Submitted:
08 April 2026
Posted:
09 April 2026
You are already at the latest version
Abstract
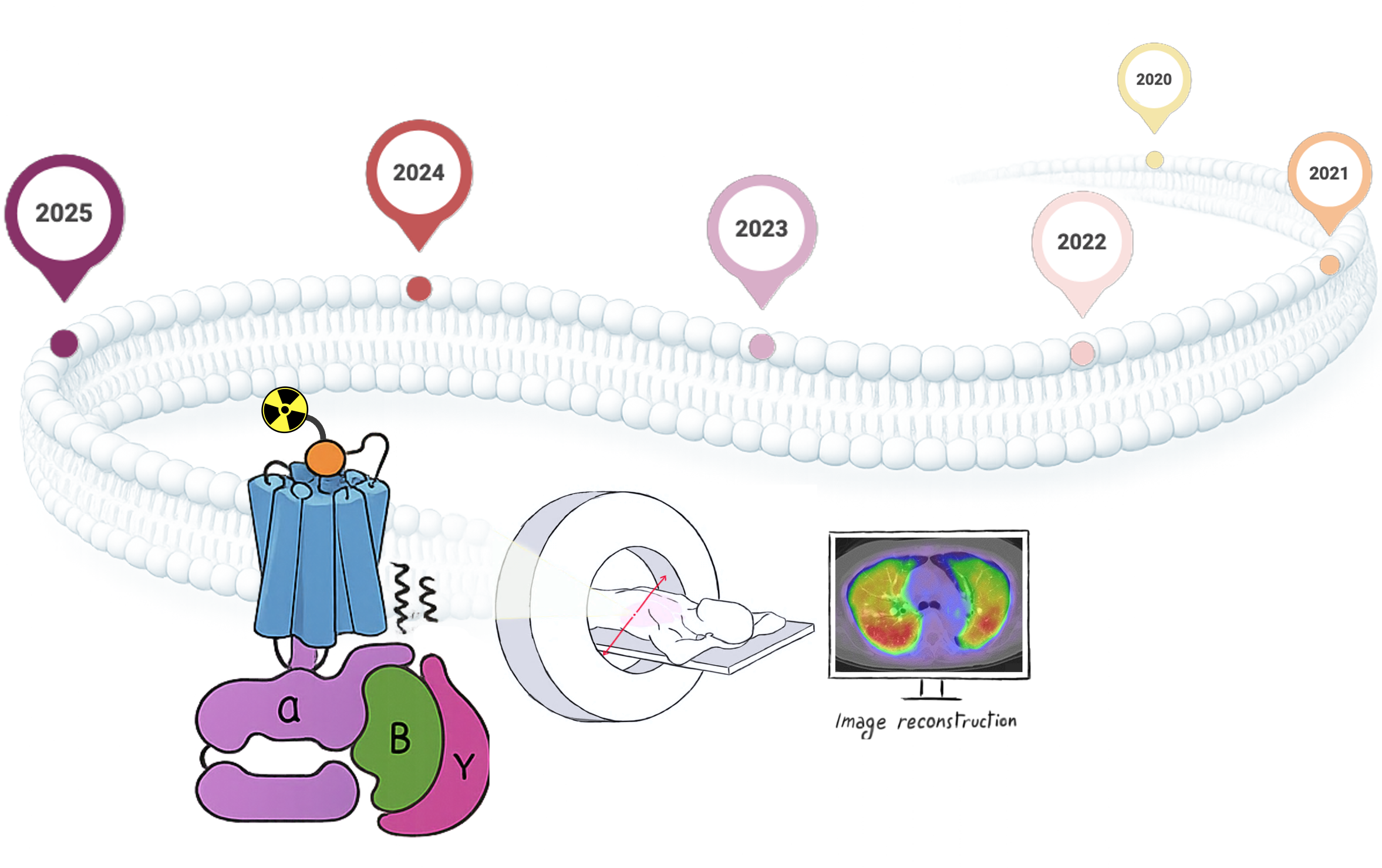
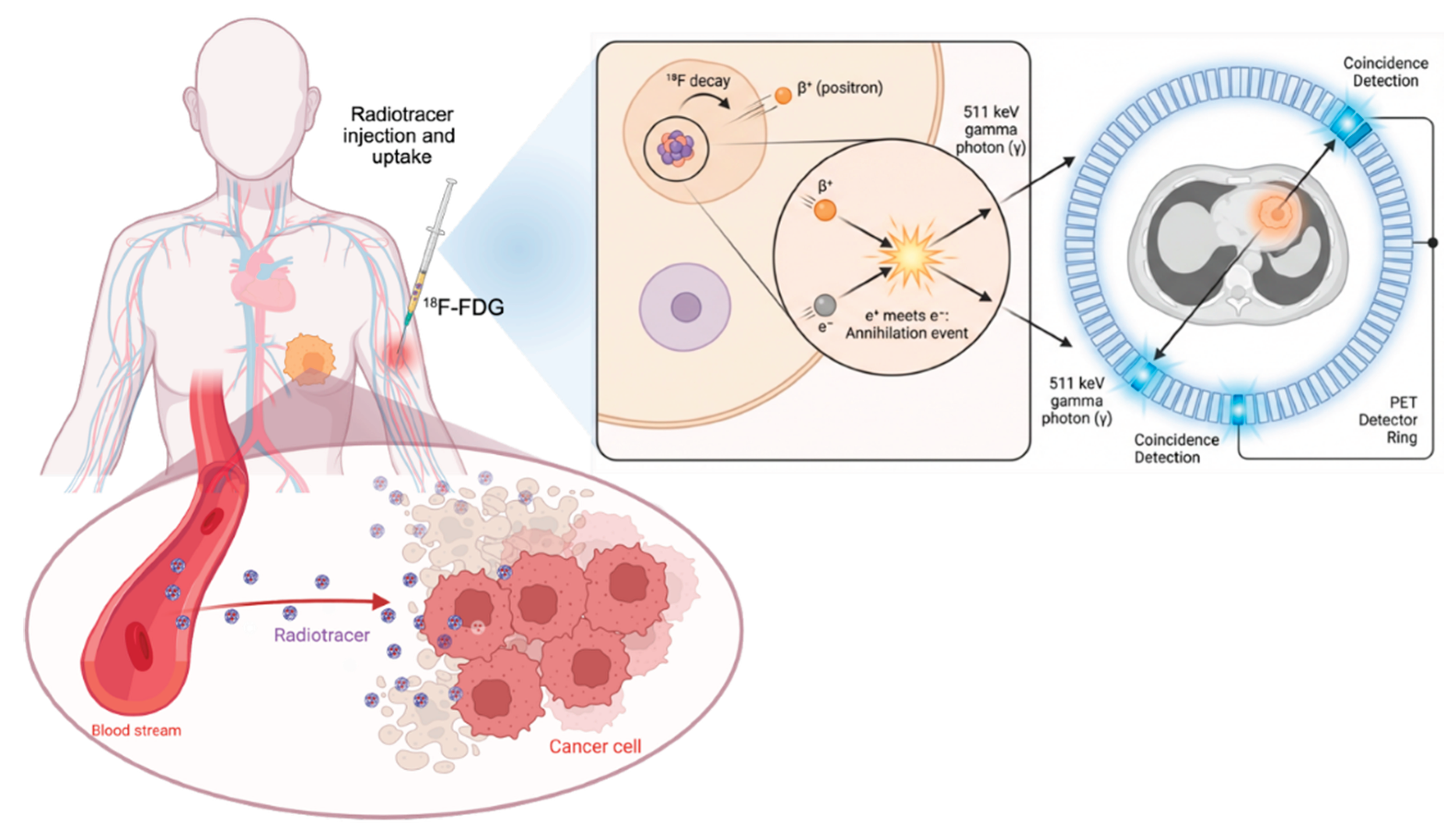
Keywords:
1. Introduction
| Isotope | Half-life | Mode of decay | Main application area | Clinical examples of PET pharmaceuticals |
|---|---|---|---|---|
| 18F | 109.8 min | β+ (97%), EC (3%) |
Oncology Neurology |
[18F]FDG – reference tracer; gold standard in oncology (solid tumors, lymphomas); also used in infection/inflammation [9]. [18F]FLT – investigational tracer for cellular proliferation and early therapy response [10]. [18F]FET – widely used for brain tumors (gliomas) [11]. [18F]NaF – bone imaging [12], and vascular disorders [13]. [18F]PSMA and [18F]fluciclovine – prostate cancer [14]. [18F]fluoroestradiol – estrogen receptor (ER)-positive breast cancer [15]. [18F]FDOPA –dopaminergic nerve terminals in the striatum of patients with suspected Parkinsonian syndrome [16]. [18F]flortaucipir –aggregated tau neurofibrillary tangles in Alzheimer’s disease [17]. |
| 11C | 20.4 min | β+ (100%) | Neurology Cardiology |
[11C]raclopride - striatal dopamine binding in autism spectrum disorder [18]. [11C]PIB - β-amyloid deposits in Alzheimer’s disease [19]. [11C]acetate – measures oxidative metabolism and myocardial perfusion; also applied in oncology (e.g., bladder carcinoma, brain tumors) [20]. |
| 13N | 10 min | β+ (100%) | Cardiology | [13N]NH₃ – myocardial perfusion imaging (rest/stress) for ischemic heart disease [21]. |
| 15O | 2 min | β+ (100%) | Cardiology & Neurology (perfusion studies) |
[15O]H2O - gold standard tracer for quantitative myocardial and cerebral perfusion imaging [22]. |
| 124I | 4.2 d | β+ (23%), EC (77%) |
Oncology | [124I]NaI – PET surrogate for 131I therapy, used for thyroid imaging, diagnosis, and dosimetry in differentiated thyroid cancer and hyperthyroidism [23,24]. |
| 44Sc | 4.0 h | β+ (94%), EC (6%) |
Oncology | [44Sc]ScDOTATOC/-TATE - NETs [25]. [44Sc]ScPSMA-617 – prostate carcinoma [26]. |
| 64Cu | 12.7 h | β+ (17%), EC (44%), β- (39%) |
Oncology | [64Cu]CuDOTATATE - NETs [27]. [64Cu]CuPSMA-617– prostate carcinoma [28]. |
| 68Ga | 67.7 min | β+ (89%), EC (11%) |
Oncology |
[68Ga]GaDOTA-TATE/-TOC/-NOC - NETs [29]. [68Ga]GaPSMA-11- prostate cancer [30]. |
| 82Rb | 1.3 min | β+ (95%), EC (5%) |
Cardiology | [82Rb]RbCl – myocardial perfusion imaging [31]. |
| 86Y | 14.7 h | β+ (32%), EC (68%) |
Oncology | [86Y]Y-DOTA-Phe¹-Tyr³-Octreotide – PET surrogate for 90Y therapy, enabling dosimetry and biodistribution assessment in NETs [32]. |
| 89Zr | 78.4 h | β+ (23%), EC (77%) |
Oncology | [89Zr]trastuzumab - HER2-positive breast cancer [33]. [89Zr]Zr-DFO-onartuzumab - PET surrogate for predicting response to Met-targeted radioligand therapy in pancreatic cancer [34]. |
2. Landmark Patents (2020–2025): Classification by Disease Area and GPCR Biomarker
2.1. Oncology
2.1.1. Somatostatin Receptors (SSTRs)
2.1.2. Cholecystokinin 2 Receptor (CCK2R)
2.1.3. Dual-Receptor Targeting Strategy: SSTR2 and CCK2R
2.1.4. CXC Chemokine Receptor Type 4 (CXCR4)
2.1.5. Neurokinin 1 Receptor (NK1R)
2.1.6. Melanocortin Type 2 Receptor (MC2R)
2.1.7. Kisspeptin Receptor (KISS1R)
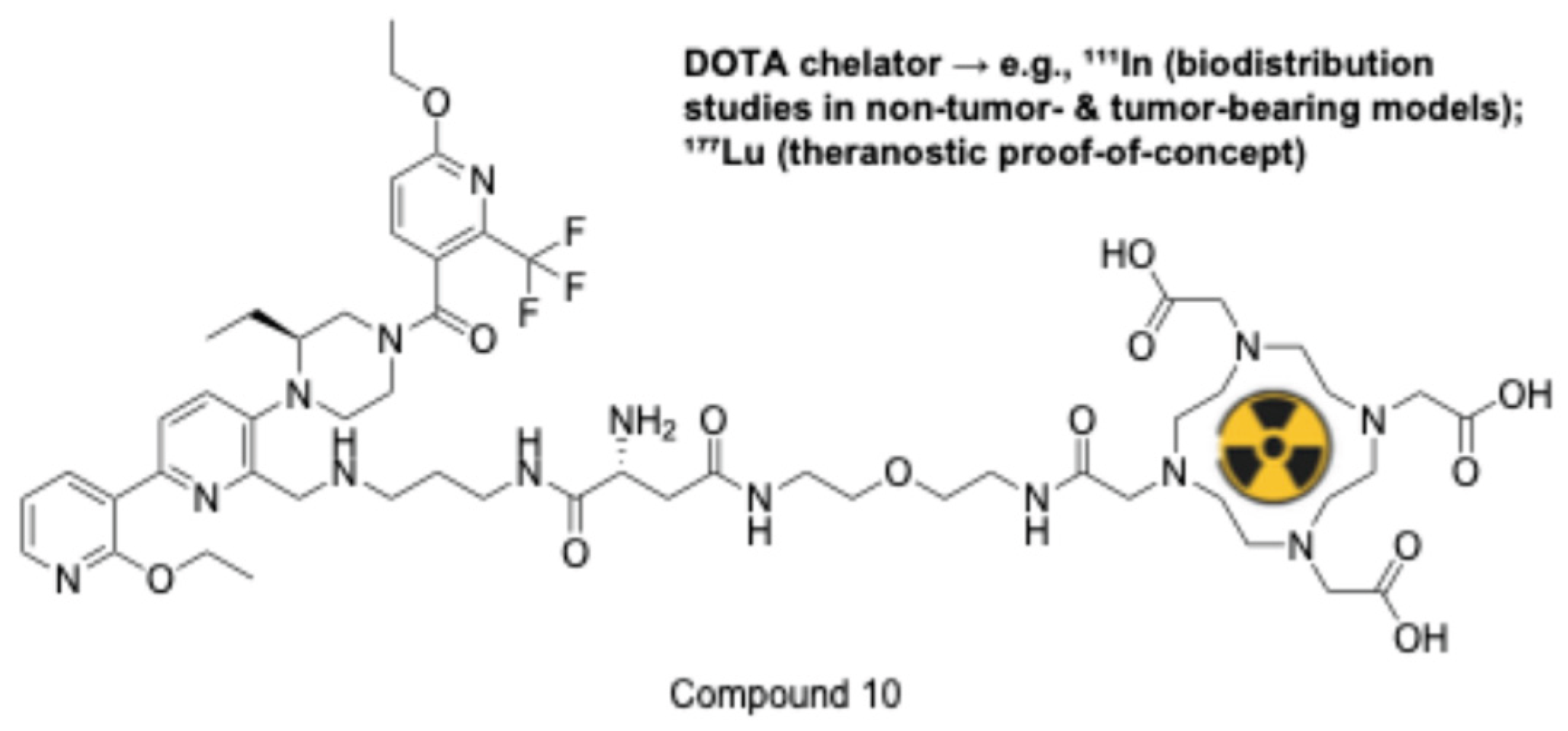
2.1.8. Neurotensin Receptor 1 (NTSR1)
2.2. CNS Diseases
2.2.1. Metabotropic glutamate receptors 2 and 3 (mGluR2 & mGluR3)
2.2.2. Metabotropic Glutamate Receptor 2 (mGluR2)
2.2.3. Metabotropic Glutamate Receptor 4 (mGluR4)
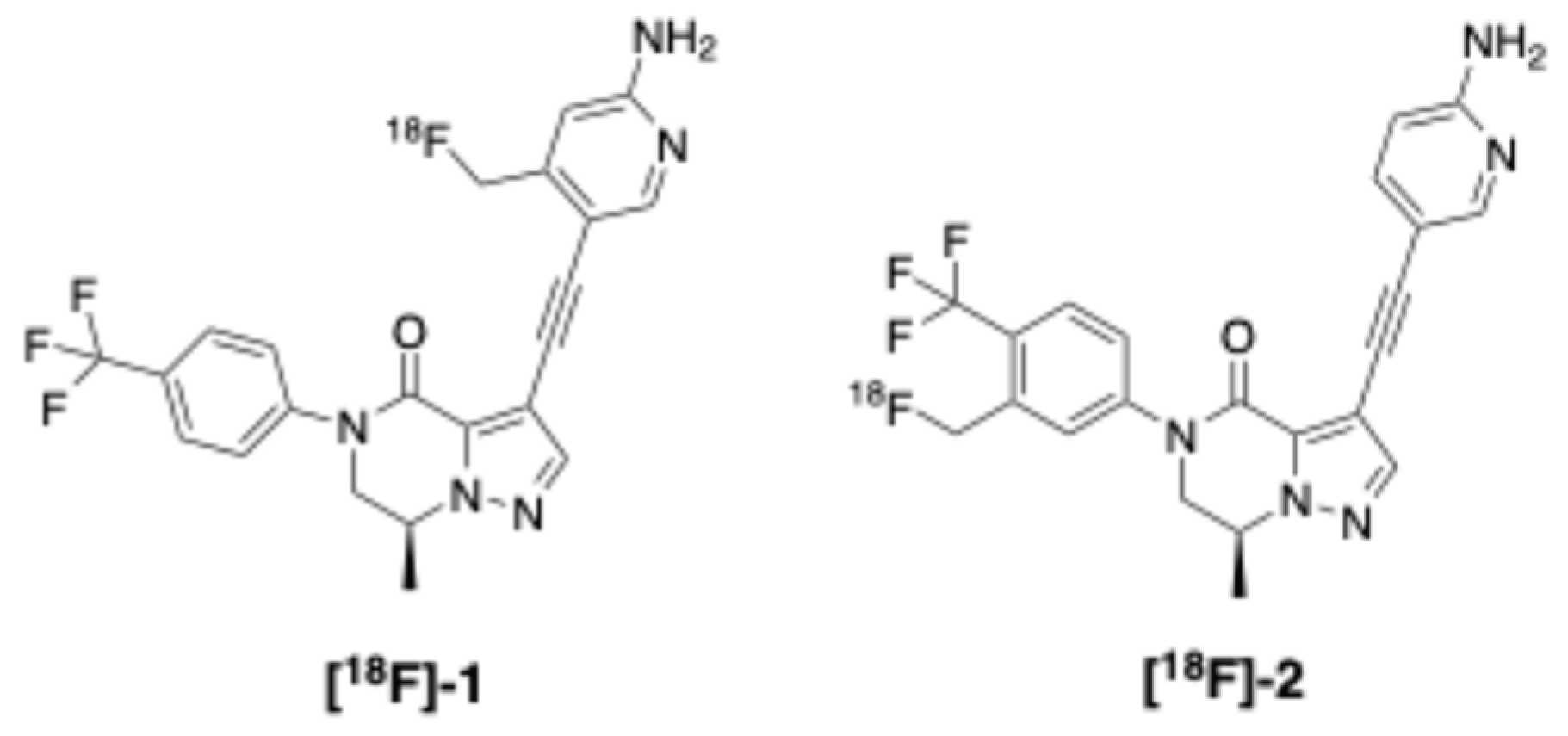
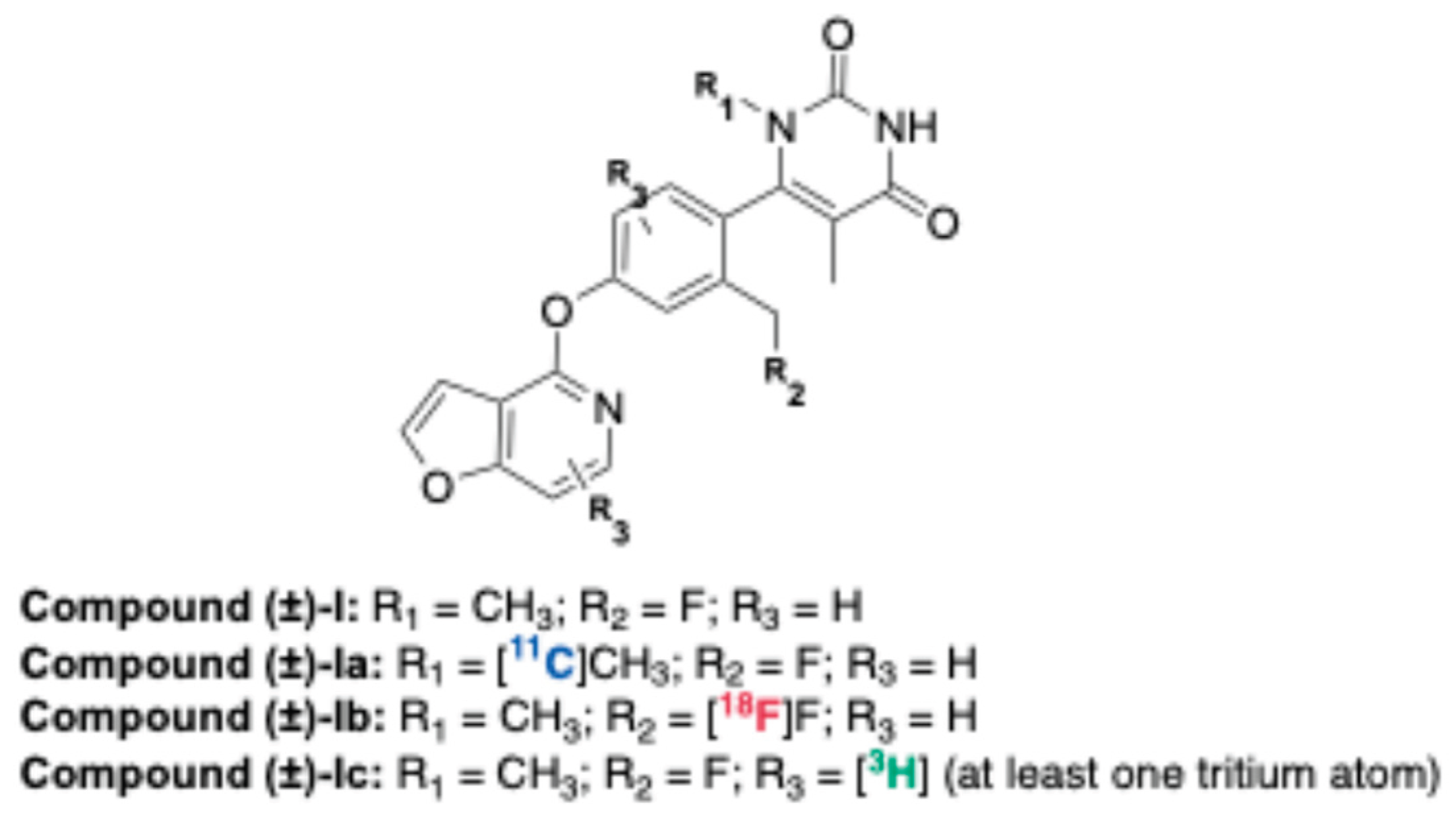
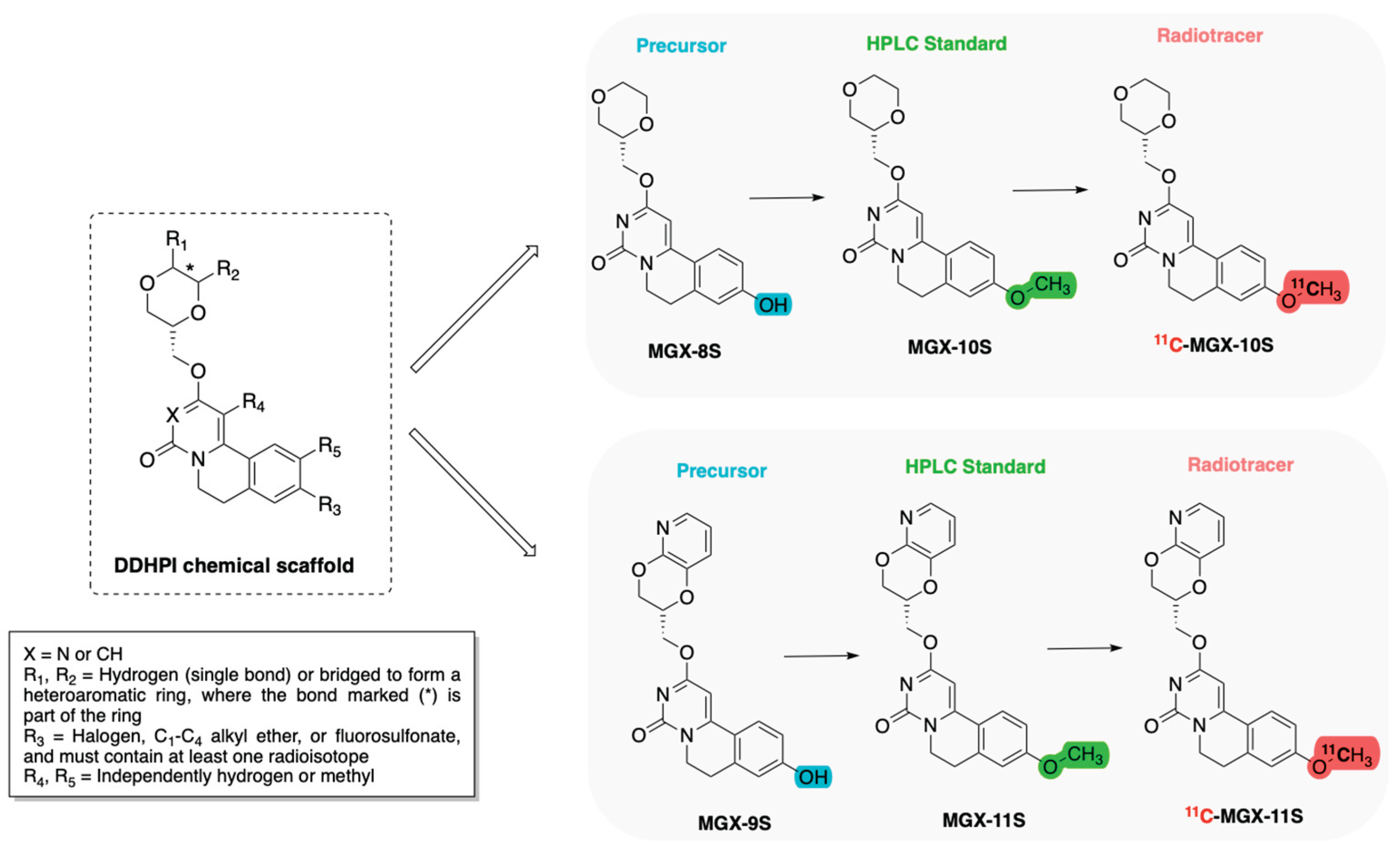
2.2.4. Dopamine D1 Receptor (D1R)
2.3. Inflammatory Diseases
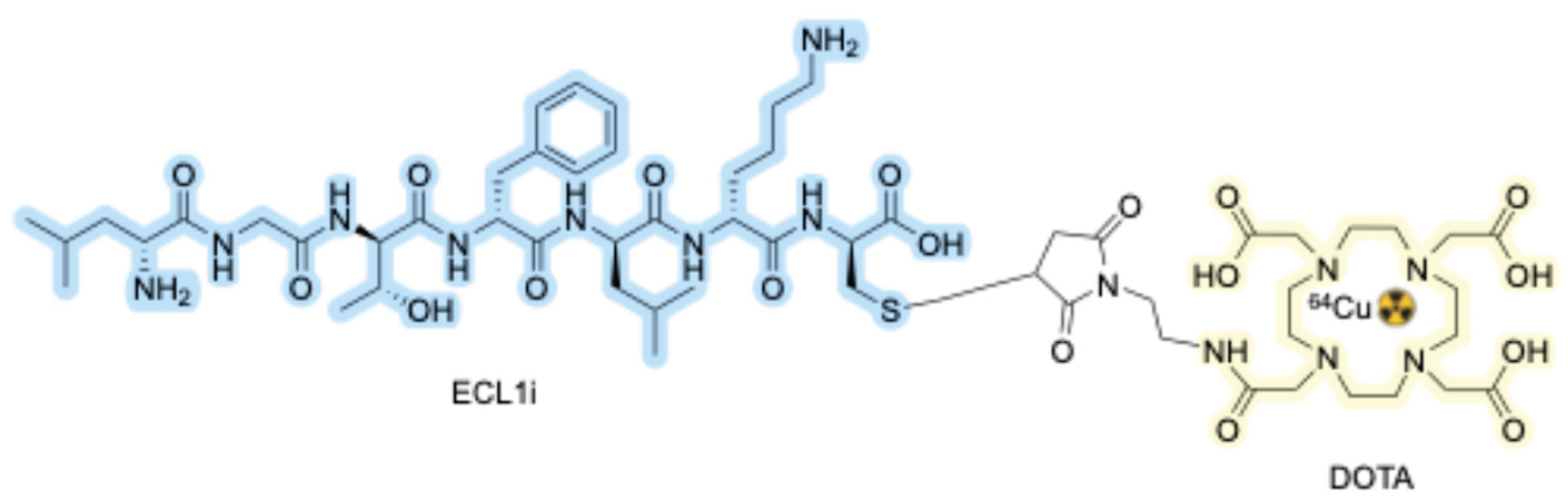
2.3.1. CC Chemokine Receptor 2 (CCR2)
2.3.2. CXC Chemokine Receptor 2 (CXCR2)
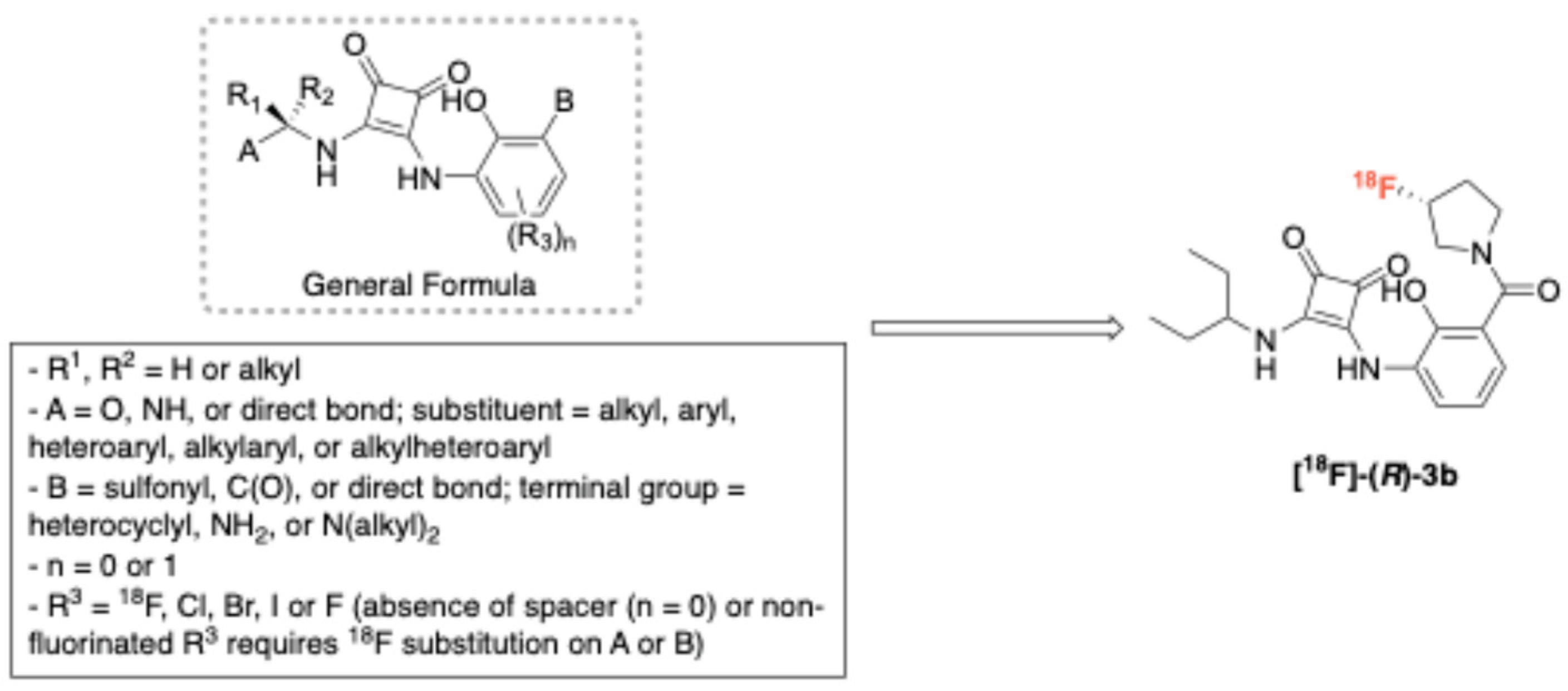
2.3.3. CXC Chemokine Receptor Type 4 (CXCR4)
2.3.4. G protein-Coupled Receptor 84 (GPR84)
3. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ametamey, S.M.; Honer, M.; Schubiger, P.A. Molecular Imaging with PET. Chem. Rev. 2008, 108, 1501–1516. [Google Scholar] [CrossRef]
- Phelps, M.E. Positron Emission Tomography Provides Molecular Imaging of Biological Processes. Proc. Natl. Acad. Sci. 2000, 97, 9226–9233. [Google Scholar] [CrossRef] [PubMed]
- Miller, P.W.; Long, N.J.; Vilar, R.; Gee, A.D. Synthesis of 11 C,18 F,15 O, and13 N Radiolabels for Positron Emission Tomography. Angew. Chem. Int. Ed. 2008, 47, 8998–9033. [Google Scholar] [CrossRef] [PubMed]
- Halder, R.; Ritter, T. 18 F-Fluorination: Challenge and Opportunity for Organic Chemists. J. Org. Chem. 2021, 86, 13873–13884. [Google Scholar] [CrossRef]
- Coenen, H.H.; Elsinga, P.H.; Iwata, R.; Kilbourn, M.R.; Pillai, M.R.A.; Rajan, M.G.R.; Wagner, H.N.; Zaknun, J.J. Fluorine-18 Radiopharmaceuticals beyond [18F]FDG for Use in Oncology and Neurosciences. Nucl. Med. Biol. 2010, 37, 727–740. [Google Scholar] [CrossRef]
- Goud, N.S.; Bhattacharya, A.; Joshi, R.K.; Nagaraj, C.; Bharath, R.D.; Kumar, P. Carbon-11: Radiochemistry and Target-Based PET Molecular Imaging Applications in Oncology, Cardiology, and Neurology. J. Med. Chem. 2021, 64, 1223–1259. [Google Scholar] [CrossRef]
- Rong, J.; Haider, A.; Jeppesen, T.E.; Josephson, L.; Liang, S.H. Radiochemistry for Positron Emission Tomography. Nat. Commun. 2023, 14, 3257. [Google Scholar] [CrossRef]
- Yordanova, A.; Eppard, E.; Kürpig, S.; Bundschuh, R.; Schönberger, S.; Gonzalez-Carmona, M.; Feldmann, G.; Ahmadzadehfar, H.; Essler, M. Theranostics in Nuclear Medicine Practice. OncoTargets Ther. 2017, Volume 10, 4821–4828. [Google Scholar] [CrossRef] [PubMed]
- Abikhzer, G.; Treglia, G.; Pelletier-Galarneau, M.; Buscombe, J.; Chiti, A.; Dibble, E.H.; Glaudemans, A.W.J.M.; Palestro, C.J.; Sathekge, M.; Signore, A.; et al. EANM/SNMMI Guideline/Procedure Standard for [18F]FDG Hybrid PET Use in Infection and Inflammation in Adults v2.0. Eur. J. Nucl. Med. Mol. Imaging 2025, 52, 510–538. [Google Scholar] [CrossRef]
- Been, L.B.; Suurmeijer, A.J.H.; Cobben, D.C.P.; Jager, P.L.; Hoekstra, H.J.; Elsinga, P.H. [18F]FLT-PET in Oncology: Current Status and Opportunities. Eur. J. Nucl. Med. Mol. Imaging 2004, 31, 1659–1672. [Google Scholar] [CrossRef] [PubMed]
- Dunet, V.; Rossier, C.; Buck, A.; Stupp, R.; Prior, J.O. Performance of18 F-Fluoro-Ethyl-Tyrosine (18 F-FET) PET for the Differential Diagnosis of Primary Brain Tumor: A Systematic Review and Metaanalysis. J. Nucl. Med. 2012, 53, 207–214. [Google Scholar] [CrossRef]
- Araz, M.; Aras, G.; Küçük, Ö.N. The Role of 18F–NaF PET/CT in Metastatic Bone Disease. J. Bone Oncol. 2015, 4, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Derlin, T.; Richter, U.; Bannas, P.; Begemann, P.; Buchert, R.; Mester, J.; Klutmann, S. Feasibility of 18F-Sodium Fluoride PET/CT for Imaging of Atherosclerotic Plaque. J. Nucl. Med. 2010, 51, 862–865. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.D.; Jethwa, K.R.; Ost, P.; Williams, S.; Kwon, E.D.; Lowe, V.J.; Davis, B.J. Prostate Cancer–Specific PET Radiotracers: A Review on the Clinical Utility in Recurrent Disease. Pract. Radiat. Oncol. 2018, 8, 28–39. [Google Scholar] [CrossRef]
- Parihar, A.S.; Vaz, S.; Sutcliffe, S.; Pant, N.; Schoones, J.W.; Ulaner, G.A. 18 F-Fluoroestradiol PET/CT for Predicting Benefit from Endocrine Therapy in Patients with Estrogen Receptor–Positive Breast Cancer: A Systematic Review and Metaanalysis. J. Nucl. Med. 2025, 66, 692–699. [Google Scholar] [CrossRef]
- Dhawan, V.; Niethammer, M.H.; Lesser, M.L.; Pappas, K.N.; Hellman, M.; Fitzpatrick, T.M.; Bjelke, D.; Singh, J.; Quatarolo, L.M.; Choi, Y.Y.; et al. Prospective F-18 FDOPA PET Imaging Study in Human PD. Nucl. Med. Mol. Imaging 2022, 56, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Mattay, V.S.; Fotenos, A.F.; Ganley, C.J.; Marzella, L. Brain Tau Imaging: Food and Drug Administration Approval of18 F-Flortaucipir Injection. J. Nucl. Med. 2020, 61, 1411–1412. [Google Scholar] [CrossRef]
- Zürcher, N.R.; Walsh, E.C.; Phillips, R.D.; Cernasov, P.M.; Tseng, C.-E.J.; Dharanikota, A.; Smith, E.; Li, Z.; Kinard, J.L.; Bizzell, J.C.; et al. A Simultaneous [11C]Raclopride Positron Emission Tomography and Functional Magnetic Resonance Imaging Investigation of Striatal Dopamine Binding in Autism. Transl. Psychiatry 2021, 11, 33. [Google Scholar] [CrossRef]
- Youn, Y.C.; Jang, J.-W.; Han, S.-H.; Kim, H.; Seok, J.W.; Byun, J.S.; Park, K.-Y.; An, S.S.; Chun, I.K.; Kim, S. 11C-PIB PET Imaging Reveals That Amyloid Deposition in Cases with Early-Onset Alzheimer’s Disease in the Absence of Known Mutations Retains Higher Levels of PIB in the Basal Ganglia. Clin. Interv. Aging 2017, Volume 12, 1041–1048. [Google Scholar] [CrossRef]
- Grassi, I.; Nanni, C.; Allegri, V.; Morigi, J.J.; Montini, G.C.; Castellucci, P.; Fanti, S. The Clinical Use of PET with 11C-Acetate.
- Singh, H.; Bhasin, D.; Gunasekaran, V.; Subedi, U.; Mittal, B.R. Potential Role of 13N-NH3 Cardiac PET in Monitoring Treatment Response in Patients with Microvascular Angina. Indian J. Nucl. Med. 2025, 40, 103–105. [Google Scholar] [CrossRef]
- Slart, R.H.J.A.; Martinez-Lucio, T.S.; Boersma, H.H.; Borra, R.H.; Cornelissen, B.; Dierckx, R.A.J.O.; Dobrolinska, M.; Doorduin, J.; Erba, P.A.; Glaudemans, A.W.J.M.; et al. [15O]H2O PET: Potential or Essential for Molecular Imaging? Semin. Nucl. Med. 2024, 54, 761–773. [Google Scholar] [CrossRef]
- Hobbs, R.F.; Wahl, R.L.; Lodge, M.A.; Javadi, M.S.; Cho, S.Y.; Chien, D.T.; Ewertz, M.E.; Esaias, C.E.; Ladenson, P.W.; Sgouros, G. 124 I PET-Based 3D-RD Dosimetry for a Pediatric Thyroid Cancer Patient: Real-Time Treatment Planning and Methodologic Comparison. J. Nucl. Med. 2009, 50, 1844–1847. [Google Scholar] [CrossRef]
- Sgouros, G.; Hobbs, R.F.; Atkins, F.B.; Van Nostrand, D.; Ladenson, P.W.; Wahl, R.L. Three-Dimensional Radiobiological Dosimetry (3D-RD) with 124I PET for 131I Therapy of Thyroid Cancer. Eur. J. Nucl. Med. Mol. Imaging 2011, 38, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Pauwels, E.; Cleeren, F.; Bormans, G.; Deroose, C.M. Somatostatin Receptor PET Ligands - the next Generation for Clinical Practice.
- Khawar, A.; Eppard, E.; Sinnes, J.P.; Roesch, F.; Ahmadzadehfar, H.; Kürpig, S.; Meisenheimer, M.; Gaertner, F.C.; Essler, M.; Bundschuh, R.A. [44Sc]Sc-PSMA-617 Biodistribution and Dosimetry in Patients With Metastatic Castration-Resistant Prostate Carcinoma. Clin. Nucl. Med. 2018, 43, 323–330. [Google Scholar] [CrossRef]
- Krasnovskaya, O.O.; Abramchuck, D.; Erofeev, A.; Gorelkin, P.; Kuznetsov, A.; Shemukhin, A.; Beloglazkina, E.K. Recent Advances in 64Cu/67Cu-Based Radiopharmaceuticals. Int. J. Mol. Sci. 2023, 24, 9154. [Google Scholar] [CrossRef] [PubMed]
- Cui, C.; Hanyu, M.; Hatori, A.; Zhang, Y.; Xie, L.; Ohya, T.; Fukada, M.; Suzuki, H.; Nagatsu, K.; Jiang, C.; et al. Synthesis and Evaluation of [64Cu]PSMA-617 Targeted for Prostate-Specific Membrane Antigen in Prostate Cancer.
- Mojtahedi, A.; Thamake, S.; Tworowska, I.; Ranganathan, D.; Delpassand, E.S. The Value of 68Ga-DOTATATE PET/CT in Diagnosis and Management of Neuroendocrine Tumors Compared to Current FDA Approved Imaging Modalities: A Review of Literature.
- Carlucci, G.; Ippisch, R.; Slavik, R.; Mishoe, A.; Blecha, J.; Zhu, S. 68Ga-PSMA-11 NDA Approval: A Novel and Successful Academic Partnership. J. Nucl. Med. 2021, 62, 149–155. [Google Scholar] [CrossRef]
- Harnett et al. Clinical Performance of Rb-82 Myocardial Perfusion PET and Tc-99m-Based SPECT in Patients with Extreme Obesity.
- Helisch, A.; Förster, GregorJ.; Reber, H.; Buchholz, H.-G.; Arnold, R.; Göke, B.; Weber, MatthiasM.; Wiedenmann, B.; Pauwels, S.; Haus, U.; et al. Pre-Therapeutic Dosimetry and Biodistribution of 86Y-DOTA-Phe1-Tyr3-Octreotide versus 111In-Pentetreotide in Patients with Advanced Neuroendocrine Tumours. Eur. J. Nucl. Med. Mol. Imaging 2004, 31. [Google Scholar] [CrossRef]
- Laforest, R.; Lapi, S.E.; Oyama, R.; Bose, R.; Tabchy, A.; Marquez-Nostra, B.V.; Burkemper, J.; Wright, B.D.; Frye, J.; Frye, S.; et al. [89Zr]Trastuzumab: Evaluation of Radiation Dosimetry, Safety, and Optimal Imaging Parameters in Women with HER2-Positive Breast Cancer. Mol. Imaging Biol. 2016, 18, 952–959. [Google Scholar] [CrossRef]
- Escorcia, F.E.; Houghton, J.L.; Abdel-Atti, D.; Pereira, P.R.; Cho, A.; Gutsche, N.T.; Baidoo, K.E.; Lewis, J.S. ImmunoPET Predicts Response to Met-Targeted Radioligand Therapy in Models of Pancreatic Cancer Resistant to Met Kinase Inhibitors. Theranostics 2020, 10, 151–165. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Heston, T.F.; Kasi, A. PET Scanning. StatPearls [Internet]. StatPearls Publishing: Treasure Island (FL), 2026. Available online: https://www.ncbi.nlm.nih.gov/books/NBK559089/.
- Piel, M.; Vernaleken, I.; Rösch, F. Positron Emission Tomography in CNS Drug Discovery and Drug Monitoring. J. Med. Chem. 2014, 57, 9232–9258. [Google Scholar] [CrossRef]
- Hauser, A.S.; Attwood, M.M.; Rask-Andersen, M.; Schiöth, H.B.; Gloriam, D.E. Trends in GPCR Drug Discovery: New Agents, Targets and Indications. Nat. Rev. Drug Discov. 2017, 16, 829–842. [Google Scholar] [CrossRef]
- Whalen, E.J.; Rajagopal, S.; Lefkowitz, R.J. Therapeutic Potential of β-Arrestin- and G Protein-Biased Agonists. Trends Mol. Med. 2011, 17, 126–139. [Google Scholar] [CrossRef]
- Sriram, K.; Insel, P.A. G Protein-Coupled Receptors as Targets for Approved Drugs: How Many Targets and How Many Drugs? Mol. Pharmacol. 2018, 93, 251–258. [Google Scholar] [CrossRef]
- Krishna Deepak, R.N.V.; Verma, R.K.; Hartono, Y.D.; Yew, W.S.; Fan, H. Recent Advances in Structure, Function, and Pharmacology of Class A Lipid GPCRs: Opportunities and Challenges for Drug Discovery. Pharmaceuticals 2021, 15, 12. [Google Scholar] [CrossRef]
- García-Nafría, J.; Tate, C.G. Structure Determination of GPCRs: Cryo-EM Compared with X-Ray Crystallography. Biochem. Soc. Trans. 2021, 49, 2345–2355. [Google Scholar] [CrossRef]
- Insel, P.A.; Sriram, K.; Gorr, M.W.; Wiley, S.Z.; Michkov, A.; Salmerón, C.; Chinn, A.M. GPCRomics: An Approach to Discover GPCR Drug Targets. Trends Pharmacol. Sci. 2019, 40, 378–387. [Google Scholar] [CrossRef] [PubMed]
- Feng, C.; Ooms, J.F.; Danen, E.H.J.; Heitman, L.H. GPCR-G Protein Signalling and Its Mutational Landscape in Cancer—Driver or Passenger. Br. J. Pharmacol. 2025, 182, 3975–3989. [Google Scholar] [CrossRef]
- O’Hayre, M.; Degese, M.S.; Gutkind, J.S. Novel Insights into G Protein and G Protein-Coupled Receptor Signaling in Cancer. Curr. Opin. Cell Biol. 2014, 27, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Insel, P.A.; Sriram, K.; Wiley, S.Z.; Wilderman, A.; Katakia, T.; McCann, T.; Yokouchi, H.; Zhang, L.; Corriden, R.; Liu, D.; et al. GPCRomics: GPCR Expression in Cancer Cells and Tumors Identifies New, Potential Biomarkers and Therapeutic Targets. Front. Pharmacol. 2018, 9, 431. [Google Scholar] [CrossRef]
- Møller, L.N.; Stidsen, C.E.; Hartmann, B.; Holst, J.J. Somatostatin Receptors. Biochim. Biophys. Acta BBA - Biomembr. 2003, 1616, 1–84. [Google Scholar] [CrossRef]
- Theodoropoulou, M.; Stalla, G.K. Somatostatin Receptors: From Signaling to Clinical Practice. Front. Neuroendocrinol. 2013, 34, 228–252. [Google Scholar] [CrossRef]
- Mikołajczak, R.; Maecke, H.R. Radiopharmaceuticals for Somatostatin Receptor Imaging. Nucl. Med. Rev. 2016, 19, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Kwekkeboom, D.; Krenning, E.P.; de Jong, M. Peptide Receptor Imaging and Therapy. [CrossRef]
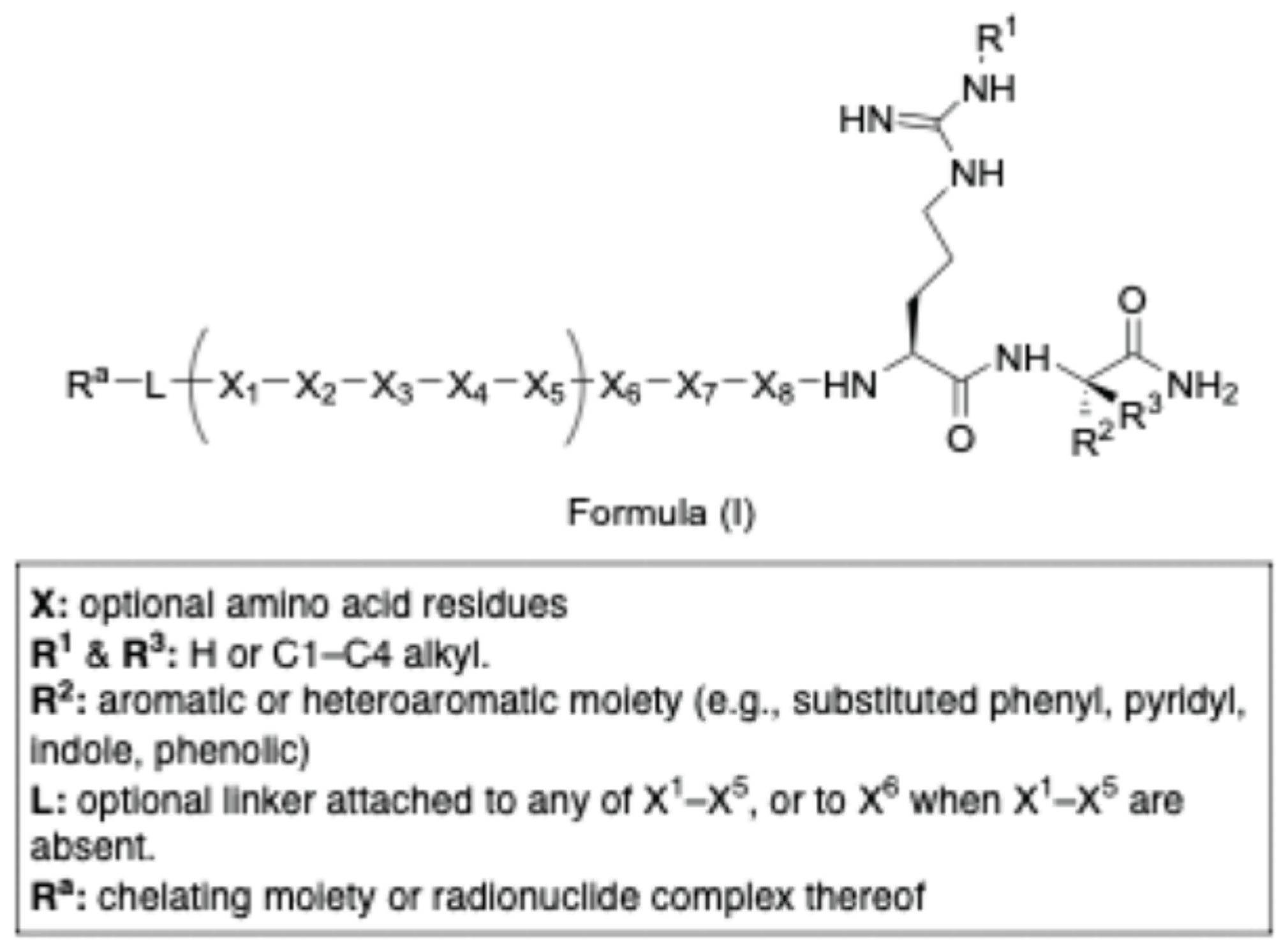
- Zhao, J.; Zhu, Y.; Chen, M. Somatostatin Subtype-2 Receptor (SST2R) Targeted Therapeutics and Uses Thereof. US20240254106, (2024).
- Oron-Herman, M.; Kirmayer, D.; Lupp, A.; Schulz, S.; Kostenich, G.; Afargan, M. Expression Prevalence and Dynamics of GPCR Somatostatin Receptors 2 and 3 as Cancer Biomarkers beyond NET: A Paired Immunohistochemistry Approach. Sci. Rep. 2023, 13, 20857. [Google Scholar] [CrossRef]
- Afargan, M.; Blum, E.; Salitra, Y. Conformational Constrained Somatostatin Receptor 3 Peptide Ligands and Their Conjugates and Uses Thereof. US20240165282, (2024).
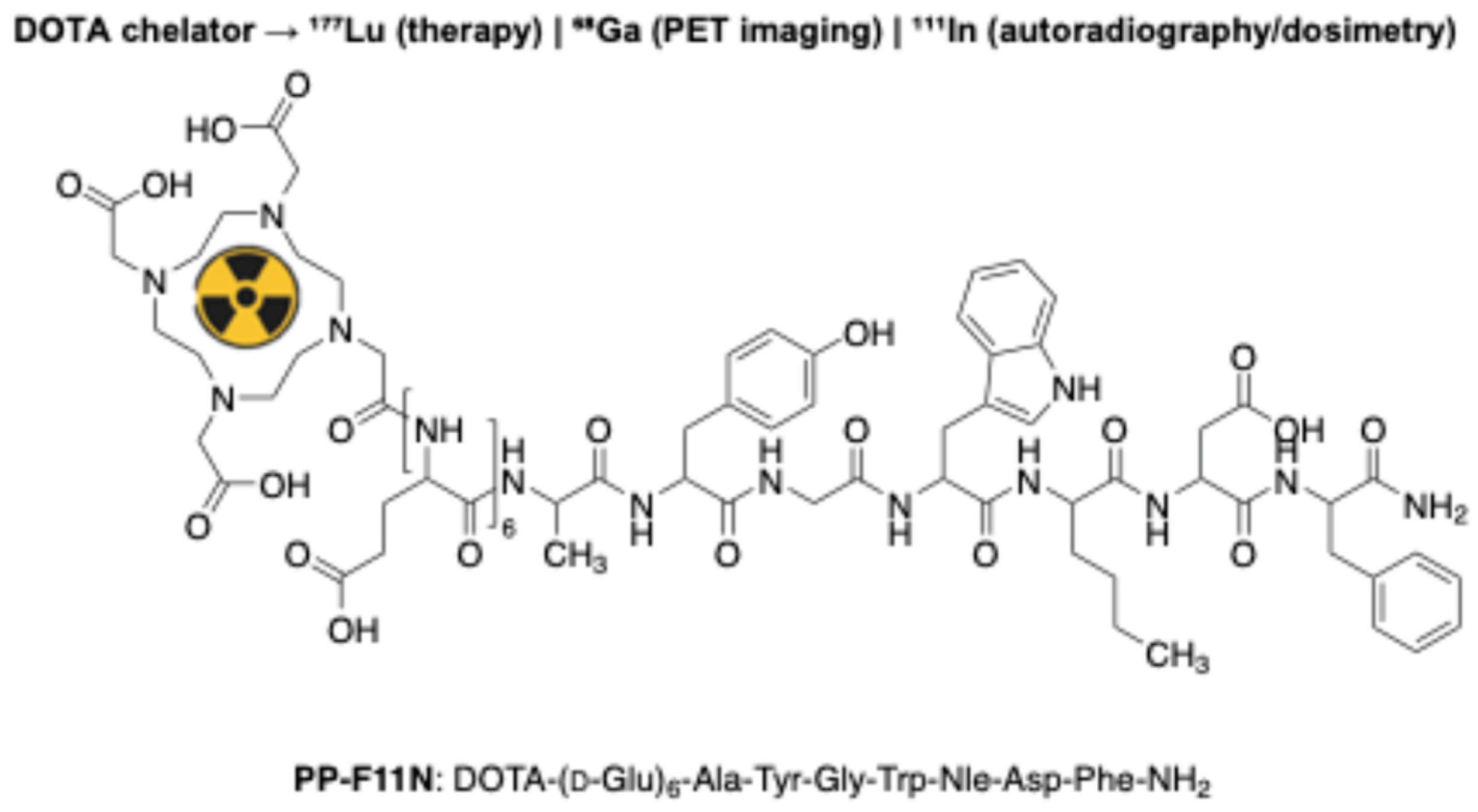
- Rottenburger, C.; Nicolas, G.P.; McDougall, L.; Kaul, F.; Cachovan, M.; Vija, A.H.; Schibli, R.; Geistlich, S.; Schumann, A.; Rau, T.; et al. Cholecystokinin 2 Receptor Agonist177 Lu-PP-F11N for Radionuclide Therapy of Medullary Thyroid Carcinoma: Results of the Lumed Phase 0a Study. J. Nucl. Med. 2020, 61, 520–526. [Google Scholar] [CrossRef] [PubMed]
- Pokorska-Bocci, A.; Attinger, A. Method for Predicting the Response of a Patient Diagnosed with Cancer to Treatment and/or Imaging with a Compound Targeting CCK2-R, and Compound for Use in Methods of Selectively Treating and/or Imaging Cancer. US20240401145, (2024).
- Behe, Martin; Schibli, Roger Mini-Gastrin Analogue, in Particular for Use in CCK2 Receptor Positive Tumour Diagnosis and/or Treatment. WO2015067473, (2015).
- Levy, F.; Attinger, A. Gallium-Labeled Gastrin Analogue and Use in a Method of Imaging CCKB-Receptor-Positive Tumors or Cancers. US20230270893, (2023).
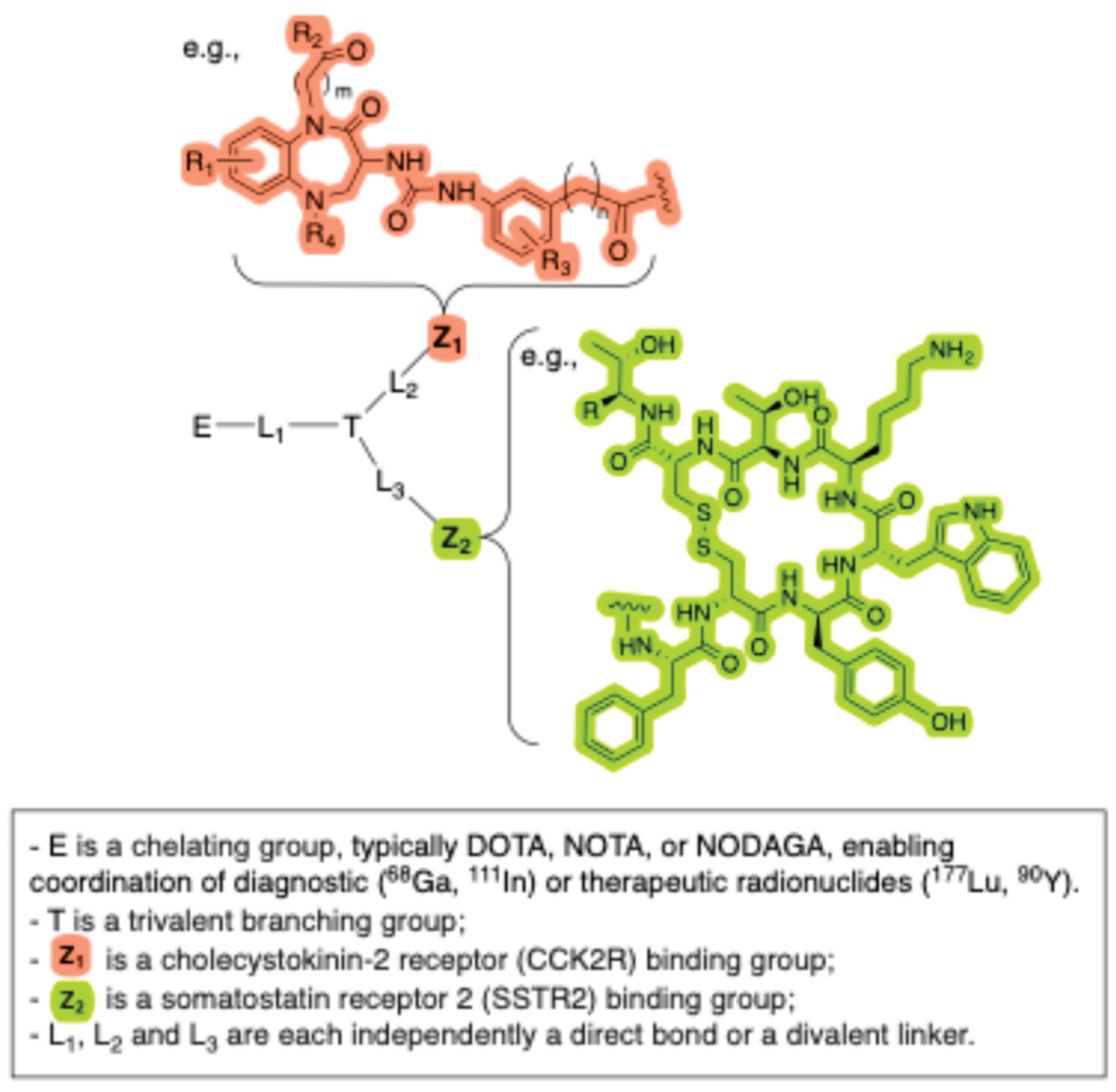
- Liu, F.; Hu, T. Dual Receptor Targeting Radioligands and Uses Thereof. WO2024083224, (2024).
- Zou, Y.-R.; Kottmann, A.H.; Kuroda, M.; Taniuchi, I.; Littman, D.R. Function of the Chemokine Receptor CXCR4 in Haematopoiesis and in Cerebellar Development. Nature 1998, 393, 595–599. [Google Scholar] [CrossRef]
- Jacobson, O.; Weiss, I.D. CXCR4 Chemokine Receptor Overview: Biology, Pathology and Applications in Imaging and Therapy. Theranostics 2013, 3, 1–2. [Google Scholar] [CrossRef]
- Yu, J.; Zhou, X.; Shen, L. CXCR4-Targeted Radiopharmaceuticals for the Imaging and Therapy of Malignant Tumors. Molecules 2023, 28, 4707. [Google Scholar] [CrossRef]
- Amodeo, P.; Vitale, R.; De Luca, S.; Scala, S.; Castello, G.; Siani, A. Cyclic Peptides Binding CXCR4 Receptor and Relative Medical and Diagnostic Uses. WO2011092575, (2011).
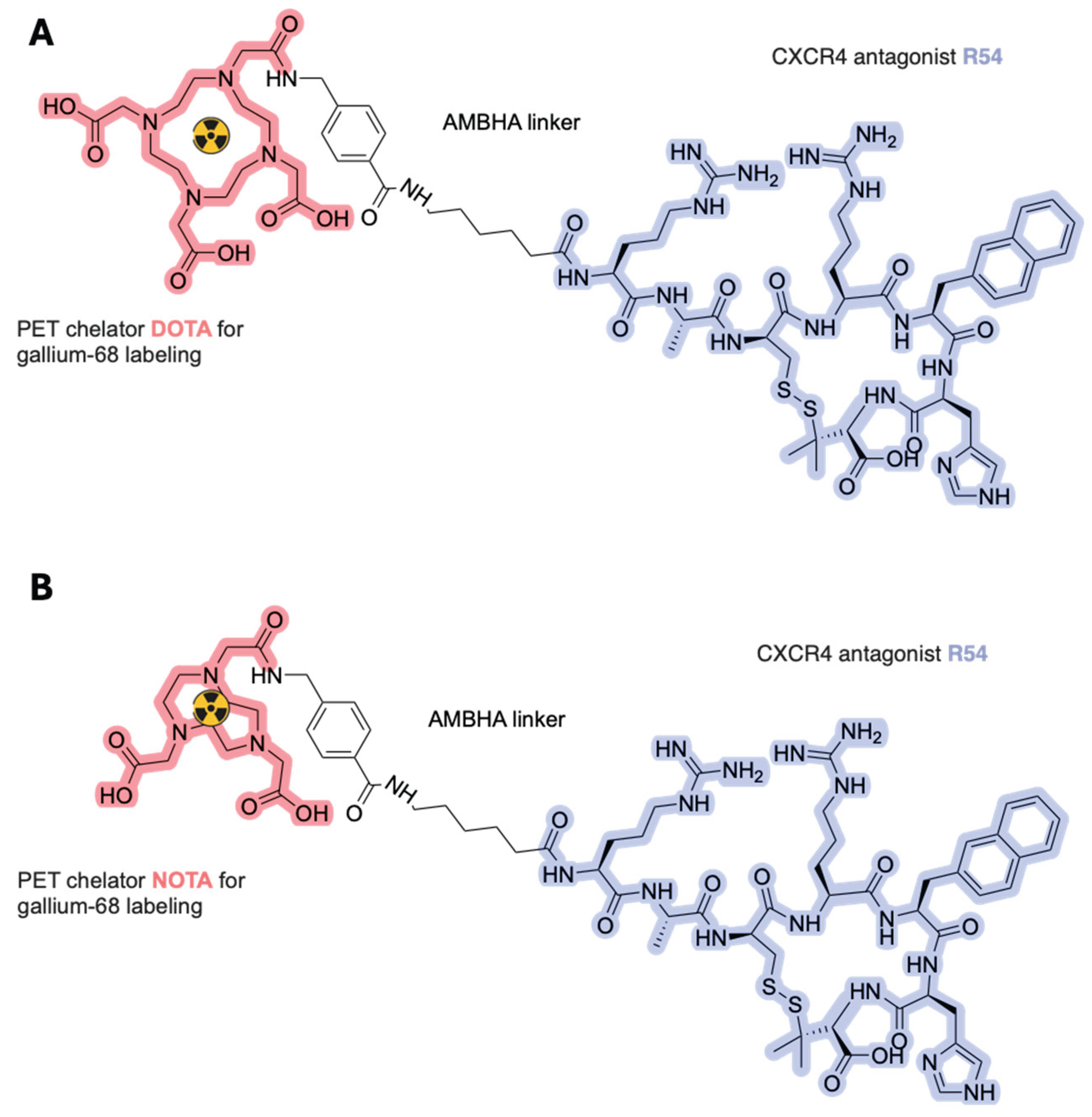
- Scala, S.; Trotta, A.M. Radiolabeled Peptides for Non-Invasive Diagnosis and Treatment of CXCR4 Expressing Tumors. WO2021130329, (2021).
- Di Maro, S.; Di Leva, F.S.; Trotta, A.M.; Brancaccio, D.; Portella, L.; Aurilio, M.; Tomassi, S.; Messere, A.; Sementa, D.; Lastoria, S.; et al. Structure–Activity Relationships and Biological Characterization of a Novel, Potent, and Serum Stable C-X-C Chemokine Receptor Type 4 (CXCR4) Antagonist. J. Med. Chem. 2017, 60, 9641–9652. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Recio, S.; Gascón, P. Biological and Pharmacological Aspects of the NK1-Receptor. BioMed Res. Int. 2015, 2015, 1–14. [Google Scholar] [CrossRef]
- Ebrahimi, S.; Mirzavi, F.; Aghaee-Bakhtiari, S.H.; Hashemy, S.I. SP/NK1R System Regulates Carcinogenesis in Prostate Cancer: Shedding Light on the Antitumoral Function of Aprepitant. Biochim. Biophys. Acta BBA - Mol. Cell Res. 2022, 1869, 119221. [Google Scholar] [CrossRef] [PubMed]
- Tattersall, F.D.; Rycrofti, W.; Francis, B.; PEARCEl, D.; Merchant’, K.; MacLEODl, A.M.; SWAINl, C.; BAKERl, R.; Ber’, E.; Metzger, J.; et al. Tachykinin NK1 Receptor Antagonists Act Centrally to Inhibit Ernesis Induced by the Chemotherapeutic Agent Cisplatin in Ferrets.
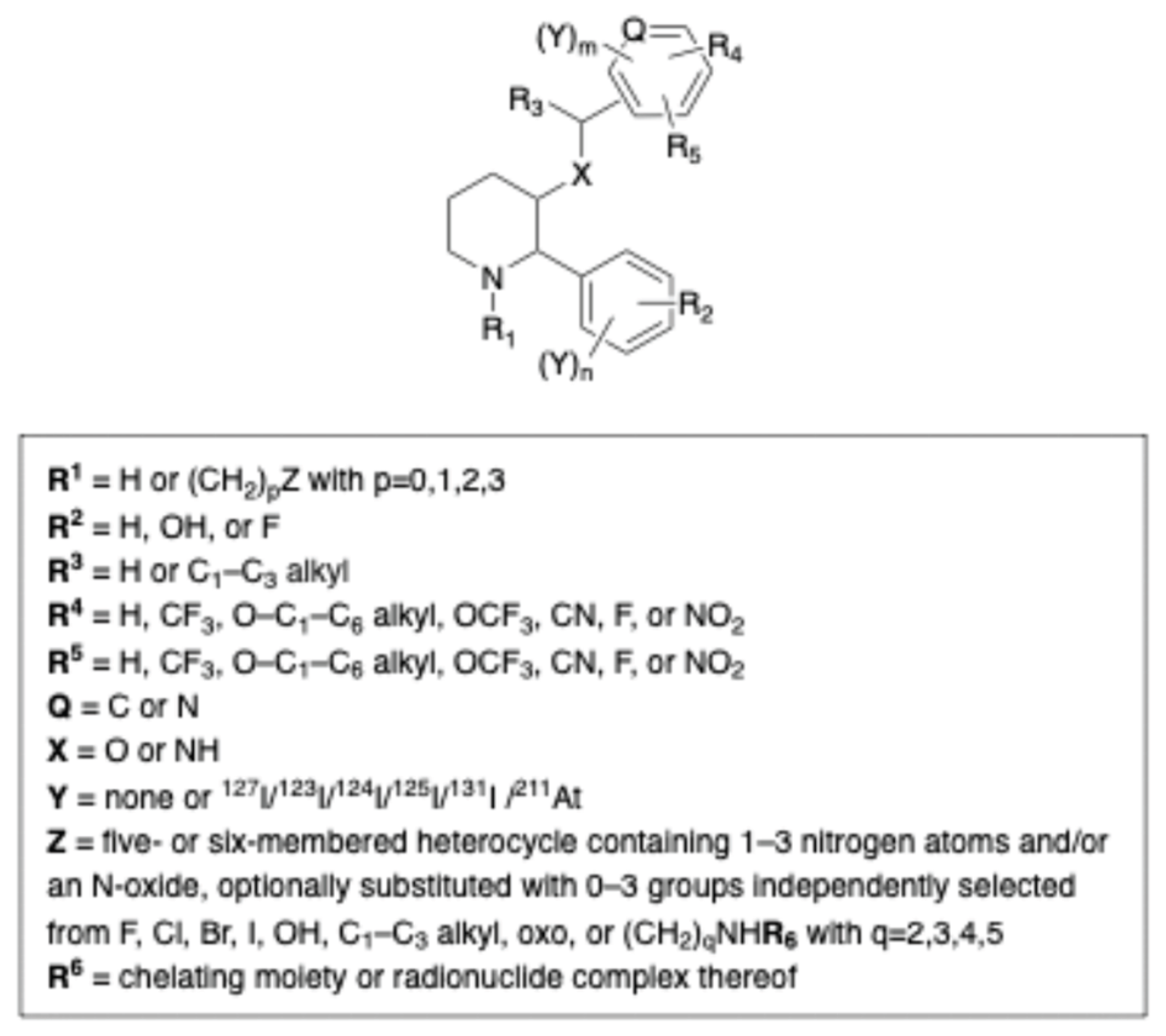
- Nestor, Marika; Bengtsson, Christoffer; Yngve, Ulrika; Begnini, Fabio; Jha, Preeti; Rosenström, Ulrika Substituted 2-Phenylpiperidine Compounds for Use in the Diagnosis, Treatment and/or Prevention of Cancer. WO2025008409, (2025).
- Fridmanis, D.; Roga, A.; Klovins, J. ACTH Receptor (MC2R) Specificity: What Do We Know About Underlying Molecular Mechanisms? Front. Endocrinol. 2017, 8. [Google Scholar] [CrossRef]
- Zhao, J.; Chen, M.; Zhu, Y.; Kim, S.H. Melanocortin Type 2 Receptor (MC2R) Targeted Therapeutics and Uses Thereof. US20230405157, (2023).
- Kirby, H.R.; Maguire, J.J.; Colledge, W.H.; Davenport, A.P. International Union of Basic and Clinical Pharmacology. LXXVII. Kisspeptin Receptor Nomenclature, Distribution, and Function. Pharmacol. Rev. 2010, 62, 565–578. [Google Scholar] [CrossRef] [PubMed]
- Navarro, V.M.; Castellano, J.M.; McConkey, S.M.; Pineda, R.; Ruiz-Pino, F.; Pinilla, L.; Clifton, D.K.; Tena-Sempere, M.; Steiner, R.A. Interactions between Kisspeptin and Neurokinin B in the Control of GnRH Secretion in the Female Rat. Am. J. Physiol.-Endocrinol. Metab. 2011, 300, E202–E210. [Google Scholar] [CrossRef] [PubMed]
- Curtis, A.E.; Cooke, J.H.; Baxter, J.E.; Parkinson, J.R.C.; Bataveljic, A.; Ghatei, M.A.; Bloom, S.R.; Murphy, K.G. A Kisspeptin-10 Analog with Greater in Vivo Bioactivity than Kisspeptin-10. Am. J. Physiol.-Endocrinol. Metab. 2010, 298, E296–E303. [Google Scholar] [CrossRef]
- Rodríguez Sarmiento, D.Y. Beyond Reproduction: Exploring the Non-Canonical Roles of the Kisspeptin System in Diverse Biological Systems. Bionatura 2023, 8, 1–6. [Google Scholar] [CrossRef]
- Rodríguez-Sarmiento, D.Y.; Rondón-Villarreal, P.; Scarpelli-Pereira, P.H.; Bouvier, M. Comprehensive Analysis of Kisspeptin Signaling: Effects on Cellular Dynamics in Cervical Cancer. Biomolecules 2024, 14, 923. [Google Scholar] [CrossRef]
- Liu, Junjie; Zhu, Yunfei; Xiong, Yifeng Kisspeptin Receptor (KISS1R) Targeted Therapeutics and Uses Thereof. WO2024206577, (2024).
- Vincent, J.-P.; Mazella, J.; Kitabgi, P. Neurotensin and Neurotensin Receptors. Trends Pharmacol. Sci. 1999, 20, 302–309. [Google Scholar] [CrossRef]
- Vincent, J.-P. Neurotensin Receptors: Binding Properties, Transduction Pathways, and Structure. Cell. Mol. Neurobiol. 1995, 15, 501–512. [Google Scholar] [CrossRef]
- Ehlers, R.A.; Kim, S.; Zhang, Y.; Ethridge, R.T.; Murrilo, C.; Hellmich, M.R.; Evans, D.B.; Townsend, C.M.; Mark Evers, B. Gut Peptide Receptor Expression in Human Pancreatic Cancers. Ann. Surg. 2000, 231, 838–848. [Google Scholar] [CrossRef]
- Gui, X.; Guzman, G.; Dobner, P.R.; Kadkol, S.S. Increased Neurotensin Receptor-1 Expression during Progression of Colonic Adenocarcinoma. Peptides 2008, 29, 1609–1615. [Google Scholar] [CrossRef]
- Souazé, F.; Dupouy, S.; Viardot-Foucault, V.; Bruyneel, E.; Attoub, S.; Gespach, C.; Gompel, A.; Forgez, P. Expression of Neurotensin and NT1 Receptor in Human Breast Cancer: A Potential Role in Tumor Progression. Cancer Res. 2006, 66, 6243–6249. [Google Scholar] [CrossRef]
- Dupouy, S.; Viardot-Foucault, V.; Alifano, M.; Souazé, F.; Plu-Bureau, G.; Chaouat, M.; Lavaur, A.; Hugol, D.; Gespach, C.; Gompel, A.; et al. The Neurotensin Receptor-1 Pathway Contributes to Human Ductal Breast Cancer Progression. PLoS ONE 2009, 4, e4223. [Google Scholar] [CrossRef] [PubMed]
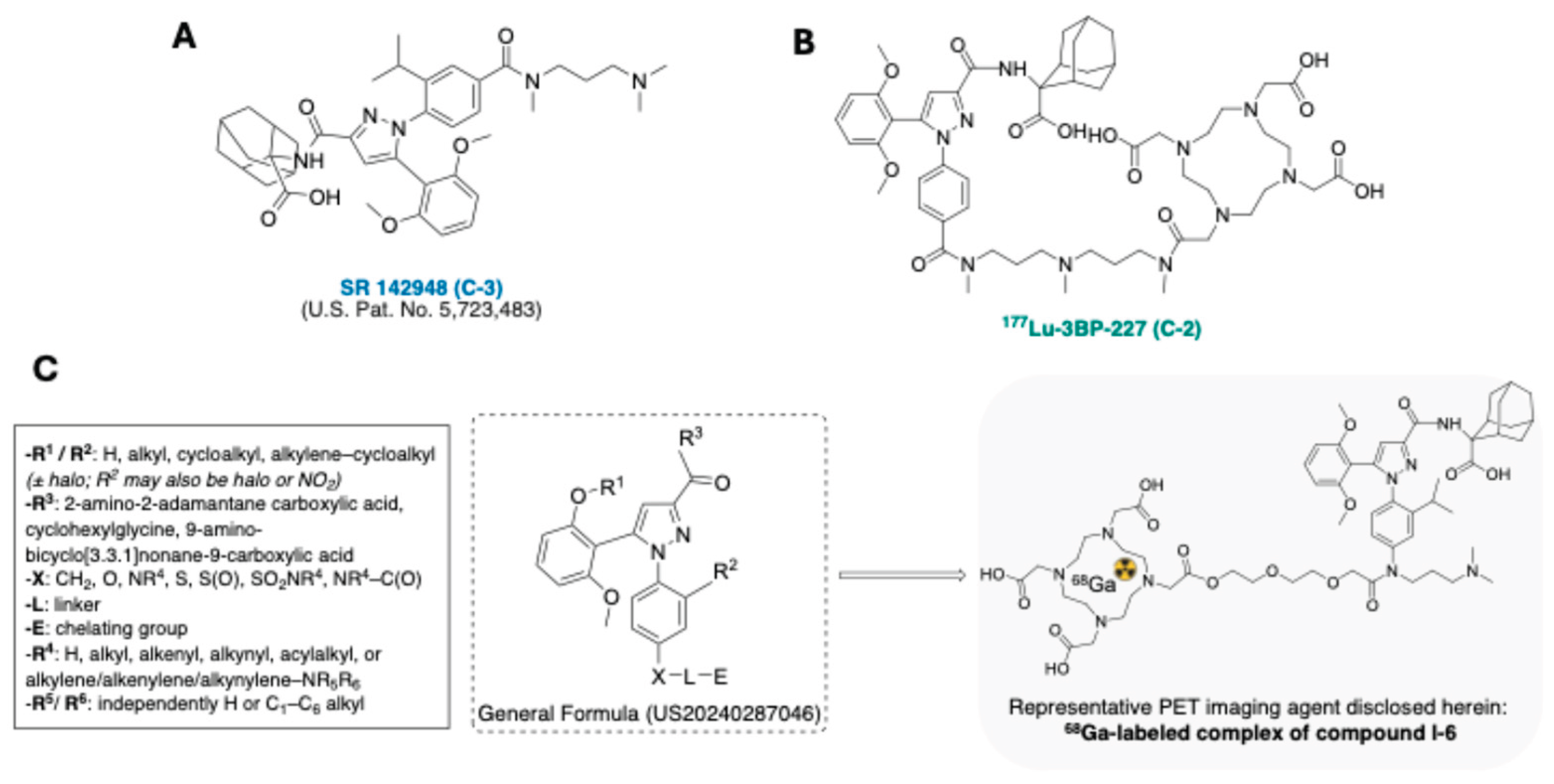
- Baum, R.P.; Singh, A.; Schuchardt, C.; Kulkarni, H.R.; Klette, I.; Wiessalla, S.; Osterkamp, F.; Reineke, U.; Smerling, C. 177 Lu-3BP-227 for Neurotensin Receptor 1–Targeted Therapy of Metastatic Pancreatic Adenocarcinoma: First Clinical Results. J. Nucl. Med. 2018, 59, 809–814. [Google Scholar] [CrossRef] [PubMed]
- D Gully Biochemical and Pharmacological Activities of SR 142948A, a New Potent Neurotensin Receptor Antagonist. The Journal of pharmacology and experimental therapeutics 1997, 280.
- Liu, F. Compounds and Radioligands for Targeting Neurotensin Receptor and Uses Thereof. US20240287046, (2024).
- Savoie, F.-A.; Arpin, D.J.; Vaillancourt, D.E. Magnetic Resonance Imaging and Nuclear Imaging of ParkinsonianDisorders: Where Do We Go from Here? Curr. Neuropharmacol. 2024, 22, 1583–1605. [Google Scholar] [CrossRef]
- Xie, L.; Zhao, J.; Li, Y.; Bai, J. PET Brain Imaging in Neurological Disorders. Phys. Life Rev. 2024, 49, 100–111. [Google Scholar] [CrossRef]
- Doroszkiewicz, J.; Groblewska, M.; Mroczko, B. Molecular Biomarkers and Their Implications for the Early Diagnosis of Selected Neurodegenerative Diseases. Int. J. Mol. Sci. 2022, 23, 4610. [Google Scholar] [CrossRef] [PubMed]
- Colom, M.; Vidal, B.; Zimmer, L. Is There a Role for GPCR Agonist Radiotracers in PET Neuroimaging? Front. Mol. Neurosci. 2019, 12, 255. [Google Scholar] [CrossRef]
- Kew, J.N.C.; Kemp, J.A. Ionotropic and Metabotropic Glutamate Receptor Structure and Pharmacology. Psychopharmacology (Berl.) 2005, 179, 4–29. [Google Scholar] [CrossRef]
- P. Jeffrey Conn; Jean-Philippe Pin. Pharmacology and Functions of Metabotropic Glutamate Receptors. 1997, 37. [CrossRef]
- Cartmell, J.; Schoepp, D.D. Regulation of Neurotransmitter Release by Metabotropic Glutamate Receptors. J. Neurochem. 2000, 75, 889–907. [Google Scholar] [CrossRef]
- Conn, P.J.; Lindsley, C.W.; Jones, C.K. Activation of Metabotropic Glutamate Receptors as a Novel Approach for the Treatment of Schizophrenia. Trends Pharmacol. Sci. 2009, 30, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Andrés-Gil, J.I.; Gool, M.L.M.V.; Bormans, G.M.R.; Verbeek, J. Radiolabelled mGluR2/3 PET Ligands. US20200306389, (2020).
- Leurquin-Sterk, G.; Celen, S.; Van Laere, K.; Koole, M.; Bormans, G.; Langlois, X.; Van Hecken, A.; Te Riele, P.; Alcázar, J.; Verbruggen, A.; et al. What We Observe In Vivo Is Not Always What We See In Vitro: Development and Validation of11 C-JNJ-42491293, A Novel Radioligand for mGluR2. J. Nucl. Med. 2017, 58, 110–116. [Google Scholar] [CrossRef]
- Yuan, Gengyang; Brownell, Anna-Liisa Chromane Imaging Ligands. WO2023278729, (2023).
- Gool, M.L., Maria Van; Andrés-Gil, J.I.; Alcázar-Vaca, M.J.; Bormans, G.M.R.; Celen, S.J.L.; Verbeek, J. Radiolabelled mGluR2 PET Ligands. US20210177995, (2021).
- Zhang, Z.; Zheng, B.; Yuan, G.; Neelamegam, R.; Brownell, A.-L. Modulators of Metabotropic Glutamate Receptor 2. US20210246140, (2021).
- Zhang, Z.; Wang, J.; Shoup, T.M.; Brownell, A.-L. Modulators of Metabotropic Glutamate Receptor 4. US20220118117, (2022).
- J W Kebabian Multiple Classes of Dopamine Receptors in Mammalian Central Nervous System: The Involvement of Dopamine-Sensitive Adenylyl Cyclase.
- Beaulieu, J.-M.; Gainetdinov, R.R. The Physiology, Signaling, and Pharmacology of Dopamine Receptors. Pharmacol. Rev. 2011, 63, 182–217. [Google Scholar] [CrossRef] [PubMed]
- J K Wamsley; D R Gehlert; F M Filloux; T M Dawson Comparison of the Distribution of D-1 and D-2 Dopamine Receptors in the Rat Brain. J Chem Neuroanat 1989.
- Marc Savasta; Alain Dubois; Bernard Scätton Autoradiographic Localization of D1 Dopamine Receptors in the Rat Brain with [3H]SCH 23390. Brain Res 1986, 375. [CrossRef]
- Chihiro Yokoyama; Hitoshi Okamura; Teruo Nakajima; Jun-Ichi Taguchi; Yasuhiko Ibata Autoradiographic Distribution of [3H]YM-09151-2, a High-Affinity and Selective Antagonist Ligand for the Dopamine D2 Receptor Group, in the Rat Brain and Spinal Cord. J Comp Neurol 1994, doi:https://doi.org/10.1002/cne.903440109Digital Object Identifier (DOI). [CrossRef]
- Prante, O.; Maschauer, S.; Banerjee, A. Radioligands for the Dopamine Receptor Subtypes. J. Label. Compd. Radiopharm. 2013, 56, 130–148. [Google Scholar] [CrossRef]
- Mercier, J.; Valade, A.; Vermeiren, C.; Wood, M.; Macquire, R. Imaging Agents. US20240343737, (2024).
- MacRitchie, N.; Frleta-Gilchrist, M.; Sugiyama, A.; Lawton, T.; McInnes, I.B.; Maffia, P. Molecular Imaging of Inflammation - Current and Emerging Technologies for Diagnosis and Treatment. Pharmacol. Ther. 2020, 211, 107550. [Google Scholar] [CrossRef]
- Calabria, F.F.; Guadagnino, G.; Cimini, A.; Leporace, M. PET/CT Imaging of Infectious Diseases: Overview of Novel Radiopharmaceuticals. Diagnostics 2024, 14, 1043. [Google Scholar] [CrossRef] [PubMed]
- Glaudemans, A.W.J.M.; De Vries, E.F.J.; Galli, F.; Dierckx, R.A.J.O.; Slart, R.H.J.A.; Signore, A. The Use of 18 F-FDG-PET/CT for Diagnosis and Treatment Monitoring of Inflammatory and Infectious Diseases. Clin. Dev. Immunol. 2013, 2013, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Wiehr, S.; Warnke, P.; Rolle, A.-M.; Schütz, M.; Oberhettinger, P.; Kohlhofer, U.; Quintanilla-Martinez, L.; Maurer, A.; Thornton, C.; Boschetti, F.; et al. New Pathogen-Specific immunoPET/MR Tracer for Molecular Imaging of a Systemic Bacterial Infection. Oncotarget 2016, 7, 10990–11001. [Google Scholar] [CrossRef]
- Mulero, F. Editorial: ImmunoPET Imaging in Disease Diagnosis and Therapy Assessment. Front. Med. 2023, 10, 1231525. [Google Scholar] [CrossRef]
- Sun, L.; Ye, R.D. Role of G Protein-Coupled Receptors in Inflammation. Acta Pharmacol. Sin. 2012, 33, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Gropler, R.; Brody, S.; Kreisel, D. Methods for Detecting CCR2 Receptors. US20200405889, (2020).
- Scholten, D.; Canals, M.; Maussang, D.; Roumen, L.; Smit, M.; Wijtmans, M.; De Graaf, C.; Vischer, H.; Leurs, R. Pharmacological Modulation of Chemokine Receptor Function. Br. J. Pharmacol. 2012, 165, 1617–1643. [Google Scholar] [CrossRef]
- Uhlén, M.; Fagerberg, L.; Hallström, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, Å.; Kampf, C.; Sjöstedt, E.; Asplund, A.; et al. Tissue-Based Map of the Human Proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef] [PubMed]
- Philip M. Murphy; H. Lee Tiffany Cloning of Complementary DNA Encoding a Functional Human Interleukin-8 Receptor. Science 1991, 253. [CrossRef]
- Decker, Michael; Higuchi, Takahiro; Spatz, Philipp 18F-CXCR2 PETtracer. WO2025149577, (2025).
- Nanki, T.; Hayashida, K.; El-Gabalawy, H.S.; Suson, S.; Shi, K.; Girschick, H.J.; Yavuz, S.; Lipsky, P.E. Stromal Cell-Derived Factor-1-CXC Chemokine Receptor 4 Interactions Play a Central Role in CD4+ T Cell Accumulation in Rheumatoid Arthritis Synovium. J. Immunol. 2000, 165, 6590–6598. [Google Scholar] [CrossRef]
- Bot, I.; Daissormont, I.T.M.N.; Zernecke, A.; Van Puijvelde, G.H.M.; Kramp, B.; De Jager, S.C.A.; Sluimer, J.C.; Manca, M.; Hérias, V.; Westra, M.M.; et al. CXCR4 Blockade Induces Atherosclerosis by Affecting Neutrophil Function. J. Mol. Cell. Cardiol. 2014, 74, 44–52. [Google Scholar] [CrossRef]
- Bénard, F.; Lin, K.-S.; Rousseau, E.; Zhang, Z.; Kwon, D.; Lau, J.; Munoz, C.U.; Lozada, J.; Perrin, D. Novel Radiolabelled CXCR4-Targeting Compounds for Diagnosis and Therapy. US20220218852, (2022).
- Bénard, François; Lin, Kuo-Shyan; Zhang, Zhengxing; Kwon, Daniel; Perrin, David; Todorovic, Mihajlo; Lozada, Jerome; Li, Lee Lee Novel CXCR4-Targeting Compounds. WO2022082312, (2022).
- James, Michelle L.; Jackson, Isaac M.; Kalita, Mausam; Nagy, Sydney C. Method for Detecting Innate Immune Action in Vivo Using GPR84-PET. WO2024220480, (2024).
- Labéguère, F.; Dupont, S.; Alvey, L.; Soulas, F.; Newsome, G.; Tirera, A.; Quenehen, V.; Mai, T.T.T.; Deprez, P.; Blanqué, R.; et al. Discovery of 9-Cyclopropylethynyl-2-(( S )-1-[1,4]Dioxan-2-Ylmethoxy)-6,7-Dihydropyrimido[6,1- a ]Isoquinolin-4-One (GLPG1205), a Unique GPR84 Negative Allosteric Modulator Undergoing Evaluation in a Phase II Clinical Trial. J. Med. Chem. 2020, 63, 13526–13545. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Chen, Z.; Gao, S.; Lian, C.; Hu, J.; Lu, J.; Zhang, L. GPR65 Is a Novel Immune Biomarker and Regulates the Immune Microenvironment in Lung Adenocarcinoma. Front. Immunol. 2025, 16, 1572757. [Google Scholar] [CrossRef] [PubMed]
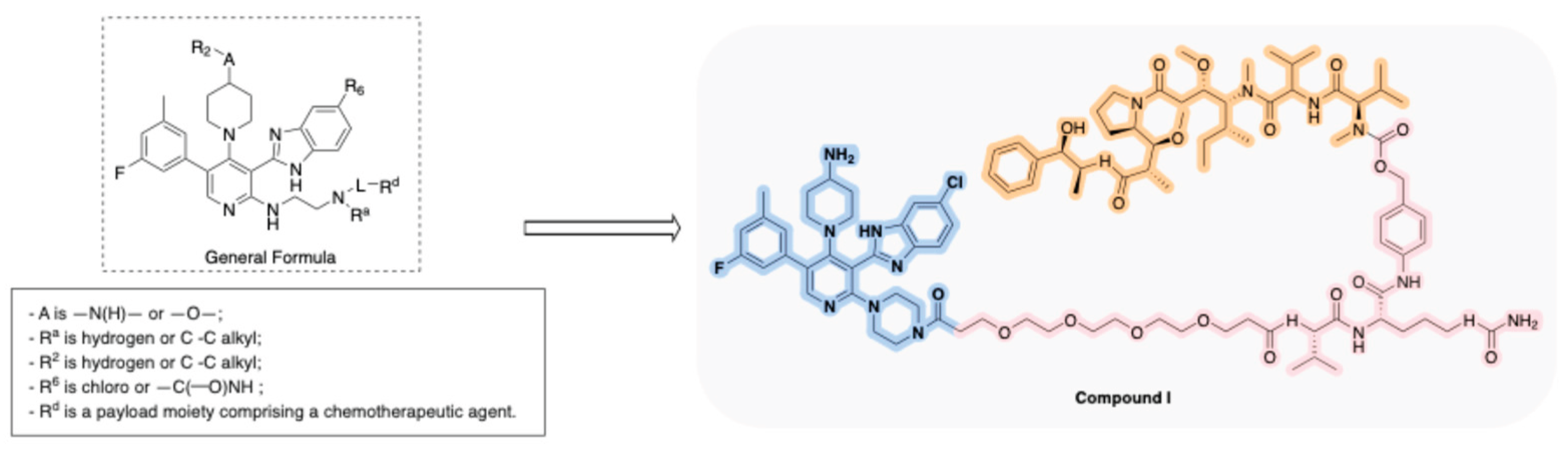
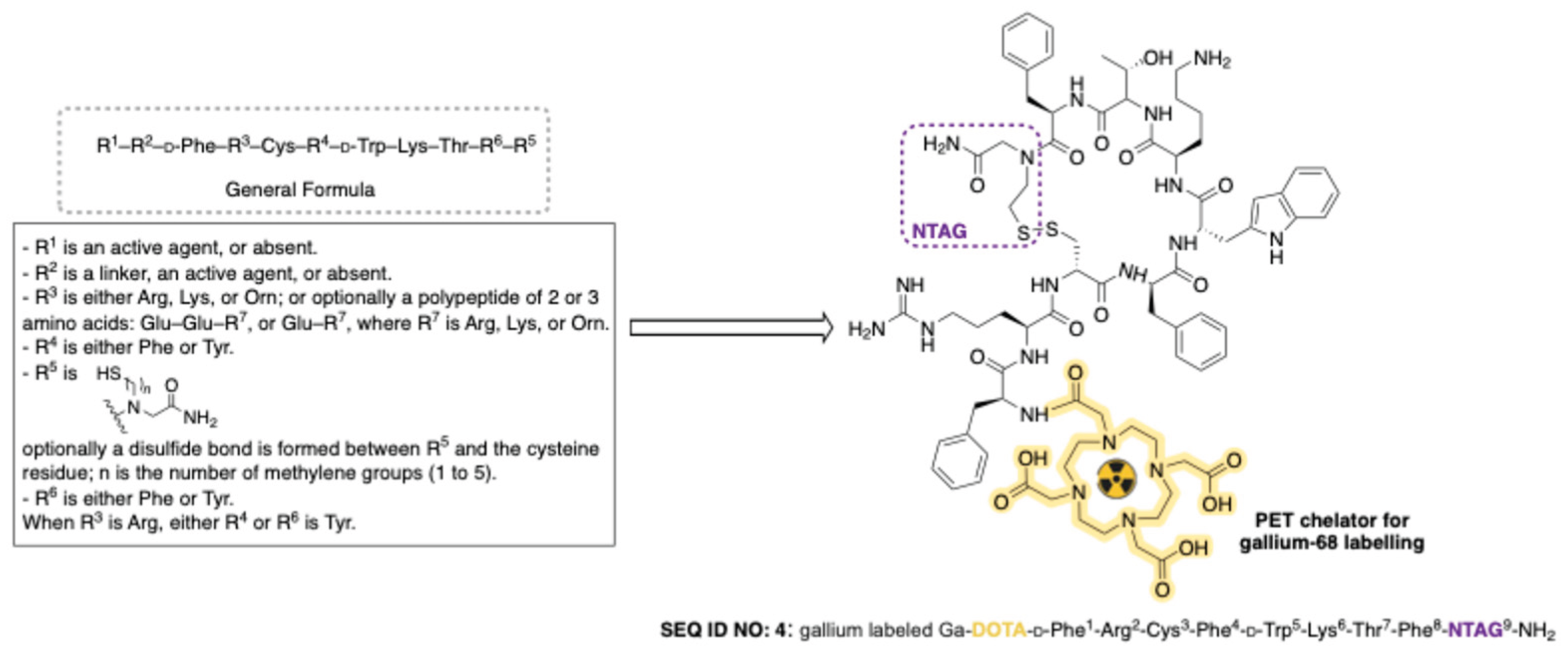
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




