Submitted:
10 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Study Design and Setting
Study Population and Sampling
Inclusion Criteria, Variables, and Clinical Examination
Scaling and Root Planing (SRP)
Saliva Collection and Handling
DNA Extraction, PCR Amplification, and 16S rRNA Sequencing
Bioinformatics and Microbiome Analysis
Statistical Analysis
Ethical Approval
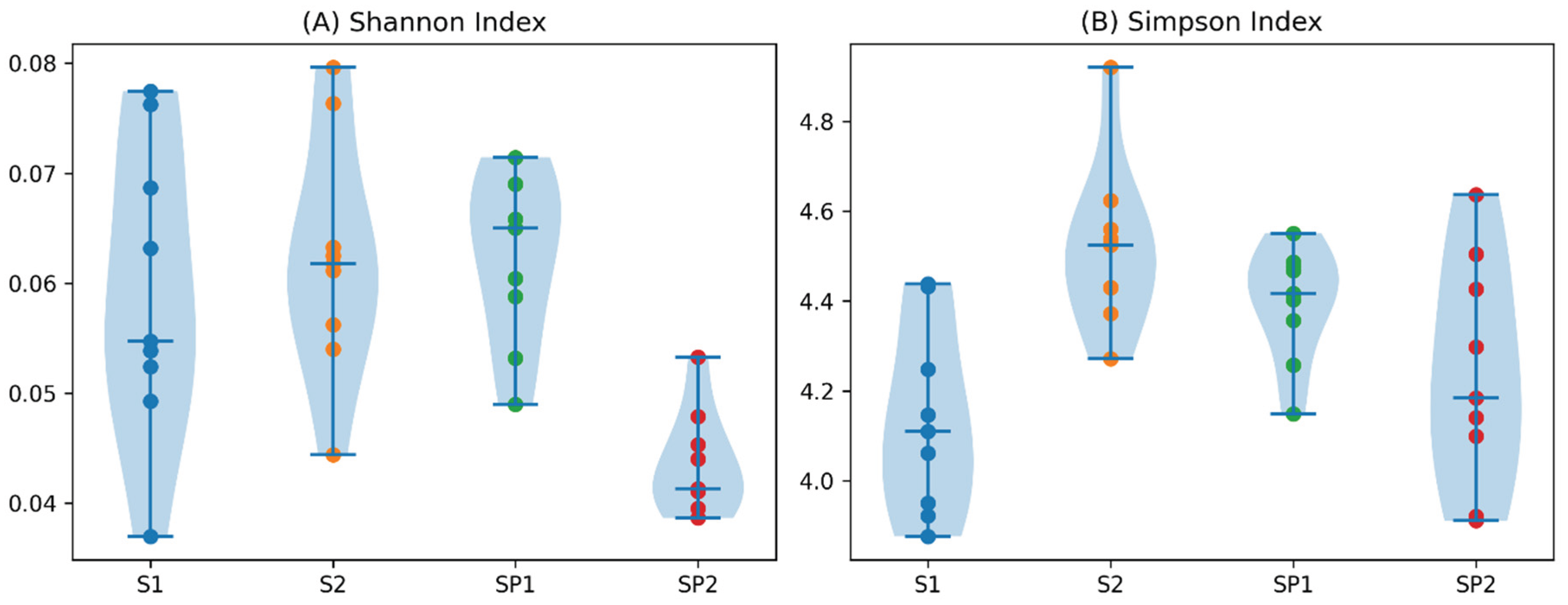
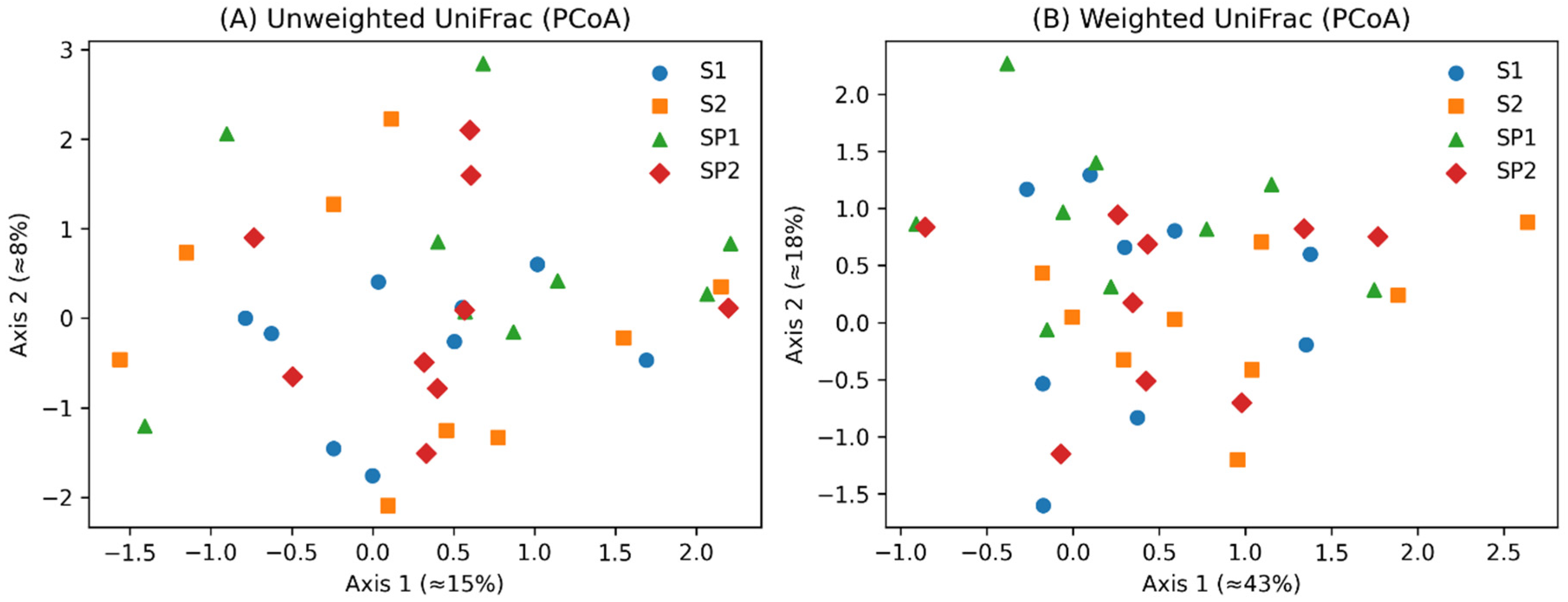
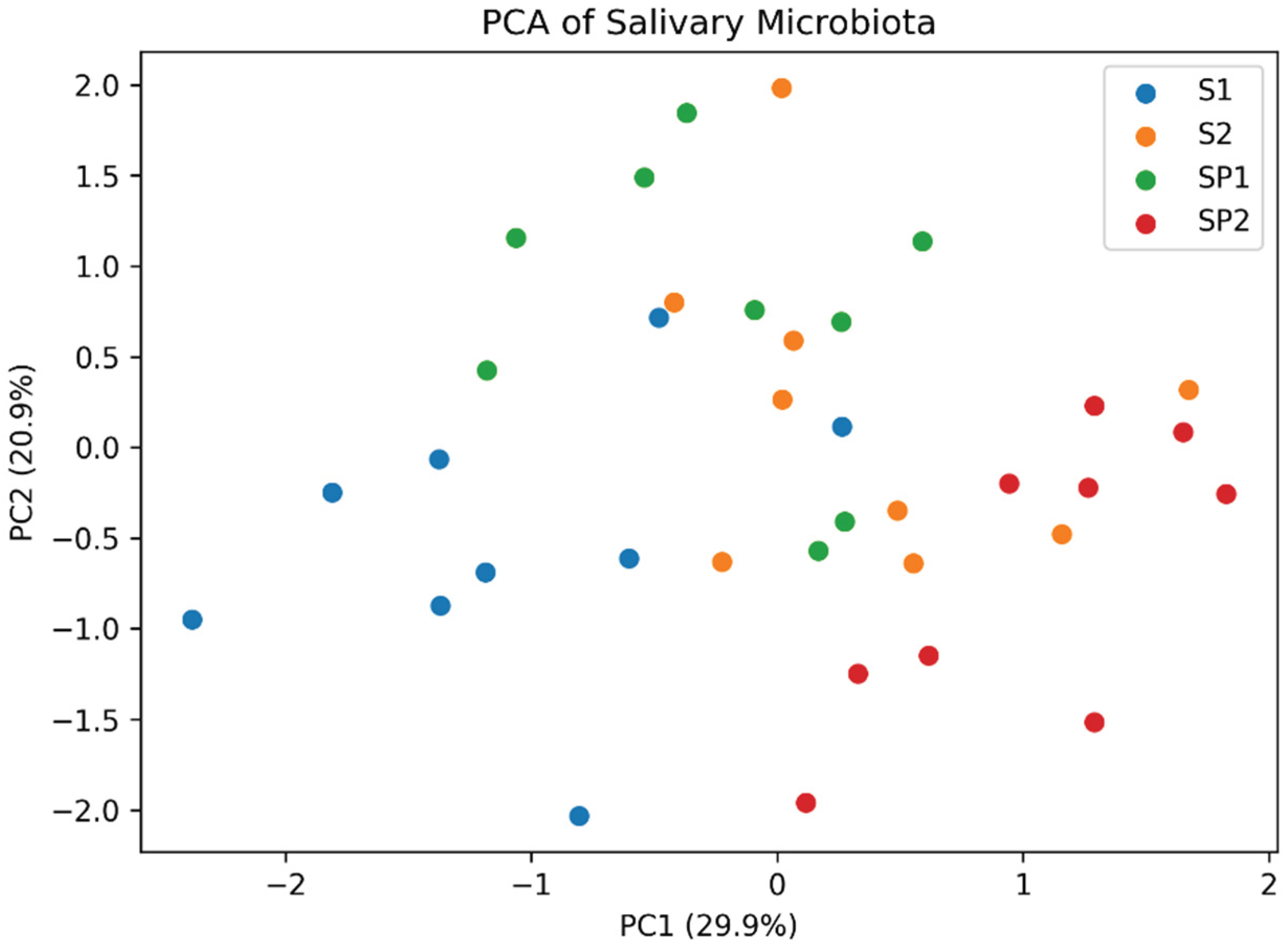
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mulyani, A.T.; Khairinisa, M.A.; Khatib, A.; Chaerunisaa, A.Y. Understanding stunting: Impact, causes, and strategy to accelerate stunting reduction—A narrative Review. Nutrients 2025, 17, 1493. [Google Scholar] [CrossRef]
- Hadi, F.; Kaeun, M.; Mursyid, F.; Melinda, V. Biological Pathways of Oral Health Inequality: A Longitudinal Analysis of Stunting, Enamel Defects, and Salivary Immunity on Caries Trajectories in Indonesian Children. Crown: Journal of Dentistry and Health Research 2024, 2, 161–171. [Google Scholar] [CrossRef]
- Nursal, D.G.A.; Kasuma, N.; Wulandari, R.W.; Ali, H.; Roza, D.; Setiawan, I.; Juwita, D.R.; Ernesto, G.; Effendi, M.D.S.; Wirza, T.R. Stunting and its effects on salivary biomarkers and oral health: a cross-sectional analysis of flow rate, pH, and buffering capacity. The Open Dentistry Journal 2025, 19. [Google Scholar] [CrossRef]
- Sedghi, L.; DiMassa, V.; Harrington, A.; Lynch, S.V.; Kapila, Y.L. The oral microbiome: Role of key organisms and complex networks in oral health and disease. Periodontology 2000 2021, 87, 107–131. [Google Scholar] [CrossRef] [PubMed]
- Caballero-Flores, G.; Pickard, J.M.; Núñez, G. Microbiota-mediated colonization resistance: mechanisms and regulation. Nature Reviews Microbiology 2023, 21, 347–360. [Google Scholar] [CrossRef]
- Scannapieco, F.A.; Dongari-Bagtzoglou, A. Dysbiosis revisited: Understanding the role of the oral microbiome in the pathogenesis of gingivitis and periodontitis: A critical assessment. Journal of periodontology 2021, 92, 1071–1078. [Google Scholar] [CrossRef]
- Morales, F.; Montserrat-De la Paz, S.; Leon, M.J.; Rivero-Pino, F. Effects of malnutrition on the immune system and infection and the role of nutritional strategies regarding improvements in children’s health status: a literature review. Nutrients 2023, 16, 1. [Google Scholar] [CrossRef]
- Vinel, A.; Al Halabi, A.; Roumi, S.; Le Neindre, H.; Millavet, P.; Simon, M.; Cuny, C.; Barthet, J.-S.; Barthet, P.; Laurencin-Dalicieux, S. Non-surgical periodontal treatment: SRP and innovative therapeutic approaches. In Periodontitis: Advances in Experimental Research; Springer, 2022; pp. 303–327. [Google Scholar]
- Farina, R.; Simonelli, A.; Trombelli, L.; Chew, R.J.J.; Tu, Y.K.; Preshaw, P.M. Clinical Efficacy of Interventions Based on Professional Mechanical Plaque Removal in the Treatment of Dental Biofilm-Induced Gingivitis: A Systematic Review and Meta-Analysis. J Clin Periodontol 2026, 53, 572–595. [Google Scholar] [CrossRef]
- Ahuja, V.; Macho, M.; Ewe, D.; Singh, M.; Saha, S.; Saurav, K. Biological and pharmacological potential of xylitol: a molecular insight of unique metabolism. Foods 2020, 9, 1592. [Google Scholar] [CrossRef]
- Boschetto, F.; Zanocco, M.; Kamei, K.; Xu, H.; Marin, E. Xylitol Modification of Electrospun Polymer Scaffolds: Impact on Physicochemical and Antibacterial Properties. Polymers 2025, 17, 3024. [Google Scholar] [CrossRef]
- Kuraji, R.; Shiba, T.; Dong, T.S.; Numabe, Y.; Kapila, Y.L. Periodontal treatment and microbiome-targeted therapy in management of periodontitis-related nonalcoholic fatty liver disease with oral and gut dysbiosis. World J Gastroenterol 2023, 29, 967–996. [Google Scholar] [CrossRef] [PubMed]
- Asakawa, M.; Furuta, M.; Asada, S.; Ando, T.; Yanagisawa, T.; Yoshikawa, E.; Kageyama, S.; Takeshita, T. Effects of 4-week xylitol tablet intake on tongue microbiota composition in children: a single-arm pilot study. Microbiology spectrum 2026, 14, e01360-01325. [Google Scholar] [CrossRef]
- Cobb, C.M.; Sottosanti, J.S. A re-evaluation of scaling and root planing. Journal of Periodontology 2021, 92, 1370–1378. [Google Scholar] [CrossRef]
- Mortazavi, H.; Yousefi-Koma, A.-A.; Yousefi-Koma, H. Extensive comparison of salivary collection, transportation, preparation, and storage methods: a systematic review. BMC Oral Health 2024, 24, 168. [Google Scholar] [CrossRef] [PubMed]
- Claassen-Weitz, S.; Gardner-Lubbe, S.; Mwaikono, K.S.; du Toit, E.; Zar, H.J.; Nicol, M.P. Optimizing 16S rRNA gene profile analysis from low biomass nasopharyngeal and induced sputum specimens. BMC Microbiol 2020, 20, 113. [Google Scholar] [CrossRef]
- Maki, K.A.; Wolff, B.; Varuzza, L.; Green, S.J.; Barb, J.J. Multi-amplicon microbiome data analysis pipelines for mixed orientation sequences using QIIME2: Assessing reference database, variable region and pre-processing bias in classification of mock bacterial community samples. Plos one 2023, 18, e0280293. [Google Scholar] [CrossRef] [PubMed]
- Loimaranta, V.; Mazurel, D.; Deng, D.; Söderling, E. Xylitol and erythritol inhibit real-time biofilm formation of Streptococcus mutans. BMC microbiology 2020, 20, 184. [Google Scholar] [CrossRef]
- He, J.; Liu, Y.; Liu, H.; Liu, H.; Xu, M.; Wei, X.; Chen, M. Combined quantitative and relative microbiome profiling reveals key salivary bacteriome shifts associated with periodontal health and different stages of periodontitis. Journal of Dentistry 2025, 106125. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, P.; Yang, Q.; Li, C.; Li, L.; Han, M. Efficacy of scaling and root planing with and without adjunct Nd: YAG laser therapy on glucose control and periodontal microecological imbalance in periodontitis patients with type 2 diabetes mellitus: a randomized controlled trial. Clinical Oral Investigations 2024, 28, 143. [Google Scholar] [CrossRef]
- Nath, S.; Pulikkotil, S.J.; Weyrich, L.; Zilm, P.; Kapellas, K.; Jamieson, L. Effect of Periodontal Interventions on Characteristics of the Periodontal Microbial Profile: A Systematic Review and Meta-Analysis. Microorganisms 2022, 10. [Google Scholar] [CrossRef]
- Relvas, M.; Regueira-Iglesias, A.; Balsa-Castro, C.; Salazar, F.; Pacheco, J.; Cabral, C.; Henriques, C.; Tomás, I. Relationship between dental and periodontal health status and the salivary microbiome: bacterial diversity, co-occurrence networks and predictive models. Scientific reports 2021, 11, 929. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Chen, J.; Zhou, X.; Li, Y. Inhibition of Streptococcus mutans biofilm formation by strategies targeting the metabolism of exopolysaccharides. Critical reviews in microbiology 2021, 47, 667–677. [Google Scholar] [CrossRef]
- de Siqueira Fraga, E.G.; Teófilo Campos, F.M.; da Silva Cavalcante, M.P.; Barbosa Martins, L.F.; Rodrigues Neto, E.M.; Mimica, M.J. Xylitol, a Promising Allied for Oral Heath. Journal of Young Pharmacists 2020, 12. [Google Scholar] [CrossRef]
- Gheisary, Z.; Mahmood, R.; Harri Shivanantham, A.; Liu, J.; Lieffers, J.R.L.; Papagerakis, P.; Papagerakis, S. The Clinical, Microbiological, and Immunological Effects of Probiotic Supplementation on Prevention and Treatment of Periodontal Diseases: A Systematic Review and Meta-Analysis. Nutrients 2022, 14. [Google Scholar] [CrossRef] [PubMed]
| Variable | SRP Group (n = 9) | SRP + Xylitol Group (n = 9) | p-value |
|---|---|---|---|
| Age (months), mean | 120.11 | 120.11 | 1.00 |
| Height (cm), mean | 119.65 | 122.07 | 0.89 |
| Sex (Male), n | 4 | 7 | — |
| Sex (Female), n | 5 | 2 | — |
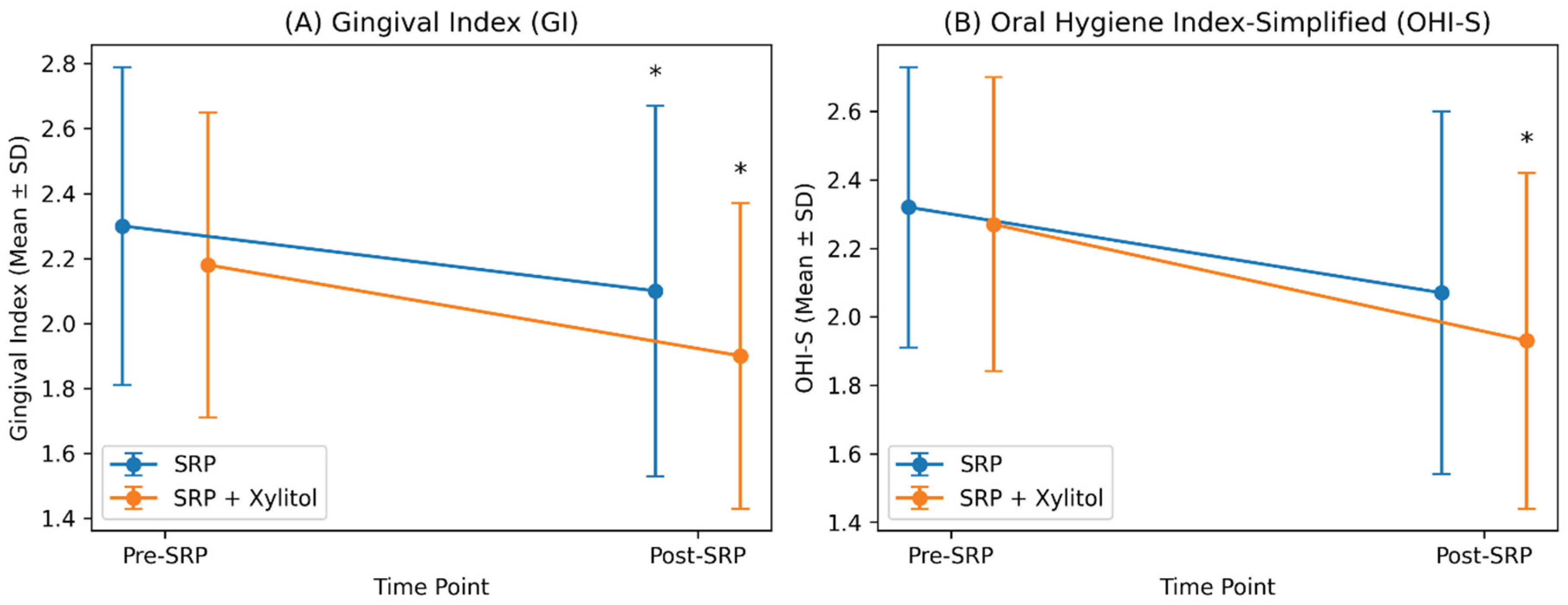
| Parameter | Group | Pre-SRP (Mean ± SD) | Post-SRP (Mean ± SD) | p-value (within group) | p-value (between groups) |
|---|---|---|---|---|---|
| Gingival Index (GI) | SRP | 2.30 ± 0.49 | 2.10 ± 0.57 | 0.005* | 0.22 |
| SRP + Xylitol | 2.18 ± 0.47 | 1.90 ± 0.47 | 0.001* | ||
| OHI-S | SRP | 2.32 ± 0.41 | 2.07 ± 0.53 | 0.065 | 0.20 |
| SRP + Xylitol | 2.27 ± 0.43 | 1.93 ± 0.49 | 0.004* |
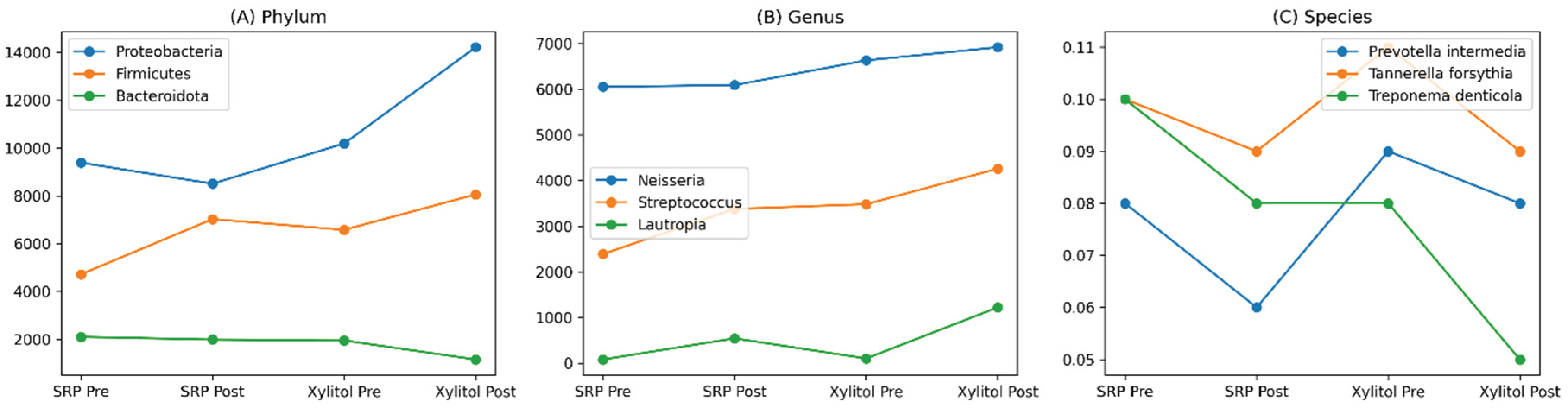
| Taxonomic level | Taxa | SRP (Pre) | SRP (Post) | p-value | SRP + Xylitol (Pre) | SRP + Xylitol (Post) | p-value |
|---|---|---|---|---|---|---|---|
| Phylum | Proteobacteria | 9388 | 8509 | >0.05 | 10190 | 14218 | <0.05* |
| Firmicutes | 4721 | 7028 | <0.05* | 6580 | 8062 | <0.05* | |
| Bacteroidota | 2103 | 1994 | >0.05 | 1955 | 1159 | <0.05* | |
| Spirochaetota | 149 | 119 | >0.05 | 419 | 165 | >0.05 | |
| Actinobacteriota | 16 | 20 | >0.05 | 15 | 32 | >0.05 | |
| Genus | Neisseria | 6054 | 6091 | >0.05 | 6633 | 6922 | >0.05 |
| Streptococcus | 2390 | 3380 | >0.05 | 3482 | 4261 | >0.05 | |
| Veillonella | 2138 | 1954 | >0.05 | 1866 | 1955 | >0.05 | |
| Haemophilus | 1135 | 1258 | >0.05 | 1290 | 1111 | >0.05 | |
| Lautropia | 80 | 544 | <0.05* | 102 | 1222 | <0.05* | |
| Species | Prevotella intermedia | 0.08 | 0.06 | 0.06 | 0.09 | 0.08 | 0.01* |
| Tannerella forsythia | 0.10 | 0.09 | 0.04* | 0.11 | 0.09 | 0.00* | |
| Treponema denticola | 0.10 | 0.08 | 0.04* | 0.08 | 0.05 | 0.00* |
| Taxonomic Level | Taxa 1 | Taxa 2 | r (Spearman) | 95% CI | p-value | FDR-adjusted p-value |
|---|---|---|---|---|---|---|
| Phylum | Firmicutes | Actinobacteriota | +0.87 | 0.53 to 0.97 | 0.002* | 0.008* |
| Firmicutes | Spirochaetota | −0.77 | −0.94 to −0.28 | 0.016* | 0.021* | |
| Spirochaetota | Actinobacteriota | −0.76 | −0.93 to −0.26 | 0.017* | 0.021* | |
| Genus | Prevotella | Veillonella | +0.67 | 0.05 to 0.92 | 0.050 | 0.050 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




