Submitted:
08 April 2026
Posted:
09 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Mechanistic Spatiotemporal Model of CAR-T Therapy for Solid Tumours
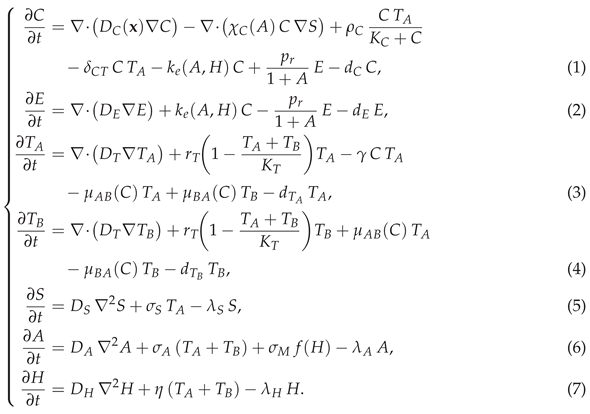 |
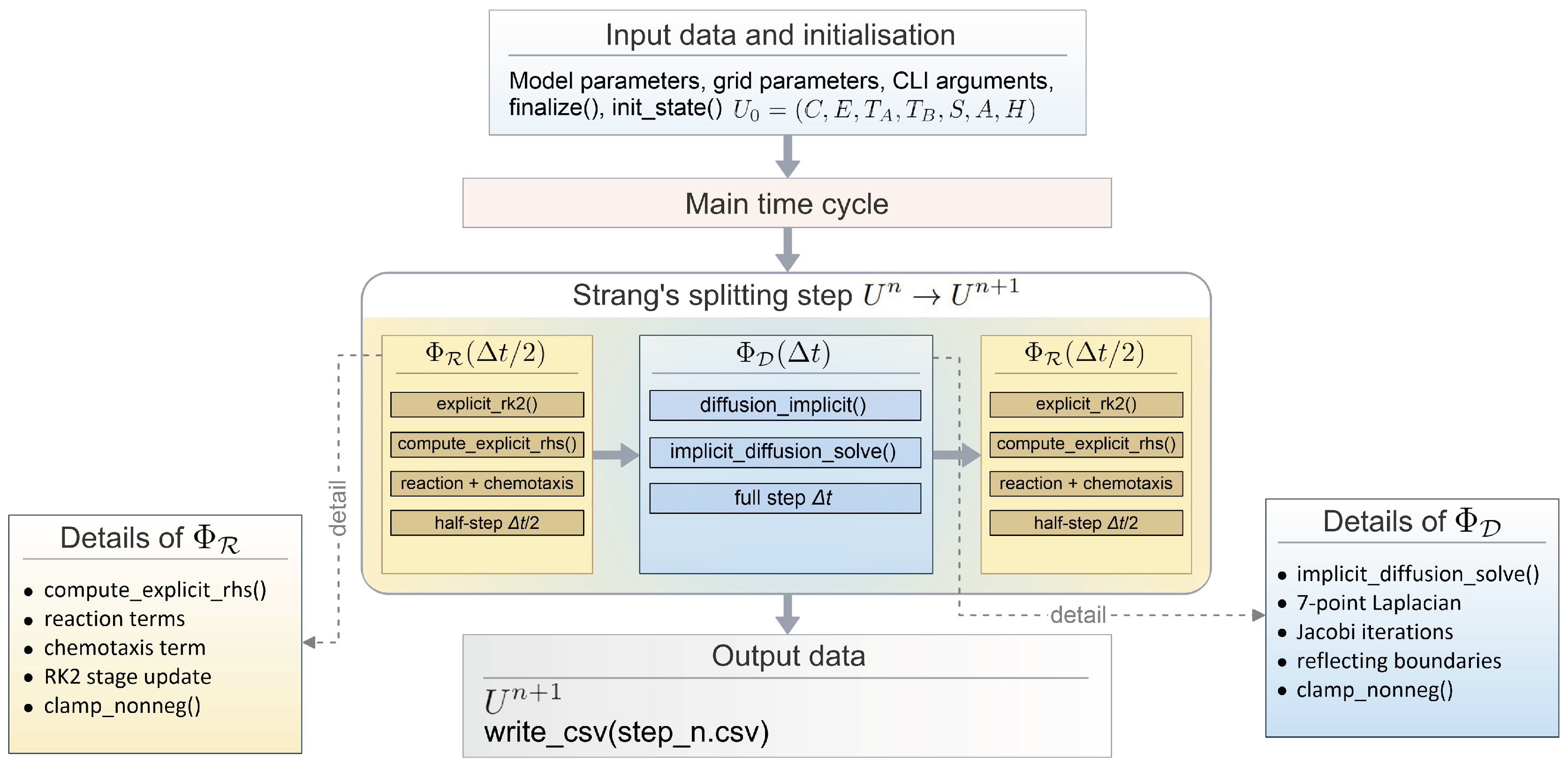
2.2. Numerical Solution of the Model and Software Implementation
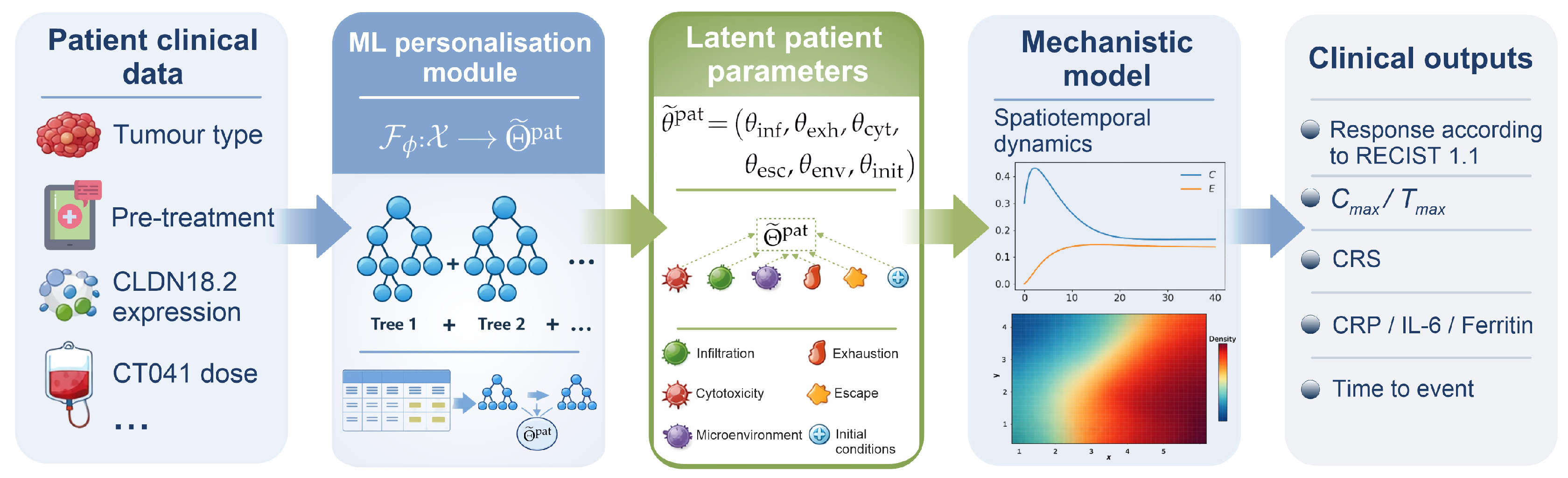
2.3. Formulation of the Limited Inverse Problem of Patient-Specific Model Personalisation
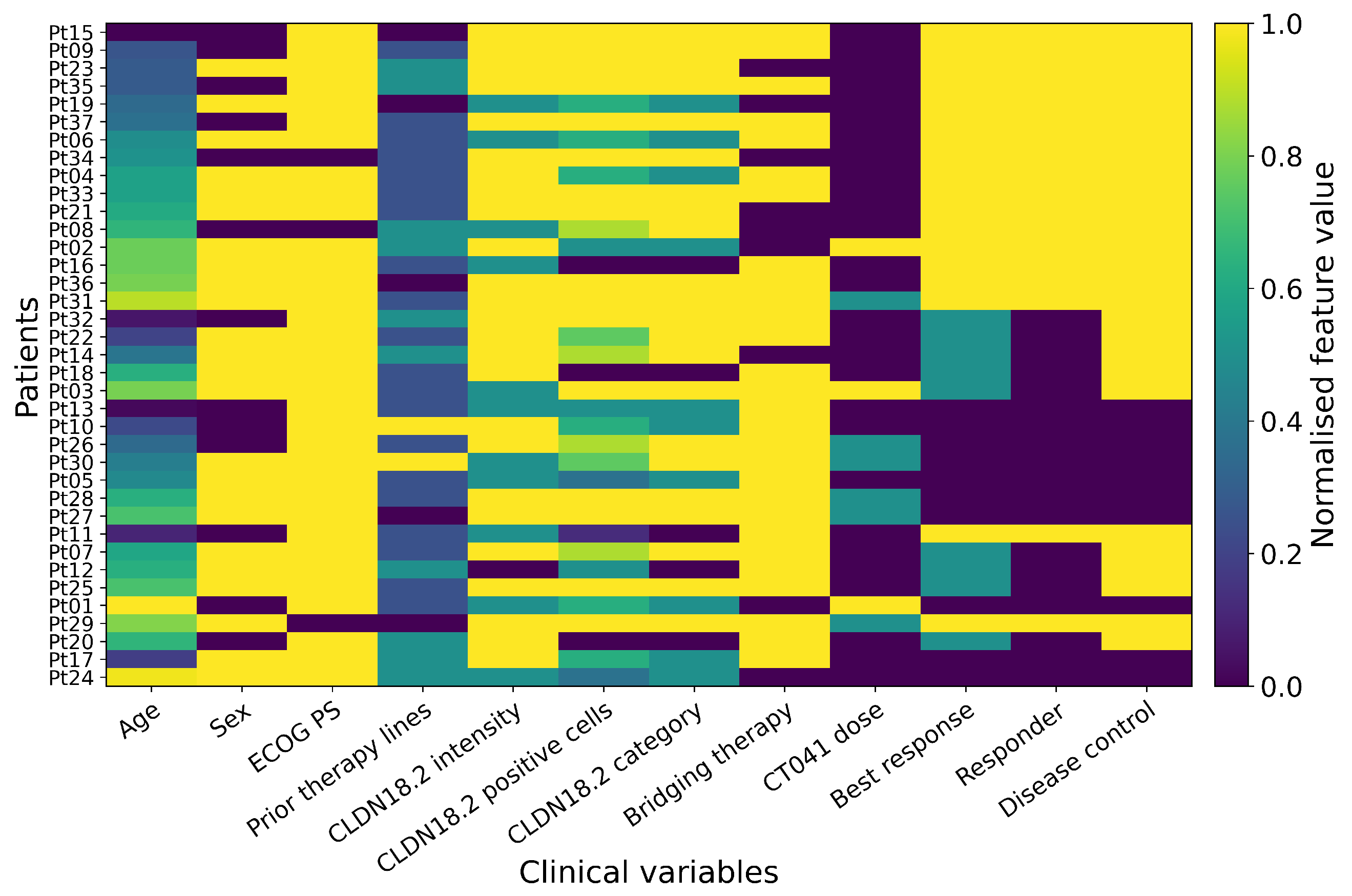
2.4. Clinical Data Source and Variable Preparation
2.5. Machine Learning for Inference of Patient-Specific Model Parameters
2.6. Validation Protocol and Performance Metrics for Latent Parameter Recovery and Baseline Prediction of Clinical Outcomes
3. Results
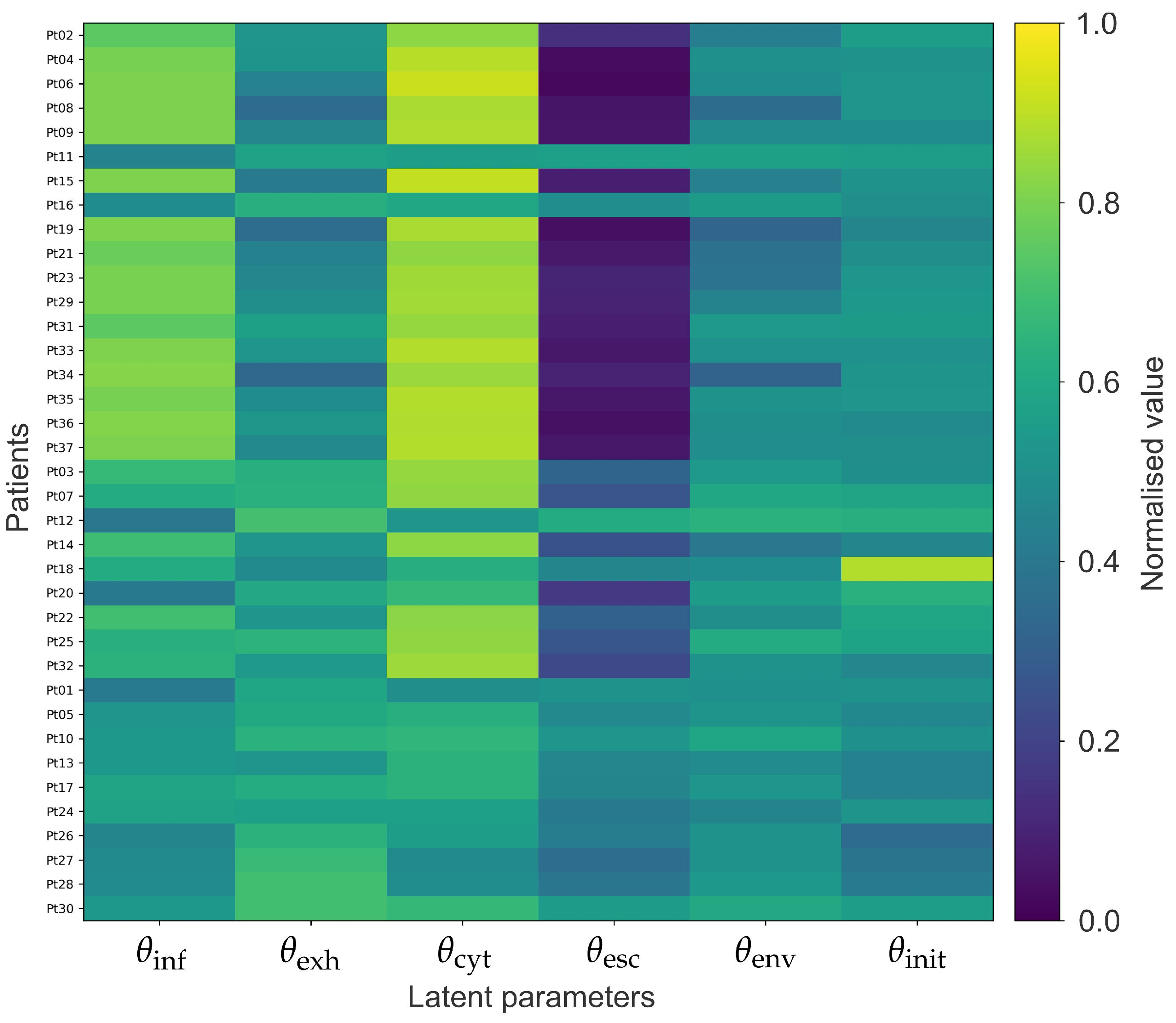
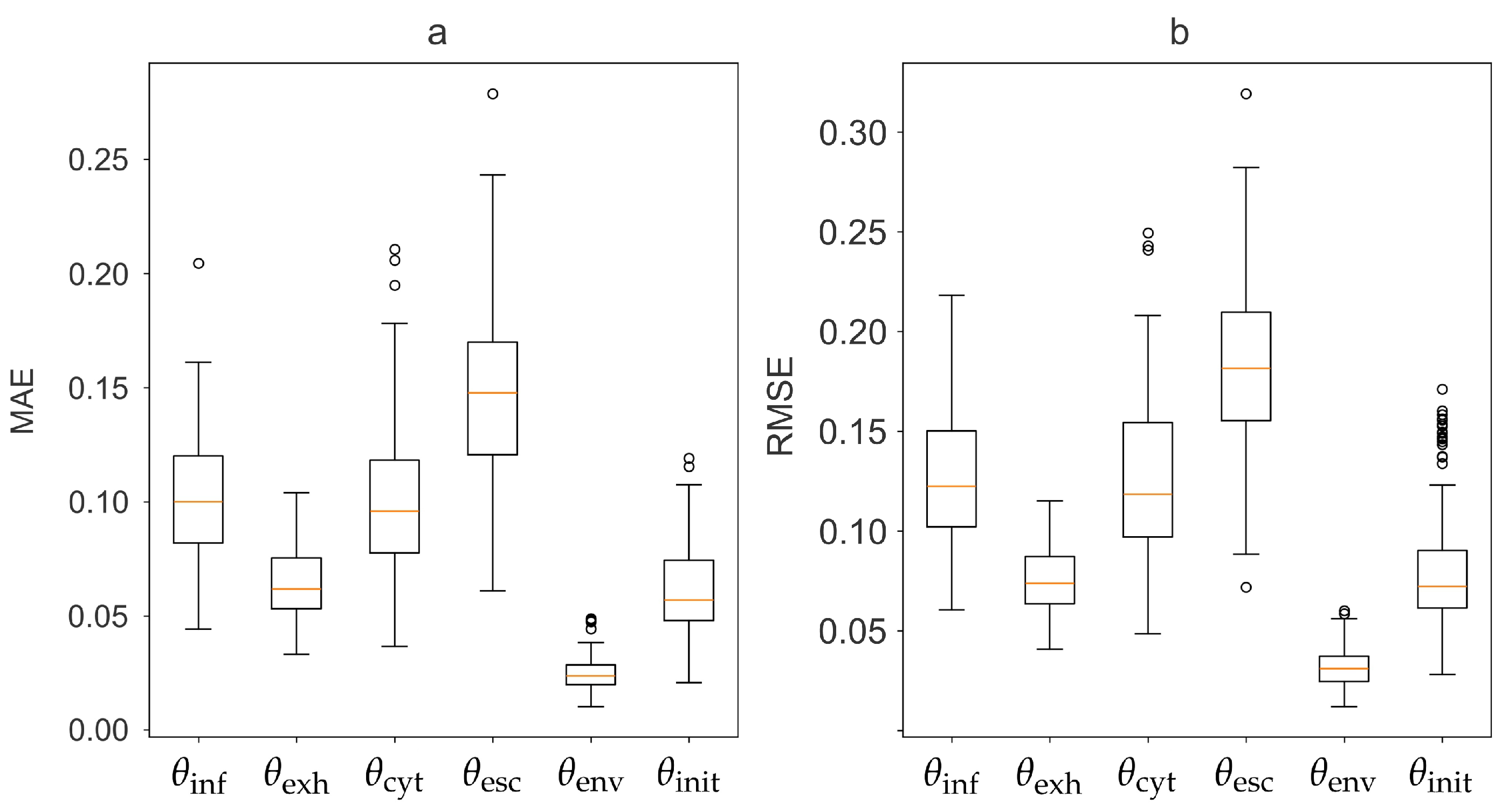
3.1. Inference of Patient-Specific Latent Model Parameters
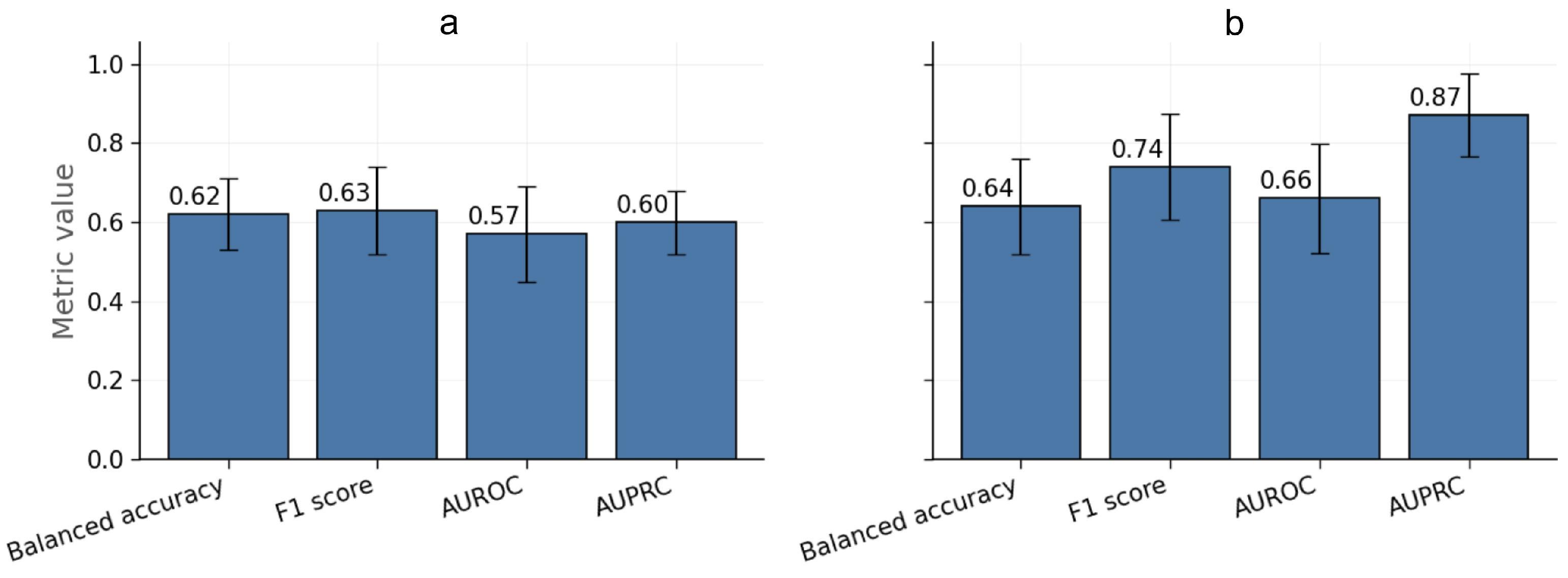
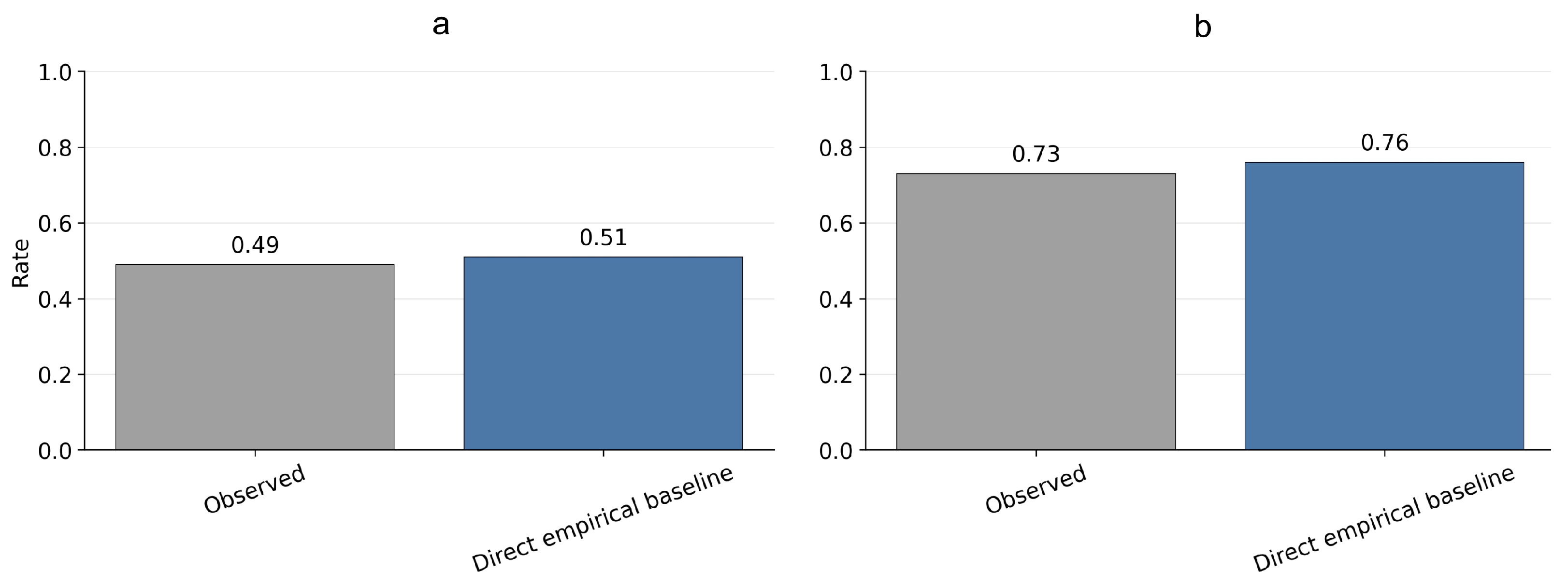
3.2. Auxiliary Empirical Baseline for Binary Clinical Outcomes
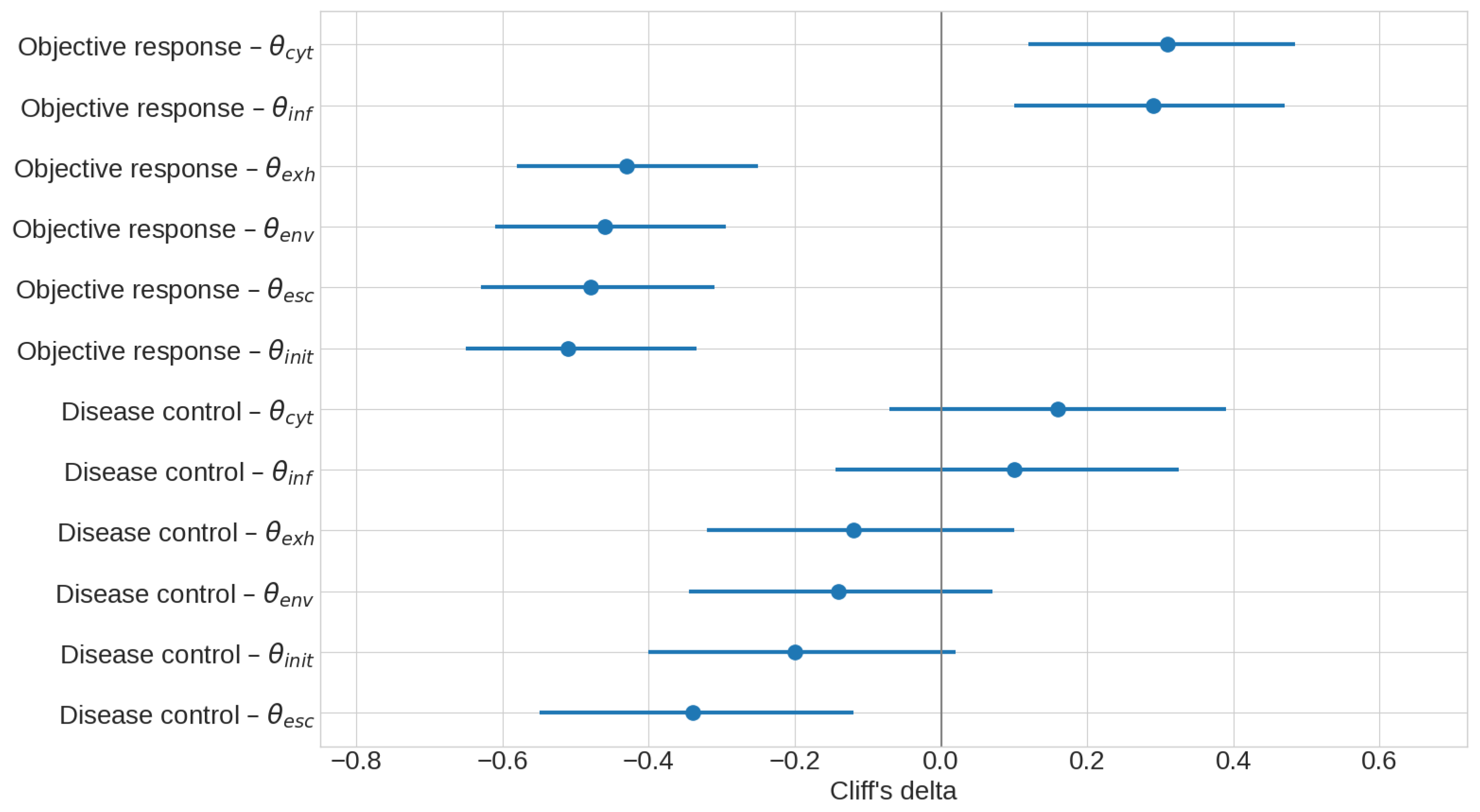
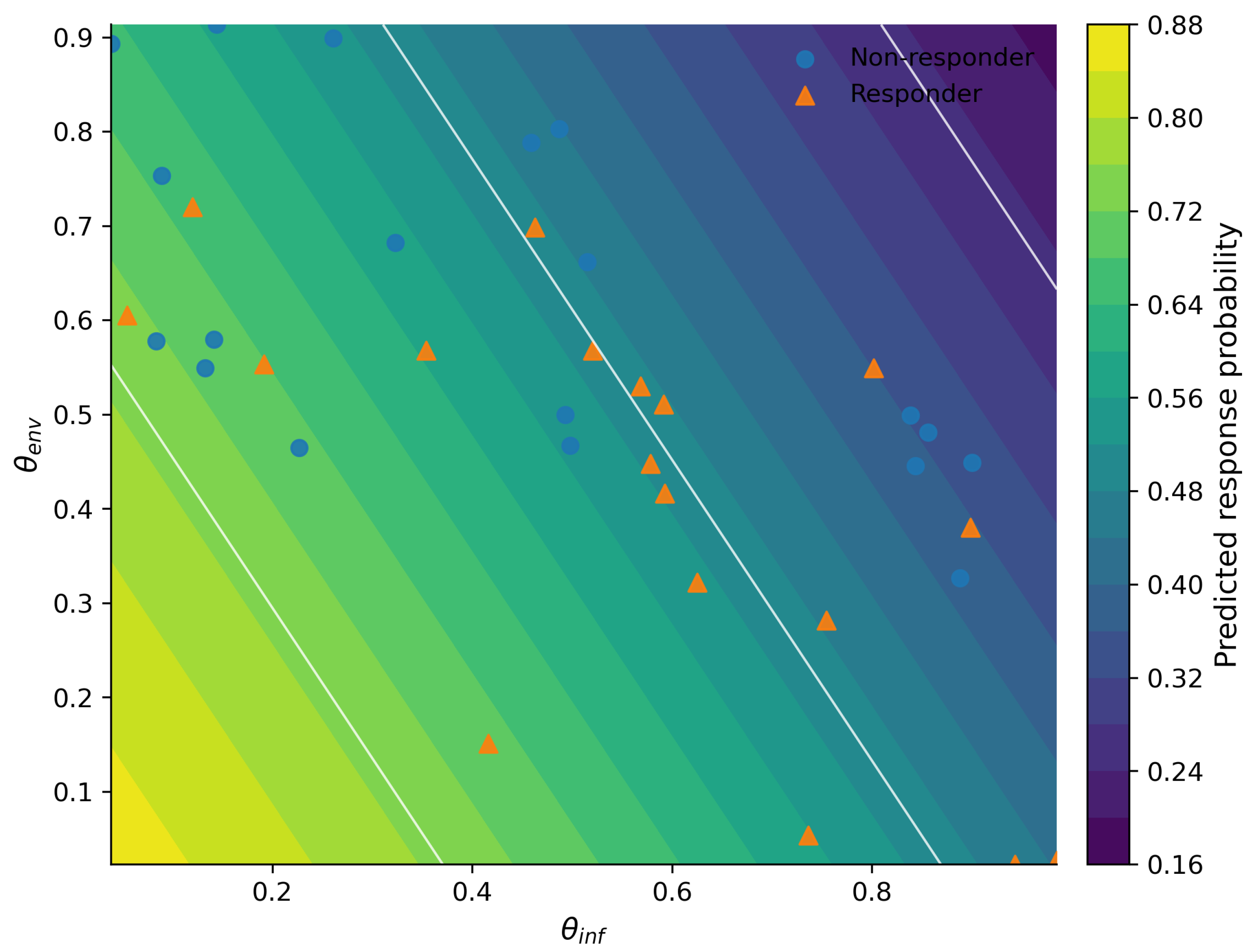
3.3. Mechanistic Interpretation of Latent Factors Underlying Response and Resistance
4. Discussion
5. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
References
- Maude, S.L.; Frey, N.; Shaw, P.A.; Aplenc, R.; Barrett, D.M.; Bunin, N.J.; Chew, A.; Gonzalez, V.E.; Zheng, Z.; Lacey, S.F.; et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N. Engl. J. Med. 2014, 371, 1507–1517. [Google Scholar] [CrossRef] [PubMed]
- Maude, S.L.; Laetsch, T.W.; Buechner, J.; et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N. Engl. J. Med. 2018, 378(5), 439–448. [Google Scholar] [CrossRef] [PubMed]
- Neelapu, S.S.; Locke, F.L.; Bartlett, N.L.; et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N. Engl. J. Med. 2017, 377(26), 2531–2544. [Google Scholar] [CrossRef] [PubMed]
- June, C.H.; O’Connor, R.S.; Kawalekar, O.U.; et al. CAR T cell immunotherapy for human cancer. Science 2018, 359(6382), 1361–1365. [Google Scholar] [CrossRef]
- Sadelain, M.; Brentjens, R.; Rivière, I. The basic principles of chimeric antigen receptor design. Cancer Discov. 2013, 3(4), 388–398. [Google Scholar] [CrossRef]
- Newick, K.; O’Brien, S.; Moon, E.; Albelda, S.M. CAR T cell therapy for solid tumors. Annu. Rev. Med. 2017, 68, 139–152. [Google Scholar] [CrossRef]
- Wagner, J.; Wickman, E.; DeRenzo, C.; Gottschalk, S. CAR T cell therapy for solid tumors: Bright future or dark reality? Mol. Ther. 2020, 28(11), 2320–2339. [Google Scholar] [CrossRef]
- Li, J.; Li, W.; Huang, K.; et al. Chimeric antigen receptor T cell (CAR-T) immunotherapy for solid tumors: Lessons learned and strategies for moving forward. J. Hematol. Oncol. 2018, 11(1), 22. [Google Scholar] [CrossRef]
- Filley, A.C.; Henriquez, M.; Dey, M. CART immunotherapy: Development, success, and translation to malignant gliomas and other solid tumors. Front. Oncol. 2018, 8, 453. [Google Scholar] [CrossRef]
- Ramakrishna, S.; Highfill, S.L.; Walsh, Z.; et al. Prospects and challenges for use of CAR T cell therapies in solid tumors. Expert Opin. Biol. Ther. 2020, 20(5), 503–516. [Google Scholar] [CrossRef]
- Rafiq, S.; Hackett, C.S.; Brentjens, R.J. Engineering strategies to overcome the current roadblocks in CAR T cell therapy. Nat. Rev. Clin. Oncol. 2020, 17(3), 147–167. [Google Scholar] [CrossRef]
- Kirtane, K.; Elmariah, H.; Chung, C.H.; Abate-Daga, D. Adoptive cellular therapy in solid tumor malignancies: Review of the literature and challenges ahead. J. Immunother. Cancer 2021, 9(7), e002723. [Google Scholar] [CrossRef] [PubMed]
- Majzner, R.G.; Mackall, C.L. Tumor antigen escape from CAR T-cell therapy. Cancer Discov. 2018, 8(10), 1219–1226. [Google Scholar] [CrossRef] [PubMed]
- Labanieh, L.; Majzner, R.G.; Mackall, C.L. Programming CAR-T cells to kill cancer. Nat. Biomed. Eng. 2018, 2(6), 377–391. [Google Scholar] [CrossRef] [PubMed]
- Petty, A.J.; Heyman, B.; Yang, Y. Chimeric antigen receptor cell therapy: Overcoming obstacles to battle cancer. Cancers 2020, 12(4), 842. [Google Scholar] [CrossRef]
- Tian, Y.; Li, Y.; Shao, Y.; Zhang, Y. Gene modification strategies for next-generation CAR T cells against solid cancers. J. Hematol. Oncol. 2020, 13(1), 54. [Google Scholar] [CrossRef]
- Andrea, A.E.; Chiron, A.; Mallah, S.; et al. Advances in CAR-T cell genetic engineering strategies to overcome hurdles in solid tumors treatment. Front. Immunol. 2022, 13, 830292. [Google Scholar] [CrossRef]
- Zhang, P.-F.; Wang, C.; Zhang, L.; Li, Q. Reversing chemokine/chemokine receptor mismatch to enhance the antitumor efficacy of CAR-T cells. Immunotherapy 2022, 14(6), 459–473. [Google Scholar] [CrossRef]
- Adachi, K.; Kano, Y.; Nagai, T.; et al. IL-7 and CCL19 expression in CAR-T cells improves immune cell infiltration and CAR-T cell survival in the tumor. Nat. Biotechnol. 2018, 36(4), 346–351. [Google Scholar] [CrossRef]
- Polyakov, M.V.; Ten, V.V. Simulation tumor growth in heterogeneous medium based on diffusion equation. Int. J. Mod. Phys. C 2024, 35(1), 2450010. [Google Scholar] [CrossRef]
- Wherry, E.J.; Kurachi, M. Molecular and cellular insights into T cell exhaustion. Nat. Rev. Immunol. 2015, 15(8), 486–499. [Google Scholar] [CrossRef]
- Schietinger, A.; Greenberg, P.D. Tolerance and exhaustion: Defining mechanisms of T cell dysfunction. Trends Immunol. 2014, 35(2), 51–60. [Google Scholar] [CrossRef] [PubMed]
- Thommen, D.S.; Schumacher, T.N. T cell dysfunction in cancer. Cancer Cell 2018, 33(4), 547–562. [Google Scholar] [CrossRef] [PubMed]
- van der Leun, A.M.; Thommen, D.S.; Schumacher, T.N. CD8+ T cell states in human cancer: Insights from single-cell analysis. Nat. Rev. Cancer 2020, 20(4), 218–232. [Google Scholar] [CrossRef] [PubMed]
- Dolina, J.S.; Van Braeckel-Budimir, N.; Thomas, G.D.; Salek-Ardakani, S. CD8+ T cell exhaustion in cancer. Front. Immunol. 2021, 12, 715234. [Google Scholar] [CrossRef]
- Schurich, A.; Magalhaes, I.; Mattsson, J. Metabolic regulation of CAR T cell function by the hypoxic microenvironment in solid tumors. Immunotherapy 2019, 11(4), 335–345. [Google Scholar] [CrossRef]
- Brown, C.E.; Hibbard, J.C.; Alizadeh, D.; et al. Locoregional delivery of IL-13Rα2-targeting CAR-T cells in recurrent high-grade glioma: A phase 1 trial. Nat. Med. 2024, 30(4), 1001–1012. [Google Scholar] [CrossRef]
- Qi, C.; Liu, C.; Gong, J.; et al. Claudin18.2-specific CAR T cells in gastrointestinal cancers: Phase 1 trial final results. Nat. Med. 2024, 30, 2224–2234. [Google Scholar] [CrossRef]
- Shi, D.; Shi, Y.; Kaseb, A.O.; et al. Chimeric antigen receptor-glypican-3 T-cell therapy for advanced hepatocellular carcinoma: Results of phase I trials. Clin. Cancer Res. 2020, 26(15), 3979–3989. [Google Scholar] [CrossRef]
- Straathof, K.; Flutter, B.; Wallace, R.; et al. Antitumor activity without on-target off-tumor toxicity of GD2-chimeric antigen receptor T cells in patients with neuroblastoma. Sci. Transl. Med. 2020, 12(571), eabd6169. [Google Scholar] [CrossRef]
- Mueller, K.T.; Waldron, E.; Grupp, S.A.; et al. Clinical pharmacology of tisagenlecleucel in B-cell acute lymphoblastic leukemia. Clin. Cancer Res. 2018, 24(24), 6175–6184. [Google Scholar] [CrossRef]
- Stein, A.M.; Grupp, S.A.; Levine, J.E.; et al. Tisagenlecleucel model-based cellular kinetic analysis of chimeric antigen receptor-T cells. CPT Pharmacometrics Syst. Pharmacol. 2019, 8(5), 285–295. [Google Scholar] [CrossRef] [PubMed]
- Chaudhury, A.; Zhu, X.; Chu, L.; et al. Chimeric antigen receptor T cell therapies: A review of cellular kinetic-pharmacodynamic modeling approaches. J. Clin. Pharmacol. 2020, 60(S1), S147–S159. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, P.; Yang, X.; Abler, D.; et al. Mathematical deconvolution of CAR T-cell proliferation and exhaustion from real-time killing assay data. J. R. Soc. Interface 2020, 17(162), 20190734. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Sahoo, P.; Wang, D.; et al. Modeling interaction of glioma cells and CAR T-cells considering multiple CAR T-cells bindings. Immunoinformatics 2023, 9, 100022. [Google Scholar] [CrossRef]
- Kara, E.; Jackson, T.L.; Jones, C.; et al. Mathematical modeling insights into improving CAR T cell therapy for solid tumors with bystander effects. NPJ Syst. Biol. Appl. 2024, 10, 105. [Google Scholar] [CrossRef]
- Polyakov, M.V.; Tuchina, E.I. Reaction-Diffusion Model of CAR-T Cell Therapy in Solid Tumours with Antigen Escape. Computation 2026, 14(1), 3. [Google Scholar] [CrossRef]
- Rohrs, J.A.; Siegler, E.L.; Wang, P.; Finley, S.D. ERK activation in CAR T cells is amplified by CD28-mediated increase in CD3ζ phosphorylation. iScience 2020, 23(4), 101023. [Google Scholar] [CrossRef]
- Logun, M.; Lyon, S.M.; Chow, V.A.; et al. Label-free in vitro assays predict the potency of anti-disialoganglioside chimeric antigen receptor T-cell products. Cytotherapy 2023, 25(6), 670–682. [Google Scholar] [CrossRef]
- Butner, J.D.; et al. Mathematical modeling of cancer immunotherapy for personalized clinical translation. Nat. Comput. Sci. 2022, 2(12), 785–796. [Google Scholar] [CrossRef]
- Wu, C.; Lorenzo, G.; Hormuth, D.A.; et al. Integrating mechanism-based modeling with biomedical imaging to build practical digital twins for clinical oncology. Biophys. Rev. 2022, 3(2), 021304. [Google Scholar] [CrossRef]
- Procopio, A.; Cesarelli, G.; Donisi, L.; et al. Combined mechanistic modeling and machine-learning approaches in systems biology – a systematic literature review. Comput. Methods Programs Biomed. 2023, 240, 107681. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, G.; Ahmed, S.R.; Hormuth, D.A.; et al. Patient-specific, mechanistic models of tumor growth incorporating artificial intelligence and big data. Annu. Rev. Biomed. Eng. 2024, 26(1), 529–560. [Google Scholar] [CrossRef] [PubMed]
- Polyakov, M. Integration of Machine Learning to Personalise a Mathematical Model of Tumour Dynamics Based on Reaction-Diffusion Equations. In 2025 7th International Conference on Control Systems, Mathematical Modeling, Automation and Energy Efficiency (SUMMA); Russian Federation: Lipetsk, Russia, 2025; pp. 776–779. [Google Scholar]
- Polyakov, M. Spatiotemporal Modelling of CAR-T Cell Therapy in Solid Tumours: Mechanisms of Antigen Escape and Immunosuppression. Computation 2026, 14(4), 87. [Google Scholar] [CrossRef]
- Qi, C.; Gong, J.; Li, J.; et al. Claudin18.2-specific CAR T cells in gastrointestinal cancers: Phase 1 trial interim results. Nat. Med. 2022, 28, 1189–1198. [Google Scholar] [CrossRef]
- Taylor, C.A.; Glover, M.; Maher, J. CAR-T cell technologies that interact with the tumour microenvironment in solid tumours. Expert Rev. Clin. Immunol. 2024, 20(8), 849–871. [Google Scholar] [CrossRef]
- Lin, H.; Yang, X.; Ye, S.; Huang, L.; Mu, W. Antigen escape in CAR-T cell therapy: Mechanisms and overcoming strategies. Biomed. Pharmacother. 2024, 178, 117252. [Google Scholar] [CrossRef]
- Katsoulakis, E.; Wang, Q.; Wu, H.; Shahriyari, L.; Fletcher, R.; Liu, J.; Achenie, L.; Liu, H.; Jackson, P.; Xiao, Y.; et al. Digital twins for health: A scoping review. npj Digit. Med. 2024, 7, 77. [Google Scholar] [CrossRef]
- De Domenico, M.; Allegri, L.; Caldarelli, G.; d’Andrea, V.; Di Camillo, B.; Rocha, L.M.; Rozum, J.; Sbarbati, R.; Zambelli, F. Challenges and opportunities for digital twins in precision medicine from a complex systems perspective. npj Digit. Med. 2025, 8, 37. [Google Scholar] [CrossRef]
- Aghamiri, S.S.; Amin, R. The Potential Use of Digital Twin Technology for Advancing CAR-T Cell Therapy. Curr. Issues Mol. Biol. 2025, 47(5), 321. [Google Scholar] [CrossRef]
| Notation | Biological meaning |
|---|---|
| C | functional CAR-T cells |
| E | exhausted CAR-T cells |
| antigen-positive tumour cells | |
| tumour cells with reduced antigen expression | |
| S | chemoattractant governing directed migration |
| A | generalised soluble immunosuppressive factor |
| H | normalised microenvironmental hypoxia |
| diffusion coefficients of the cellular populations | |
| diffusion coefficients of the chemoattractant, the immunosuppressive factor, and the hypoxia variable | |
| baseline chemotactic sensitivity and its suppression by immunosuppression | |
| parameters of antigen-dependent CAR-T cell proliferation | |
| intensity of cytotoxic interaction and associated functional loss of CAR-T cells | |
| parameters of exhaustion and partial recovery of CAR-T cells | |
| half-saturation parameter of the hypoxic effect | |
| natural death rates of functional and exhausted CAR-T cells | |
| tumour growth parameters | |
| natural death rates of antigen-positive and antigen-reduced tumour cells | |
| parameters of antigen escape and reverse phenotypic switching | |
| production of the chemoattractant, immunosuppressive factors, and hypoxia accumulation | |
| decay of the microenvironmental fields |
| Parameter | MAE | RMSE | ||
|---|---|---|---|---|
| 0.569 | 0.100 | 0.130 | 0.182 | |
| 0.585 | 0.062 | 0.076 | 0.388 | |
| 0.563 | 0.095 | 0.126 | 0.230 | |
| 0.300 | 0.154 | 0.192 | -0.027 | |
| 0.858 | 0.021 | 0.031 | 0.831 | |
| -0.023 | 0.068 | 0.100 | -0.357 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




