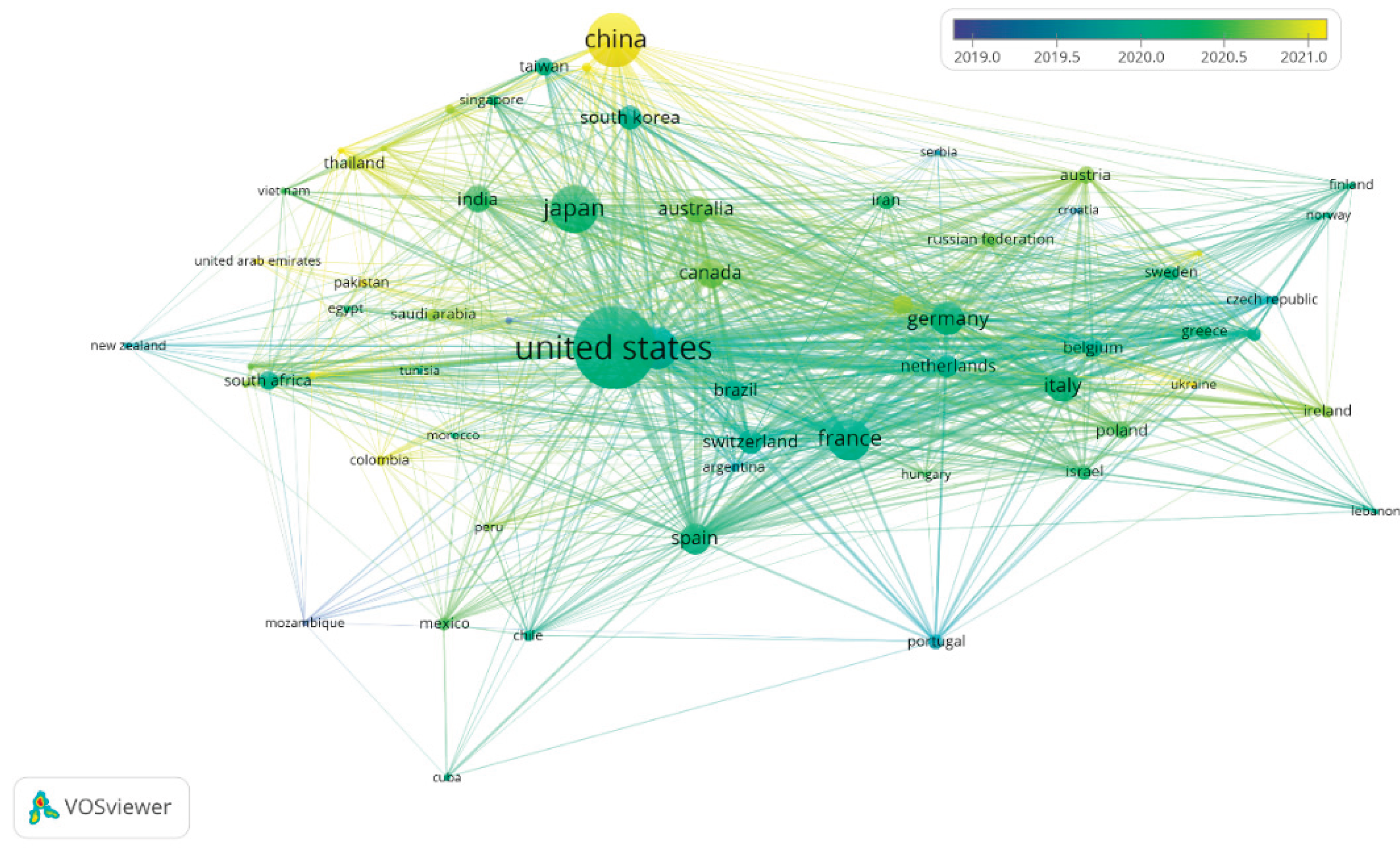
3.4. Most Productive Countries
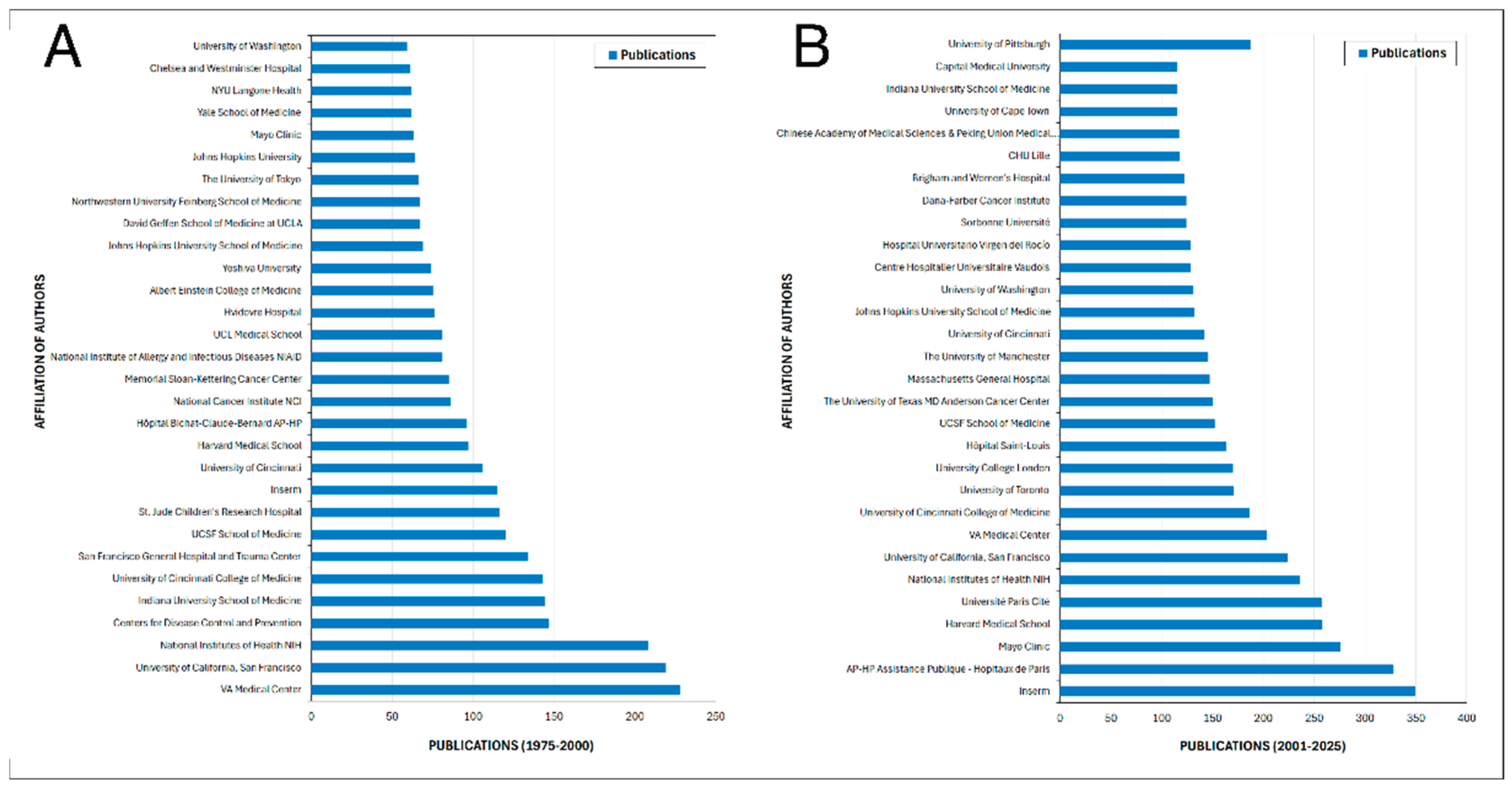
3.5. Most Productive Institutions
3.6. Journals
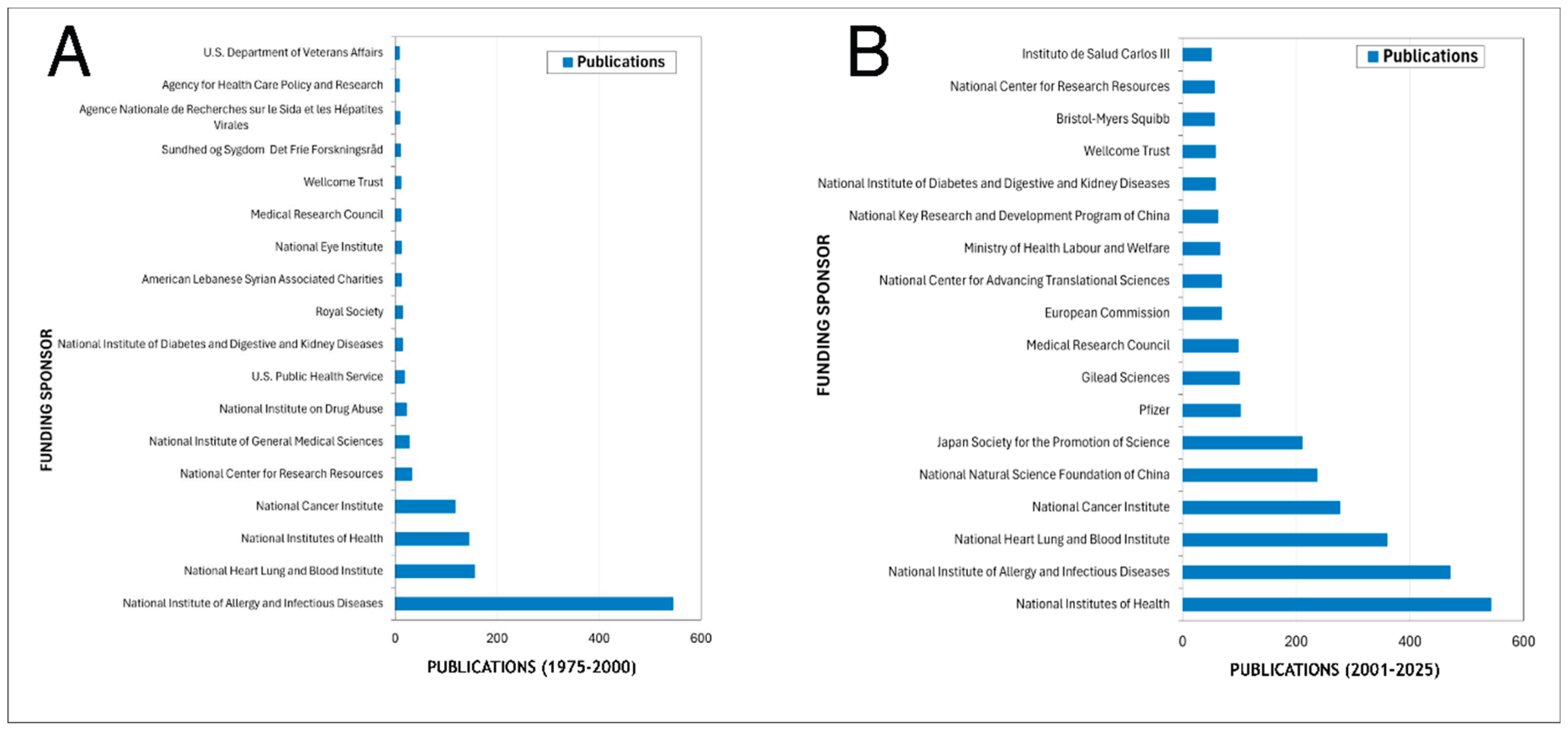
3.7. Sponsor
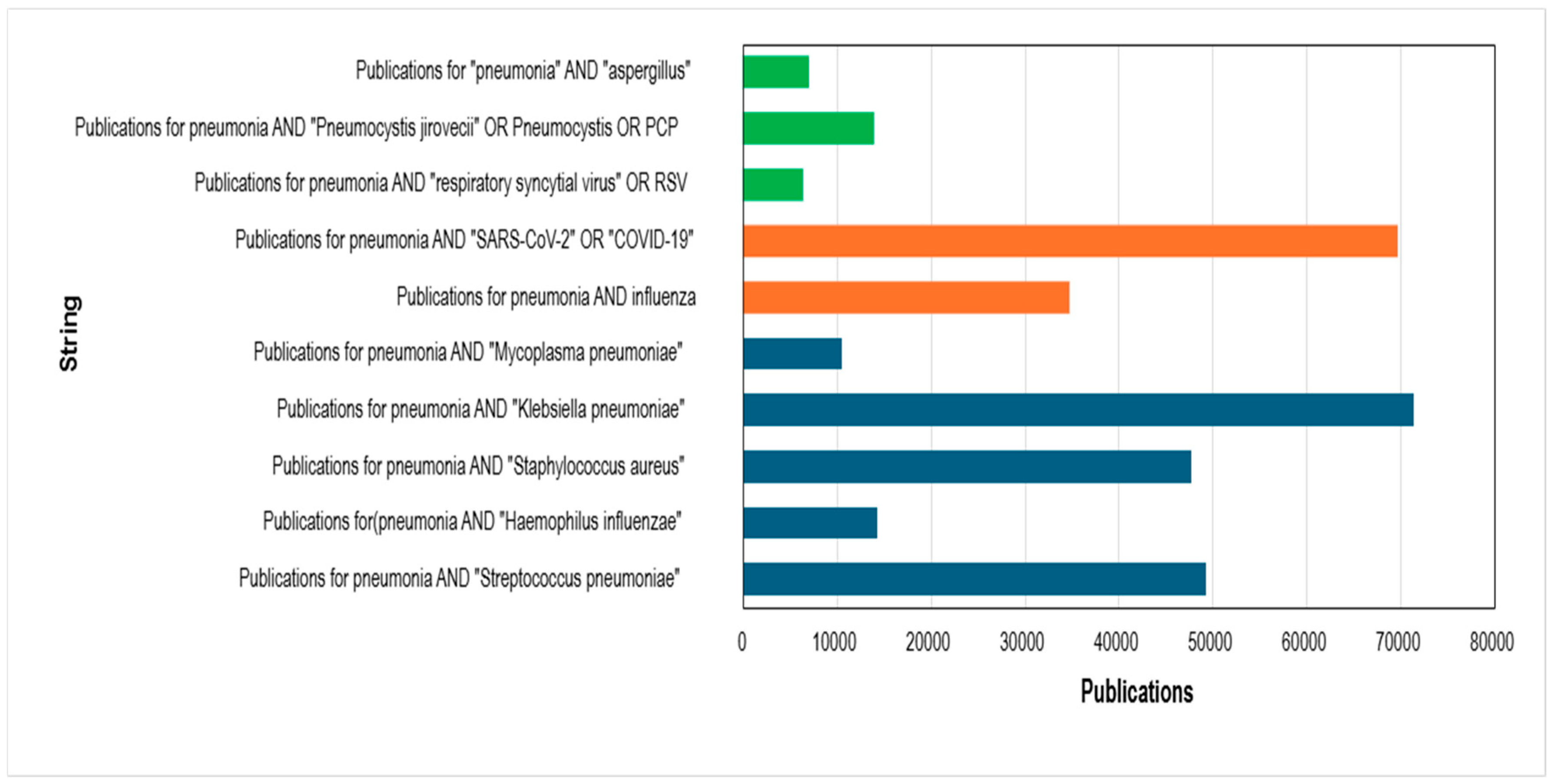
3.8. Bibliometric Positioning of Pneumocystis jirovecii Relative to Other Pneumonia-Associated Pathogens
4. Final Considerations
5. Conclusion
6. Limitations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stringer, J.R.; Beard, C.B.; Miller, R.F.; Cushion, M.T. A new name (Pneumocystis jiroveci) for Pneumocystis from human. Emerging Infectious Diseases 2003, 9(2), 277–80. [Google Scholar] [CrossRef]
- Wilkin, A.; Feinberg, J. Pneumocystis carinii pneumonia: a clinical review. American family physician 1999, 60(6), 1699–708. [Google Scholar]
- Catherinot, E.; Lanternier, F.; Bougnoux, M.-E.; Lecuit, M.; Couderc, L.-J.; Lortholary, O. Pneumocystis jirovecii pneumonia. Infectious Disease Clinics 2010, 24(1), 107–38. [Google Scholar] [CrossRef]
- Kim, SJ; Lee, J; Cho, Y-J; Park, YS; Lee, C-H; Yoon, H.I.; et al. Prognostic factors of Pneumocystis jirovecii pneumonia in patients without HIV infection. Journal of infection 2014, 69(1), 88–95. [Google Scholar] [CrossRef]
- Salzer, HJ; Schäfer, G; Hoenigl, M; Günther, G; Hoffmann, C; Kalsdorf, B.; et al. Clinical, diagnostic, and treatment disparities between HIV-infected and non-HIV-infected immunocompromised patients with Pneumocystis jirovecii pneumonia. Respiration 2018, 96(1), 52–65. [Google Scholar] [CrossRef]
- Stringer, J.R. Pneumocystis carinii: what is it, exactly? Clinical microbiology reviews 1996, 9(4), 489–98. [Google Scholar] [CrossRef]
- Masur, H; Michelis, MA; Greene, JB; Onorato, I; Vande Stouwe, RA; Holzman, R.S.; et al. An outbreak of community-acquired Pneumocystis carinii pneumonia: initial manifestation of cellular immune dysfunction. New England Journal of Medicine 1981, 305(24), 1431–8. [Google Scholar] [CrossRef] [PubMed]
- Morris, A; Lundgren, JD; Masur, H; Walzer, PD; Hanson, DL; Frederick, T.; et al. Current epidemiology of Pneumocystis pneumonia. Emerging infectious diseases 2004, 10(10), 1713. [Google Scholar] [CrossRef] [PubMed]
- Xue, T.; Kong, X.; Ma, L. Trends in the epidemiology of Pneumocystis pneumonia in immunocompromised patients without HIV infection. Journal of Fungi 2023, 9(8), 812. [Google Scholar] [CrossRef]
- Ibrahim, A; Chattaraj, A; Iqbal, Q; Anjum, A; Rehman, M; Aijaz, Z.; et al. Pneumocystis jiroveci pneumonia: a review of management in human immunodeficiency virus (HIV) and non-HIV immunocompromised patients. Avicenna J Med. 2023, 13(1), 23–34. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.A.; Ismail, M.; Albirair, M.; Nail, A.M.A.; Denning, D.W. Fungal infections in Sudan: An underestimated health problem. PLoS Negl Trop Dis. 2023, 17(9), e0011464. [Google Scholar] [CrossRef]
- Organization, W.H. WHO fungal priority pathogens list to guide research, development and public health action; World Health Organization, 2022. [Google Scholar]
- Nations. UNTowTAfSDU.
- Vera, C.; Rueda, Z.V. Transmission and colonization of Pneumocystis jirovecii. Journal of Fungi 2021, 7(11), 979. [Google Scholar] [CrossRef]
- Cushion, M.T.; Tisdale-Macioce, N.; Sayson, S.G.; Porollo, A. The persistent challenge of Pneumocystis growth outside the mammalian lung: past and future approaches. Frontiers in Microbiology 2021, 12, 681474. [Google Scholar] [CrossRef] [PubMed]
- Brown, L; Alanio, A; Cruciani, M; Barnes, R; Donnelly, JP; Loeffler, J.; et al. Strengths and limitations of molecular diagnostics for Pneumocystis jirovecii pneumonia. Expert Review of Molecular Diagnostics 2024, 24(10), 899–911. [Google Scholar] [CrossRef] [PubMed]
- Schmoldt, S; Schuhegger, R; Wendler, T; Huber, I; Sȯllner, H; Hogardt, M.; et al. Molecular evidence of nosocomial Pneumocystis jirovecii transmission among 16 patients after kidney transplantation. Journal of clinical microbiology 2008, 46(3), 966–71. [Google Scholar] [CrossRef]
- Singh, Y; Mirdha, BR; Guleria, R; Kabra, SK; Mohan, A; Chaudhry, R.; et al. Genetic polymorphisms associated with treatment failure and mortality in pediatric Pneumocystosis. Scientific Reports 2019, 9(1), 1192. [Google Scholar] [CrossRef]
- Liu, Y.; Fahle, G.A.; Kovacs, J.A. Inability to culture Pneumocystis jirovecii. MBio 2018, 9(3). [Google Scholar] [CrossRef] [PubMed]
- Calderón, E.J.; De Armas, Y.; Panizo, M.M.; Wissmann, G. Pneumocystis jirovecii pneumonia in Latin America. A public health problem? Expert Review of Anti-infective Therapy. 2013, 11(6), 565–70. [Google Scholar]
- Rodríguez, YDA; Wissmann, G; Müller, A; Pederiva, MA; Brum, M; Brackmann, R.; et al. Pneumocystis jirovecii pneumonia in developing countries. Parasite: journal de la Société Française de Parasitologie 2011, 18(3), 219. [Google Scholar] [CrossRef]
- Clarivate™. Plataforma Web of Science. 2024. Available online: https://clarivate.libguides.com/webofscienceplatform/introduction (accessed on 1 January 2024).
- Scopus®. Available online: https://assets.ctfassets.net/o78em1y1w4i4/28v2L8eQgAGxOnnvZlqJWh/7947feb83982b078ec1d70c297055c34/ELSV_15617_Scopus_Fact_Sheet_Update_WEB.pdf.
- Falagas, M.E.; Pitsouni, E.I.; Malietzis, G.A.; Pappas, G. Comparison of PubMed, Scopus, web of science, and Google scholar: strengths and weaknesses. The FASEB journal 2008, 22(2), 338–42. [Google Scholar] [CrossRef]
- Palella, FJ, Jr.; Delaney, KM; Moorman, AC; Loveless, MO; Fuhrer, J; Satten, G.A.; et al. Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. New England Journal of Medicine 1998, 338(13), 853–60. [Google Scholar] [CrossRef] [PubMed]
- Mocroft, A; Ledergerber, B; Katlama, C; Kirk, O; Reiss, dP; Monforte, A.d.A.; et al. Decline in the AIDS and death rates in the EuroSIDA study: an observational study. The Lancet 2003, 362(9377), 22–9. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, JE; Benson, C; Holmes, KK; Brooks, JT; Pau, A; Masur, H.; et al. Guidelines for prevention and treatment of opportunistic infections in HIV-infected adults and adolescents. MMWR Recomm Rep. 2009, 58(4), 1–207. [Google Scholar]
- Wickramasekaran, R.N.; Jewell, M.P.; Sorvillo, F.; Kuo, T. The changing trends and profile of pneumocystosis mortality in the United States, 1999-2014. Mycoses 2017, 60(9), 607–15. [Google Scholar] [CrossRef]
- Thomas, C.F.; Limper, A.H. Pneumocystis pneumonia. N Engl J Med. 2004, 350(24), 2487–98. [Google Scholar] [CrossRef]
- Morris, A.; Wei, K.; Afshar, K.; Huang, L. Epidemiology and clinical significance of Pneumocystis colonization. The Journal of infectious diseases 2008, 197(1), 10–7. [Google Scholar] [CrossRef] [PubMed]
- Tamburrini, E; Mencarini, P; Visconti, E; Zolfo, M; Marinaci, S; Zinzi, D., IV; et al. Potential impact of Pneumocystis genetic diversity on the molecular detection of the parasite in human host. FEMS Immunology & Medical Microbiology 1998, 22(1-2), 37–49. [Google Scholar] [CrossRef]
- Flori, P; Bellete, B; Durand, F; Raberin, H; Cazorla, C; Hafid, J.; et al. Comparison between real-time PCR, conventional PCR and different staining techniques for diagnosing Pneumocystis jiroveci pneumonia from bronchoalveolar lavage specimens. Journal of medical microbiology 2004, 53(7), 603–7. [Google Scholar] [CrossRef]
- Helweg-Larsen, J. Pneumocystis jiroveci. Applied molecular microbiology, epidemiology and diagnosis Dan Med Bull. 2004, 51(3), 251–73. [Google Scholar]
- Frenkel, J. Pneumocystis jiroveci n. sp. From Man: Morphology, Physiology, and Immunology in Relation to. National Cancer Institute Monograph 1976, 43, 13. [Google Scholar]
- Calderón, E.J.; Armas Rodríguez Yd Capó de Paz, V. Pneumocystis jirovecii: One hundred years of history; 2011. [Google Scholar]
- Keely, S.P.; Fischer, J.M.; Stringer, J.R. Evolution and speciation of Pneumocystis. The Journal of eukaryotic microbiology 2003, 50(6), 624–6. [Google Scholar] [CrossRef]
- Armbruster, C.; Hassl, A.; Kriwanek, S. Pneumocystis carinii colonization in the absence of immunosuppression. Scandinavian journal of infectious diseases 1997, 29(6), 591–3. [Google Scholar] [CrossRef]
- Llibre, JM; Revollo, B; Vanegas, S; Lopez-Nuñez, JJ; Ornelas, A; Marin, J.M.; et al. Pneumocystis jirovecii pneumonia in HIV-1-infected patients in the late-HAART era in developed countries. Scandinavian journal of infectious diseases 2013, 45(8), 635–44. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A; Chattaraj, A; Iqbal, Q; Anjum, A; Rehman, MEU; Aijaz, Z.; et al. Pneumocystis jiroveci pneumonia: a review of management in human immunodeficiency virus (HIV) and non-HIV immunocompromised patients. Avicenna Journal of Medicine 2023, 13(01), 023–34. [Google Scholar] [CrossRef] [PubMed]
- Ekstrand, ML; Shet, A; Chandy, S; Singh, G; Shamsundar, R; Madhavan, V.; et al. Suboptimal adherence associated with virological failure and resistance mutations to first-line highly active antiretroviral therapy (HAART) in Bangalore, India. International health 2011, 3(1), 27–34. [Google Scholar] [CrossRef] [PubMed]
- Pneumocystis jirovecii infections among COVID-19 patients: a case series and literature review. In Open Forum Infectious Diseases; Amstutz, P, Bahr, N.C., Snyder, K., Shoemaker, D.M., Eds.; Oxford University Press US, 2023. [Google Scholar]
- Yu, S.; Yang, T. Non-HIV Immunocompetent Patient with COVID-19 and Severe Pneumocystis jirovecii Pneumonia Co-Infection. Emerging infectious diseases 2024, 30(9), 1948. [Google Scholar] [CrossRef]
- Watson, A.L.; Woodford, J.; Britton, S.; Gupta, R.; Whiley, D.; McCarthy, K. Determining Pneumocystis jirovecii colonisation from infection using PCR-based diagnostics in HIV-negative individuals. Diagnostics 2024, 14(1), 114. [Google Scholar] [CrossRef]
- Lee, R; Huh, K; Kang, CK; Kim, YC; Kim, JH; Kim, H.; et al. Diagnosis of Pneumocystis jirovecii pneumonia in non-HIV immunocompromised patient in Korea: a review and algorithm proposed by expert consensus group. Infection & Chemotherapy 2025, 57(1), 45. [Google Scholar] [CrossRef]
- Jaramillo Cartagena, A.; Asowata, O.E.; Ng, D.; Babady, N.E. An overview of the laboratory diagnosis of Pneumocystis jirovecii pneumonia. Journal of Clinical Microbiology 2025, 63(3), e00361-24. [Google Scholar] [CrossRef]
- Cederwall, S.; Ottander, E.; Björkhem, D.; Oldberg, K.; Påhlman, L.I. Performance of Quantitative PCR to Distinguish Pneumocystis jirovecii Pneumonia From Colonisation in Immunocompromised Patients. Mycoses 2025, 68(10), e70120. [Google Scholar] [CrossRef]
- Costa, VS; Cidade, JP; Medeiros, I; Fidalgo, P; Moreira, H; Miranda, T.; et al. Pneumocystis jirovecii Pneumonia Diagnosis with Oropharyngeal Wash PCR in Immunocompromised Patients—A Systematic Review. Journal of Clinical Medicine 2025, 14(18), 6572. [Google Scholar] [CrossRef] [PubMed]
- Yi, J; Wang, N; Wu, J; Tang, Y; Zhang, J; Zhu, L.; et al. Development of a droplet digital polymerase chain reaction for sensitive detection of Pneumocystis jirovecii in respiratory tract specimens. Frontiers in Medicine 2021, 8, 761788. [Google Scholar] [CrossRef]
- Zhan, Y.; Gao, X.; Li, S.; Si, Y.; Li, Y.; Han, X. Development and evaluation of rapid and accurate CRISPR/Cas13-based RNA diagnostics for Pneumocystis jirovecii pneumonia. Front Cell Infect Microbiol 2022. [Google Scholar] [CrossRef]
- Del Corpo, O.; Butler-Laporte, G.; Sheppard, D.C.; Cheng, M.P.; McDonald, E.G.; Lee, T.C. Diagnostic accuracy of serum (1-3)-β-D-glucan for Pneumocystis jirovecii pneumonia: a systematic review and meta-analysis. Clinical Microbiology and Infection 2020, 26(9), 1137–43. [Google Scholar] [CrossRef]
- Low-dose TMP-SMX in the treatment of Pneumocystis jirovecii pneumonia: a systematic review and meta-analysis. In Open forum infectious diseases; Butler-Laporte, G., Smyth, E., Amar-Zifkin, A., Cheng, M.P., McDonald, E.G., Lee, T.C., Eds.; Oxford University Press US, 2020. [Google Scholar]
- Dimeas, I.E.; Dimeas, G.E.; Zakynthinos, G.E.; Tsolaki, V. Individualized Trimethoprim-Sulfamethoxazole Dosing in Non-HIV Patients with Pneumocystis Pneumonia: A Narrative Review of Current Evidence. Journal of Personalized Medicine 2025, 15(7), 311. [Google Scholar] [CrossRef]
- Rivero, L; de la Horra, C; Montes-Cano, MA; Rodríguez-Herrera, A; Respaldiza, N; Friaza, V.; et al. Pneumocystis jirovecii transmission from immunocompetent carriers to infant. Emerging Infectious Diseases 2008, 14(7), 1116. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y; Xu, X; Huang, Z; Lai, X; Li, C; Chen, J.; et al. Diagnostic predictive evaluation of pneumocystis jirovecii pneumonia using digital chest CT analysis combined with clinical features. Frontiers in Physiology 2025, 16, 1616791. [Google Scholar] [CrossRef]
- Lian, Q; Song, X; Yang, J; Wang, L; Xu, P; Wang, X.; et al. Alterations of lung microbiota in lung transplant recipients with pneumo cystis jirovecii pneumonia. Respiratory Research 2024, 25(1), 125. [Google Scholar] [CrossRef] [PubMed]
| Top 10 most prolific authors in Scopus (1975–2000) | Top 10 most prolific authors in Scopus (2001-2025) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rank | Author/ Scopus ID | Affiliation | Country | n pub | Citations / Mean citations | Hi | Rank | Author | Affiliation | Country | n pub | Citations / Mean citations | Hi |
| 1 | Walter T. Hughes 7202094239 |
St. Jude Children's Research Hospital | United States | 115 | 4,911 / 42.7 | 34 | 1 | Andrew Harold Limper 7006099760 |
Mayo Clinic | United States |
112 | 5,153 / 46 | 31 |
| 2 | Peter D. Walzer 7007081824 |
University of Cincinnati |
United States | 101 | 3,697 / 36.6 | 37 | 2 | David W. Denning 7102640098 |
The University of Manchester | United Kingdom | 109 | 10,534 | 33 |
| 3 | Henry Masur 56744299400 |
NIH Clinical Center (CC) | United States | 100 | 7,440 / 74.4 | 42 | 3 | Melanie T. Cushion 35474601800 |
University of Cincinnati College of Medicine | United States | 97 | 2,297 | 29 |
| 4 | Marilyn S. Bartlett 7102151507 |
Indiana University School of Medicine | United States | 94 | 2766 / 29.4 | 29 | 4 | Laurence Huang 56144978300 |
UCSF School of Medicine | United States | 90 | 4,327 | 33 |
| 5 | Queener, Sherry F. 7006547726 |
Indiana University School of Medicine | United States | 91 | 3,382 / 37.1 | 35 | 5 | Gilles Nevez 7003589276 |
Université de Bretagne Occidentale | France | 73 | 1,369 / 18.7 | 21 |
| 6 | James W. Smith 9844027900 |
Indiana University School of Medicine | United States | 90 | 2,728 / 30.3 | 30 | 6 | Joseph A. Kovacs 7202246288 |
National Institutes of Health Clinical Center | United States | 71 | 2,684 / 37.8 | 28 |
| 7 | Robert F Miller 55547118657 |
University College London | United Kingdom | 69 | 2000 / 29 | 25 | 7 | Eduardo Dei-Cas 7006604602 |
Center for Infection and Immunity of Lille (CIIL) | France | 60 | 1,981 / 33 | 26 |
| 8 | Eduardo Dei-Cas 7006604602 |
Center for Infection and Immunity of Lille (CIIL)The institution will open in a new tab, Lille, France | France | 65 | 1,774 / 27 | 27 | 8 | Chaohung Lee 7410145264 |
Indiana University School of Medicine | United States | 58 | 1,141 / 19.6 | 20 |
| 9 | Melanie T. Cushion 35474601800 |
University of Cincinnati College of Medicine | United States | 62 | 1,959 / 31.5 | 28 | 9 | Theodore J. Kottom 6603350625 |
Mayo Clinic, Rochester | United States | 55 | 1,079 / 19.6 | 17 |
| 10 | Joseph A. Kovacs 7202246288 |
National Institutes of Health Clinical Center | United States | 60 | 5, 097 / 84.9 | 34 | 10 | Robert F. Miller 55547118657 |
University College London | United Kingdom | 55 | 2, 331 / 42.3 | 24 |
| Top 10 most prolific journals in Scopus (1975–2000) | Top 10 most prolific journals in Scopus (2001-2025) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ranking | Journal | Pub. n | Total citations / mean citations (1996–2000) * | Quartile (Scimago) |
Ranking | Journal | Pub. n | Total citations / mean citations (2001-2025) |
Quartile (Scimago) |
| 1 | Chest | 326 | 9,032 / 27.7 | Q1 | 1 | Clinical Infectious Diseases | 239 | 16,762 / 77.6 | Q1 |
| 2 | Clinical Infectious Diseases | 228 | 9,290 / 40 | Q1 | 2 | Transplant Infectious Disease | 228 | 4,325 / 18.9 | Q2 |
| 3 | New England Journal of Medicine | 216 | 40,602 / 187 | Q1 | 3 | Bone Marrow Transplantation | 210 | 7,383 / 35.1 | Q1 |
| 4 | Journal of Eukaryotic Microbiology | 202 | 2,771 / 13.7 | Q2 | 4 | AIDS | 165 | 6,491 / 42.7 | Q1 |
| 5 | AIDS | 193 | 7,289 / 37.7 | Q1 | 5 | BMJ Case Reports | 159 | 673 / 4.2 | Q4 |
| 6 | Lancet | 191 | 7,497 / 39.2 | Q1 | 6 | Transplantation Proceedings | 153 | 1,817 / 11.8 | Q3 |
| 7 | Annals of Internal Medicine | 180 | 14,542 / 80.7 | Q1 | 7 | Biology of Blood and Marrow Transplantation | 152 | 8,916 / 58.6 | Q1 |
| 8 | Journal of Infectious Diseases | 159 | 8,487 / 53.3 | Q1 | 8 | Journal of Eukaryotic Microbiology | 137 | 1,302 / 9.5 | Q1 |
| 9 |
Antimicrobial Agents and Chemotherapy |
134 | 5, 264 / 39 | Q1 | 9 | BMC Infectious Diseases | 130 | 2,144 / 16.4 | Q1 |
| 10 | Journal of Protozoology | 125 | 1,562 / 12.7 | Q1 | 10 | Leukemia and Lymphoma | 121 | 2,316 / 19.1 | Q2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




