Submitted:
10 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Mechanistic Elucidation of Glycohypoxia-Mediated Nitric Oxide Dysregulation in T2DM Vascular Complications
3. Objective and Methods
3.1. Data Sources and Study Selection
3.2. Data Extraction and Normalization
3.3. Meta-Regression Analysis
3.4. Physiologic Integration
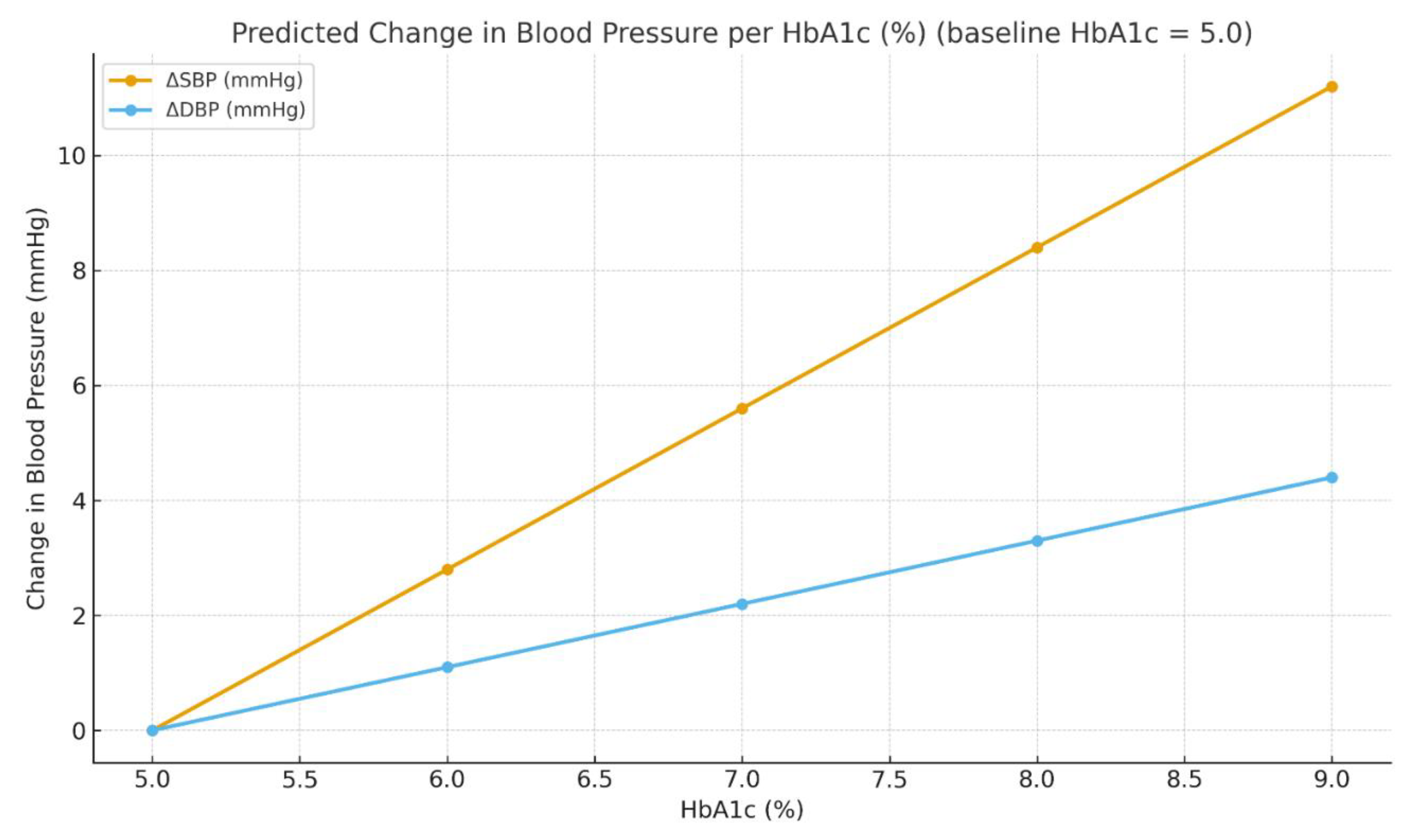
4. Results
4.1. Data Extraction and Quantitative Framework
4.2. Meta-Regression and Pooled Estimation
| Parameter | Pooled β (mmHg/%) | 95% CI | p-value | I² (%) | Interpretation |
| ΔSBP | +2.8 | +1.9 to +3.7 | <0.001 | 46.3 | Hemodynamic compensation |
| ΔDBP | +1.1 | +0.6 to +1.7 | <0.001 | 41.5 | Peripheral resistance component |
4.3. Mechanistic Correlation with Oxygen Unloading
4.4. Subgroup and Consistency Analyses
- The slope of blood pressure elevation was steeper among individuals with established type 2 diabetes compared with prediabetic or normoglycemic cohorts (ΔSBP ≈ 3.2 mmHg/% vs. 1.9 mmHg/%), indicating progressive sensitivity to glycohypoxic burden.
- Exclusion of any single dataset produced minimal deviation in pooled estimates (variance < 0.15 mmHg/%), confirming model stability.
- Funnel-plot symmetry and Egger’s test indicated no significant publication bias.
4.5. Quantitative Summary and Physiological Interpretation
5. Discussion
6. Conclusion
References
- Akalu, Y.; Belsti, Y. Hypertension and Its Associated Factors Among Type 2 Diabetes Mellitus Patients at Debre Tabor General Hospital, Northwest Ethiopia. Diabetes, metabolic syndrome and obesity: targets and therapy 2020, 13, 1621–1631. [Google Scholar] [CrossRef]
- Liu, F.; Wu, M.; Feng, Y. H.; Zhong, H.; Cui, T. L.; Huang, Y. Q.; Liang, Y. P.; Diao, Y. S.; Zang, L.; Li, L.; Zang, J.; Qiu, H. Y.; Huang, S. M.; Fu, P. Influence of HbA1c on short-term blood pressure variability in type 2 diabetic patients with diabetic nephropathy. Journal of Zhejiang University. Science. B 2013, 14(11), 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y. C.; Chang, C. T.; Chen, R. H.; Wang, T. Y.; Chen, C. C. HbA1c and systolic blood pressure variation to predict all-cause mortality in patients with type 2 diabetes mellitus. Primary care diabetes 2024, 18(2), 146–150. [Google Scholar] [CrossRef] [PubMed]
- Akl, Maher M.; *; †; Ahmed, Amr2. Glycohypoxia: a hypothesis linking chronic hyperglycemia to functional hypoxia and diabetic complications in type 2 diabetes 10.4103/mgr.MEDGASRES-D-25-00137. Medical Gas Research () 2026. [Google Scholar] [CrossRef] [PubMed]
- Janaszak-Jasiecka, A.; Siekierzycka, A.; Płoska, A.; Dobrucki, I. T.; Kalinowski, L. Endothelial Dysfunction Driven by Hypoxia-The Influence of Oxygen Deficiency on NO Bioavailability. Biomolecules 2021, 11(7), 982. [Google Scholar] [CrossRef]
- El-Remessy, A. B.; Abou-Mohamed, G.; Caldwell, R. W.; Caldwell, R. B. High glucose-induced tyrosine nitration in endothelial cells: role of eNOS uncoupling and aldose reductase activation. Investigative ophthalmology & visual science 2003, 44(7), 3135–3143. [Google Scholar] [CrossRef]
- Sasaki, N.; Yamashita, T.; Takaya, T.; Shinohara, M.; Shiraki, R.; Takeda, M.; Emoto, N.; Fukatsu, A.; Hayashi, T.; Ikemoto, K.; Nomura, T.; Yokoyama, M.; Hirata, K.; Kawashima, S. Augmentation of vascular remodeling by uncoupled endothelial nitric oxide synthase in a mouse model of diabetes mellitus. Arteriosclerosis, thrombosis, and vascular biology 2008, 28(6), 1068–1076. [Google Scholar] [CrossRef]
- Santhanam, A. V.; d’Uscio, L. V.; Smith, L. A.; Katusic, Z. S. Uncoupling of eNOS causes superoxide anion production and impairs NO signaling in the cerebral microvessels of hph-1 mice. Journal of neurochemistry 2012, 122(6), 1211–1218. [Google Scholar] [CrossRef]
- Schramm, A.; Matusik, P.; Osmenda, G.; Guzik, T. J. Targeting NADPH oxidases in vascular pharmacology. Vascular pharmacology 2012, 56(5-6), 216–231. [Google Scholar] [CrossRef]
- Zou, M. H.; Shi, C.; Cohen, R. A. Oxidation of the zinc-thiolate complex and uncoupling of endothelial nitric oxide synthase by peroxynitrite. The Journal of clinical investigation 2002, 109(6), 817–826. [Google Scholar] [CrossRef]
- Mahdi, A.; Tengbom, J.; Alvarsson, M.; Wernly, B.; Zhou, Z.; Pernow, J. Red Blood Cell Peroxynitrite Causes Endothelial Dysfunction in Type 2 Diabetes Mellitus via Arginase. Cells 2020, 9(7), 1712. [Google Scholar] [CrossRef]
- Bar-Shai, M.; Reznick, A. Z. Peroxynitrite induces an alternative NF-kappaB activation pathway in L8 rat myoblasts. Antioxidants & redox signaling 2006, 8(3-4), 639–652. [Google Scholar] [CrossRef]
- González, P.; Lozano, P.; Ros, G.; Solano, F. Hyperglycemia and Oxidative Stress: An Integral, Updated and Critical Overview of Their Metabolic Interconnections. International Journal of Molecular Sciences 2023, 24(11), 9352. [Google Scholar] [CrossRef] [PubMed]
- Bakker, W.; Eringa, E.C.; Sipkema, P.; et al. Endothelial dysfunction and diabetes: roles of hyperglycemia, impaired insulin signaling and obesity. Cell Tissue Res 2009, 335, 165–189. [Google Scholar] [CrossRef] [PubMed]
- Tunedal, K.; Viola, F.; Garcia, B. C.; Bolger, A.; Nyström, F. H.; Östgren, C. J.; Engvall, J.; Lundberg, P.; Dyverfeldt, P.; Carlhäll, C. J.; Cedersund, G.; Ebbers, T. Haemodynamic effects of hypertension and type 2 diabetes: Insights from a 4D flow MRI-based personalized cardiovascular mathematical model. The Journal of physiology 2023, 601(17), 3765–3787. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, Y.; Liu, S.; et al. Diabetic vascular diseases: molecular mechanisms and therapeutic strategies. Sig Transduct Target Ther 2023, 8, 152. [Google Scholar] [CrossRef]
- Thum, T.; Fraccarollo, D.; Schultheiss, M.; Froese, S.; Galuppo, P.; Widder, J. D.; Tsikas, D.; Ertl, G.; Bauersachs, J. Endothelial nitric oxide synthase uncoupling impairs endothelial progenitor cell mobilization and function in diabetes. Diabetes 2007, 56(3), 666–674. [Google Scholar] [CrossRef]
- An, Y.; Xu, B. T.; Wan, S. R.; Ma, X. M.; Long, Y.; Xu, Y.; Jiang, Z. Z. The role of oxidative stress in diabetes mellitus-induced vascular endothelial dysfunction. Cardiovascular diabetology 2023, 22(1), 237. [Google Scholar] [CrossRef]
- Gui, F.; You, Z.; Fu, S.; Wu, H.; Zhang, Y. Endothelial Dysfunction in Diabetic Retinopathy. Frontiers in endocrinology 2020, 11, 591. [Google Scholar] [CrossRef]
- Blanton, R. M. cGMP Signaling and Modulation in Heart Failure. Journal of cardiovascular pharmacology 2020, 75(5), 385–398. [Google Scholar] [CrossRef]
- Giles, T. D.; Sander, G. E.; Nossaman, B. D.; Kadowitz, P. J. Impaired vasodilation in the pathogenesis of hypertension: focus on nitric oxide, endothelial-derived hyperpolarizing factors, and prostaglandins. Journal of clinical hypertension (Greenwich, Conn.) 2012, 14(4), 198–205. [Google Scholar] [CrossRef]
- Condello, I.; Federico, M.; Condello, S. The dynamic interplay between endothelial reactivity, temperature, and vascular resistance in cardiopulmonary bypass. Annals of medicine and surgery (2012) 2025, 87(8), 4695–4700. [Google Scholar] [CrossRef]
- Boese, M.; Busse, R.; Mülsch, A.; Schini-Kerth, V. Effect of cyclic GMP-dependent vasodilators on the expression of inducible nitric oxide synthase in vascular smooth muscle cells: role of cyclic AMP. British journal of pharmacology 1996, 119(4), 707–715. [Google Scholar] [CrossRef]
- Thangarajah, H.; Yao, D.; Chang, E. I.; Shi, Y.; Jazayeri, L.; Vial, I. N.; Galiano, R. D.; Du, X. L.; Grogan, R.; Galvez, M. G.; Januszyk, M.; Brownlee, M.; Gurtner, G. C. The molecular basis for impaired hypoxia-induced VEGF expression in diabetic tissues. Proceedings of the National Academy of Sciences of the United States of America 2009, 106(32), 13505–13510. [Google Scholar] [CrossRef]
- Zhang, D.; Lv, F. L.; Wang, G. H. Effects of HIF-1α on diabetic retinopathy angiogenesis and VEGF expression. European review for medical and pharmacological sciences 2018, 22(16), 5071–5076. [Google Scholar] [CrossRef] [PubMed]
- Thomas, H.Y.; Ford Versypt, A.N. Pathophysiology of mesangial expansion in diabetic nephropathy: mesangial structure, glomerular biomechanics, and biochemical signaling and regulation. J Biol Eng 2022, 16, 19. [Google Scholar] [CrossRef] [PubMed]
- Noonan, J.; Cardoso, L.; Bobik, A.; Peter, K. Atherosclerotic Plaque Instability and Rupture: Recommended Mouse Models to Empower Clinically Relevant Discoveries, Diagnostics, and Therapeutics. Arteriosclerosis, thrombosis, and vascular biology 2025, 45(10), 1707–1714. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Mohamed, A. S.; Zhou, S. H. Oxidized low density lipoprotein, stem cells, and atherosclerosis. Lipids in health and disease 2012, 11, 85. [Google Scholar] [CrossRef]
- Huang, X.; Qin, C.; Guo, X.; Cao, F.; Tang, C. Association of hemoglobin A1c with the incidence of hypertension: A large prospective study. Frontiers in endocrinology 2023, 13, 1098012. [Google Scholar] [CrossRef]
- Luo, B.; Xu, W.; Luo, F.; Zhang, S.; Li, X. Association between elevated glycosylated hemoglobin levels and hypertension among US adults: NHANES 2011-2018. BMC cardiovascular disorders 2025, 25(1), 58. [Google Scholar] [CrossRef]
- Pu, L. J.; Shen, Y.; Lu, L.; Zhang, R. Y.; Zhang, Q.; Shen, W. F. Increased blood glycohemoglobin A1c levels lead to overestimation of arterial oxygen saturation by pulse oximetry in patients with type 2 diabetes. Cardiovascular diabetology 2012, 11, 110. [Google Scholar] [CrossRef]
- O’Riordan, J. F.; Goldstick, T. K.; Ditzel, J.; Ernest, J. T. Diabetic oxygen-hemoglobin equilibrium curves evaluated by nonlinear regression of the Hill equation. Advances in experimental medicine and biology 1984, 169, 187–198. [Google Scholar] [CrossRef]
- Alshnbari, A. S.; Millar, S. A.; O’Sullivan, S. E.; Idris, I. Effect of Sodium-Glucose Cotransporter-2 Inhibitors on Endothelial Function: A Systematic Review of Preclinical Studies. Diabetes therapy: research, treatment and education of diabetes and related disorders 2020, 11(9), 1947–1963. [Google Scholar] [CrossRef]
- Kostov, K. The Causal Relationship between Endothelin-1 and Hypertension: Focusing on Endothelial Dysfunction, Arterial Stiffness, Vascular Remodeling, and Blood Pressure Regulation. Life (Basel, Switzerland) 2021, 11(9), 986. [Google Scholar] [CrossRef]
- Banecki, K. M. R. M.; Dora, K. A. Endothelin-1 in Health and Disease. International Journal of Molecular Sciences 2023, 24(14), 11295. [Google Scholar] [CrossRef]
| Dataset | Population | N | Key Findings Utilized |
|---|---|---|---|
| CHNS 2011–2015 [29] | Chinese adults | 4,074 | HR = 1.10 per 1% HbA1c ↑ (Cox regression) |
| NHANES 2011–2018 [30] | U.S. adults | 10,503 | OR = 1.22 (95% CI 1.07–1.39) per 1% HbA1c ↑ |
| Pu et al., 2012 [31] | Ventilated T2DM | 261 | ΔSpO₂–SaO₂ ≈ 1.83%, r = 0.307 → ΔP₅₀ ≈ −0.20 mmHg /% HbA1c |
| Mechanistic Variable | Derived Relation | Physiological Interpretation |
|---|---|---|
| ΔP₅₀ (mmHg) | −0.19 × ΔHbA1c | Reduced O₂ release affinity |
| %ΔO₂ unloading | −0.8% × ΔHbA1c | Cellular oxygen deficit |
| ΔSBP (mmHg) | 2.8 × ΔHbA1c | Compensatory pressure response |
| ΔDBP (mmHg) | 1.1 × ΔHbA1c | Resistance-mediated adaptation |
| Study / Dataset | Population | N | HbA1c Range (%) | BP Association | Derived ΔSBP (mmHg/%) | Derived ΔDBP (mmHg/%) | Model Type |
|---|---|---|---|---|---|---|---|
| CHNS (2011–2015) [29] | Chinese adults | 4,074 | 4.8–10.1 | HR = 1.10 per 1% HbA1c↑ | +2.6 | +1.0 | Cox regression |
| NHANES (2011–2018) [30] | U.S. adults | 10,503 | 4.5–9.8 | OR = 1.22 (95% CI 1.07–1.39) per 1%↑ | +2.9 | +1.2 | Logistic regression |
| Pu et al. (2012) [31] | Ventilated T2DM | 261 | ≤7 / >7 | ΔSpO₂–SaO₂ ≈ 1.83%, r = 0.307 | +3.0 (inferred) | +1.1 | Linear correlation |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




