Submitted:
09 April 2026
Posted:
10 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction—Chemosensory Plasticity Beyond Synapses
2. Chemosensory Circuit Architecture as a Plastic Substrate
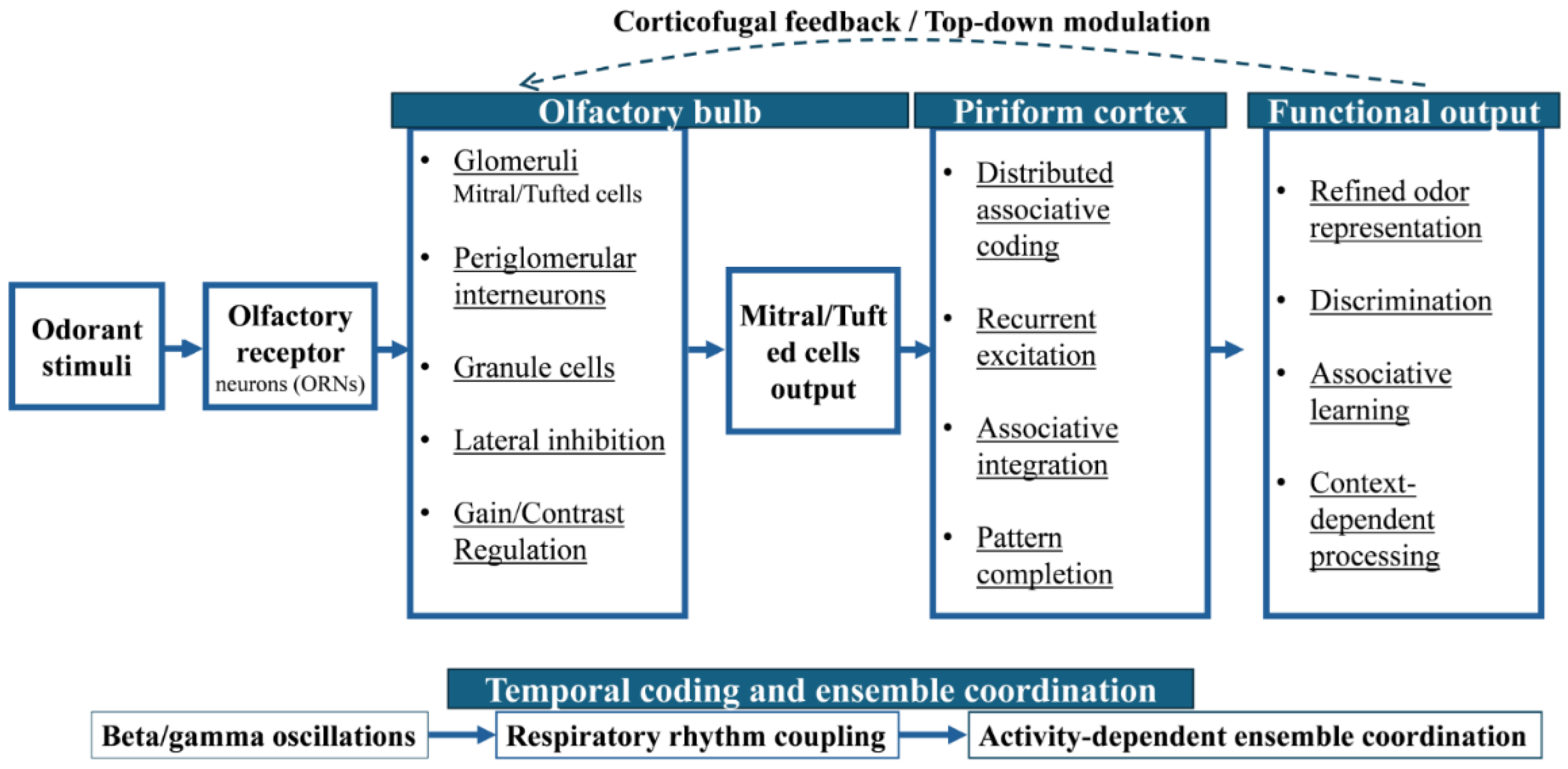
2.1. Organization of Chemosensory Circuits
2.2. Recurrent Connectivity and Feedback Pathways
2.3. Activity-Dependent Circuit Reconfiguration
2.4. Chemosensory Circuits as Dynamic Processing Systems
3. Experience-Dependent Plasticity in Chemosensory Systems
3.1. Behavioral and Circuit-Level Adaptation to Sensory Experience
3.2. Molecular and Cellular Mechanisms Underlying Experience-Dependent Plasticity
4. Cortical Feedback as a Driver of Circuit Reconfiguration
4.1. Anatomical and Functional Organization of Cortical Feedback Pathways
4.2. Mechanisms of Cortical Modulation of Sensory Processing
4.3. Cortical Feedback in Learning and Predictive Processing
5. Glial Regulation of Chemosensory Circuit States
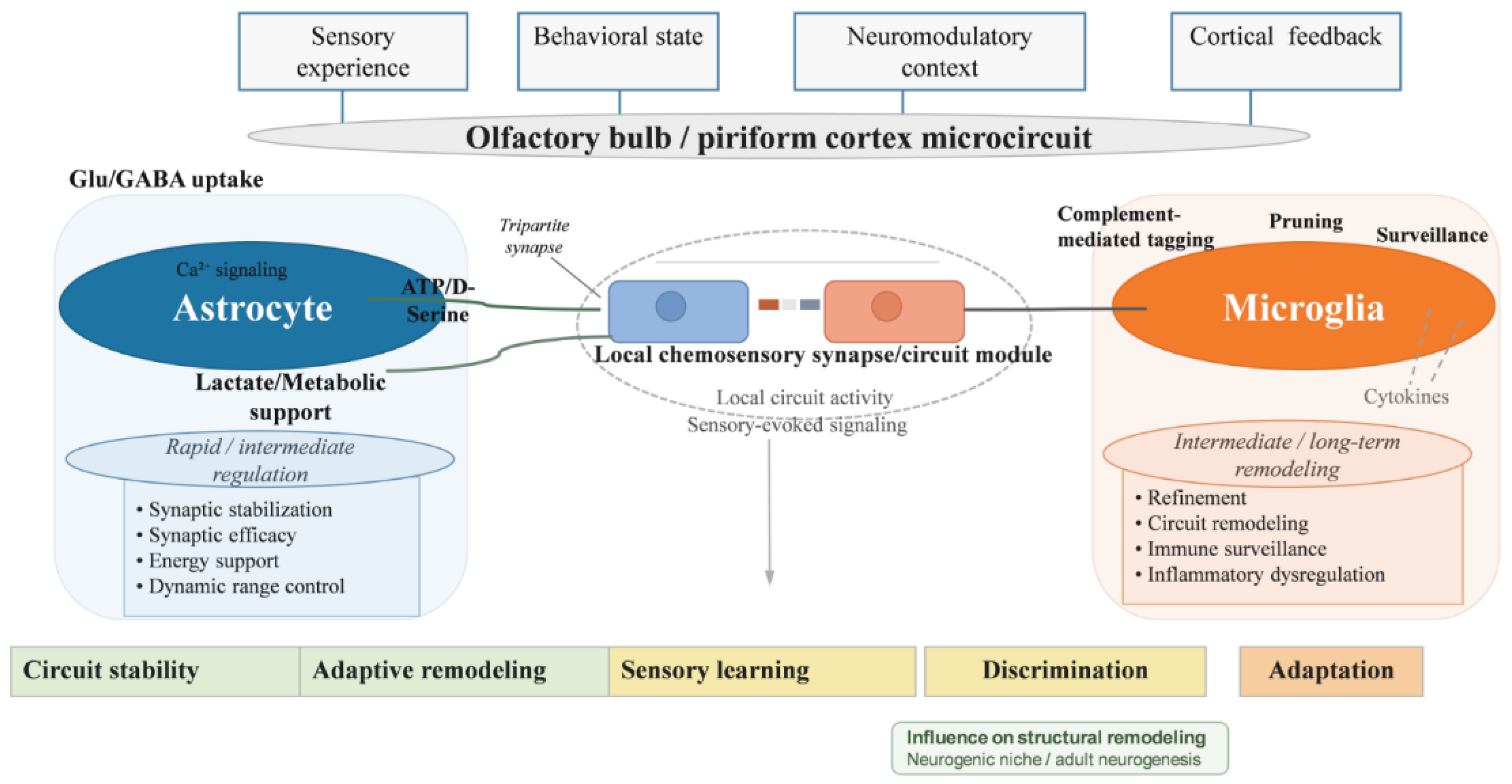
5.1. Astrocytes in Chemosensory Circuit Modulation
5.2. Microglial Roles in Synaptic Remodeling and Immune Signaling
5.3. Glial Contributions to Circuit Stability and Plasticity Balance
5.4. Glial Regulation in the Context of Chemosensory Plasticity
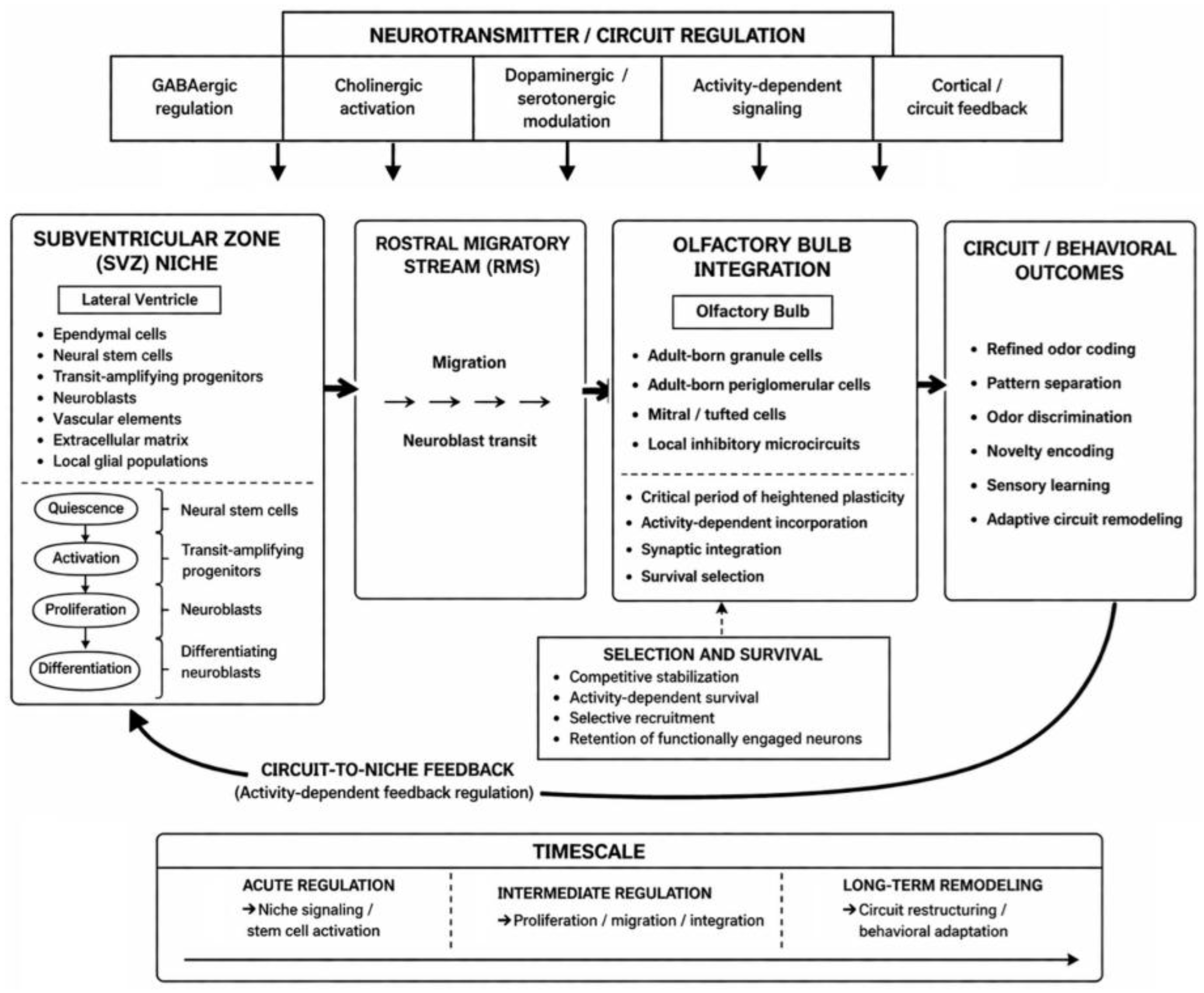
6. Neural Stem Cells as Structural Plasticity Engines
7. Circuit Control of Neural Stem Cell Dynamics
7.1. Neurotransmitter and Activity-Dependent Regulation of Neural Stem Cells
7.2. Circuit Feedback to the Stem Cell Niche and Bidirectional Coupling
8. Integration of Newborn Neurons in Sensory Learning
8.1. Functional Incorporation of Adult-Born Neurons into Olfactory Circuits
8.2. Contributions to Odor Discrimination and Pattern Separation
8.3. Impact on Network Dynamics and Sensory Performance
9. Cortical Glial and Neural Stem Cell Coupling in Chemosensory Plasticity
9.1. Rationale for a Multiscale Coupling Framework
9.2. Cortical Regulation of Circuit Dynamics
9.3. Glial Regulation of the Synaptic and Metabolic Environment
9.4. Neural Stem Cells as Structural Effectors of Plasticity
9.5. Integrated Multiscale Coupling in Chemosensory Plasticity
10. Implications for Chemosensory Dysfunction and Disease
Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Artificial Intelligence Generated Content Declaration
References
- Berners-Lee, A., et al., Experience-dependent evolution of odor mixture representations in piriform cortex. PLoS Biol, 2023. 21(4): p. e3002086.
- Kogan, J.F. and A. Fontanini, Learning enhances representations of taste-guided decisions in the mouse gustatory insular cortex. Curr Biol, 2024. 34(9): p. 1880-1892 e5.
- Dossat, A.M., et al., Glucagon-Like Peptide-1 Receptors in the Gustatory Cortex Influence Food Intake. J Neurosci, 2023. 43(23): p. 4251-4261.
- Subramanian, N., et al., Recent odor experience selectively modulates olfactory sensitivity across the glomerular output in the mouse olfactory bulb. Chem Senses, 2025. 50.
- Bao, S., et al., Signaling mechanisms underlying activity-dependent integration of adult-born neurons in the mouse olfactory bulb. Genesis, 2024. 62(2): p. e23595.
- Zak, J.D., et al., Distinct information conveyed to the olfactory bulb by feedforward input from the nose and feedback from the cortex. Nat Commun, 2024. 15(1): p. 3268.
- Zhao, D., M. Hu, and S. Liu, Glial cells in the mammalian olfactory bulb. Front Cell Neurosci, 2024. 18: p. 1426094.
- Hernandez, D.E., et al., Fast updating feedback from piriform cortex to the olfactory bulb relays multimodal identity and reward contingency signals during rule-reversal. Nat Commun, 2025. 16(1): p. 937.
- Gomez-Sotres, P., et al., Olfactory bulb astrocytes link social transmission of stress to cognitive adaptation in male mice. Nat Commun, 2024. 15(1): p. 7103.
- Dejou, J., N. Mandairon, and A. Didier, Olfactory neurogenesis plays different parts at successive stages of life, implications for mental health. Front Neural Circuits, 2024. 18: p. 1467203.
- Wang, P., S. Li, and A. Li, Odor representation and coding by the mitral/tufted cells in the olfactory bulb. J Zhejiang Univ Sci B, 2024. 25(10): p. 824-840.
- Federman, N., et al., Acquisition of non-olfactory encoding improves odour discrimination in olfactory cortex. Nat Commun, 2024. 15(1): p. 5572.
- Chen, K., J.F. Kogan, and A. Fontanini, Spatially Distributed Representation of Taste Quality in the Gustatory Insular Cortex of Behaving Mice. Curr Biol, 2021. 31(2): p. 450.
- Puche, A.C., C. Hook, and F.W. Zhou, Cell type-specific and frequency-dependent centrifugal modulation in olfactory bulb output neurons in vivo. J Neurophysiol, 2024. 131(6): p. 1226-1239.
- Bolding, K.A., et al., Recurrent circuitry is required to stabilize piriform cortex odor representations across brain states. Elife, 2020. 9.
- Terral, G., et al., Endogenous cannabinoids in the piriform cortex tune olfactory perception. Nat Commun, 2024. 15(1): p. 1230.
- Terrier, C., et al., Long-term olfactory enrichment promotes non-olfactory cognition, noradrenergic plasticity and remodeling of brain functional connectivity in older mice. Neurobiol Aging, 2024. 136: p. 133-156.
- A, A.D., et al., A mechanosensory feedback that uncouples external and self-generated sensory responses in the olfactory cortex. Cell Rep, 2024. 43(4): p. 114013.
- Geng, C., et al., Noradrenergic inputs from the locus coeruleus to anterior piriform cortex and the olfactory bulb modulate olfactory outputs. Nat Commun, 2025. 16(1): p. 260.
- Gonzalez, J., et al., Communication subspace dynamics of the canonical olfactory pathway. iScience, 2024. 27(12): p. 111275.
- Schiff, H.C., et al., Experience-dependent plasticity of gustatory insular cortex circuits and taste preferences. Sci Adv, 2023. 9(2): p. eade6561.
- Gonzalez, J., P. Torterolo, and A.B.L. Tort, Mechanisms and functions of respiration-driven gamma oscillations in the primary olfactory cortex. Elife, 2023. 12.
- Okumura, T., et al., Semantic context-dependent neural representations of odors in the human piriform cortex revealed by 7T MRI. Hum Brain Mapp, 2024. 45(6): p. e26681.
- Yu, B., et al., Cholinergic feedback for modality- and context-specific modulation of sensory representations. Science, 2025. 388(6753): p. 1324-1329.
- Boyd, A.M., et al., Cortical feedback control of olfactory bulb circuits. Neuron, 2012. 76(6): p. 1161-74.
- Burton, S.D., C.M. Malyshko, and N.N. Urban, Fast-spiking interneuron detonation drives high-fidelity inhibition in the olfactory bulb. PLoS Biol, 2024. 22(8): p. e3002660.
- Chae, H., et al., Long-range functional loops in the mouse olfactory system and their roles in computing odor identity. Neuron, 2022. 110(23): p. 3970-3985 e7.
- Chen, Z. and K. Padmanabhan, Top-down feedback enables flexible coding strategies in the olfactory cortex. Cell Rep, 2022. 38(12): p. 110545.
- Otazu, G.H., et al., Cortical Feedback Decorrelates Olfactory Bulb Output in Awake Mice. Neuron, 2015. 86(6): p. 1461-77.
- Lindeman, S., et al., Value-related learning in the olfactory bulb occurs through pathway-dependent perisomatic inhibition of mitral cells. PLoS Biol, 2024. 22(3): p. e3002536.
- Gnaedinger, A., et al., Multisensory learning between odor and sound enhances beta oscillations. Sci Rep, 2019. 9(1): p. 11236.
- Norden, F., et al., The human olfactory bulb communicates perceived odor valence to the piriform cortex in the gamma band and receives a refined representation back in the beta band. PLoS Biol, 2024. 22(10): p. e3002849.
- Garg, R., Q. Qiu, and C.R. Yu, Basal forebrain cholinergic input mediates adaptive attention allocation to enhance olfactory discrimination. PLoS Biol, 2025. 23(9): p. e3003374.
- Jacobson, G.A., P. Rupprecht, and R.W. Friedrich, Experience-Dependent Plasticity of Odor Representations in the Telencephalon of Zebrafish. Curr Biol, 2018. 28(1): p. 1-14 e3.
- Lyons, S.H. and J.A. Gottfried, Predictive coding in the human olfactory system. Trends Cogn Sci, 2025. 29(9): p. 814-826.
- Rajabi, N., et al., Exploring the feasibility of olfactory brain-computer interfaces. Sci Rep, 2025. 15(1): p. 18404.
- Zelano, C., A. Mohanty, and J.A. Gottfried, Olfactory predictive codes and stimulus templates in piriform cortex. Neuron, 2011. 72(1): p. 178-87.
- Ferreira, A., et al., Distinct forms of structural plasticity of adult-born interneuron spines in the mouse olfactory bulb induced by different odor learning paradigms. Commun Biol, 2024. 7(1): p. 420.
- Mazo, C., et al., Long-range GABAergic projections contribute to cortical feedback control of sensory processing. Nat Commun, 2022. 13(1): p. 6879.
- Maier, J.X., A. Idris, and B.A. Christensen, Taste-Odor Association Learning Alters the Dynamics of Intraoral Odor Responses in the Posterior Piriform Cortex of Awake Rats. eNeuro, 2023. 10(3).
- Ung, K., et al., Olfactory bulb astrocytes mediate sensory circuit processing through Sox9 in the mouse brain. Nat Commun, 2021. 12(1): p. 5230.
- Xu, R., et al., Olfactory bulb microglia activation mediates neuronal pyroptosis in ozone-exposed mice with olfactory and cognitive dysfunction. J Hazard Mater, 2025. 485: p. 136901.
- Leier, H.C., et al., Glia control experience-dependent plasticity in an olfactory critical period. Elife, 2025. 13.
- Li, W.L., et al., Adult-born neurons facilitate olfactory bulb pattern separation during task engagement. Elife, 2018. 7.
- Akinlaja, Y.O. and A. Nishiyama, Glial modulation of synapse development and plasticity: oligodendrocyte precursor cells as a new player in the synaptic quintet. Front Cell Dev Biol, 2024. 12: p. 1418100.
- Noriega-Prieto, J.A. and A. Araque, Sensing and Regulating Synaptic Activity by Astrocytes at Tripartite Synapse. Neurochem Res, 2021. 46(10): p. 2580-2585.
- Sanz-Galvez, R., et al., The role of astrocytes from synaptic to non-synaptic plasticity. Front Cell Neurosci, 2024. 18: p. 1477985.
- Andersen, J.V., The Glutamate/GABA-Glutamine Cycle: Insights, Updates, and Advances. J Neurochem, 2025. 169(3): p. e70029.
- Andersen, J.V., A. Schousboe, and P. Wellendorph, Astrocytes regulate inhibitory neurotransmission through GABA uptake, metabolism, and recycling. Essays Biochem, 2023. 67(1): p. 77-91.
- Limon, I.D., et al., Disturbance of the Glutamate-Glutamine Cycle, Secondary to Hepatic Damage, Compromises Memory Function. Front Neurosci, 2021. 15: p. 578922.
- Lei, L., et al., Novel insight into astrocyte-mediated gliotransmission modulates the synaptic plasticity in major depressive disorder. Life Sci, 2024. 355: p. 122988.
- Won, W., et al., Astrocytes as Key Regulators of Neural Signaling in Health and Disease. Annu Rev Neurosci, 2025. 48(1): p. 251-276.
- Kim, Y., S.E. Dube, and C.B. Park, Brain energy homeostasis: the evolution of the astrocyte-neuron lactate shuttle hypothesis. Korean J Physiol Pharmacol, 2025. 29(1): p. 1-8.
- Marty-Lombardi, S., et al., Neuron-astrocyte metabolic coupling facilitates spinal plasticity and maintenance of inflammatory pain. Nat Metab, 2024. 6(3): p. 494-513.
- Pang, Y., et al., Metabolic interactions in the brain: the crucial roles of neurons, astrocytes, and microglia in health and disease. Front Neurosci, 2026. 20: p. 1731771.
- Chamaa, F., P.J. Magistretti, and H. Fiumelli, Astrocyte-derived lactate in stress disorders. Neurobiol Dis, 2024. 192: p. 106417.
- Miguel-Quesada, C., et al., Astrocytes adjust the dynamic range of cortical network activity to control modality-specific sensory information processing. Cell Rep, 2023. 42(8): p. 112950.
- Ogino, T., et al., Astrocytic activation increases blood flow in the adult olfactory bulb. Mol Brain, 2024. 17(1): p. 52.
- Rosa, J.M. and J. Aguilar, Rethinking Sensory Information Processing: The Essential Role of Astrocytes. J Neurochem, 2025. 169(6): p. e70113.
- Duran Laforet, V. and D.P. Schafer, Microglia: Activity-dependent regulators of neural circuits. Ann N Y Acad Sci, 2024. 1533(1): p. 38-50.
- Meller, S.J. and C.A. Greer, Olfactory Development and Dysfunction: Involvement of Microglia. Physiology (Bethesda), 2025. 40(2): p. 0.
- Zhao, S., A.D. Umpierre, and L.J. Wu, Tuning neural circuits and behaviors by microglia in the adult brain. Trends Neurosci, 2024. 47(3): p. 181-194.
- Nandi, S., et al., Synaptic Pruning by Microglia: Lessons from Genetic Studies in Mice. Dev Neurosci, 2025. 47(5): p. 362-382.
- Rogerson-Wood, L., A. Sawatari, and C.A. Leamey, Microglia: Mediators of experience-driven corrective neuroplasticity. IBRO Neurosci Rep, 2025. 19: p. 91-100.
- Grier, B.D., L. Belluscio, and C.E. Cheetham, Olfactory Sensory Activity Modulates Microglial-Neuronal Interactions during Dopaminergic Cell Loss in the Olfactory Bulb. Front Cell Neurosci, 2016. 10: p. 178.
- Tillmon, H., et al., Complement and microglia activation mediate stress-induced synapse loss in layer 2/3 of the medial prefrontal cortex in male mice. Nat Commun, 2024. 15(1): p. 9803.
- Yang, G., et al., Microglia-orchestrated neuroinflammation and synaptic remodeling: roles of pro-inflammatory cytokines and receptors in neurodegeneration. Front Cell Neurosci, 2025. 19: p. 1700692.
- Pena-Ortega, F., Microglial modulation of neuronal network function and plasticity. J Neurophysiol, 2025. 133(2): p. 661-680.
- Khakh, B.S., On astrocyte-neuron interactions: Broad insights from the striatum. Neuron, 2025. 113(19): p. 3079-3107.
- Faust, T.E., et al., Microglia-astrocyte crosstalk regulates synapse remodeling via Wnt signaling. Cell, 2025. 188(19): p. 5212-5230 e21.
- Lines, J., et al., The Duality of Astrocyte Neuromodulation: Astrocytes Sense Neuromodulators and Are Neuromodulators. J Neurochem, 2025. 169(4): p. e70054.
- Rasmussen, R.N., et al., Astrocytes: integrators of arousal state and sensory context. Trends Neurosci, 2023. 46(6): p. 418-425.
- Taylor, C.R., et al., Cortical astrocyte histamine-1-receptors regulate intracellular calcium and extracellular adenosine dynamics across sleep and wake. PLoS Biol, 2025. 23(10): p. e3003376.
- Naffaa, M.M., Glial-Dopamine crosstalk: Astrocytic and microglial gatekeepers of neuroinflammation, plasticity, and motivation. AIMS Neuroscience, 2026. 13: p. 64-118.
- Naffaa, M.M., Mechanisms of astrocytic and microglial purinergic signaling in homeostatic regulation and implications for neurological disease. Exploration of Neuroscience, 2025. 4.
- Imrie, G. and I. Farhy-Tselnicker, Astrocyte regulation of behavioral outputs: the versatile roles of calcium. Front Cell Neurosci, 2025. 19: p. 1606265.
- Naffaa, M.M., Neurogenesis dynamics in the olfactory bulb: deciphering circuitry organization, function, and adaptive plasticity. Neural Regen Res, 2025. 20(6): p. 1565-1581.
- Gregory, J.D., et al., New Neurons in the Postnatal Olfactory System: Functions in the Healthy and Regenerating Brain. Brain Sci, 2025. 15(6).
- Li, K., et al., The subventricular zone structure, function and implications for neurological disease. Genes Dis, 2025. 12(3): p. 101398.
- Purvis, E.M., et al., A three-dimensional tissue-engineered rostral migratory stream as an in vitro platform for subventricular zone-derived cell migration. Front Bioeng Biotechnol, 2024. 12: p. 1410717.
- Shani-Narkiss, H., et al., Young adult-born neurons improve odor coding by mitral cells. Nat Commun, 2020. 11(1): p. 5867.
- Tufo, C., et al., Functional Maturation and Experience-Dependent Plasticity in Adult-Born Olfactory Bulb Dopaminergic Neurons. Eur J Neurosci, 2025. 62(1): p. e70188.
- Chen, Y., et al., Olfactory bulb neurogenesis depending on signaling in the subventricular zone. Cereb Cortex, 2023. 33(22): p. 11102-11111.
- Nath, S., et al., Interaction between subventricular zone microglia and neural stem cells impacts the neurogenic response in a mouse model of cortical ischemic stroke. Nat Commun, 2024. 15(1): p. 9095.
- Alonso, M., et al., Olfactory discrimination learning increases the survival of adult-born neurons in the olfactory bulb. J Neurosci, 2006. 26(41): p. 10508-13.
- Nakamura, Y., et al., Histological changes in the olfactory bulb and rostral migratory stream due to interruption of olfactory input. Auris Nasus Larynx, 2024. 51(3): p. 517-524.
- Kelsch, W., et al., A critical period for activity-dependent synaptic development during olfactory bulb adult neurogenesis. J Neurosci, 2009. 29(38): p. 11852-8.
- Hanson, E., J. Swanson, and B.R. Arenkiel, Sensory experience shapes the integration of adult-born neurons into the olfactory bulb. J Nat Sci, 2017. 3(8).
- Forest, J., et al., Short-term availability of adult-born neurons for memory encoding. Nat Commun, 2019. 10(1): p. 5609.
- Cummings, D.M., et al., Adult neurogenesis is necessary to refine and maintain circuit specificity. J Neurosci, 2014. 34(41): p. 13801-10.
- Naffaa, M.M., Neural circuit regulation of postnatal and adult subventricular zone neurogenesis: Mechanistic insights, functional models, and circuit-based neurological disorders. Archives of Stem Cell and Therapy, 2024. 5(1).
- Hanson, E., J. Swanson, and B.R. Arenkiel, GABAergic Input From the Basal Forebrain Promotes the Survival of Adult-Born Neurons in the Mouse Olfactory Bulb. Front Neural Circuits, 2020. 14: p. 17.
- Chaker, Z., E. Makarouni, and F. Doetsch, The Organism as the Niche: Physiological States Crack the Code of Adult Neural Stem Cell Heterogeneity. Annu Rev Cell Dev Biol, 2024. 40(1): p. 381-406.
- Miyamoto, T., et al., High spatial resolution gene expression profiling and characterization of neuroblasts migrating in the peri-injured cortex using photo-isolation chemistry. Front Neurosci, 2024. 18: p. 1504047.
- Baig, S., et al., Identity and nature of neural stem cells in the adult human subventricular zone. iScience, 2024. 27(4): p. 109342.
- Quaresima, S., et al., Assessing the Role of Ependymal and Vascular Cells as Sources of Extracellular Cues Regulating the Mouse Ventricular-Subventricular Zone Neurogenic Niche. Front Cell Dev Biol, 2022. 10: p. 845567.
- Alfonso, J., et al., Diazepam binding inhibitor promotes progenitor proliferation in the postnatal SVZ by reducing GABA signaling. Cell Stem Cell, 2012. 10(1): p. 76-87.
- Liu, X., et al., Nonsynaptic GABA signaling in postnatal subventricular zone controls proliferation of GFAP-expressing progenitors. Nat Neurosci, 2005. 8(9): p. 1179-87.
- Young, S.Z., et al., GABA(A) Increases Calcium in Subventricular Zone Astrocyte-Like Cells Through L- and T-Type Voltage-Gated Calcium Channels. Front Cell Neurosci, 2010. 4: p. 8.
- Naffaa, M.M., et al., Cortical regulation of neurogenesis and cell proliferation in the ventral subventricular zone. Cell Rep, 2023. 42(7): p. 112783.
- Naffaa, M.M. and H.H. Yin, A cholinergic signaling pathway underlying cortical circuit activation of quiescent neural stem cells in the lateral ventricle. Sci Signal, 2024. 17(855): p. eadk8810.
- Hitoshi, S., et al., Antidepressant drugs reverse the loss of adult neural stem cells following chronic stress. J Neurosci Res, 2007. 85(16): p. 3574-85.
- Lao, C.L., C.S. Lu, and J.C. Chen, Dopamine D3 receptor activation promotes neural stem/progenitor cell proliferation through AKT and ERK1/2 pathways and expands type-B and -C cells in adult subventricular zone. Glia, 2013. 61(4): p. 475-89.
- O’Keeffe, G.C. and R.A. Barker, Dopamine stimulates epidermal growth factor release from adult neural precursor cells derived from the subventricular zone by a disintegrin and metalloprotease. Neuroreport, 2011. 22(18): p. 956-8.
- Delgado, A.C., et al., Release of stem cells from quiescence reveals gliogenic domains in the adult mouse brain. Science, 2021. 372(6547): p. 1205-1209.
- Naffaa, M.M., Significance of the anterior cingulate cortex in neurogenesis plasticity: Connections, functions, and disorders across postnatal and adult stages. Bioessays, 2024. 46(3).
- Corotto, F.S., J.R. Henegar, and J.A. Maruniak, Odor deprivation leads to reduced neurogenesis and reduced neuronal survival in the olfactory bulb of the adult mouse. Neuroscience, 1994. 61(4): p. 739-44.
- Grelat, A., et al., Adult-born neurons boost odor-reward association. Proc Natl Acad Sci U S A, 2018. 115(10): p. 2514-2519.
- Moreno, M.M., et al., Olfactory perceptual learning requires adult neurogenesis. Proc Natl Acad Sci U S A, 2009. 106(42): p. 17980-5.
- Breton-Provencher, V., et al., Interneurons produced in adulthood are required for the normal functioning of the olfactory bulb network and for the execution of selected olfactory behaviors. J Neurosci, 2009. 29(48): p. 15245-57.
- Bugeon, S., et al., Intrinsic Neuronal Activity during Migration Controls the Recruitment of Specific Interneuron Subtypes in the Postnatal Mouse Olfactory Bulb. J Neurosci, 2021. 41(12): p. 2630-2644.
- Wu, A., et al., Context-dependent plasticity of adult-born neurons regulated by cortical feedback. Sci Adv, 2020. 6(42).
- Li, K., et al., Endogenous but not sensory-driven activity controls migration, morphogenesis and survival of adult-born juxtaglomerular neurons in the mouse olfactory bulb. Cell Mol Life Sci, 2023. 80(4): p. 98.
- Dasgupta, D., et al., Coupling of Mouse Olfactory Bulb Projection Neurons to Fluctuating Odor Pulses. J Neurosci, 2022. 42(21): p. 4278-4296.
- Pardillo-Diaz, R., et al., The subventricular zone neurogenic niche provides adult born functional neurons to repair cortical brain injuries in response to diterpenoid therapy. Stem Cell Res Ther, 2025. 16(1): p. 1.
- Ware, K., et al., Inhibition of TGF-beta signaling in microglia stimulates hippocampal adult neurogenesis and reduces anxiety-like behavior in adult mice. Nat Commun, 2026. 17(1): p. 1440.
- Zhou, H., et al., Cathepsin B overexpression and lysosomal leakage in inflammatory microglia promote neuroinflammation in olfactory dysfunction by triggering mitochondrial dysfunction and pyroptosis. Brain Behav Immun, 2026. 132: p. 106188.
- Zhang, Y.J., J.Y. Lee, and K.M. Igarashi, Circuit dynamics of the olfactory pathway during olfactory learning. Front Neural Circuits, 2024. 18: p. 1437575.
- Martin-Lopez, E., et al., Inflammatory Response and Defects on Myelin Integrity in the Olfactory System of K18hACE2 Mice Infected with SARS-CoV-2. eNeuro, 2024. 11(6).
- Mou, Y., et al., P2X7 receptor of olfactory bulb microglia plays a pathogenic role in stress-related depression in mice with allergic rhinitis. Neurobiol Dis, 2024. 192: p. 106432.
- Chang, K., et al., Mechanisms of COVID-19-associated olfactory dysfunction. Neuropathol Appl Neurobiol, 2024. 50(2): p. e12960.
- Simonini, L., et al., A Comprehensive Review of COVID-19-Related Olfactory Deficiency: Unraveling Associations with Neurocognitive Disorders and Magnetic Resonance Imaging Findings. Diagnostics (Basel), 2024. 14(4).
- Tsukahara, T., D.H. Brann, and S.R. Datta, Mechanisms of SARS-CoV-2-associated anosmia. Physiol Rev, 2023. 103(4): p. 2759-2766.
- Wang, K., et al., Rbm24/Notch1 signaling regulates adult neurogenesis in the subventricular zone and mediates Parkinson-associated olfactory dysfunction. Theranostics, 2024. 14(11): p. 4499-4518.
- Naffaa, M.M., Disruptions in Adult Neurogenesis: Mechanisms, Pathways, and Therapeutic Strategies for Cognitive Decline and Neurodegenerative Diseases in Aging. Nature Cell and Science, 2025. 3(1).
- Elhabbari, K., et al., Olfactory deficits in aging and Alzheimer’s-spotlight on inhibitory interneurons. Front Neurosci, 2024. 18: p. 1503069.
- Greco-Vuilloud, J., et al., 12 months is a pivotal age for olfactory perceptual learning and its underlying neuronal plasticity in aging mice. Neurobiol Aging, 2022. 114: p. 73-83.
- Xie, Y., et al., Aging and chronic inflammation: impacts on olfactory dysfunction-a comprehensive review. Cell Mol Life Sci, 2025. 82(1): p. 199.
- Ballotta, D., et al., The olfactory functional network in the Alzheimer’s disease continuum: a resting state fMRI study. Front Aging Neurosci, 2025. 17: p. 1744413.
- Cieri, F., et al., Functional connectivity differences of the olfactory network in Parkinson’s Disease, mild cognitive impairment and cognitively normal individuals: A resting-state fMRI study. Neuroscience, 2024. 559: p. 8-16.
- Ielo, A., et al., Structural and functional connectomics of the olfactory system in Parkinson’s disease: a systematic review. Parkinsonism Relat Disord, 2025. 132: p. 107230.
- Kedzia, D., et al., Microglia, Astrocytes, and Oligodendrocytes in Parkinson’s Disease: Neuroinflammatory Crosstalk and Emerging Therapeutic Strategies. Biomolecules, 2026. 16(1).
- Liu, K., et al., The decreased astrocyte-microglia interaction reflects the early characteristics of Alzheimer’s disease. iScience, 2024. 27(3): p. 109281.
- Naffaa, M.M., The Fate and Dynamics of Neural Stem Cells (NSCs) and Their Neurogenic Decline in Alzheimer’s Disease. Lifespan Development and Mental Health, 2025. 1(2).
- Brosse, S., et al., Specific structural changes in Parkinson’s disease-related olfactory dysfunction compared to others forms of olfactory dysfunction. Front Neural Circuits, 2024. 18: p. 1503841.
- Peiris, S., et al., Olfactory Network Functional Connectivity as a Marker for Parkinson’s Disease Severity. Life (Basel), 2025. 15(8).
- Abraham, J.N., et al., Alpha-synuclein pathology and Parkinson’s disease-related olfactory dysfunctions: an update on preclinical models and therapeutic approaches. Mamm Genome, 2025. 36(2): p. 444-464.
- He, X., et al., Adult neurogenesis dysfunction in Parkinson’s disease: Molecular pathology and functional implications. Pathol Res Pract, 2026. 278: p. 156340.
- Li, H., et al., Potential convergence of olfactory dysfunction in Parkinson’s disease and COVID-19: The role of neuroinflammation. Ageing Res Rev, 2024. 97: p. 102288.
- Leon, M., E.T. Troscianko, and C.C. Woo, Inflammation and olfactory loss are associated with at least 139 medical conditions. Front Mol Neurosci, 2024. 17: p. 1455418.
- Kay, L.M., COVID-19 and olfactory dysfunction: a looming wave of dementia? J Neurophysiol, 2022. 128(2): p. 436-444.
- Chen, Y.N. and J.K. Kostka, Beyond anosmia: olfactory dysfunction as a common denominator in neurodegenerative and neurodevelopmental disorders. Front Neurosci, 2024. 18: p. 1502779.
- Meyer, C., et al., Early Locus Coeruleus noradrenergic axon loss drives olfactory dysfunction in Alzheimer’s disease. Nat Commun, 2025. 16(1): p. 7338.
- Fruholz, I. and M. Meyer-Luehmann, The intricate interplay between microglia and adult neurogenesis in Alzheimer’s disease. Front Cell Neurosci, 2024. 18: p. 1456253.
- Hashimoto, K., M. Gotoh, and H. Ikeshima-Kataoka, Astrocytic and microglial cell functions in neuroinflammatory diseases and their animal models. Front Cell Neurosci, 2025. 19: p. 1708775.
- Alonso, M., A.C. Petit, and P.M. Lledo, The impact of adult neurogenesis on affective functions: of mice and men. Mol Psychiatry, 2024. 29(8): p. 2527-2542.
- Kim, Y.K., et al., High-fat diet triggers transcriptomic changes in the olfactory bulb. Heliyon, 2025. 11(3): p. e42196.
- Pecoraro, S., et al., Adult human subventricular zone microglia promote a pro-neurogenic niche for neuronal progenitors in Parkinson’s disease. Brain Behav Immun, 2025. 129: p. 318-334.
- Iravani, B., et al., Metabolic state modulates neural processing of odors in the human olfactory bulb. Biol Psychol, 2024. 187: p. 108770.
- Lee, D.H. and J. Song, Impaired olfactory system in metabolic imbalance-related neuropathology. Life Sci, 2024. 355: p. 122967.
- Nettore, I.C., et al., Flavor and taste recognition impairments in people with type 1 diabetes. Nutr Diabetes, 2024. 14(1): p. 57.
- Kim, D.H., et al., Strategies to Develop Regenerative Medicine Approaches for Olfactory Disorders. Clin Exp Otorhinolaryngol, 2025. 18(3): p. 204-209.
- Yi, K.I., et al., Stem Cells and Cell-Free Therapies for Olfactory Epithelium Regeneration: Insights from Experimental Models. Int J Mol Sci, 2025. 26(18).
- De Cleene, N., et al., Olfactory dysfunction as potential biomarker in neurodegenerative diseases: a narrative review. Front Neurosci, 2024. 18: p. 1505029.
- Ekstrom, I., et al., Blood-based biomarkers of Alzheimer’s disease and olfactory decline over 15 years in older adults. Geroscience, 2025.
- Walters, B.N., et al., Longitudinal imaging of the taste bud in vivo with two-photon laser scanning microscopy. PLoS One, 2024. 19(12): p. e0309366.
| Level of plasticity | Primary mechanism | Representative components | Functional outcome |
| Synaptic plasticity | Activity-dependent modification of synaptic strength and efficacy [16,38] | Long-term potentiation, long-term depression, inhibitory–excitatory balance, calcium-dependent signaling [16,39] | Modulates neuronal responsiveness, sensory gain, and associative encoding [16,40] |
| Circuit-level plasticity | Reconfiguration of ensemble activity, recurrent connectivity, temporal coding, and top-down modulation [8,16] | Olfactory bulb microcircuits, piriform cortex networks, corticofugal feedback, beta/gamma oscillations [8,32] | Refines sensory representations, discrimination, and context-dependent processing [8,32] |
| Cellular / glial plasticity | Regulation of synaptic and metabolic environments by non-neuronal cells [41,42] | Astrocytes, microglia, tripartite synapse, gliotransmission, synaptic pruning [41,43] | Stabilizes circuits while permitting adaptive remodeling and sensory learning [41,43] |
| Structural / neurogenic plasticity | Activity-dependent generation and integration of new neurons into existing circuits [38] | Subventricular zone neural stem cells, rostral migratory stream, adult-born olfactory interneurons [38] | Supports long-term circuit remodeling, pattern separation, and sensory adaptation [38,44] |
| Integrated multiscale plasticity | Coordinated interaction of synaptic, circuit, cellular, and structural mechanisms [8,38] | Cortical feedback, glial regulation, neural stem cell dynamics, neuromodulatory signaling [8,41] | Enables adaptive chemosensory function across multiple temporal and organizational scales [8,38] |
| Component | Primary mode of action | Timescale | Effect on plasticity | Interaction with other components |
| Cortical feedback | Top-down modulation of sensory circuits through regulation of excitability, inhibition, temporal coordination, and contextual signaling [8,19] | Rapid to intermediate | Refines sensory representations, supports learning-dependent circuit reconfiguration, and aligns processing with behavioral relevance [6,16] | Shapes local circuit states that influence glial responses and activity-dependent neurogenesis [9,41,112] |
| Astrocytes | Regulation of neurotransmitter uptake, synaptic environment, gliotransmission, and metabolic support [9,41,58] | Intermediate | Stabilizes synaptic function while permitting activity-dependent modulation of circuit responsiveness [9,41,58] | Modulates how cortical and local circuit signals are expressed at synapses and within the neurogenic niche [9,41,112] |
| Microglia | Activity-dependent synaptic remodeling, immune surveillance, and inflammatory signaling [116,117] | Intermediate to long-term | Contributes to pruning, refinement, and restructuring of circuit connectivity [61,116,117] | Responds to circuit activity and influences the structural environment in which neurogenesis and synaptic plasticity occur [61,116,117] |
| Neural stem cells / adult-born neurons | Activity-dependent neurogenesis, neuronal integration, and structural remodeling of olfactory circuits [61,82,115,116,117] | Long-term | Supports sustained circuit adaptation, pattern separation, and updating of sensory representations [38,44,112] | Regulated by circuit activity and glial microenvironment; newly integrated neurons reshape the circuits that influence their generation [41,82,112] |
| Condition | Circuit-level alterations | Glial / inflammatory features | Neurogenesis / stem cell changes | Functional chemosensory outcome |
| Anosmia / hyposmia | Altered olfactory bulb activity, reduced cortical feedback, disrupted synaptic organization [6,8] | Local inflammatory responses and glial activation may impair circuit stability [7,140] | May be secondarily affected depending on severity and duration of dysfunction [10,123] | Reduced odor detection, impaired discrimination, diminished sensory adaptation [121,122] |
| Post-viral dysfunction / SARS-CoV-2 | Peripheral sensory disruption with possible secondary central alterations in olfactory pathways [122,123] | Prominent inflammatory and cellular disruption involving peripheral and central mechanisms [119,121] | Potential impairment of neurogenic and reparative processes remains under investigation [119,123] | Persistent anosmia, hyposmia, distorted odor perception, delayed sensory recovery [122,141] |
| Aging | Progressive decline in circuit plasticity, altered synaptic regulation, reduced adaptive responsiveness [126,128] | Age-related changes in glial activity and inflammatory tone [61,128] | Reduced neural stem cell activity and diminished integration of adult-born olfactory interneurons [10,93] | Decline in odor sensitivity, discrimination, and adaptation to novel sensory input [126,128,142] |
| Parkinson’s disease | Early alterations in olfactory bulb circuitry and cortical processing; impaired circuit-level plasticity [131,135,137] | Increased microglial and astrocytic activation, neuroinflammatory disruption of local microenvironment [61,132] | Reduced adult neurogenesis and impaired structural remodeling capacity have been reported [124,137] | Early olfactory dysfunction, impaired odor identification and discrimination [137,142] |
| Alzheimer’s disease | Disrupted olfactory pathway connectivity and altered network activity [129,143] | Neuroinflammatory processes involving astrocytic and microglial responses [133,144,145] | Neurogenesis may be altered, though functional significance remains incompletely resolved [144,146] | Early olfactory impairment, reduced odor recognition, progressive sensory decline [126,143] |
| Metabolic / systemic disorders | Altered neuronal excitability and impaired integration of sensory signals [55,147] | Hormonal, metabolic, and inflammatory dysregulation affecting local circuit environment [55,61,147] | Potential disruption of neurogenic niche regulation and adaptive remodeling [10,148] | Altered smell and taste perception, reduced sensory flexibility, impaired adaptive responses [142,147] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




