Submitted:
10 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Literature Selection and Data Extraction
| Study ID | Design | HbA1c Range (%) | % Glycated Insulin | Bioactivity Loss (% vs. Native) | Key Covariates |
|---|---|---|---|---|---|
| Hunter (2003) | HPLC-MS + Euglycemic Clamp (n=11) | 8.1 | 9% | 70% (low-dose clamp) | Age 50–60, Duration 5–10 yrs |
| Wautier et al. (2010) | In vitro MALDI-TOF (IR peptide binding) | N/A (20–60 mM glucose) | 14% (peptides) | 33% ↓ receptor binding | Glucose levels only |
| Rabbani et al. (2016) | CHO-IR-GLUT4 cell assay | Simulated hyperglycemia | Variable (dose-dependent) | 50–70% ↓ AKT phosphorylation | ROS markers, Losartan modulation |
| Zanella et al. (2019) | Vascular relaxation (RBCs, diabetic cohort) | Variable | N/A (Hb proxy: high glycation) | ↓ HbSNO 40% | Tissue O₂, BMI |
| Pfeifer et al. (2023) | Cross-sectional cohort (n=1362 T2DM) | 5.3–15.2 | Indirect (HbA1c proxy) | N/A (r = −0.15 SpO₂-glucose) | BMI 27.6, Age 66, eGFR |
| Wang et al. (2019) | Leukocyte adhesion (PMNs, diabetic) | Variable (T1/T2DM) | Indirect (O-glycation ↑3×) | 3-fold ↑ adhesion | PKC inhibition (LY379196) |
2.2. Quantitative Modeling
- : residual insulin bioactivity (% baseline)
- : total circulating insulin (100%)
- : slope coefficient (fitted = 0.82 ± 0.07)
-
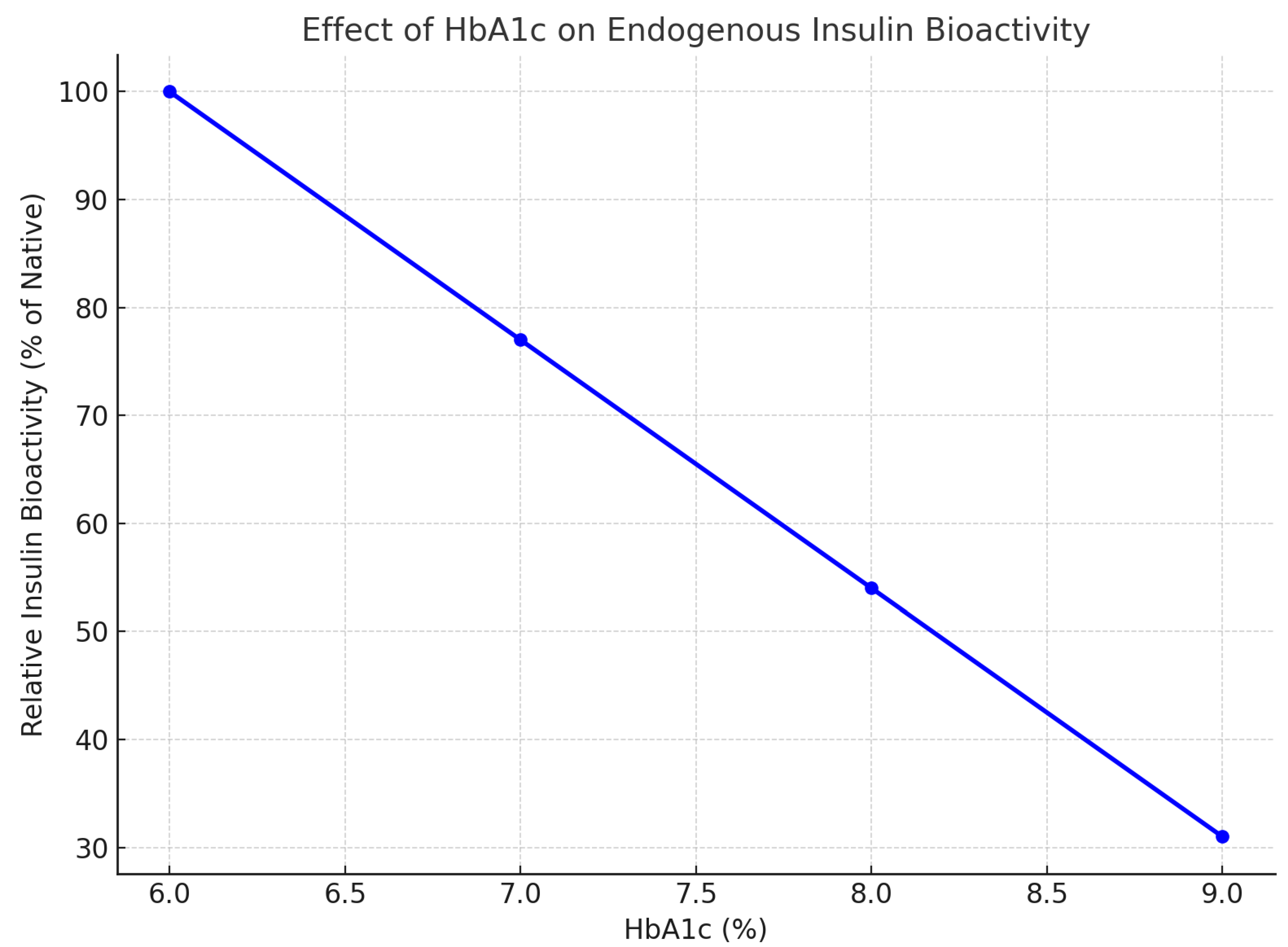
: HbA1c inflection for 50% activity loss = 7.6 ± 0.2%Model comparison favored the sigmoidal over linear regression (ΔAIC = −12.4), indicating a threshold-like decline beyond 7.5–8.0% HbA1c, consistent with the onset of microvascular complications (Table 3).
2.3. Operational Definitions
3. Evidence Synthesis: Molecular and Clinical Insights
4. Pathophysiologic Model: The Glycohypoxic Cycle and Transition to Insulin Failure
4.1. Self-Amplifying Loop
4.2. Integration with Complications
4.3. Phase-Threshold Mapping Defines the Biochemical Trajectory:
- Compensation (HbA1c <7%) – Minimal glycation (≤3%), reversible resistance.
- Onset (7–8%) – Emerging glycohypoxia (5–8%), 30–50% signaling loss.
- Failure (>8.5%) – Nonlinear acceleration (9–10% glycation, ≥70% loss).
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Banday, M.Z.; Sameer, A.S.; Nissar, S. Pathophysiology of diabetes: An overview. Avicenna journal of medicine 2020, 10, 174–188. [Google Scholar] [CrossRef]
- Młynarska, E.; Czarnik, W.; Dzieża, N.; Jędraszak, W.; Majchrowicz, G.; Prusinowski, F.; Stabrawa, M.; Rysz, J.; Franczyk, B. Type 2 Diabetes Mellitus: New Pathogenetic Mechanisms, Treatment and the Most Important Complications. International Journal of Molecular Sciences 2025, 26, 1094. [Google Scholar] [CrossRef]
- Khalid, M.; Petroianu, G.; Adem, A. Advanced Glycation End Products and Diabetes Mellitus: Mechanisms and Perspectives. Biomolecules 2022, 12, 542. [Google Scholar] [CrossRef] [PubMed]
- Leow, M.K. Glycated Hemoglobin (HbA1c): Clinical Applications of a Mathematical Concept. Acta informatica medica: AIM: journal of the Society for Medical Informatics of Bosnia & Herzegovina: casopis Drustva za medicinsku informatiku BiH 2016, 24, 233–238. [Google Scholar] [CrossRef]
- Akl, Maher M.1,*,†; Ahmed, Amr2. Glycohypoxia: a hypothesis linking chronic hyperglycemia to functional hypoxia and diabetic complications in type 2 diabetes. Medical Gas Research ():10.4103/mgr.MEDGASRES-D-25-00137, March 14, 2026. [CrossRef]
- LIVIO LUZI, CONCETTA MACRÌ, ANNA FERRULLI, CESARE C. BERRA, CRISTINA ROMANO, STEFANO MASSARINI; 1400-P: Low Blood Oxygen Saturation Is Associated with Impaired Glucose Control in Diabetes. Diabetes 2023, 72 (Supplement_1), 1400–P. [CrossRef]
- Hunter, S.J.; Boyd, A.C.; O'Harte, F.P.; McKillop, A.M.; Wiggam, M.I.; Mooney, M.H.; McCluskey, J.T.; Lindsay, J.R.; Ennis, C.N.; Gamble, R.; Sheridan, B.; Barnett, C.R.; McNulty, H.; Bell, P.M.; Flatt, P.R. Demonstration of glycated insulin in human diabetic plasma and decreased biological activity assessed by euglycemic-hyperinsulinemic clamp technique in humans. Diabetes 2003, 52, 492–498. [Google Scholar] [CrossRef]
- Rhinesmith, T.; Turkette, T.; Root-Bernstein, R. Rapid Non-Enzymatic Glycation of the Insulin Receptor under Hyperglycemic Conditions Inhibits Insulin Binding In Vitro: Implications for Insulin Resistance. International Journal of Molecular Sciences 2017, 18, 2602. [Google Scholar] [CrossRef]
- Chibber, R.; Ben-Mahmud, B.M.; Mann, G.E.; Zhang, J.J.; Kohner, E.M. Protein kinase C beta2-dependent phosphorylation of core 2 GlcNAc-T promotes leukocyte-endothelial cell adhesion: a mechanism underlying capillary occlusion in diabetic retinopathy. Diabetes 2003, 52, 1519–1527. [Google Scholar] [CrossRef] [PubMed]
- James, P.E.; Lang, D.; Tufnell-Barret, T.; Milsom, A.B.; Frenneaux, M.P. Vasorelaxation by red blood cells and impairment in diabetes: reduced nitric oxide and oxygen delivery by glycated hemoglobin. Circulation research 2004, 94, 976–983. [Google Scholar] [CrossRef] [PubMed]
- Rhinesmith, T.; Turkette, T.; Root-Bernstein, R. Rapid Non-Enzymatic Glycation of the Insulin Receptor under Hyperglycemic Conditions Inhibits Insulin Binding In Vitro: Implications for Insulin Resistance. International journal of molecular sciences 2017, 18, 2602. [Google Scholar] [CrossRef]
- Ansari, N.A.; Dash, D. Amadori glycated proteins: role in production of autoantibodies in diabetes mellitus and effect of inhibitors on non-enzymatic glycation. Aging and disease 2013, 4, 50–56. [Google Scholar]
- Huang, J.P.; Huang, S.S.; Deng, J.Y.; Hung, L.M. Impairment of insulin-stimulated Akt/GLUT4 signaling is associated with cardiac contractile dysfunction and aggravates I/R injury in STZ-diabetic rats. Journal of biomedical science 2009, 16, 77. [Google Scholar] [CrossRef]
- Walke, P.B.; Bansode, S.B.; More, N.P.; Chaurasiya, A.H.; Joshi, R.S.; Kulkarni, M.J. Molecular investigation of glycated insulin-induced insulin resistance via insulin signaling and AGE-RAGE axis. Biochimica et biophysica acta. Molecular basis of disease 2021, 1867, 166029. [Google Scholar] [CrossRef]
- Rojas, A.; Lindner, C.; Gonzàlez, I.; Morales, M.A. Advanced-glycation end-products axis: A contributor to the risk of severe illness from COVID-19 in diabetes patients. World journal of diabetes 2021, 12, 590–602. [Google Scholar] [CrossRef]
- Walke, P.B.; Bansode, S.B.; More, N.P.; Chaurasiya, A.H.; Joshi, R.S.; Kulkarni, M.J. Molecular investigation of glycated insulin-induced insulin resistance via insulin signaling and AGE-RAGE axis. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2021, 1867, 166029. [Google Scholar] [CrossRef] [PubMed]
- Bahaedin M. Ben-Mahmud, Giovanni E. Mann, Alessandro Datti, Aldo Orlacchio, Eva M. Kohner, Rakesh Chibber; Tumor Necrosis Factor-α in Diabetic Plasma Increases the Activity of Core 2 GlcNAc-T and Adherence of Human Leukocytes to Retinal Endothelial Cells: Significance of Core 2 GlcNAc-T in Diabetic Retinopathy. Diabetes 2004, 53, 2968–2976. [CrossRef]
- Curran, C.S.; Kopp, J.B. RAGE pathway activation and function in chronic kidney disease and COVID-19. Frontiers in medicine 2022, 9, 970423. [Google Scholar] [CrossRef]
- Lu, Z.; Fan, B.; Li, Y.; Zhang, Y. RAGE plays key role in diabetic retinopathy: a review. Biomedical engineering online 2023, 22, 128. [Google Scholar] [CrossRef] [PubMed]
- Woo, J.R.; Bae, S.H.; Wales, T.E.; Engen, J.R.; Lee, J.; Jang, H.; Park, S. The serine phosphorylations in the IRS-1 PIR domain abrogate IRS-1 and IR interaction. Proceedings of the National Academy of Sciences of the United States of America 2024, 121, e2401716121. [Google Scholar] [CrossRef]
- Gieroba, B.; Kryska, A.; Sroka-Bartnicka, A. Type 2 diabetes mellitus - conventional therapies and future perspectives in innovative treatment. Biochemistry and biophysics reports 2025, 42, 102037. [Google Scholar] [CrossRef]
- Michalani, M.L.E.; Passarelli, M.; Machado, U.F. Nuclear Factor-Kappa-B Mediates the Advanced Glycation End Product-Induced Repression of Slc2a4 Gene Expression in 3T3-L1 Adipocytes. International journal of molecular sciences 2024, 25, 8242. [Google Scholar] [CrossRef]
- Jonny Violetta, L.; Kartasasmita, A.S.; Supriyadi, R.; Rita, C. Circulating Biomarkers to Predict Diabetic Retinopathy in Patients with Diabetic Kidney Disease. Vision (Basel, Switzerland) 2023, 7, 34. [Google Scholar] [CrossRef]
- Shah, H.S.; McGill, J.B.; Hirsch, I.B.; Wu, C.; Galecki, A.; de Boer, I.H.; Mauer, M.; Doria, A. Poor Glycemic Control Is Associated With More Rapid Kidney Function Decline After the Onset of Diabetic Kidney Disease. The Journal of clinical endocrinology and metabolism 2024, 109, 2124–2135. [Google Scholar] [CrossRef]
- Baldimtsi, E.; Amezcua, S.; Ulander, M.; et al. HbA1c and the risk of developing peripheral neuropathy in childhood-onset type 1 diabetes: a follow-up study over 3 decades. Diabetes Metab Res Rev. 2024, e3825. [Google Scholar] [CrossRef]
- Chen, J.; Yin, D.; Dou, K. Intensified glycemic control by HbA1c for patients with coronary heart disease and Type 2 diabetes: a review of findings and conclusions. Cardiovasc Diabetol 2023, 22, 146. [Google Scholar] [CrossRef] [PubMed]
- Wan, E.Y.F.; Yu, E.Y.T.; Mak, I.L.; Youn, H.M.; Chan, K.S.; Chan, E.W.Y.; Wong, I.C.K.; Lam, C.L.K. Diabetes with poor-control HbA1c is cardiovascular disease 'risk equivalent' for mortality: UK Biobank and Hong Kong population-based cohort study. BMJ open diabetes research & care 2023, 11, e003075. [Google Scholar] [CrossRef]
- Alhawiti, N.M.; Elsokkary, E.M.; Aldali, J.A.; et al. Investigating the impact of glycated hemoglobin levels on stroke severity in patients with acute ischemic stroke. Sci Rep 2025, 15, 12114. [Google Scholar] [CrossRef]
- Elizabeth Selvin, Keattiyoat Wattanakit, Michael W. Steffes, Josef Coresh, A. Richey Sharrett; HbA1c and Peripheral Arterial Disease in Diabetes: The Atherosclerosis Risk in Communities study. Diabetes Care 2006, 29, 877–882. [CrossRef]
- Miyake, T.; Furukawa, S.; Matsuura, B.; Yoshida, O.; Miyazaki, M.; Shiomi, A.; Kanamoto, A.; Nakaguchi, H.; Nakamura, Y.; Imai, Y.; Koizumi, M.; Watanabe, T.; Yamamoto, Y.; Koizumi, Y.; Tokumoto, Y.; Hirooka, M.; Kumagi, T.; Takesita, E.; Ikeda, Y.; Abe, M.; Hiasa, Y. Glycemic Control Is Associated with Histological Findings of Nonalcoholic Fatty Liver Disease. Diabetes & metabolism journal 2024, 48, 440–448. [Google Scholar] [CrossRef]
- Xiao, Y.; Hong, X.; Neelagar, R.; et al. Association between glycated hemoglobin A1c levels, control status, and cognitive function in type 2 diabetes: a prospective cohort study. Sci Rep 2025, 15, 5011. [Google Scholar] [CrossRef]
- Akyüz, S.; Bahçecioğlu Mutlu, A.B.; Guven, H.E.; Başak, A.M.; Yilmaz, K.B. Elevated HbA1c level associated with disease severity and surgical extension in diabetic foot patients. Diyabetik ayak hastalarında yüksek HbA1c düzeyi ile hastalık şiddeti ve cerrahi seviye ilişkisi. Ulusal travma ve acil cerrahi dergisi = Turkish journal of trauma & emergency surgery: TJTES 2023, 29, 1013–1018. [Google Scholar] [CrossRef]
- Hou, Z.; Wang, H.; He, B.; et al. The association between glycemic indicators and bone mineral density and osteoporosis: a cross-sectional study. Sci Rep 2025, 15, 28302. [Google Scholar] [CrossRef]
- Habous, M.; Tal, R.; Tealab, A.; Soliman, T.; Nassar, M.; Mekawi, Z.; Mahmoud, S.; Abdelwahab, O.; Elkhouly, M.; Kamr, H.; Remeah, A.; Binsaleh, S.; Ralph, D.; Mulhall, J. Defining a glycated haemoglobin (HbA1c) level that predicts increased risk of penile implant infection. BJU international 2018, 121, 293–300. [Google Scholar] [CrossRef]
- Rhoden, E.L.; Ribeiro, E.P.; Riedner, C.E.; Teloken, C.; Souto, C.A. Glycosylated haemoglobin levels and the severity of erectile function in diabetic men. BJU international 2005, 95, 615–617. [Google Scholar] [CrossRef] [PubMed]
- Christman, A.L.; Selvin, E.; Margolis, D.J.; Lazarus, G.S.; Garza, L.A. Hemoglobin A1c predicts healing rate in diabetic wounds. Journal of Investigative Dermatology 2011, 131, 2121–2127. [Google Scholar] [CrossRef] [PubMed]
| Phase | HbA1c Range (%) | Key Mechanisms | % Glycated Insulin | Clinical Implication |
|---|---|---|---|---|
| Compensation | <7 | Receptor-level resistance (IRS-1/2 desensitization) | ≤3% | Oral therapy effective; adaptive hyperinsulinemia |
| Complication Onset | 7–8 | ROS ignition, endothelial breach, early glycohypoxia | 5–8% | Emerging microvascular injury; partial bioactivity loss |
| Post-Receptor Failure | 8–9 | Glycohypoxia and AGE–RAGE activation | 9–10% | 70–80% loss in PI3K/Akt signaling; exogenous shift |
| Exogenous Dependence | >9 | β-cell dedifferentiation, oxidative exhaustion | >10% | Complete ligand failure; multi-organ complications |
| HbA1c Range (%) | Predicted SpO₂ (%) | Relative Insulin Bioactivity (% of I₀) | Dominant Pathophysiological Process |
|---|---|---|---|
| 5.0–6.0 | 97–98 | 100–90 | Normal oxidative metabolism |
| 6.0–7.0 | 96–95 | 90–77 | Early glycation & mild ROS activation |
| 7.0–8.0 | 94–92 | 77–55 | RAGE–ROS–HIF-1α activation (pre-hypoxia) |
| 8.0–9.0 | 92–90 | 55–30 | Glycohypoxic transition & endothelial dysfunction |
| >9.0 | <90 | <30 | Established glycohypoxic vasculopathy |
| HbA1c (%) | % Glycated Insulin | Bioactivity Loss (Linear) | Bioactivity Loss (Sigmoid) | Interpretation |
|---|---|---|---|---|
| 6.0 | ≤3% | 0% | 5% | Baseline – No complications |
| 7.5 | 5% | 13% | 25% | Threshold – Adaptive failure |
| 8.1 | 9% | 70% | 70% | Inflection – Functional collapse |
| 9.0 | 10% | 80% | 85% | Advanced failure |
| 10.0 | >10% | >90% | 92% | Saturation – End-stage dysfunction |
| Trigger | Mechanism | Effect on Insulin Bioactivity | Linked Complication | Evidence |
|---|---|---|---|---|
| HbA1c Elevation | Amadori → AGE (+164 Da B-chain adduct) | ~9% glycated insulin | Microangiopathy / ↓ O₂ unloading | [5,7] |
| Tissue Hypoxia | ↓ SpO₂ / HbSNO / ↑ HIF-1α / ROS | 50–70% ↓ GLUT4 translocation | Vascular occlusion / Retinopathy | [6,9,10] |
| Glycated Insulin | RAGE activation → NF-κB ↑ / IRS-1 Ser307 ↑ | ↓ IR/AKT phosphorylation | Nephropathy / Oxidative stress | [7,8] |
| IR Glycation | Steric hindrance → 33% ↓ binding | Post-receptor resistance | Metabolic failure | [7,8] |
| PKCβ₂ Activation | Core 2 GlcNAc-T phosphorylation | ↑ Adhesion / Inflammation | Retinopathy / β-cell stress | [9,17] |
| Complication | Typical HbA1c Onset (%) | Key Pathophysiologic Driver | Mechanistic Notes / Biomarkers | Ref. |
|---|---|---|---|---|
| Retinopathy | 7.5–8.0 | PKCβ₂ activation + O-glycosylation | ↑ PSGL-1 glycosylation → leukocyte adhesion → capillary occlusion | [23] |
| Nephropathy | 8.0–8.5 | RAGE–TGFβ axis | NF-κB activation → fibrosis, basement-membrane thickening | [24] |
| Neuropathy | 8.5–9.0 | Oxidative stress, ischemic demyelination | ↓ NO, ↑ ROS, axonal degeneration | [25] |
| Macroangiopathy / Atherosclerosis | 7.8–8.5 | Endothelial AGE crosslinking | ↓ eNOS, ↑ VCAM-1, ↑ oxidized LDL | [26] |
| Cardiomyopathy | 8.5–9.0 | Mitochondrial dysfunction, RAGE–MAPK | ↓ ATP generation, ↑ apoptosis | [27] |
| Stroke (Ischemic) | 8.5–9.0 | Hypercoagulability + endothelial damage | ↑ fibrinogen, ↓ NO bioavailability | [28] |
| Peripheral Artery Disease (PAD) | 8.0–8.5 | AGE crosslinking + elastin fragmentation | ↑ arterial stiffness; ABI < 0.9 | [29] |
| NAFLD / NASH | 7.5–8.0 | Insulin resistance + oxidative glycation | ↑ TNF-α, ↑ hepatic ROS, lipotoxicity | [30] |
| Cognitive Decline / Alzheimer’s-like | 8.0–8.5 | Brain insulin glycation, amyloid crosslinking | ↓ IDE activity, ↑ Aβ–AGE complexes | [31] |
| Diabetic Foot / Ulceration | 8.5–9.0 | Ischemia + poor angiogenesis | ↓ VEGF response, ↓ keratinocyte migration | [32] |
| Osteopathy (Bone fragility) | 8.0–8.5 | Collagen AGE accumulation | ↓ osteocalcin, brittle bone architecture | [33] |
| Immune Dysfunction / Infection Risk | 7.8–8.3 | Neutrophil glycation + ROS exhaustion | ↓ chemotaxis, ↓ phagocytosis | [34] |
| Erectile Dysfunction | 8.0–8.5 | eNOS uncoupling + ROS-NO trapping | ↓ cGMP, ↓ cavernous vasodilation | [35] |
| Wound-Healing Delay | 8.0–8.6 | Fibroblast glycation + angiogenesis block | ↓ TGF-β1, ↓ collagen deposition | [36] |
| Insulin Failure (This Study) | ≈ 8.0 | Glycohypoxia-driven insulin glycation | 9% monoglycated insulin; 70% bioactivity loss; RAGE/ROS amplification |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




