Submitted:
08 April 2026
Posted:
09 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. The Transformative Impact of Fifty Years of Vaccine Innovation
1.2. Evolving Infectious Disease Threats and the Expanding Value of Prevention
1.3. Innovation Pathways: Breakthrough Discoveries and Incremental Progress
1.4. Horizon Scanning and the Role of Vaccine Pipelines
2. Materials and Methods
2.1. Data Source and Scope
2.2. Data Collection and Classification
- Classification by target population. Candidates were categorised according to the clinical trial population(s) in which they were being evaluated. Population groups included paediatric, adult, and older adult populations, and candidates spanning multiple age groups were counted under each applicable population category. Maternal immunisation candidates were captured within adult populations and described separately where applicable.
- Classification by pathogen / disease area. Each candidate was categorised according to its primary pathogen target or disease indication, enabling analysis by infectious disease area and comparison across pipeline segments (e.g., transmission routes, travel-related, zoonotic threats). Candidates addressing multiple pathogens or diseases were captured under each relevant section.
- Classification by public health challenge addressed. To support horizon scanning and policy relevance, candidates were additionally labelled according to the public health challenges they address. Standardised labels were assigned to reflect thematic priorities such as AMR (i.e., priority pathogens as defined by WHO [23] or climate change-related threats, zoonoses, and pandemic preparedness (i.e., mentioned by GAVI [26]).
- Classification by technology. Candidates were classified by immunisation technology to analyse innovation trends and the diversity of scientific approaches represented in clinical pipelines.
2.3. Longitudinal Comparison
3. Results
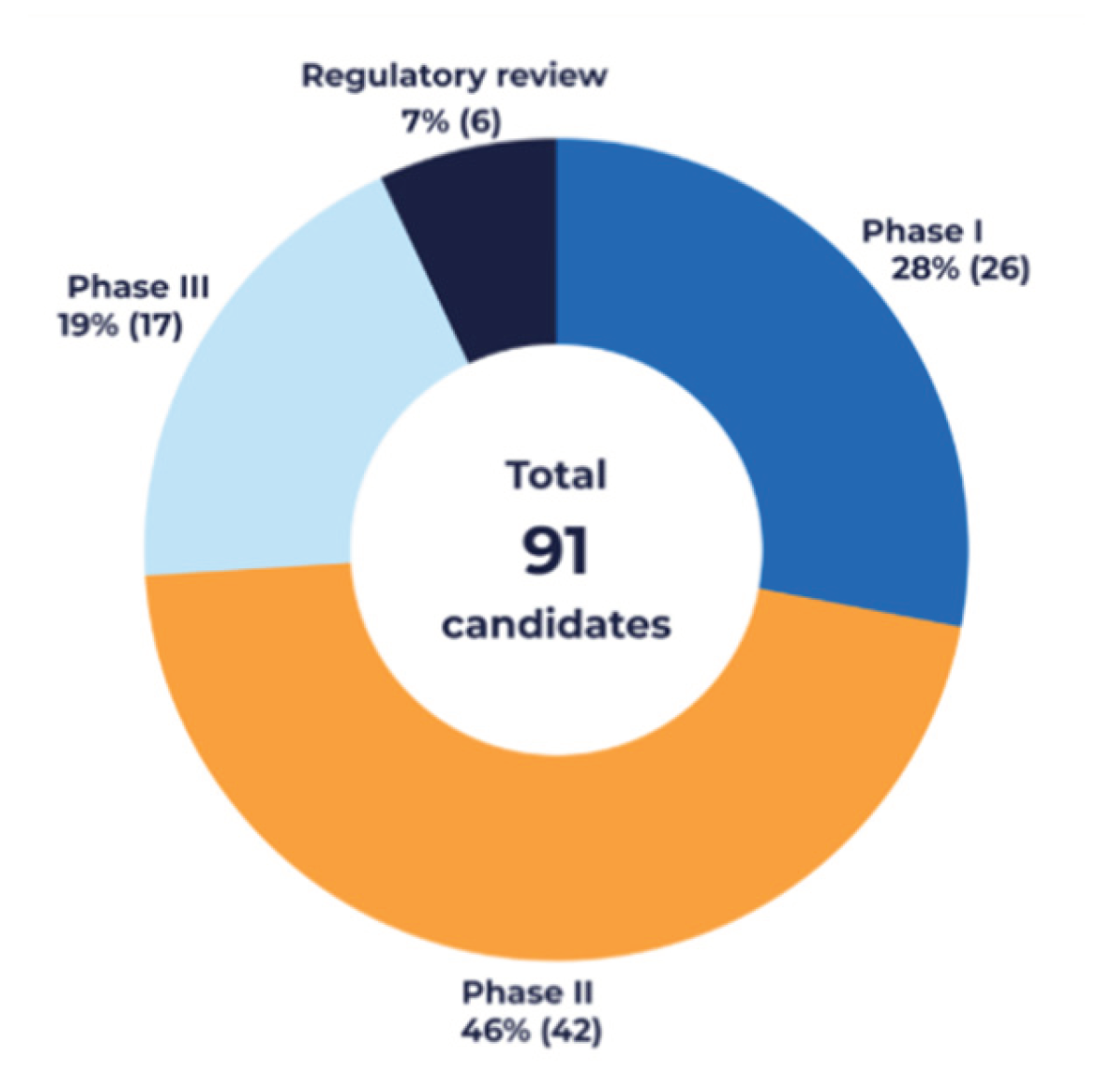
3.1. Overview of the European Vaccine Development Pipeline
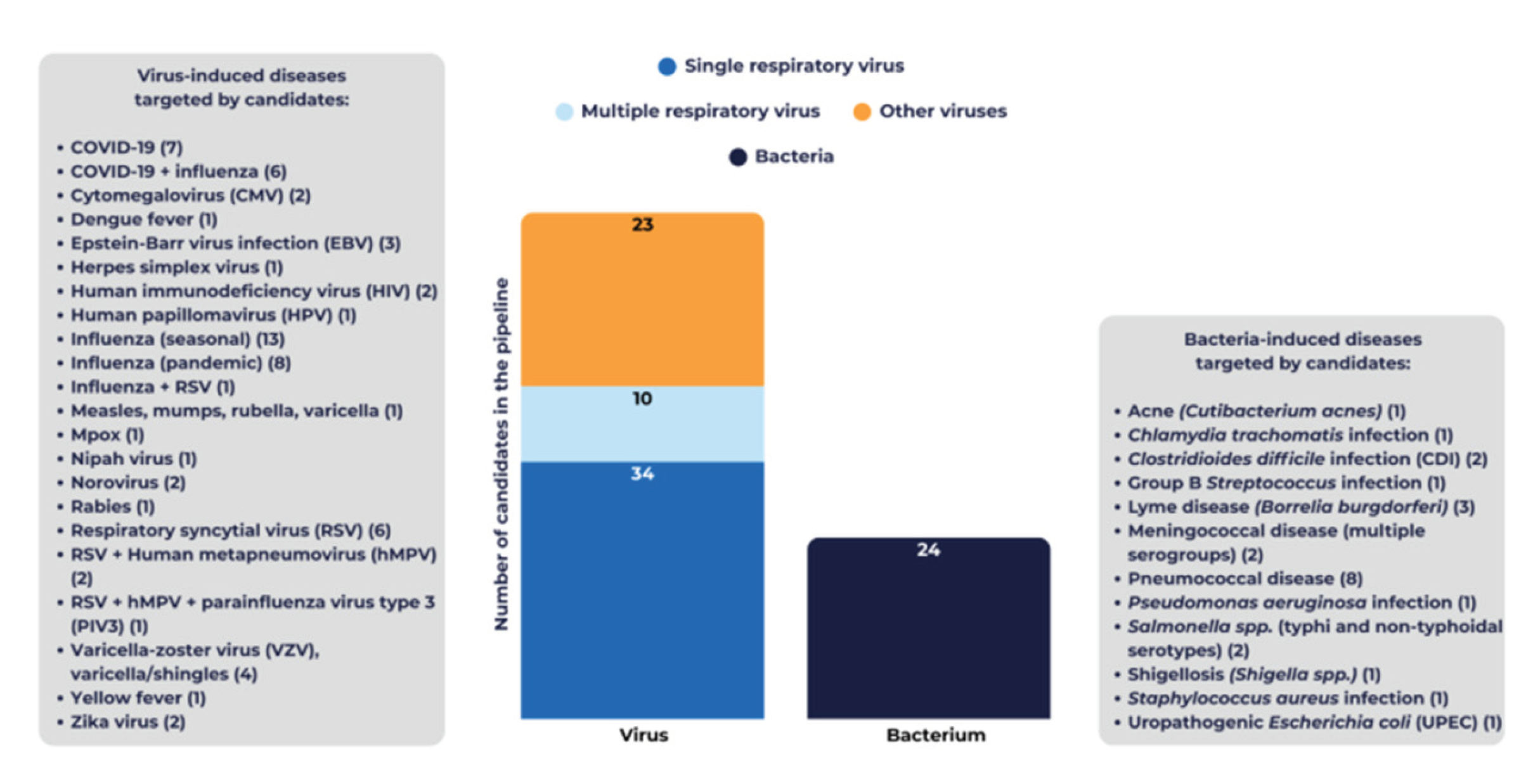
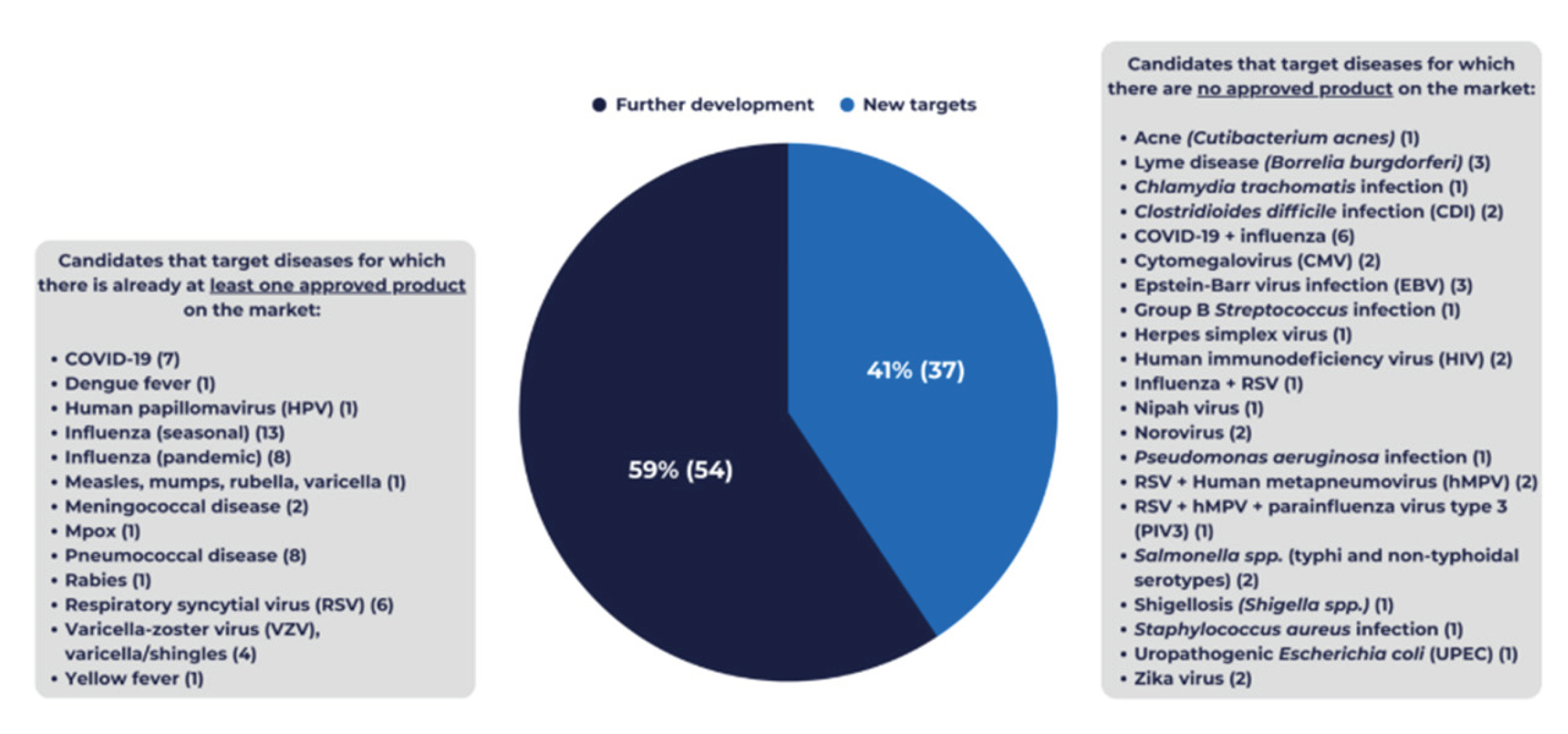
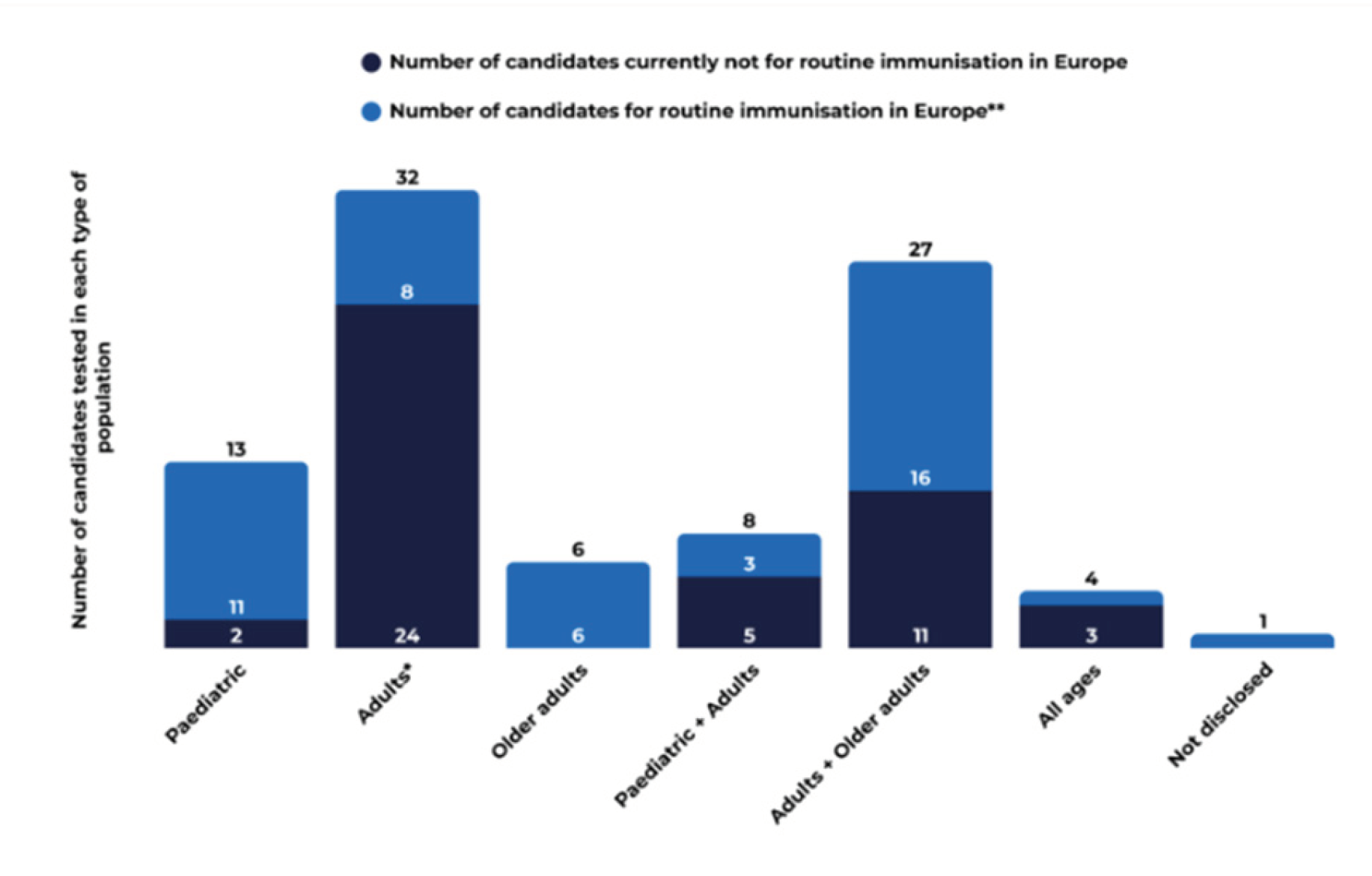
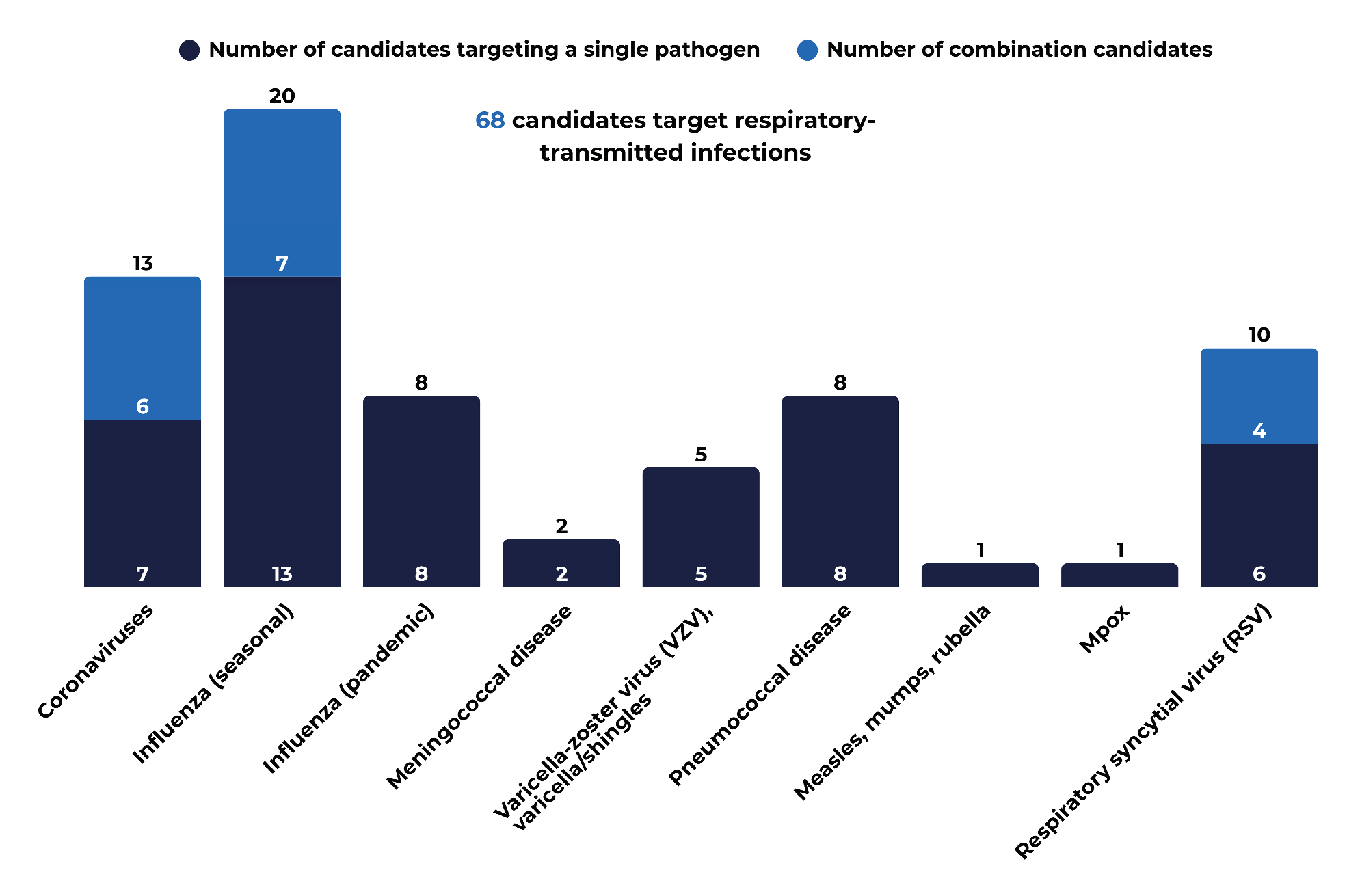
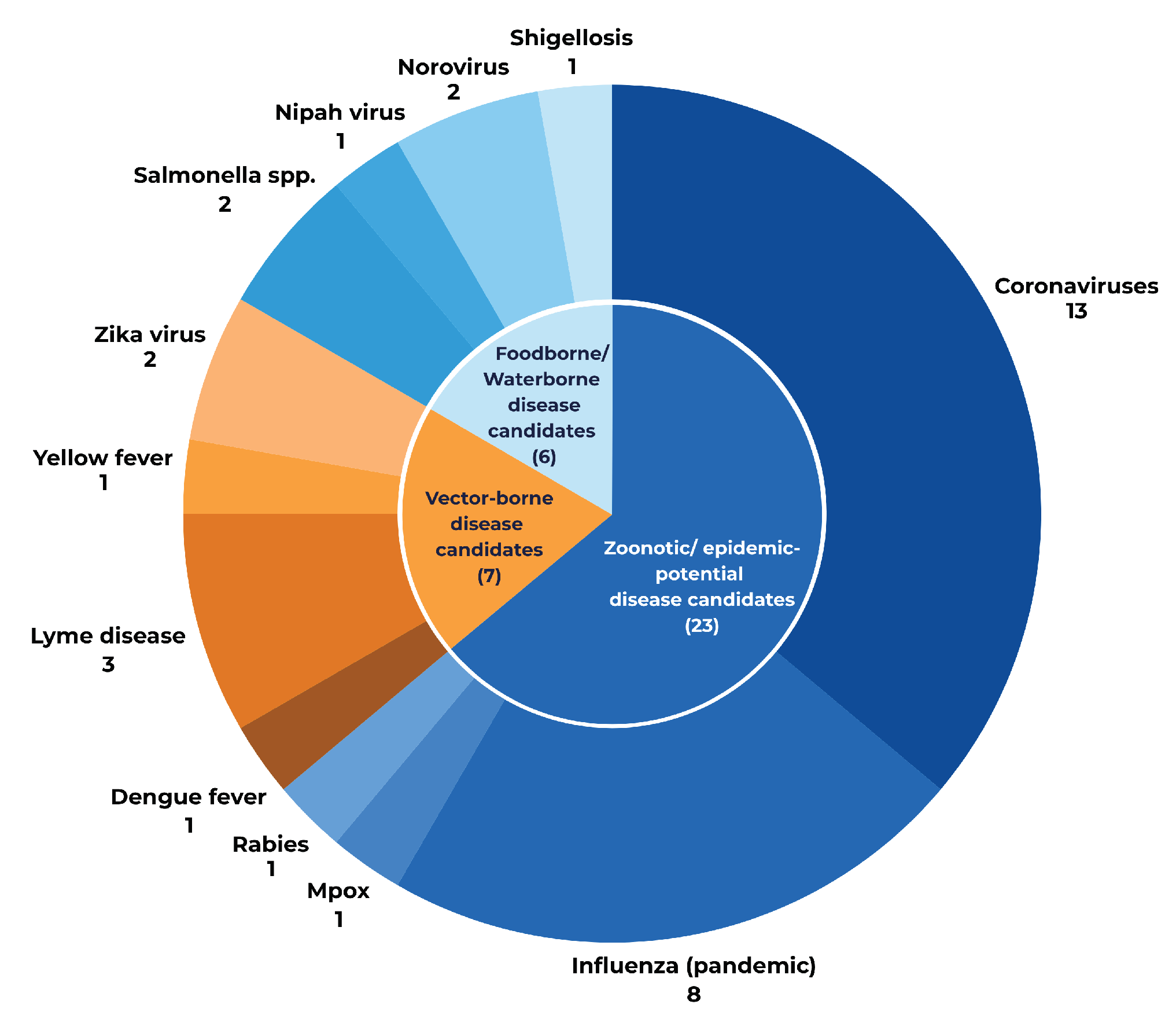
3.2. Public Health Priorities Reflected in the 2025 Pipeline
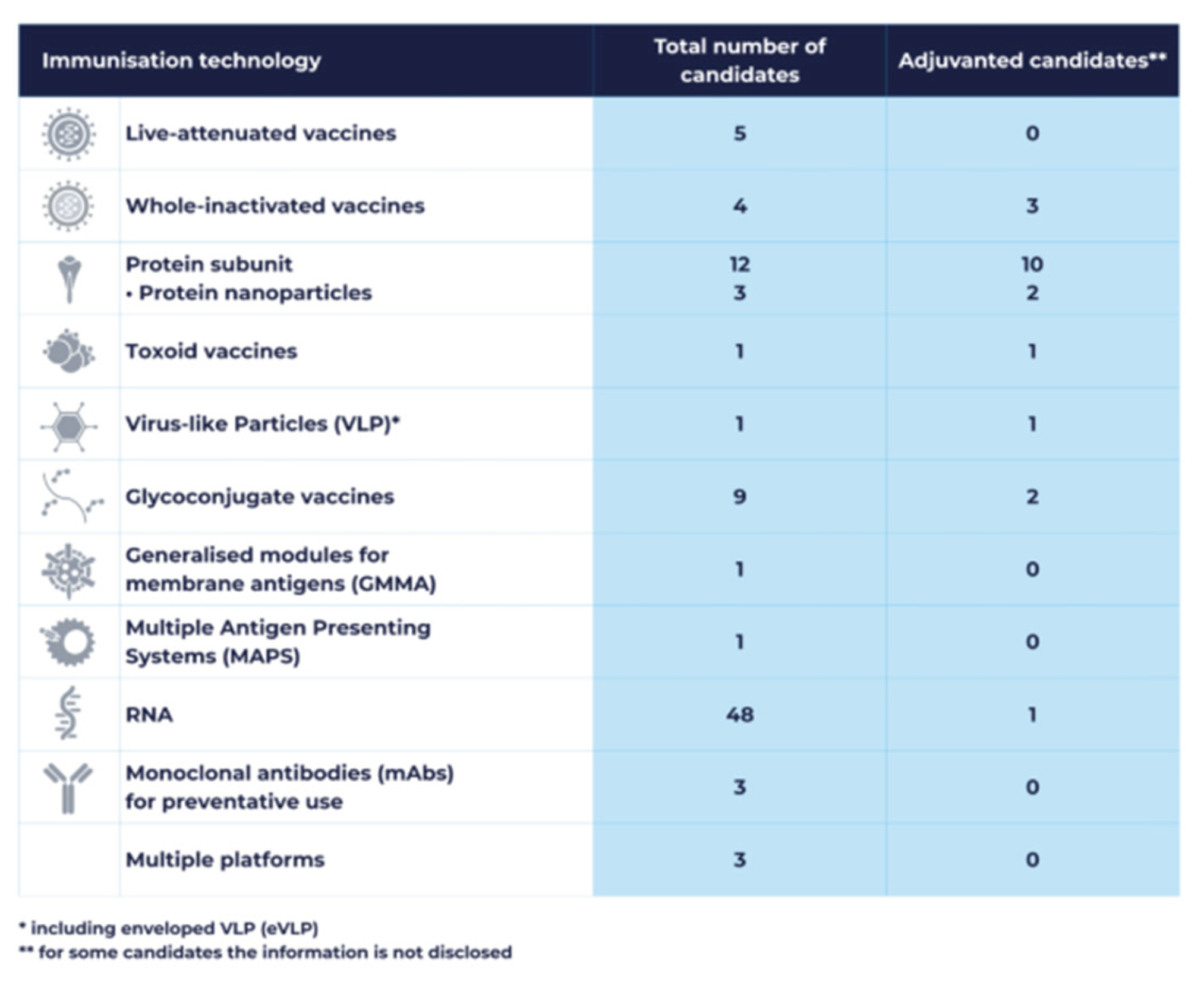
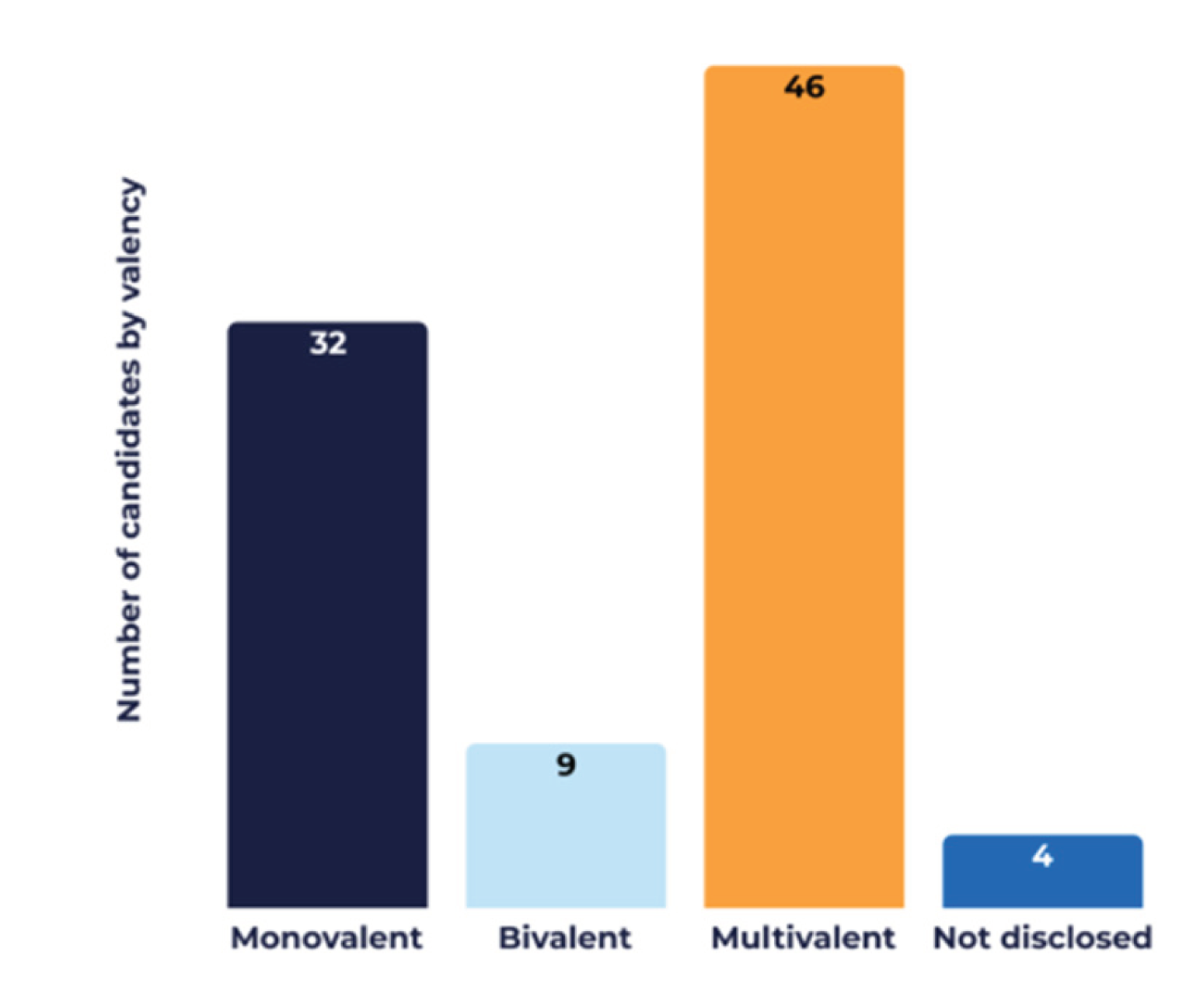
3.3. Technological Innovation Strategies
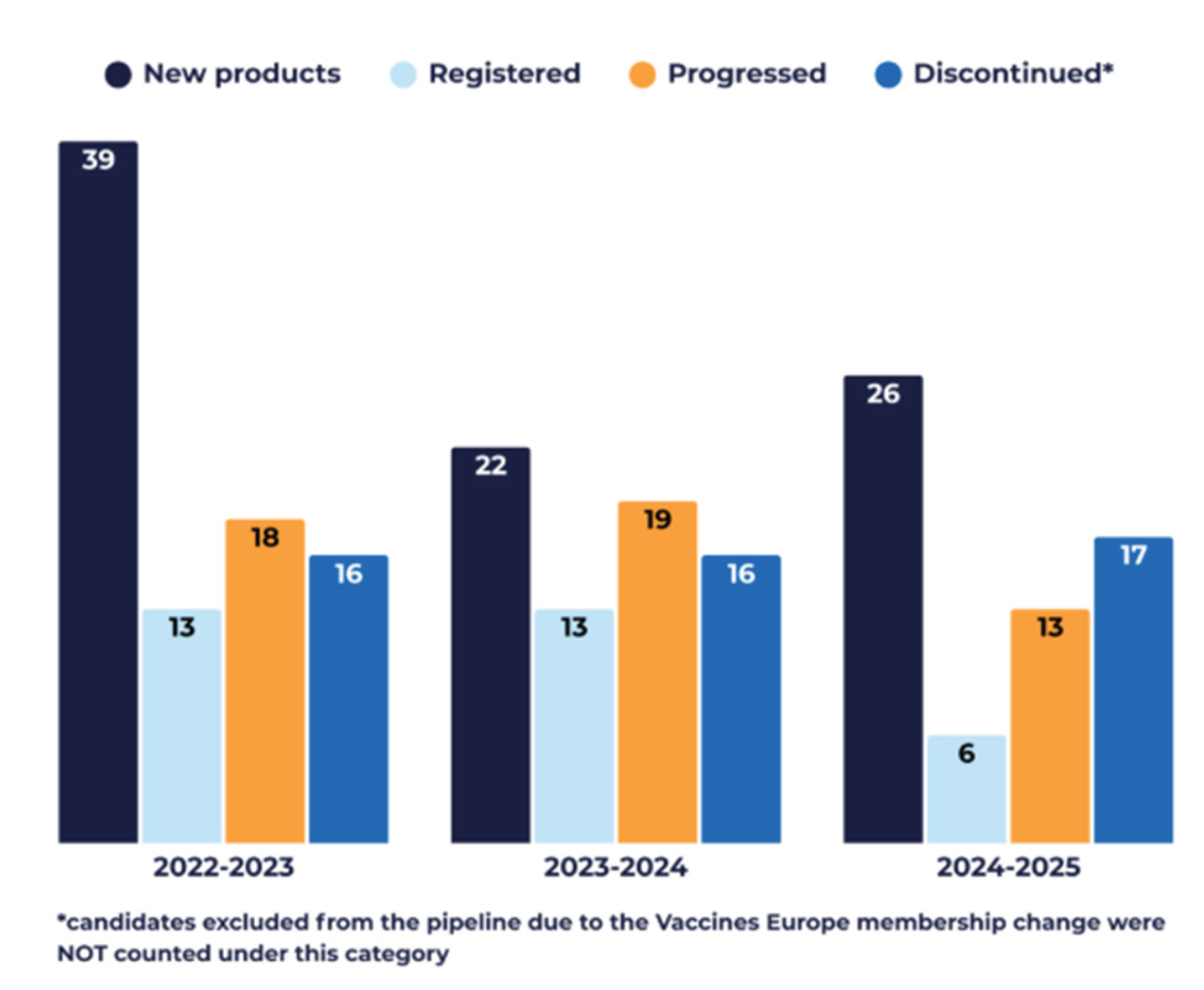
3.4. Pipeline Evolution over Time (2022–2025)
- The annual attrition rate remained relatively stable across the period at approximately 17% (range: 15.5–17.4%).
- Over the same timeframe, the annual registration rate averaged approximately 9% (range: 6.6–12.6%), reflecting the proportion of candidates receiving marketing authorisation each year.
- Progression across clinical development phases also remained consistent, with an average of 17% (range: 13.3–18.8%) of the pipeline advancing annually, indicating continued movement of candidates through the pipeline despite discontinuations.
- Pipeline entry was highest in the 2022–2023 interval, with 38% (39) new candidates entering clinical development, reflecting accelerated activity in the context of the COVID-19 pandemic. In subsequent years, entry stabilised, with 24% (24) candidates entering the pipeline in 2023–2024 and 29% (26) in 2024–2025.
4. Discussion
4.1. Interpretation of Pipeline Trends
4.2. Limitations of the Analysis
4.3. Drivers and Constraints Shaping the Pipeline
4.4. Implications for Policy and Public Health Strategy
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Global immunization efforts have saved at least 154 million lives over the past 50 years. Available online: https://www.who.int/news/item/24-04-2024-global-immunization-efforts-have-saved-at-least-154-million-lives-over-the-past-50-years/ (accessed on 06 February 2026).
- Razzini, J.L.; Giné-Vázquez, I.; Jin, J.; Santiago-Pérez, M.I.; Pérez-Martínez, O.; Otero-Barrós, M.T.; Suárez-Gaiche, N.; Kramer, R.; Platero-Alonso, L.; Álvarez-Gil, R.M.; et al. Impact of universal nirsevimab prophylaxis in infants on hospital and primary care outcomes across two respiratory syncytial virus seasons in Galicia, Spain (NIRSE-GAL): a population-based prospective observational study. Lancet Infect Dis 2026. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, P. Advances in preventing RSV in children. Lancet Microbe 2024, 5, e421. [Google Scholar] [CrossRef] [PubMed]
- Falcaro, M.; Soldan, K.; Ndlela, B.; Sasieni, P. Effect of the HPV vaccination programme on incidence of cervical cancer and grade 3 cervical intraepithelial neoplasia by socioeconomic deprivation in England: population based observational study. Bmj 2024, 385, e077341. [Google Scholar] [CrossRef] [PubMed]
- WHO. Vaccinating at every age is key to unlocking the full potential of immunization. Available online: https://www.who.int/news/item/05-06-2025-vaccinating-at-every-age-is-key-to-unlocking-the-full-potential-of-immunization (accessed on 06 February 2026).
- WHO. COVID-19 Vaccination Insights Report - 4 March 2024. Available online: https://www.who.int/publications/m/item/covid-19-vaccination-insights-report-4-march-2024 (accessed on 06 February 2026).
- Heidecker, B.; Libby, P.; Vassiliou, V.S.; Roubille, F.; Vardeny, O.; Hassager, C.; Gatzoulis, M.A.; Mamas, M.A.; Cooper, L.T.; Schoenrath, F.; et al. Vaccination as a new form of cardiovascular prevention: a European Society of Cardiology clinical consensus statement: With the contribution of the European Association of Preventive Cardiology (EAPC), the Association for Acute CardioVascular Care (ACVC), and the Heart Failure Association (HFA) of the ESC. European Heart Journal 2025, 46, 3518–3531. [Google Scholar] [PubMed]
- Tsanani, S.E.; Yorav, S.; Yaron, S.; Razi, T.; Yechezkel, M.; Arbel, R.; Yamin, D. Effectiveness of influenza vaccination in preventing severe COPD exacerbations and pneumonia before, during, and after the COVID-19 pandemic: a retrospective cohort study. Lancet Reg Health Eur 2025, 53, 101307. [Google Scholar] [CrossRef] [PubMed]
- de Martel, C.; Georges, D.; Bray, F.; Ferlay, J.; Clifford, G.M. Global burden of cancer attributable to infections in 2018: a worldwide incidence analysis. Lancet Glob Health 2020, 8, e180–e190. [Google Scholar] [CrossRef] [PubMed]
- Onisiforou, A.; Zanos, P. From Viral Infections to Alzheimer’s Disease: Unveiling the Mechanistic Links Through Systems Bioinformatics. J Infect Dis 2024, 230, S128–s140. [Google Scholar] [CrossRef] [PubMed]
- Bjornevik, K.; Münz, C.; Cohen, J.I.; Ascherio, A. Epstein-Barr virus as a leading cause of multiple sclerosis: mechanisms and implications. Nat Rev Neurol 2023, 19, 160–171. [Google Scholar] [CrossRef] [PubMed]
- WHO. Ageing and health. Available online: https://www.who.int/news-room/fact-sheets/detail/ageing-and-health (accessed on 09 February 2026).
- Eurostat. Demography of Europe – 2025 edition. Available online: https://ec.europa.eu/eurostat/web/interactive-publications/demography-2025 (accessed on 09 February 2026).
- Klugman, K.P.; Black, S. Impact of existing vaccines in reducing antibiotic resistance: Primary and secondary effects. Proc Natl Acad Sci U S A 2018, 115, 12896–12901. [Google Scholar] [CrossRef] [PubMed]
- WHO. Estimating the impact of vaccines in reducing antimicrobial resistance and antibiotic use. Available online: https://www.who.int/teams/immunization-vaccines-and-biologicals/product-and-delivery-research/anti-microbial-resistance (accessed on 13 February 2026).
- Bluedot Intelligence Report Year-to-Year Event-Based Surveillance Overview 2024 & 2025. Available online: https://mcusercontent.com/ab84a833923e562d0999bf440/files/0df69ebb-2773-6cc9-af24-d6452d12ae44/BlueDot_Year_to_Year_EBS_Overview_2024_2025_.pdf (accessed on 09 February 2026).
- de Souza, W.M.; Weaver, S.C. Effects of climate change and human activities on vector-borne diseases. Nat Rev Microbiol 2024, 22, 476–491. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, A.; Waltenburg, M.A.; Hall, A.; Kile, J.; Killerby, M.; Knust, B.; Negron, M.; Nichols, M.; Wallace, R.M.; Behravesh, C.B.; et al. Vaccine Preventable Zoonotic Diseases: Challenges and Opportunities for Public Health Progress. Vaccines (Basel) 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Hossain, A.; Monem, M.A.; Rahman, M.; Raza, R. Mpox (monkeypox): a comprehensive updated of current epidemic evidence. Sci One Health 2025, 4, 100100. [Google Scholar] [CrossRef] [PubMed]
- The Value of Prevention for Economic Growth and the Sustainability of Healthcare, Social, and Welfare Systems. Available online: https://www.ambrosetti.eu/en/news/the-value-of-prevention-for-economic-growth-and-the-sustainability-of-healthcare-social-and-welfare-systems/ (accessed on 17 March 2026).
- Socio-Economic Value of Adult Immunisation Programmes. Available online: https://www.ohe.org/publications/the-socio-economic-value-of-adult-immunisation-programmes/ (accessed on 24 February 2026).
- McLellan, J.S.; Ray, W.C.; Peeples, M.E. Structure and function of respiratory syncytial virus surface glycoproteins. Curr Top Microbiol Immunol 2013, 372, 83–104. [Google Scholar] [PubMed]
- WHO bacterial priority pathogens list, 2024: Bacterial pathogens of public health importance to guide research, development and strategies to prevent and control antimicrobial resistance. Available online: https://www.who.int/publications/i/item/9789240093461 (accessed on 18 February 2026).
- Europe, Vaccines. Vaccines Europe reveals its first pipeline review. Available online: https://www.vaccineseurope.eu/media-hub/blogs/vaccines-europe-reveals-its-first-pipeline-review/ (accessed on February 2026).
- Europe, Vaccines. Vaccines Europe pipeline review. Available online: https://www.vaccineseurope.eu/vaccines-ecosystem/vaccines-pipeline/ (accessed on 17 March 2026).
- GAVI. Six global health threats to watch in 2026. Available online: https://www.gavi.org/sites/default/files/2026/gavi-insight-paper-global-health-threats.pdf (accessed on 26 February 2026).
- Pharmaprojects® Citeline 2026. Available online: https://www.citeline.com/en (accessed on 12 February 2026).
- Trialtrove®Citeline 2026. Available online: https://www.citeline.com/en/products-services/clinical/trialtrove (accessed on 18 February 2026).
- Global burden of bacterial antimicrobial resistance 1990-2021: a systematic analysis with forecasts to 2050. Lancet 2024, 404, 1199–1226. [CrossRef] [PubMed]
- ECDC. World Mosquito Day 2025: Europe sets new records for mosquito-borne diseases. Available online: https://www.ecdc.europa.eu/en/news-events/world-mosquito-day-2025-europe-sets-new-records-mosquito-borne-diseases (accessed on 20 March 2026).
- Gouglas, D.; Thanh Le, T.; Henderson, K.; Kaloudis, A.; Danielsen, T.; Hammersland, N.C.; Robinson, J.M.; Heaton, P.M.; Røttingen, J.A. Estimating the cost of vaccine development against epidemic infectious diseases: a cost minimisation study. Lancet Glob Health 2018, 6, e1386–e1396. [Google Scholar] [CrossRef] [PubMed]
- EFPIA; Vaccines Europe. Assessing the clinical trial ecosystem in Europe. Available online: https://www.vaccineseurope.eu/wp-content/uploads/2024/10/EFPIA-VE-CT-Report-221024-final.pdf (accessed on 18 March 2026).

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



