Submitted:
10 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Clinical Cases
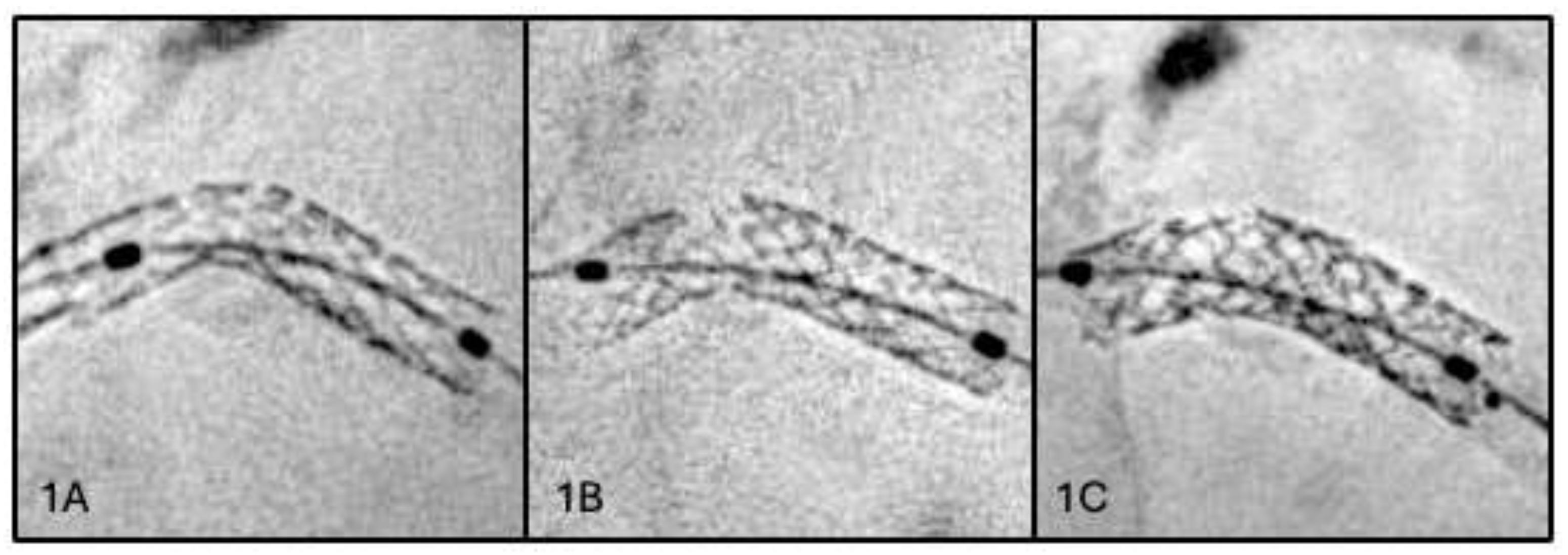
2.1. First Scenario: Stent Fracture
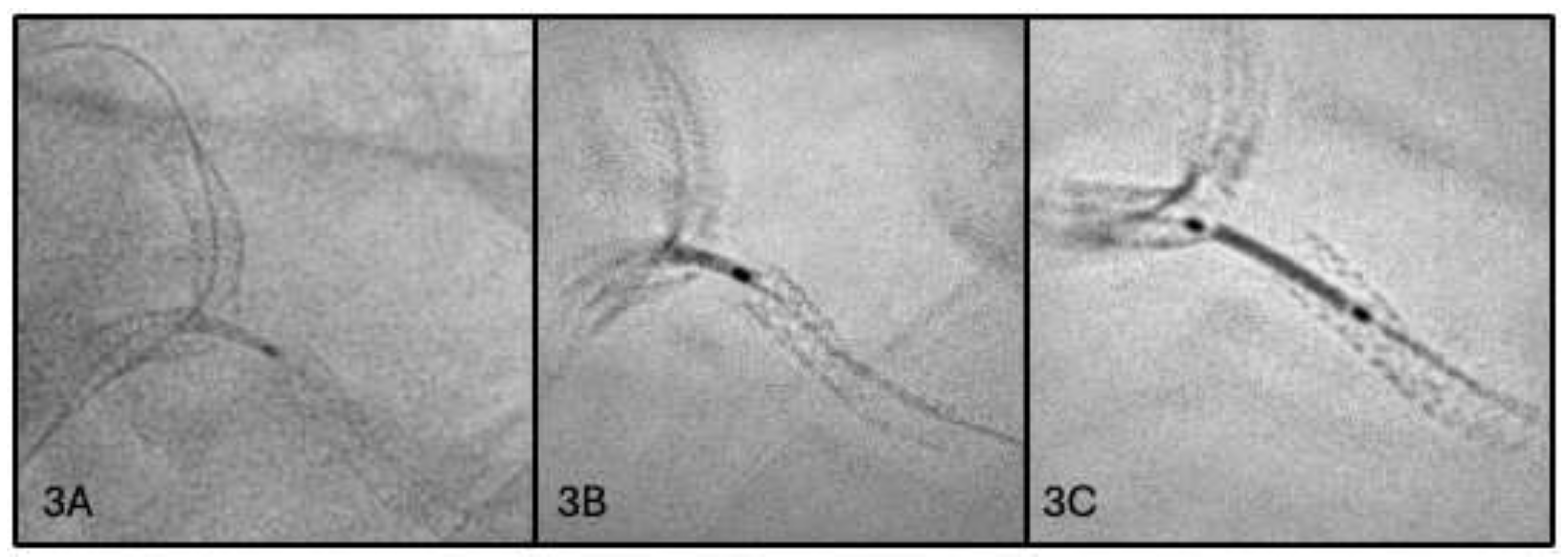
2.2. Second Scenario: Stent Loss
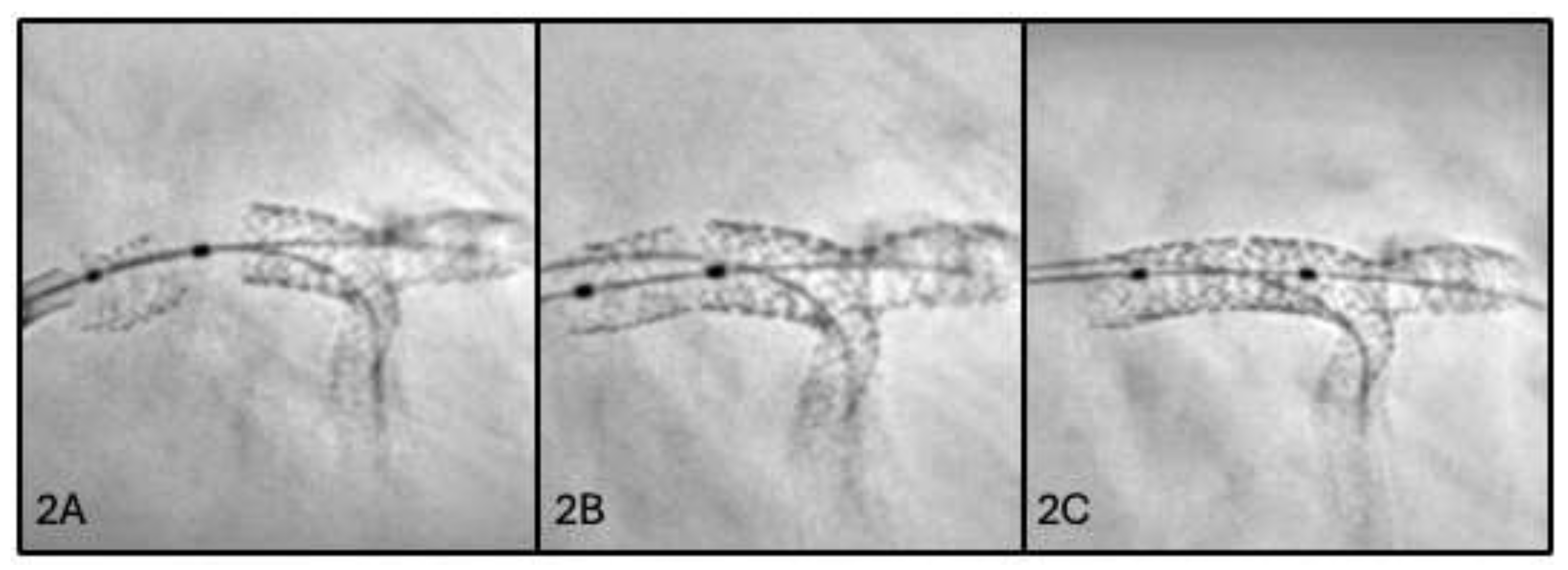
2.3. Third Scenario: Stent Dislodgement
2.4. Fourth Scenario: Stent Underexpansion
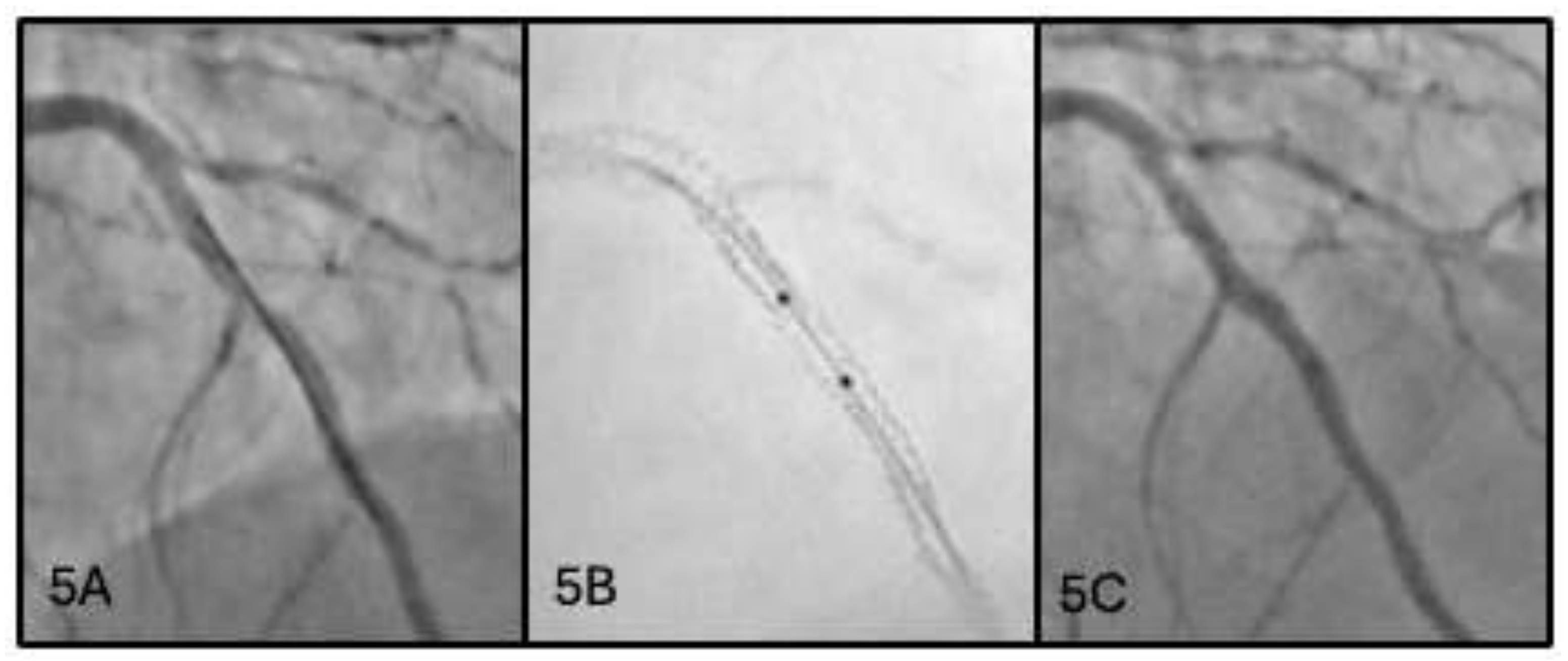
2.5. Fifth Scenario: Missed Overlap
3. Conclusions
4. Future Perspectives
Abbreviations
| PCI | Percutaneous coronary intervention |
| ESV | Enhanced stent visualization |
| IVUS | Intravascular ultrasound |
| OCT | Optical coherence tomography |
| LM | Left main |
| ACS | Acute coronary syndrome |
| STEMI | ST-elevation myocardial infarction |
| NSTEMI | Non-ST-elevation myocardial infarction |
| TLR | Target lesion restenosis |
| CABG | Coronary artery bypass grafting |
| LAD | Left anterior descending artery |
| DES | Drug-eluting stent |
| RCA | Right coronary artery |
| LCx | Left circumflex artery |
| TAP | T and protrusion |
| DCB | Drug-coated balloon |
| IVL | Intravascular lithotripsy |
| MLA | Minimum lumen area |
| D1 | Diagonal branch |
| POT | Proximal optimization technique |
References
- Ali, ZA; Maehara, A; Généreux, P; Shlofmitz, RA; Fabbiocchi, F; Nazif, TM; et al. Optical coherence tomography compared with intravascular ultrasound and with angiography to guide coronary stent implantation (ILUMIEN III: OPTIMIZE PCI): a randomised controlled trial. Lancet 2016, 388, 2618–2628. [Google Scholar] [CrossRef] [PubMed]
- Kang, DY; Ahn, JM; Yun, SC; Hur, SH; Cho, YK; Lee, CH; et al. Guiding Intervention for Complex Coronary Lesions by Optical Coherence Tomography or Intravascular Ultrasound. J Am Coll Cardiol. 2024, 83, 401–413. [Google Scholar] [CrossRef] [PubMed]
- Tufaro, V; Jaffer, FA; Serruys, PW; Onuma, Y; van der Steen, AFW; Stone, GW; et al. Emerging Hybrid Intracoronary Imaging Technologies and Their Applications in Clinical Practice and Research. JACC Cardiovasc Interv. 2024, 17, 1963–1979. [Google Scholar] [CrossRef] [PubMed]
- Truesdell, AG; Alasnag, MA; Kaul, P; Rab, ST; Riley, RF; Young, MN; et al. Intravascular Imaging During Percutaneous Coronary Intervention: JACC State-of-the-Art Review. J Am Coll Cardiol. 2023, 81, 590–605. [Google Scholar] [CrossRef] [PubMed]
- Scalia, L; Squillace, M; Popolo Rubbio, A; Poletti, E; Agnello, F; Sisinni, A; et al. Modern Imaging Techniques for Percutaneous Coronary Intervention Guidance: A Focus on Intravascular Ultrasound and Optical Coherence Tomography. J Clin Med. 2025, 14(24). [Google Scholar] [CrossRef] [PubMed]
- Rao, SV; O’Donoghue, ML; Ruel, M; Rab, T; Tamis-Holland, JE; Alexander, JH; et al. 2025 ACC/AHA/ACEP/NAEMSP/SCAI Guideline for the Management of Patients With Acute Coronary Syndromes: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2025, 85, 2135–2237. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y; Jing, L; Pan, S; Yan, S; Wang, F; Yu, H; et al. Optical coherence tomography: evaluating the effects of stent boost subtract imaging on stent underexpansion in STEMI patients. BMC Cardiovasc Disord. 2022, 22, 62. [Google Scholar] [CrossRef] [PubMed]
- Oh, DJ; Choi, CU; Kim, S; Im, SI; Na, JO; Lim, HE; et al. Effect of StentBoost imaging guided percutaneous coronary intervention on mid-term angiographic and clinical outcomes. Int J Cardiol. 2013, 168, 1479–1484. [Google Scholar] [CrossRef] [PubMed]
- Mansour, HM; Mohamed, AM; Ibrahim, SG; Ibrahim, AM; Mohamed, RG. Value of stent boost imaging in decision making after coronary stenting. Int J Cardiovasc Imaging 2023, 39, 2557–2566. [Google Scholar] [CrossRef] [PubMed]
- Biscaglia, S; Tebaldi, M; Tumscitz, C; Pavasini, R; Marchesini, J; Gallo, F; et al. Prospective Identification of Stent Fracture by Enhanced Stent Visualization System During Percutaneous Coronary Intervention. Circ J. 2016, 81, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Blicq, E; Georges, JL; Elbeainy, E; Gibault-Genty, G; Benjemaa, K; Jerbi, B; et al. Detection of Stent Underdeployment by StentBoost Imaging. J Interv Cardiol. 2013, 26, 444–453. [Google Scholar] [CrossRef] [PubMed]
- Sant Bakshsingh, V; Bundhoo, SS; Protty, MB. The role of enhanced stent visualization imaging in percutaneous coronary intervention: a systematic review of efficacy and clinical outcomes. Expert Rev Cardiovasc Ther. 2025, 23, 915–921. [Google Scholar] [CrossRef] [PubMed]
- Fysal, Z; Hyde, T; Barnes, E; McCrea, W; Ramcharitar, S. Evaluating stent optimisation technique (StentBoost®) in a dedicated bifurcation stent (the Tryton™). Cardiovasc Revasc Med. 2014, 15, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z; Yang, S; Jing, L; Liu, H. Impact of StentBoost subtract imaging on patient radiation exposure during percutaneous coronary intervention. Int J Cardiovasc Imaging 2013, 29, 1207–1213. [Google Scholar] [CrossRef] [PubMed]
- Shabbir, A; Ali, Z; Colletti, G; Dudek, D; Garbo, R; Hellig, F; et al. Ultra-Low-Contrast PCI: A Structured Approach to Reducing Dependence on Contrast Vessel Opacification in PCI. JACC Cardiovasc Interv. 2025, 18, 409–424. [Google Scholar] [CrossRef] [PubMed]
- Alqawasmi M, Blan.

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



