Submitted:
08 April 2026
Posted:
10 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Location and Plant Material
2.2. Spectra Collection
2.3. Spectra Processing
2.4. Soil Moisture and Watering Regime
2.5. Chlorophyll Fluorescence (Fv/Fm)
2.6. Statistical Analysis
3. Results
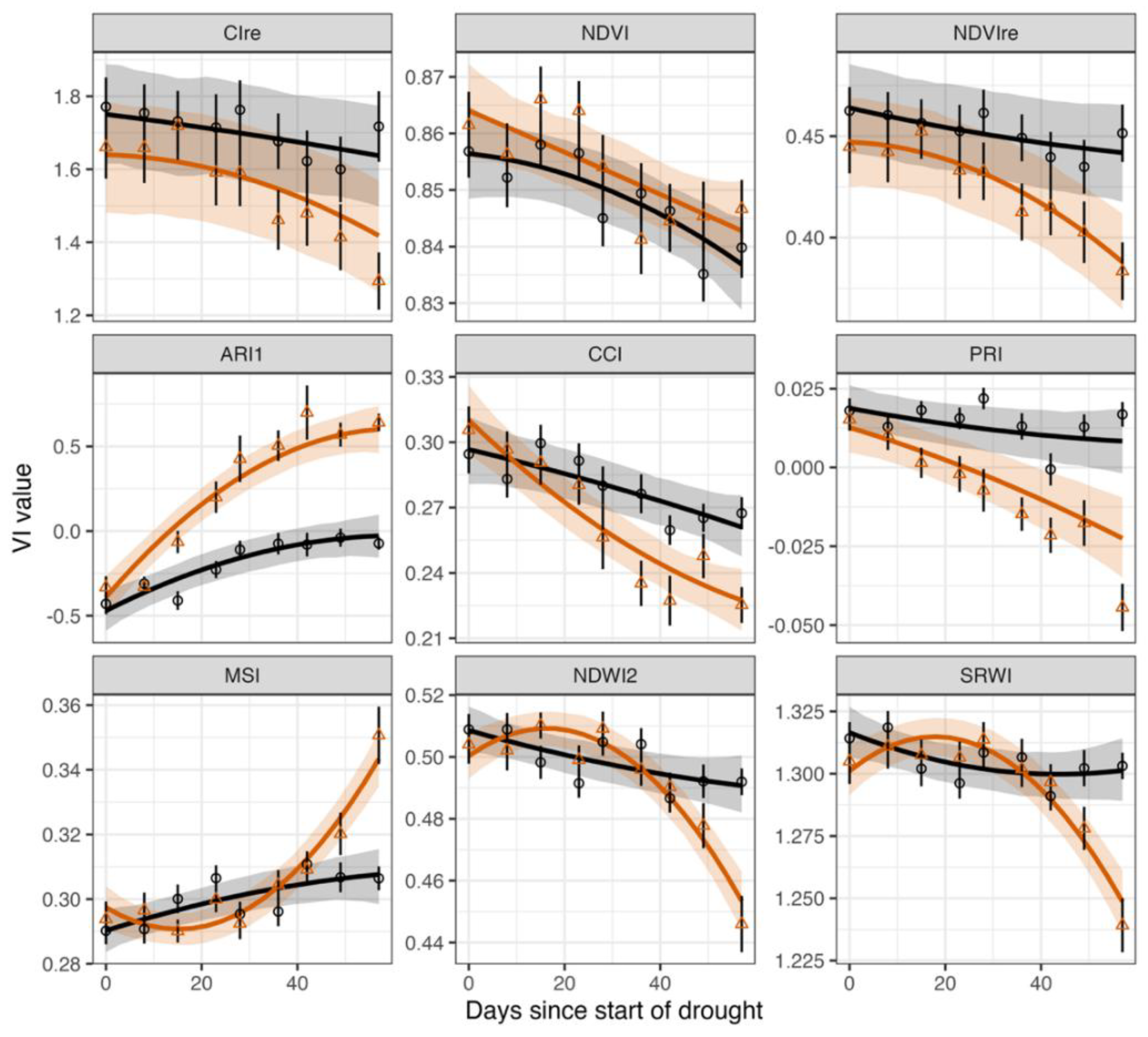
3.1. Temporal Trends
3.2. Vegetation Indices
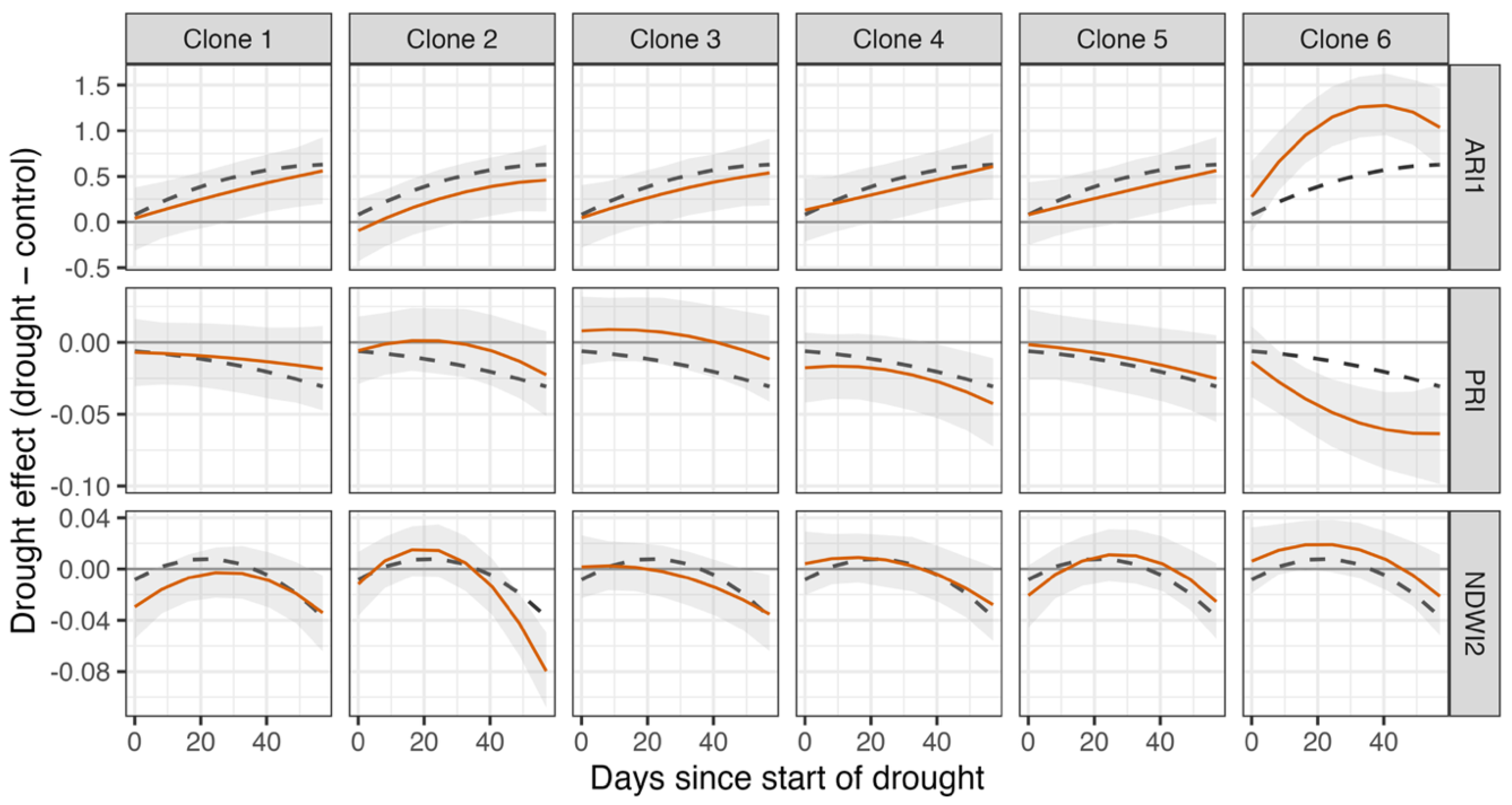
3.3. Clone Effects
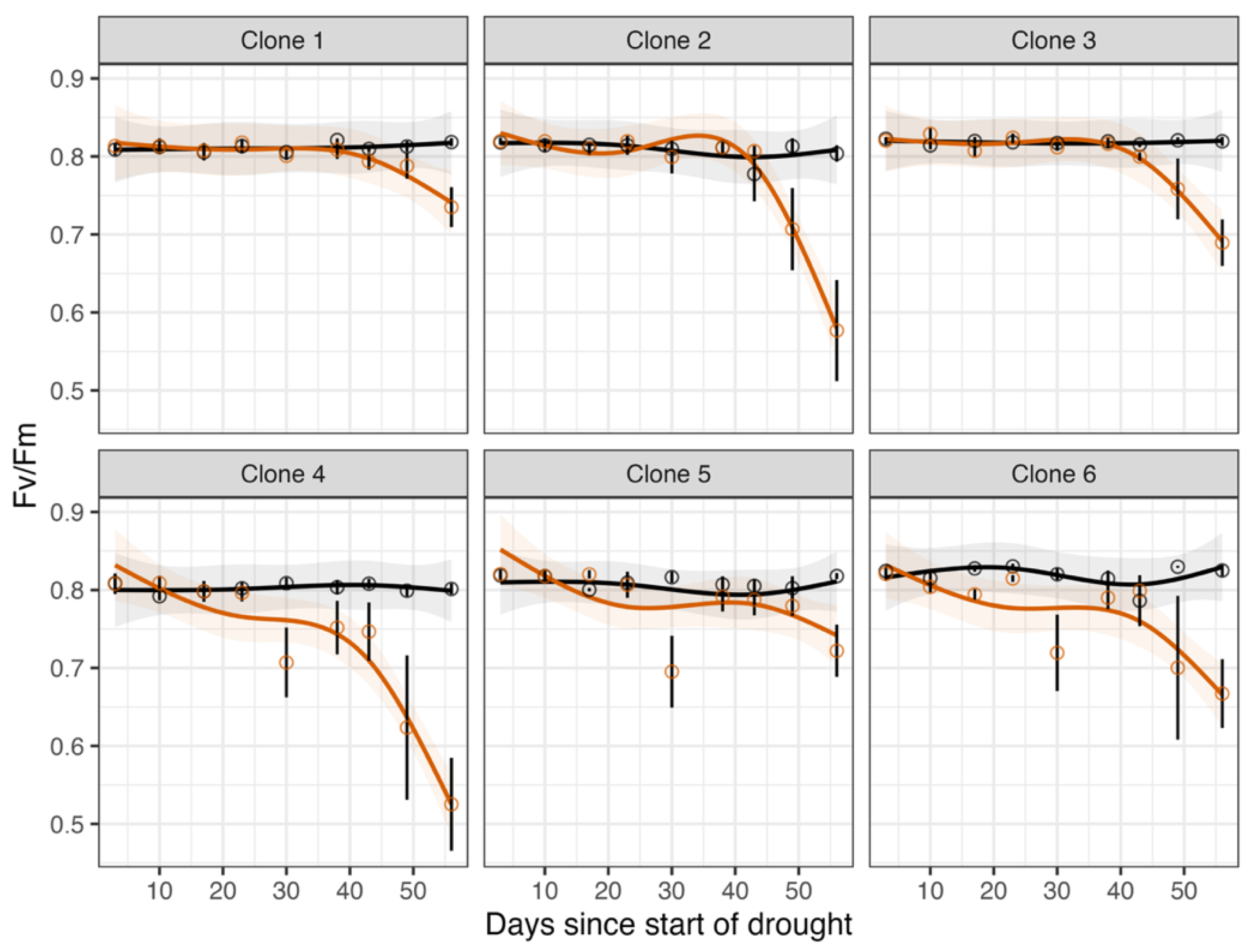
3.4. Chlorophyll Fluorescence (Fv/Fm)
4. Discussion
4.1. Chlorophyll and Greenness Response
4.2. Photoprotective Pigment Dynamics
4.3. Foliar Water Dynamics
4.4. Maximal Photosynthetic Capacity and Clonal Variation
4.5. Clonal Differences in Drought Strategy
4.6. Seasonal and Temporal Trends
4.7. Implications for Breeding and Management
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Fv/Fm | Maximal photochemical efficiency of photosystem II |
| VI | Vegetation Index |
| VIg | Greenness vegetation index |
| VIp | Pigment vegetation index |
| VIw | Water content vegetation index |
| FC | Field capacity |
| AIC | Akaike’s Information Criterion |
References
- Xenakis, G.; Ash, A.; Siebicke, L.; Perks, M.; Morison, J.I.L. Comparison of the Carbon, Water, and Energy Balances of Mature Stand and Clear-Fell Stages in a British Sitka Spruce Forest and the Impact of the 2018 Drought. Agric. For. Meteorol. 2021, 306, 108437. [Google Scholar] [CrossRef]
- Burke, E.J.; Perry, R.H.J.; Brown, S.J. An Extreme Value Analysis of UK Drought and Projections of Change in the Future. J. Hydrol. 2010, 388, 131–143. [Google Scholar] [CrossRef]
- Met Office UK Climate Projections: Headline Findings; 2021; pp. 1–12.
- Locatelli, T.; Beauchamp, K.; Perks, M.; Xenakis, G.; Nicoll, B.; Morison, J. Drought Risk in Scottish Forests 2021.
- Mason, W. Implementing Continuous Cover Forestry in Planted Forests: Experience with Sitka Spruce (Picea Sitchensis) in the British Isles. Forests 2015, 6, 879–902. [Google Scholar] [CrossRef]
- Samuel, S.; Fletcher, A.M.; Lines, R. Choice of Sitka Spruce Seed Origins for Use in British Forests; Bulletin / Forestry Commission; Forestry Commission: Edinburgh, 2007; ISBN 978-0-85538-727-3. [Google Scholar]
- Green, S.; Hendry, S.; Redfern, D. Drought Damage to Pole-Stage Sitka in NE Scotland. Scott. For. 2008, 62, 10–18. [Google Scholar]
- Green, S.; Ray, D. Potential Impacts of Drought and Disease on Forestry in Scotland. For. Res. 2009, 1–8. [Google Scholar]
- Betts, R.A.; Brown, K. The Third UK Climate Change Risk Assessment (CCRA3) Technical Reportduction. UK Clim. Risk 2021. [Google Scholar]
- Lee, S. Improving the Timber Quality of Sitka Spruce through Selection and Breeding. Forestry 1999, 72, 123–146. [Google Scholar] [CrossRef]
- Davies, S.; Bathgate, S.; Petr, M.; Gale, A.; Patenaude, G.; Perks, M. Drought Risk to Timber Production – A Risk versus Return Comparison of Commercial Conifer Species in Scotland. For. Policy Econ. 2020, 117, 102189. [Google Scholar] [CrossRef]
- Schiop, S.T.; Al Hassan, M.; Sestras, A.F.; Boscaiu, M.; Sestras, R.E.; Vicente, O. Biochemical Responses to Drought, at the Seedling Stage, of Several Romanian Carpathian Populations of Norway Spruce (Picea Abies L. Karst). Trees - Struct. Funct. 2017, 31, 1479–1490. [Google Scholar] [CrossRef]
- Serbin, S.P.; Singh, A.; McNeil, B.E.; Kingdon, C.C.; Townsend, P.A. Spectroscopic Determination of Leaf Morphological and Biochemical Traits for Northern Temperate and Boreal Tree Species. Ecol. Appl. 2014, 24, 1651–1669. [Google Scholar] [CrossRef] [PubMed]
- Wu, Qiaoli.; Song, C.; Song, Jinling.; Wang, Jindi.; Chen, Shaoyuan.; Yu, Bo. Impacts of Leaf Age on Canopy Spectral Signature Variation in Evergreen Chinese Fir Forests. Remote Sens. 2018, 10, 262. [Google Scholar] [CrossRef]
- O’Neill, A.L.; Kupiec, J.A.; Curran, P.J. Biochemical and Reflectance Variation throughout a Sitka Spruce Canopy. Remote Sens. Environ. 2002, 80, 134–142. [Google Scholar] [CrossRef]
- Gamon, J.A.; Huemmrich, K.F.; Wong, C.Y.S.; Ensminger, I.; Garrity, S.; Hollinger, D.Y.; Noormets, A.; Peñuelask, J. A Remotely Sensed Pigment Index Reveals Photosynthetic Phenology in Evergreen Conifers. Proc. Natl. Acad. Sci. U. S. A. 2016, 113, 13087–13092. [Google Scholar] [CrossRef]
- Nadal-Sala, D.; Grote, R.; Birami, B.; Knüver, T.; Rehschuh, R.; Schwarz, S.; Ruehr, N.K. Leaf Shedding and Non-Stomatal Limitations of Photosynthesis Mitigate Hydraulic Conductance Losses in Scots Pine Saplings During Severe Drought Stress. Front. Plant Sci. 2021, 12, 715127. [Google Scholar] [CrossRef]
- Clevers, J.G.P.W.; Gitelson, A.A. Remote Estimation of Crop and Grass Chlorophyll and Nitrogen Content Using Red-Edge Bands on Sentinel-2 and -3. Int. J. Appl. Earth Obs. Geoinformation 2013, 23, 344–351. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Merzlyak, M.N. Quantitative Estimation of Chlorophyll-a Using Reflectance Spectra: Experiments with Autumn Chestnut and Maple Leaves. J. Photochem. Photobiol. B 1994, 22, 247–252. [Google Scholar] [CrossRef]
- Horler, D.N.H.; Dockray, M.; Barber, J. The Red Edge of Plant Leaf Reflectance. Int. J. Remote Sens. 1983, 4, 273–288. [Google Scholar] [CrossRef]
- Brodersen, C.R.; Vogelmann, T.C. Do Changes in Light Direction Affect Absorption Profiles in Leaves? Funct. Plant Biol. 2010, 37, 403–412. [Google Scholar] [CrossRef]
- Liu, J.; van Iersel, M.W. Photosynthetic Physiology of Blue, Green, and Red Light: Light Intensity Effects and Underlying Mechanisms. Front. Plant Sci. 2021, 12, 619987. [Google Scholar] [CrossRef]
- Demmig-Adams, B. Survey of Thermal Energy Dissipation and Pigment Composition in Sun and Shade Leaves. Plant Cell Physiol. 1998, 39, 474–482. [Google Scholar] [CrossRef]
- Jiang, C.-D.; Gao, H.-Y.; Zou, Q.; Jiang, G.-M.; Li, L.-H. Leaf Orientation, Photorespiration and Xanthophyll Cycle Protect Young Soybean Leaves against High Irradiance in Field. Environ. Exp. Bot. 2006, 55, 87–96. [Google Scholar] [CrossRef]
- Takahashi, S.; Badger, M.R. Photoprotection in Plants: A New Light on Photosystem II Damage. Trends Plant Sci. 2011, 16, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Filella, I.; Porcar-Castell, A.; Munné-Bosch, S.; Bäck, J.; Garbulsky, M.F.; Peñuelas, J. PRI Assessment of Long-Term Changes in Carotenoids/Chlorophyll Ratio and Short-Term Changes in de-Epoxidation State of the Xanthophyll Cycle. Int. J. Remote Sens. 2009, 30, 4443–4455. [Google Scholar] [CrossRef]
- Hernández-Clemente, R.; Hornero, A.; Mottus, M.; Penuelas, J.; González-Dugo, V.; Jiménez, J.C.; Suárez, L.; Alonso, L.; Zarco-Tejada, P.J. Early Diagnosis of Vegetation Health From High-Resolution Hyperspectral and Thermal Imagery: Lessons Learned From Empirical Relationships and Radiative Transfer Modelling. Curr. For. Rep. 2019, 5, 169–183. [Google Scholar] [CrossRef]
- Mõttus, M.; Hernández-Clemente, R.; Perheentupa, V.; Markiet, V. In Situ Measurement of Scots Pine Needle PRI. Plant Methods 2017, 13, 35. [Google Scholar] [CrossRef]
- Peguero-Pina, J.J.; Camarero, J.J.; Abadía, A.; Martín, E.; González-Cascón, R.; Morales, F.; Gil-Pelegrín, E. Physiological Performance of Silver-Fir (Abies Alba Mill.) Populations under Contrasting Climates near the South-Western Distribution Limit of the Species. Flora Morphol. Distrib. Funct. Ecol. Plants 2007, 202, 226–236. [Google Scholar] [CrossRef]
- Wong, C.Y.S.; Gamon, J.A. Three Causes of Variation in the Photochemical Reflectance Index (PRI) in Evergreen Conifers. New Phytol. 2015, 206, 187–195. [Google Scholar] [CrossRef]
- Cirillo, V.; D’Amelia, V.; Esposito, M.; Amitrano, C.; Carillo, P.; Carputo, D.; Maggio, A. Anthocyanins Are Key Regulators of Drought Stress Tolerance in Tobacco. Biology 2021, 10, 139. [Google Scholar] [CrossRef]
- Landi, M.; Tattini, M.; Gould, K.S. Multiple Functional Roles of Anthocyanins in Plant-Environment Interactions. Environ. Exp. Bot. 2015, 119, 4–17. [Google Scholar] [CrossRef]
- Steyn, W.J.; Wand, S.J.E.; Holcroft, D.M.; Jacobs, G. Anthocyanins in Vegetative Tissues: A Proposed Unified Function in Photoprotection. New Phytol. 2002, 155, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Gould, K.S. Nature’s Swiss Army Knife: The Diverse Protective Roles of Anthocyanins in Leaves. J. Biomed. Biotechnol. 2004, 2004, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Kyparissis, A.; Grammatikopoulos, G.; Manetas, Y. Leaf Morphological and Physiological Adjustments to the Spectrally Selective Shade Imposed by Anthocyanins in Prunus Cerasifera. Tree Physiol. 2007, 27, 849–857. [Google Scholar] [CrossRef]
- Hernández, I.; Alegre, L.; Van Breusegem, F.; Munné-Bosch, S. How Relevant Are Flavonoids as Antioxidants in Plants? Trends Plant Sci. 2009, 14, 125–132. [Google Scholar] [CrossRef]
- Stimson, H.C.; Breshears, D.D.; Ustin, S.L.; Kefauver, S.C. Spectral Sensing of Foliar Water Conditions in Two Co-Occurring Conifer Species: Pinus Edulis and Juniperus Monosperma. Remote Sens. Environ. 2005, 96, 108–118. [Google Scholar] [CrossRef]
- Dao, P.D.; He, Y.; Proctor, C. Plant Drought Impact Detection Using Ultra-High Spatial Resolution Hyperspectral Images and Machine Learning. Int. J. Appl. Earth Obs. Geoinformation 2021, 102, 102364. [Google Scholar] [CrossRef]
- Andrews, S.F.; Flanagan, L.B.; Sharp, E.J.; Cai, T. Variation in Water Potential, Hydraulic Characteristics and Water Source Use in Montane Douglas-Fir and Lodgepole Pine Trees in Southwestern Alberta and Consequences for Seasonal Changes in Photosynthetic Capacity. Tree Physiol. 2012, 32, 146–160. [Google Scholar] [CrossRef]
- Irvine, J.; Perks, M.P.; Magnani, F.; Grace, J. The Response of Pinus Sylvestris to Drought: Stomatal Control of Transpiration and Hydraulic Conductance. Tree Physiol. 1998, 18, 393–402. [Google Scholar] [CrossRef]
- Black, K.; Philk, D.; Mc Grath, J.; Doherty, P.; Bruce, O. Interactive Effects of Irradiance and Water Availability on the Photosynthetic Performance of Picea Sitchensis Seedlings: Implications for Seedling Establishment under Different Management Practices. Ann. For. Sci. 2005, 64, 219–228. [Google Scholar] [CrossRef]
- Matisons, R.; Krišāns, O.; Jansons, Ā.; Kondratovičs, T.; Elferts, D.; Ievinsh, G. Norway Spruce Seedlings from an Eastern Baltic Provenance Show Tolerance to Simulated Drought. Forests 2021, 12, 82. [Google Scholar] [CrossRef]
- Hetherington, A.M.; Woodward, F.I. The Role of Stomata in Sensing and Driving Environmental Change. Nature 2003, 424, 901–908. [Google Scholar] [CrossRef]
- Ensminger, I.; Yao-Yun Chang, C.; Bräutigam, K. Tree Responses to Environmental Cues. In Advances in Botanical Research; Elsevier, 2015; Vol. 74, pp. 229–263. ISBN 978-0-12-398548-4. [Google Scholar]
- Balekoglu, S.; Caliskan, S.; Dirik, H.; Rosner, S. Response to Drought Stress Differs among Pinus Pinea Provenances. For. Ecol. Manag. 2023, 531, 120779. [Google Scholar] [CrossRef]
- López, R.; Rodríguez-Calcerrada, J.; Gil, L. Physiological and Morphological Response to Water Deficit in Seedlings of Five Provenances of Pinus Canariensis: Potential to Detect Variation in Drought-Tolerance. Trees - Struct. Funct. 2009, 23, 509–519. [Google Scholar] [CrossRef]
- Sánchez-Gómez, D.; Velasco-Conde, T.; Cano-Martín, F.J.; Ángeles Guevara, M.; Teresa Cervera, M.; Aranda, I. Inter-Clonal Variation in Functional Traits in Response to Drought for a Genetically Homogeneous Mediterranean Conifer. Environ. Exp. Bot. 2011, 70, 104–109. [Google Scholar] [CrossRef]
- Taïbi, K.; Del Campo, A.D.; Vilagrosa, A.; Bellés, J.M.; López-Gresa, M.P.; Pla, D.; Calvete, J.J.; López-Nicolás, J.M.; Mulet, J.M. Drought Tolerance in Pinus Halepensis Seed Sources As Identified by Distinctive Physiological and Molecular Markers. Front. Plant Sci. 2017, 8, 1202. [Google Scholar] [CrossRef]
- Grant, O.M.; Montero Ribeiro, A.F.; Glombik, P.; O’Reilly, C. Impact of Limited Water Availability on Growth and Biomass Production of a Range of Full-Sibling Sitka Spruce (Picea Sitchensis (Bong.) Carr.) Families. For. Int. J. For. Res. 2018, 91, 83–97. [Google Scholar] [CrossRef]
- Čepl, J.; Stejskal, J.; Lhotáková, Z.; Holá, D.; Korecký, J.; Lstibůrek, M.; Tomášková, I.; Kočová, M.; Rothová, O.; Palovská, M.; et al. Heritable Variation in Needle Spectral Reflectance of Scots Pine (Pinus Sylvestris L.) Peaks in Red Edge. Remote Sens. Environ. 2018, 219, 89–98. [Google Scholar] [CrossRef]
- George, J.-P.; Grabner, M.; Campelo, F.; Karanitsch-Ackerl, S.; Mayer, K.; Klumpp, R.T.; Schüler, S. Intra-Specific Variation in Growth and Wood Density Traits under Water-Limited Conditions: Long-Term-, Short-Term-, and Sudden Responses of Four Conifer Tree Species. Sci. Total Environ. 2019, 660, 631–643. [Google Scholar] [CrossRef]
- Barakat, M.; El-Hendawy, S.; Al-Suhaibani, N.; Elshafei, A.; Al-Doss, A.; Al-Ashkar, I.; Ahmed, E.; Al-Gaadi, K. The Genetic Basis of Spectral Reflectance Indices in Drought-Stressed Wheat. Acta Physiol. Plant. 2016, 38, 227. [Google Scholar] [CrossRef]
- El-Hendawy, S.E.; Alotaibi, M.; Al-Suhaibani, N.; Al-Gaadi, K.; Hassan, W.; Dewir, Y.H.; Emam, M.A.E.-G.; Elsayed, S.; Schmidhalter, U. Comparative Performance of Spectral Reflectance Indices and Multivariate Modeling for Assessing Agronomic Parameters in Advanced Spring Wheat Lines Under Two Contrasting Irrigation Regimes. Front. Plant Sci. 2019, 10, 1537. [Google Scholar] [CrossRef]
- Ensminger, I.; Sveshnikov, D.; Campbell, D.A.; Funk, C.; Jansson, S.; Lloyd, J.; Shibistova, O.; Öquist, G. Intermittent Low Temperatures Constrain Spring Recovery of Photosynthesis in Boreal Scots Pine Forests. Glob. Change Biol. 2004, 10, 995–1008. [Google Scholar] [CrossRef]
- Qi, Y.; Dennison, P.E.; Jolly, W.M.; Kropp, R.C.; Brewer, S.C. Spectroscopic Analysis of Seasonal Changes in Live Fuel Moisture Content and Leaf Dry Mass. Remote Sens. Environ. 2014, 150, 198–206. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



