Submitted:
06 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Literature Review
2.1. Deep Learning in Medical Imaging
2.2. Pneumonia Detection Studies
2.3. Dataset and Challenges
2.4. Research Gap
3. Materials and Methods
3.1. Dataset Description
3.2. Data Preprocessing
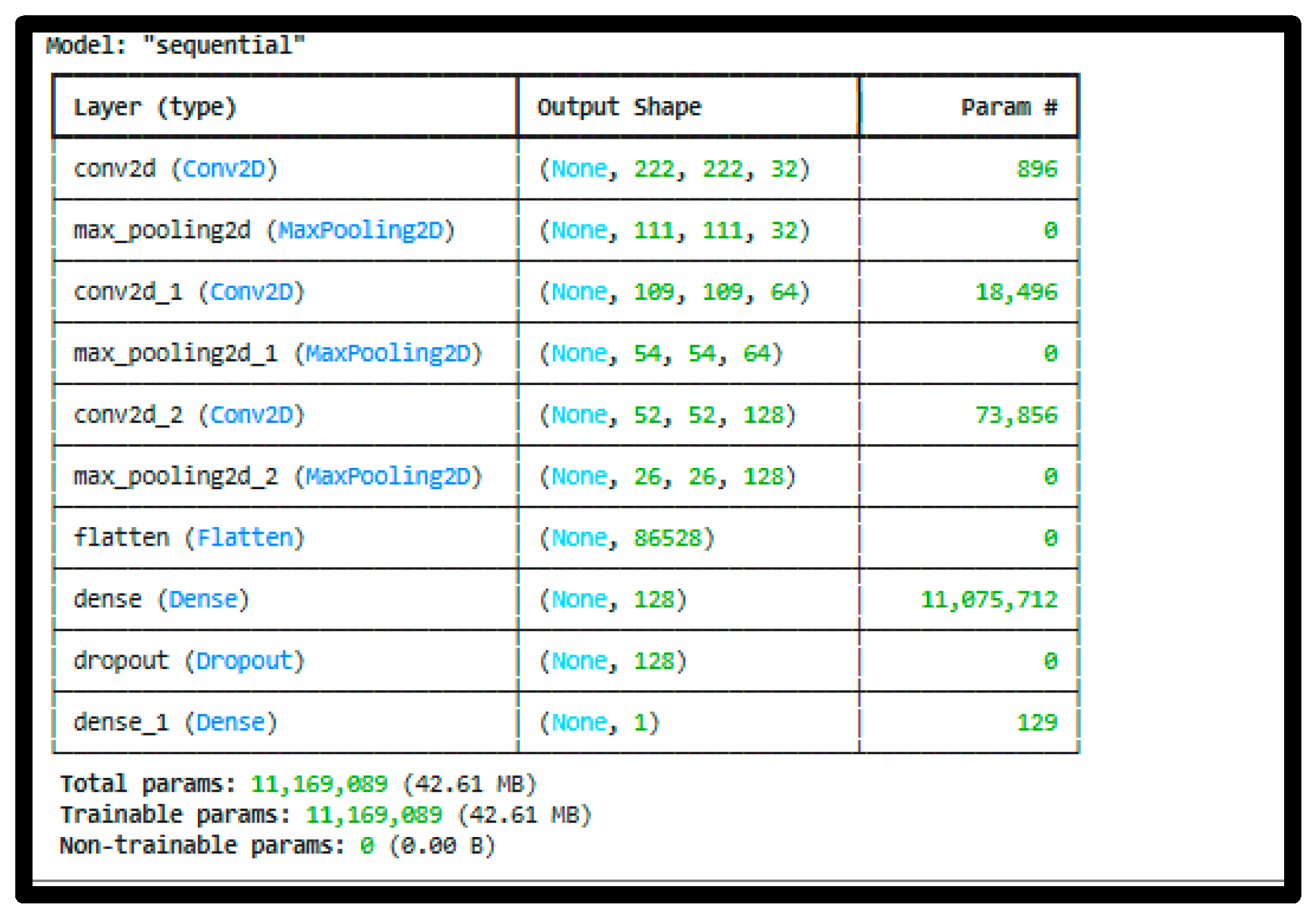
3.3. Model Architecture
3.4. Training Setup
3.5. Evaluation Metrics
3.6. Robotic Intervention System
4. Results
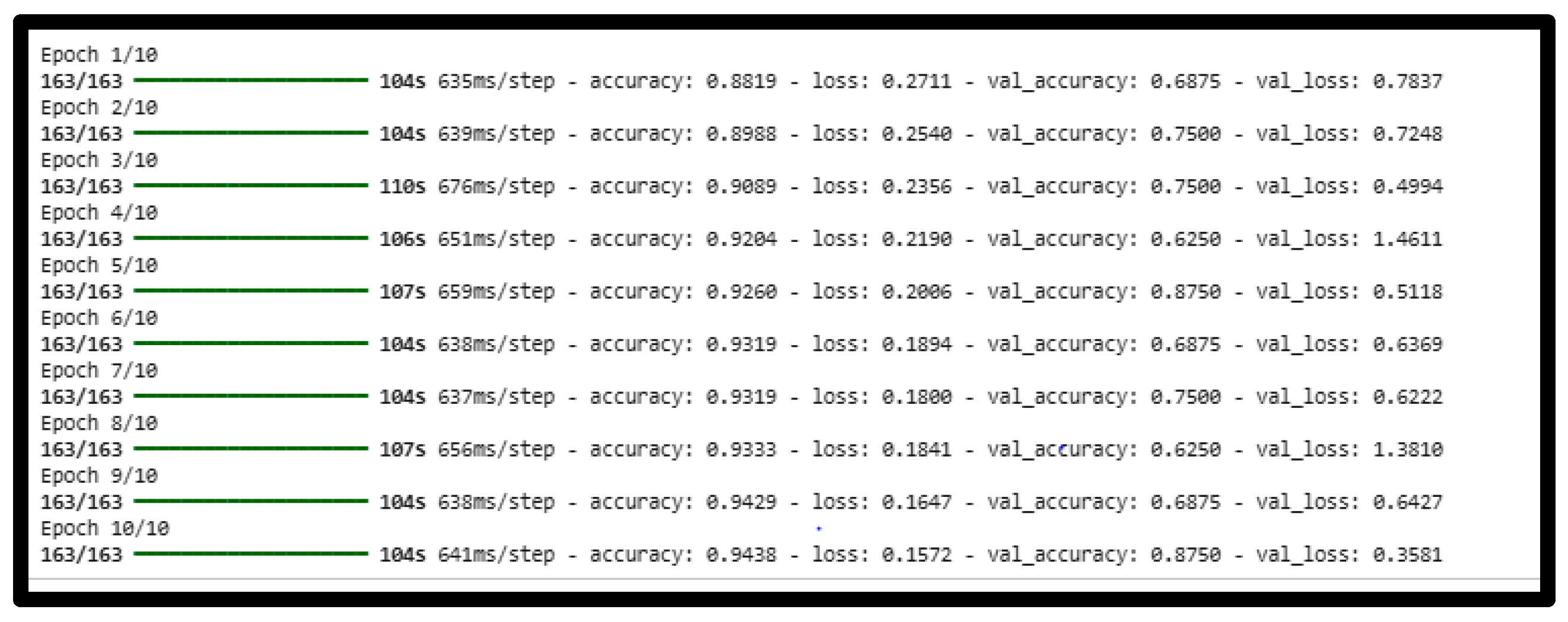
4.1. Training Performance
4.2. Test Performance
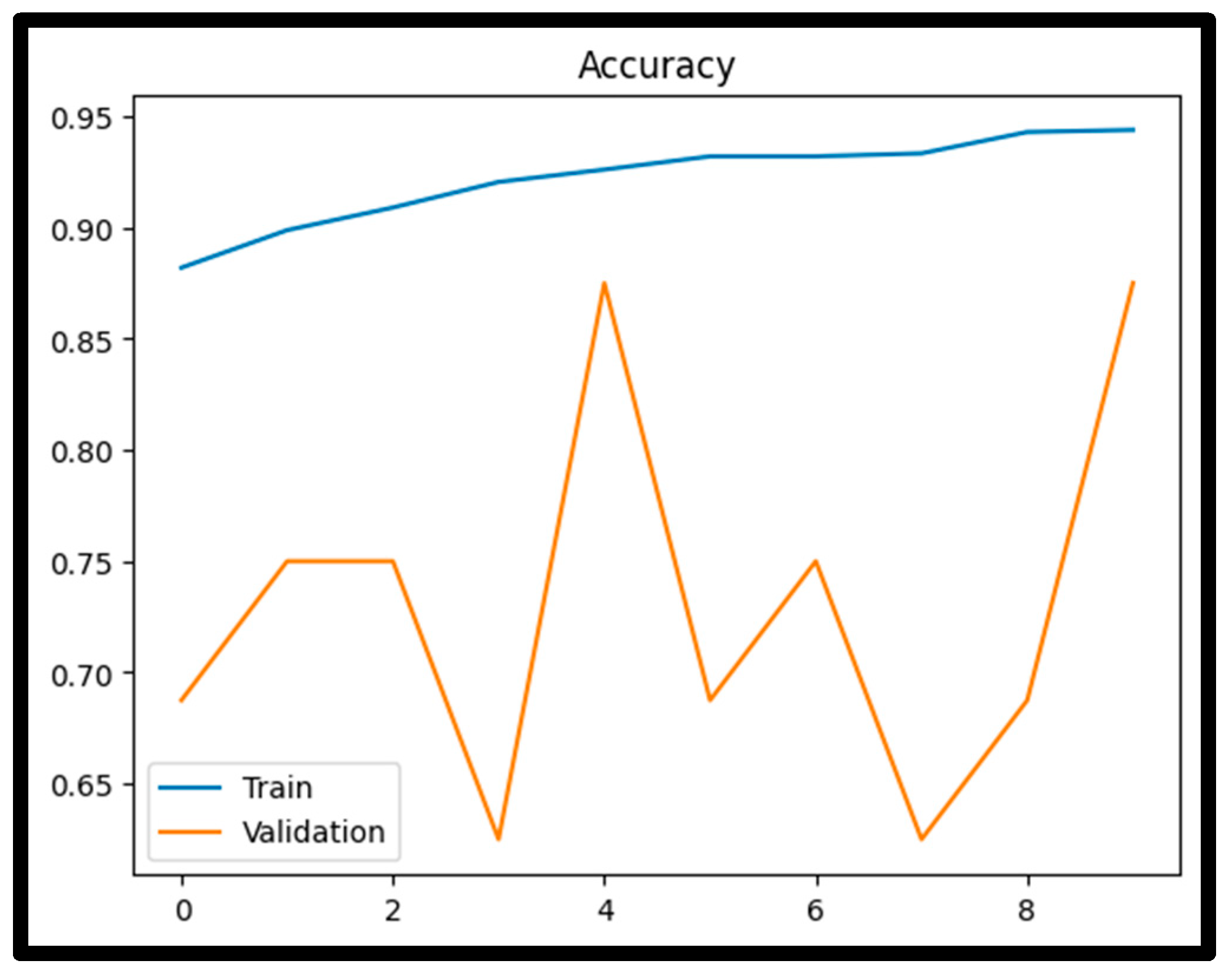
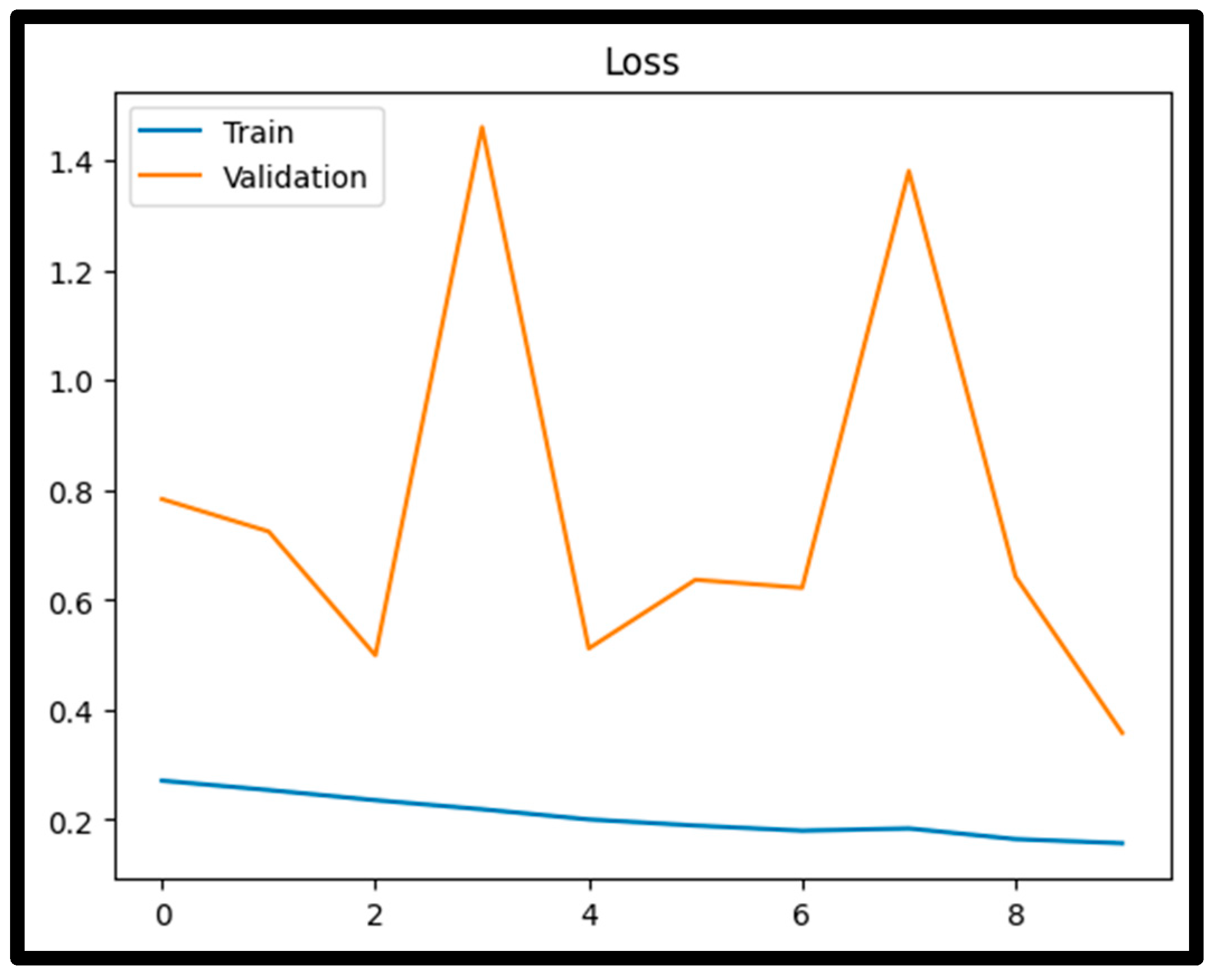
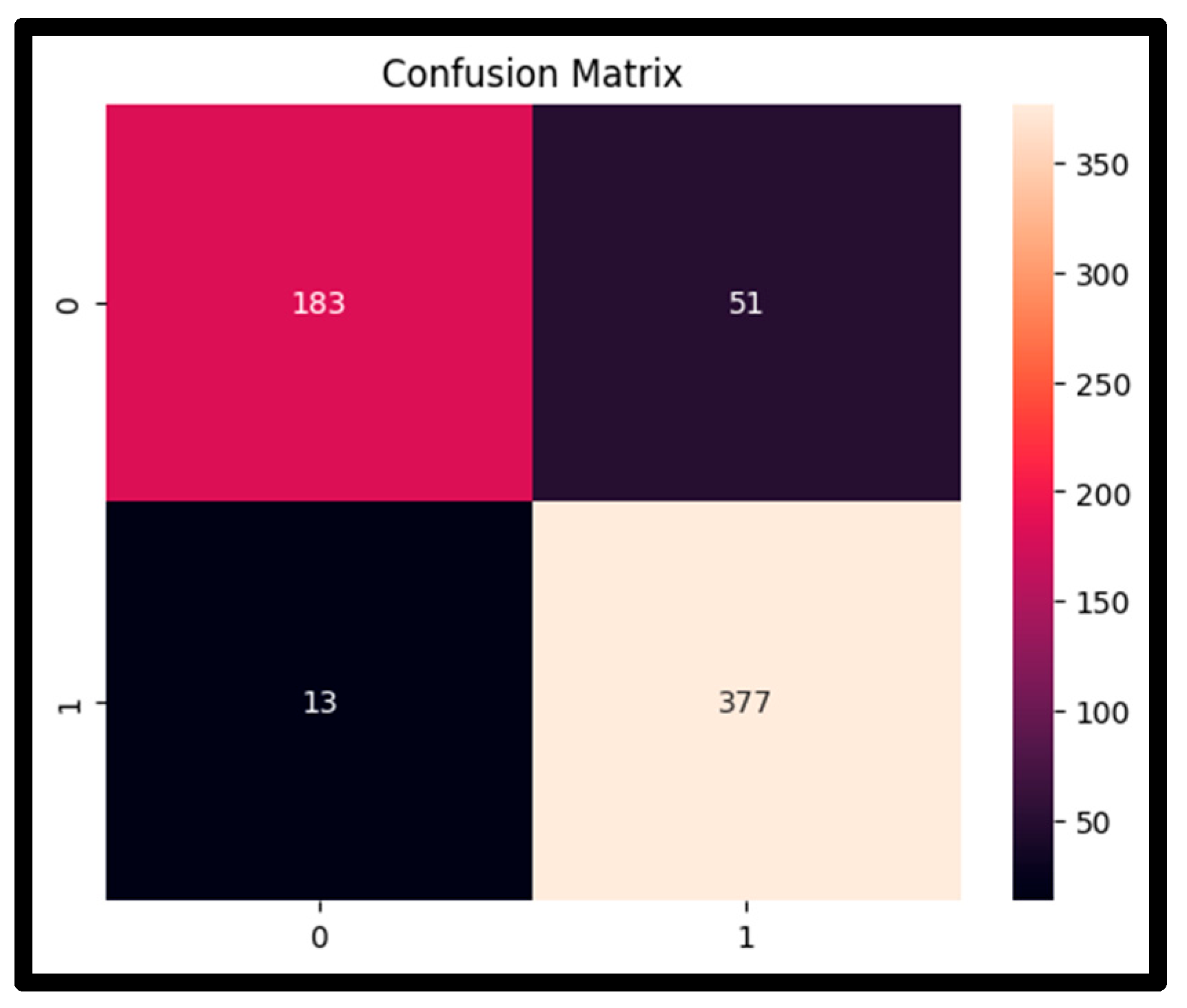
4.3. Confusion Matrix Analysis
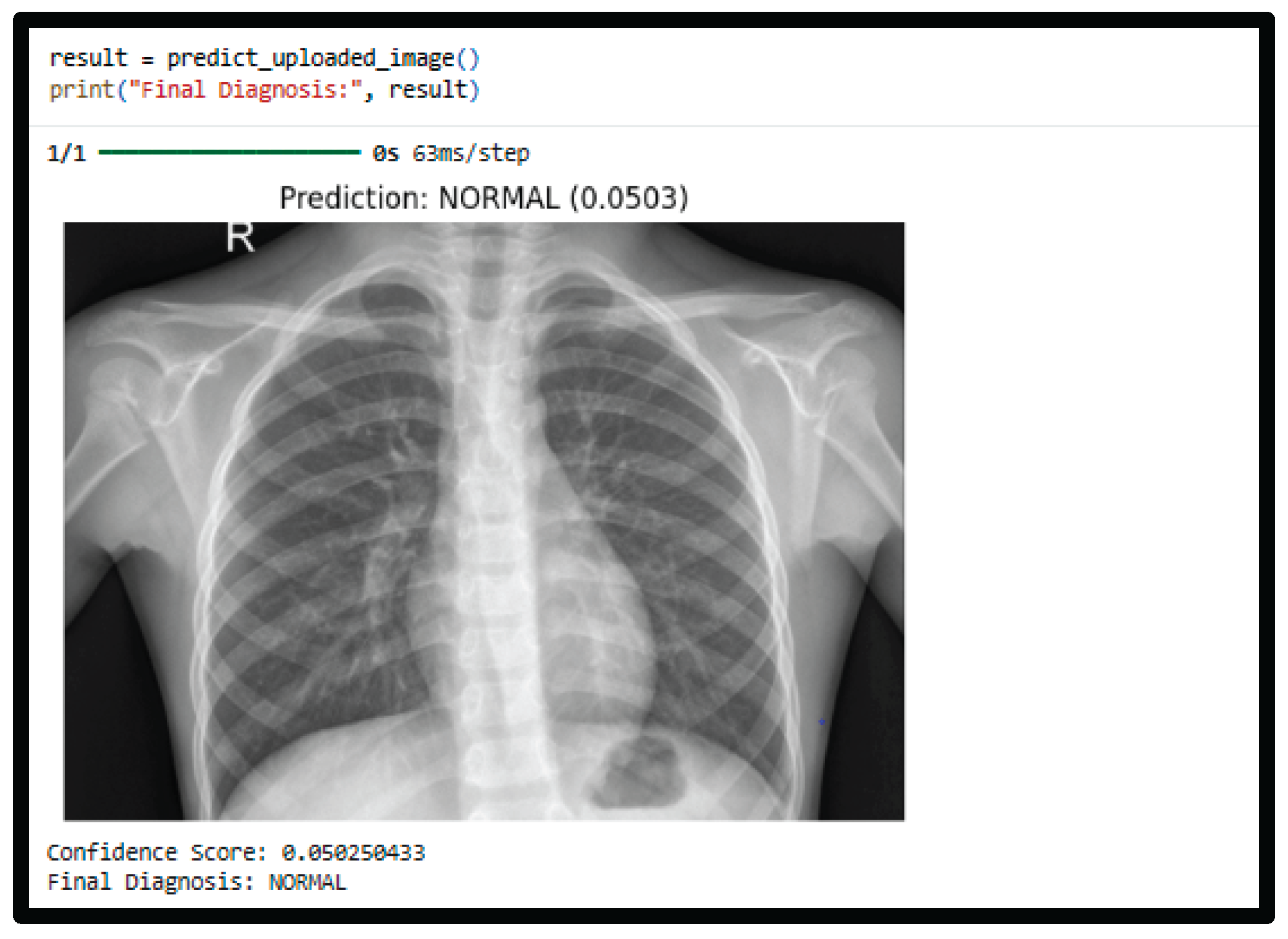
4.4. Prediction Example
5. Discussion
6. Conclusions
Appendix
References
- Application wise, the proposed framework can have tremendous potential in smart hospital setups and remote healthcare systems in which specialist expertise is not readily available. The system, though presently in the form of a simulation, forms a scalable base to deploy in the real-world, and it will promote an increase in diagnostic efficiency, decrease in clinical workload, and better patient outcomes.ReferencesNouh, M. I., & Nouran, M. Perspective of Pneumonia in the Health-Care Setting.
- Uddin, A. Z. M. J., Begum, M. R., Akib, A. A. S., Islam, K., Hasib, A., Giri, A., & Shahi, A. (2025, December). LungNet: An Interpretable Machine Learning Framework for Early Lung Cancer Detection Using Structured Clinical Data. In 2025 IEEE 13th Conference on Systems, Process & Control (ICSPC) (pp. 181-186). IEEE.
- Rabbah, J.; Ridouani, M.; Hassouni, L. Improving pneumonia diagnosis with high-accuracy CNN-Based chest X-ray image classification and integrated gradient. Biomedical Signal Processing and Control 2025, 101, 107239. [Google Scholar] [CrossRef]
- An, Q.; Rahman, S.; Zhou, J.; Kang, J. J. A comprehensive review on machine learning in healthcare industry: classification, restrictions, opportunities and challenges. Sensors 2023, 23(9), 4178. [Google Scholar] [CrossRef] [PubMed]
- Abad, M.; Casas-Roma, J.; Prados, F. Generalizable disease detection using model ensemble on chest X-ray images. Scientific Reports 2024, 14(1), 5890. [Google Scholar] [CrossRef] [PubMed]
- Giri, A., Das, S. R., Joy, A. Z. M. J. U., Akib, A. S. M., Misat, M. M. H., Khadgi, M., ... & Shahi, B. (2025). Smart IoT Egg Incubator System with Machine Learning for Damaged Egg Detection. In International conference on WorldS4 (pp. 236-245). Springer, Cham.
- Mabrouk, A.; Diaz Redondo, R. P.; Dahou, A.; Abd Elaziz, M.; Kayed, M. Pneumonia detection on chest X-ray images using ensemble of deep convolutional neural networks. Applied Sciences 2022, 12(13), 6448. [Google Scholar] [CrossRef]
- Arun Prakash, J.; Asswin, C. R.; Ravi, V.; Sowmya, V.; Soman, K. P. Pediatric pneumonia diagnosis using stacked ensemble learning on multi-model deep CNN architectures. Multimedia tools and applications 2023, 82(14), 21311–21351. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Ren, F.; Li, Y.; Na, L.; Ma, Y. Pneumonia detection from chest X-ray images based on convolutional neural network. Electronics 2021, 10(13), 1512. [Google Scholar] [CrossRef]
- Giri, A., Akib, A. A. S., Hasib, A., Acharya, A., Prithibi, M. A., Rahman, R. H., ... & Taha, H. I. C. (2025, April). Design and development of a cost effective and modular cnc plotter for educational and prototyping applications. In 2025 IEEE 4th International Conference on Computing and Machine Intelligence (ICMI) (pp. 1-6). IEEE.
- Kundu, R.; Das, R.; Geem, Z. W.; Han, G. T.; Sarkar, R. Pneumonia detection in chest X-ray images using an ensemble of deep learning models. PloS one 2021, 16(9), e0256630. [Google Scholar] [CrossRef] [PubMed]
- Ayan, E.; Karabulut, B.; Ünver, H. M. Diagnosis of pediatric pneumonia with ensemble of deep convolutional neural networks in chest X-ray images. Arabian Journal for Science and Engineering 2022, 47(2), 2123–2139. [Google Scholar] [CrossRef] [PubMed]
- Al-qaness, M. A.; Zhu, J.; AL-Alimi, D.; Dahou, A.; Alsamhi, S. H.; Abd Elaziz, M.; Ewees, A. A. Chest X-ray Images for Lung Disease Detection Using Deep Learning Techniques: A Comprehensive Survey: MAA Al-qaness et al. Archives of Computational Methods in Engineering 2024, 31(6), 3267–3301. [Google Scholar] [CrossRef]
- Arora, A., Alderman, J. E., Palmer, J., Ganapathi, S., Laws, E., Mccradden, M. D., ... & Liu, X. (2023). The value of standards for health datasets in artificial intelligence-based applications. Nature medicine, 29(11), 2929-2938. [CrossRef] [PubMed]
- Giri, A., Hasib, A., Islam, M., Tazim, M. F., Rahman, M. S., Khadgi, M., & Akib, A. A. S. (2025, June). Real-time human fall detection using yolov5 on raspberry pi: An edge ai solution for smart healthcare and safety monitoring. In International Conference on Data Analytics & Management (pp. 493-507). Cham: Springer Nature Switzerland.
- Prakash, J. A.; Ravi, V.; Sowmya, V.; Soman, K. P. Stacked ensemble learning based on deep convolutional neural networks for pediatric pneumonia diagnosis using chest X-ray images. Neural Computing and Applications 2023, 35(11), 8259–8279. [Google Scholar] [CrossRef] [PubMed]
- An, Q.; Chen, W.; Shao, W. A deep convolutional neural network for pneumonia detection in x-ray images with attention ensemble. Diagnostics 2024, 14(4), 390. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.; Zeng, Z.; Zhang, J. Robust Multiclass Pneumonia Classification via Multi-Head Attention and Transfer Learning Ensemble. Applied Sciences 2025, 15(21), 11426. [Google Scholar] [CrossRef]
- Račić, L., Popović, T., & Šandi, S. (2021, February). Pneumonia detection using deep learning based on convolutional neural network. In 2021 25th International Conference on Information Technology (IT) (pp. 1-4). IEEE.
- Hussain, A.; Amin, S. U.; Lee, H.; Khan, A.; Khan, N. F.; Seo, S. An automated chest X-ray image analysis for covid-19 and pneumonia diagnosis using deep ensemble strategy. IEEE Access 2023, 11, 97207–97220. [Google Scholar] [CrossRef]
- Masud, M.; Bairagi, A. K.; Nahid, A. A.; Sikder, N.; Rubaiee, S.; Ahmed, A.; Anand, D. A pneumonia diagnosis scheme based on hybrid features extracted from chest radiographs using an ensemble learning algorithm. Journal of Healthcare Engineering 2021, 2021(1), 8862089. [Google Scholar] [CrossRef] [PubMed]
- Kheirdoust, A.; Barzanouni, F.; Rasoulian, A.; Behrouzi, F.; Esmailzadeh, A.; Ghaddaripouri, K.; Mazaheri Habibi, M. R. Evaluation of Machine Learning Methods Developed for Prediction and Diagnosis of Pneumonia: A Systematic Review. Health Science Reports 2025, 8(12), e71446. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Guleria, K. A deep learning based model for the detection of pneumonia from chest X-ray images using VGG-16 and neural networks. Procedia Computer Science 2023, 218, 357–366. [Google Scholar] [CrossRef]
- Akib, A. A. S., Giri, A., Islam, M., Sifa, F. J., Elahi, T. A., Aktia, A. N., ... & Khanna, A. (2024, October). Design and simulation of a quadruped robot. In International Conference on Data-Processing and Networking (pp. 373-385). Singapore: Springer Nature Singapore.
| Metric | Value |
|---|---|
| True Positive (TP) | 377 |
| True Negative (TN) | 183 |
| False Positive (FP) | 51 |
| False Negative (FN) | 13 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




