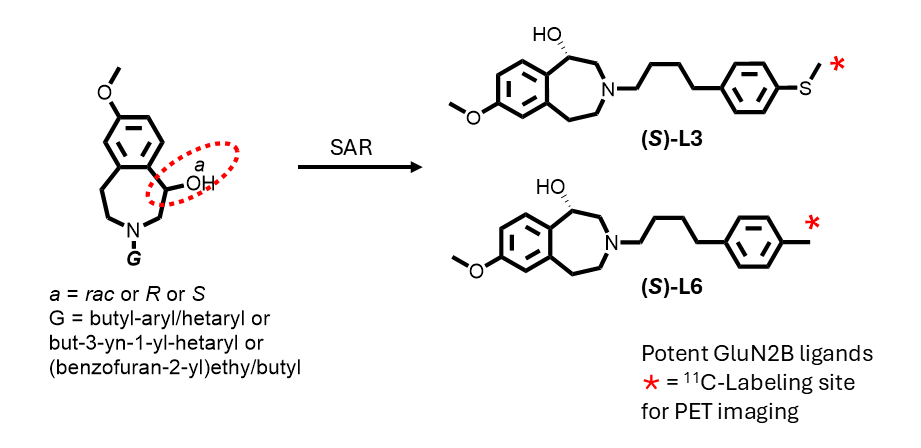
N-Methyl-D-aspartate (NMDA) receptors are ligand- and voltage-gated ion channels essential for synaptic plasticity, learning, and memory. The GluN2B subunit, highly expressed in the forebrain and spinal cord, is implicated in multiple neurological and psychiatric disorders, making it an attractive target for positron emission tomography (PET) imaging. However, the development of selective GluN2B PET radioligands remains challenging. Here, we describe the design, synthesis, and evaluation of eighteen 3-alkylaryl derivatives of 7-methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol, including enantiomerically resolved compounds, as candidate PET radioligands. Structure–activity relationship studies show that binding affinity is largely insensitive to electronic and steric variation at the terminal aryl group but strongly dependent on alkyl linker length, with a four-carbon chain providing optimal affinity. Binding affinity does not correlate with calculated lipophilicity, suggesting hydrophobicity is not the primary determinant of receptor interaction. Absolute configuration was established using vibrational circular dichroism and infrared spectroscopy, and docking studies provided insight into enantiomer-specific binding modes. Two ligands, L3 and L6, and their enantiomers exhibited high GluN2B affinity, favorable physicochemical properties, and suitability for carbon-11 labeling. PET imaging confirmed strong and specific brain binding of the radiolabeled compounds. These findings establish this scaffold as a promising platform for GluN2B PET ligand development.