Submitted:
07 April 2026
Posted:
08 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
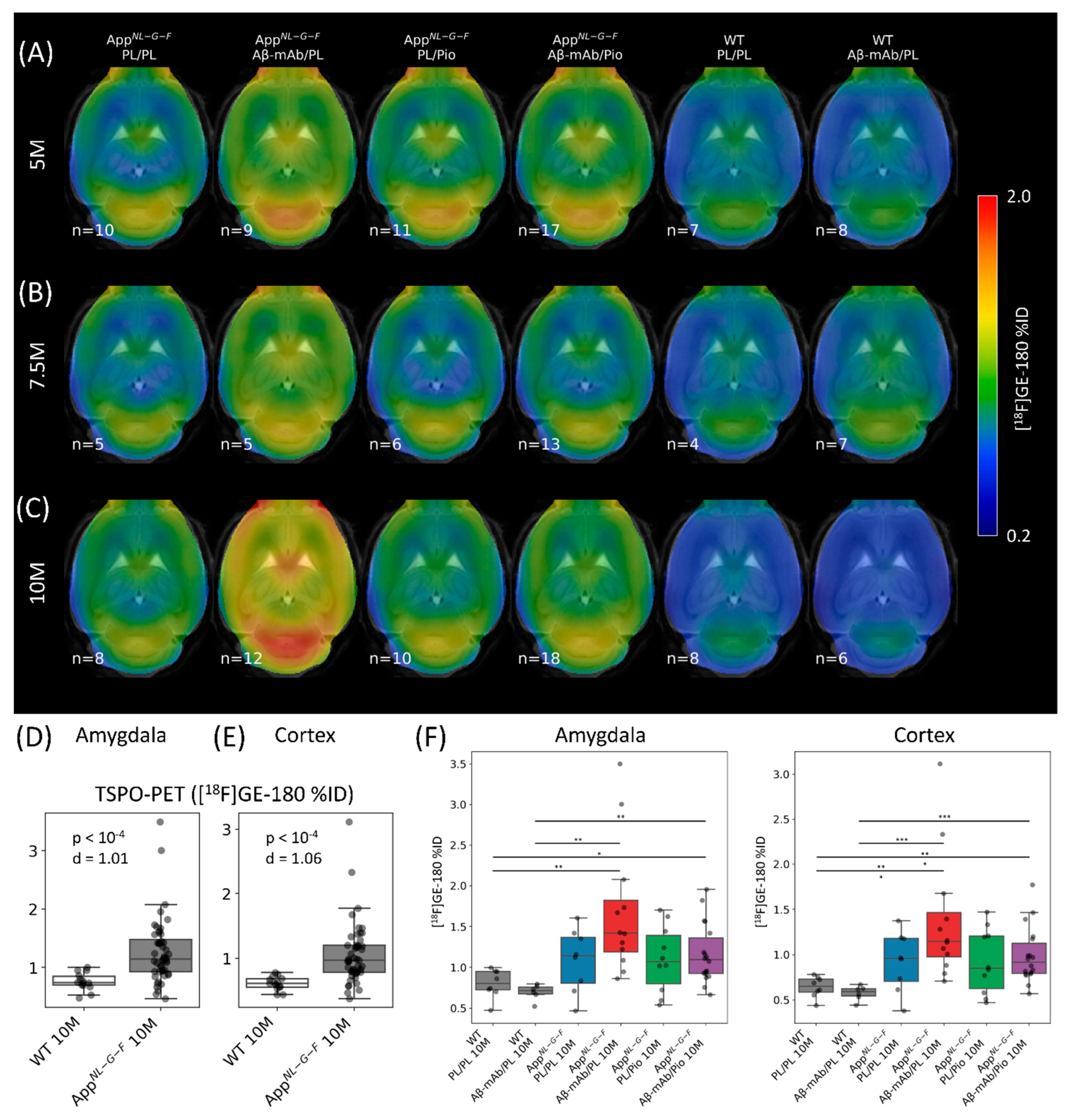
2.1. TSPO-PET Signals Are Increased in AppNL-G-F Mice Compared to Wild-Type Mice and Indicate Exploratory Treatment-Related Differences in the AppNL-G-F Mouse Model
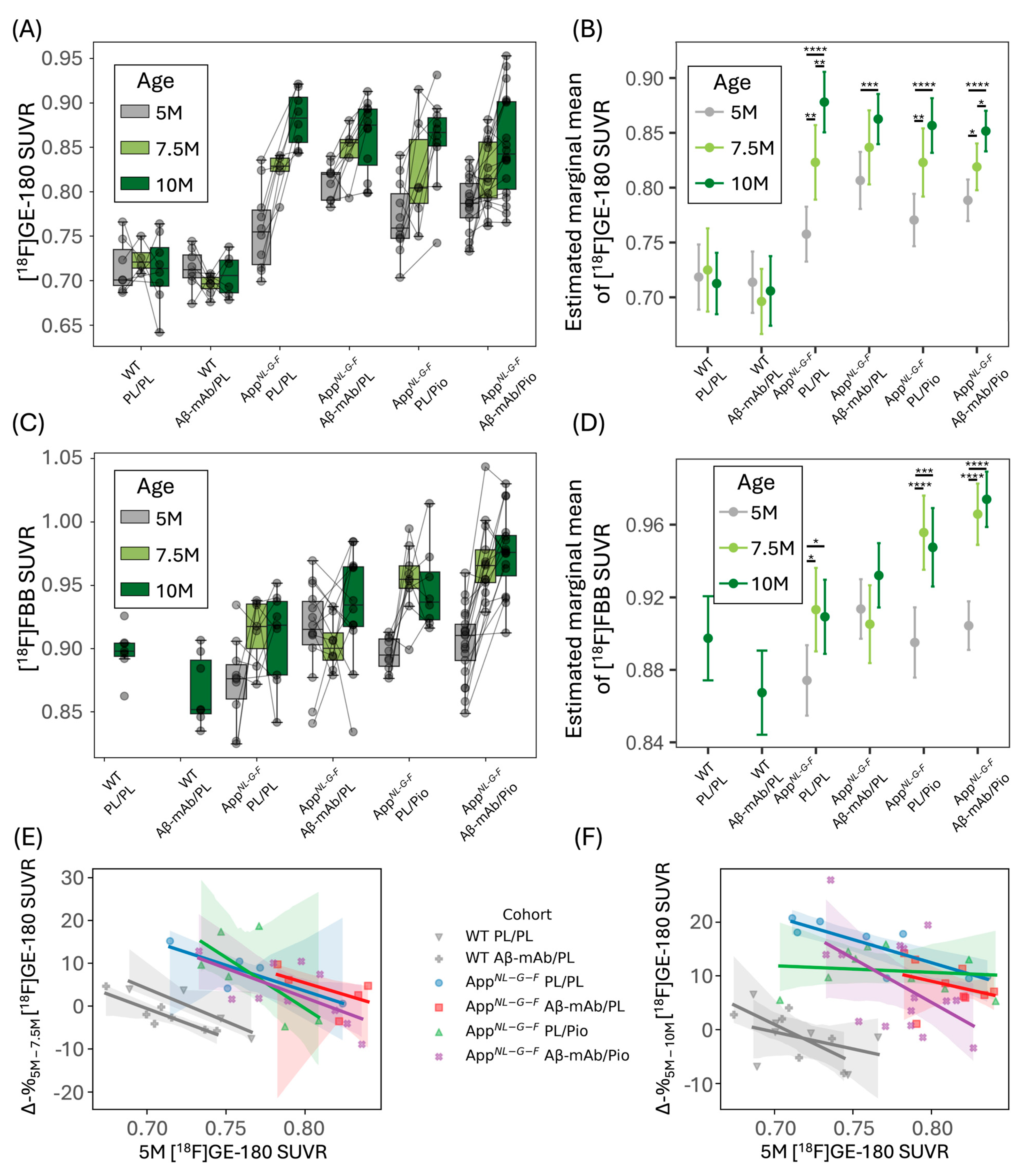
2.2. Treatments with Anti-Aβ-mAb and Pioglitazone Ameliorate Progression of Neuroinflammation in the AppNL-G-F Mouse Model
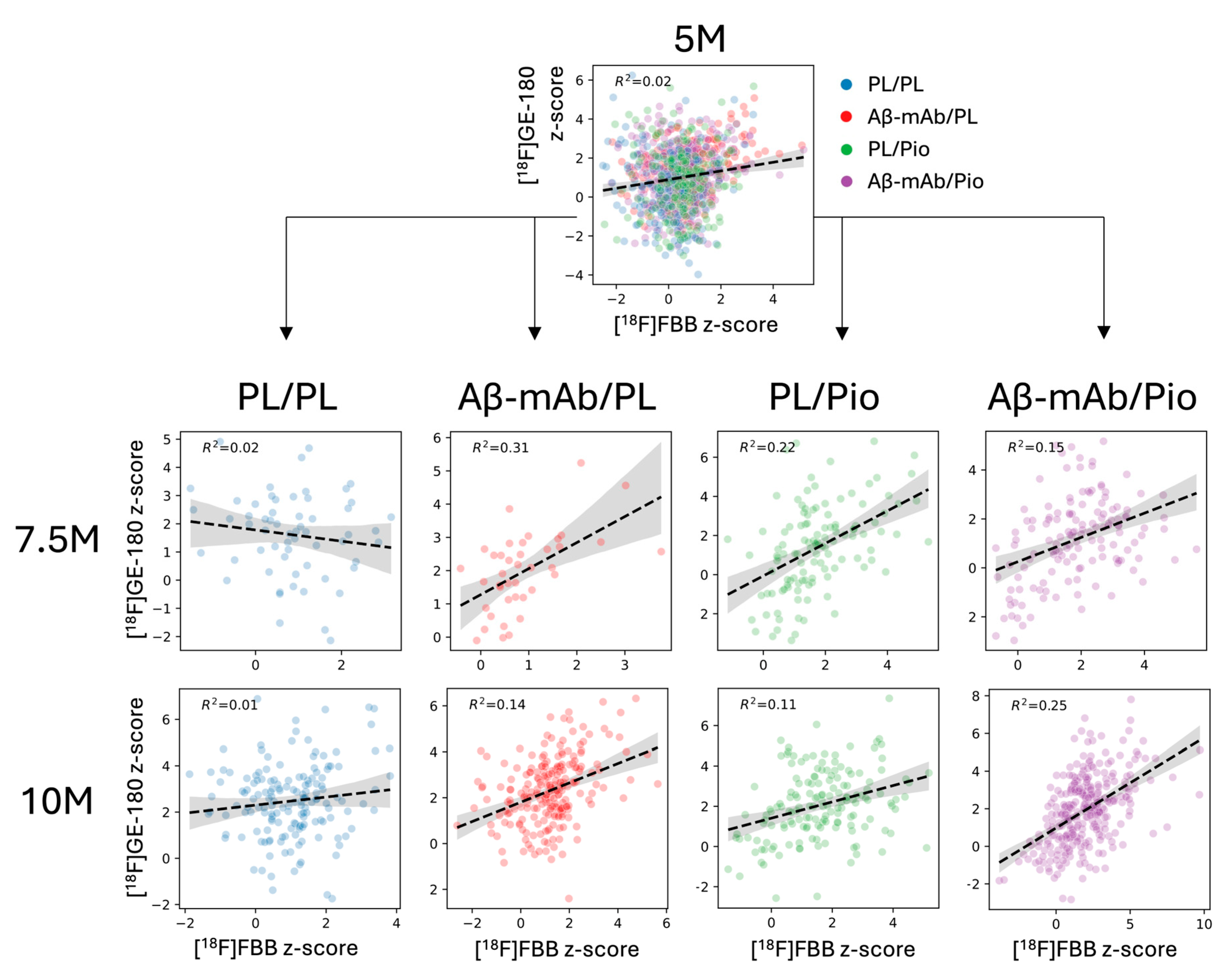
2.3. Pioglitazone Treatment Induced Early Neuroinflammatory Response Against Fibrillary Amyloid in the AppNL-G-F Mouse Model
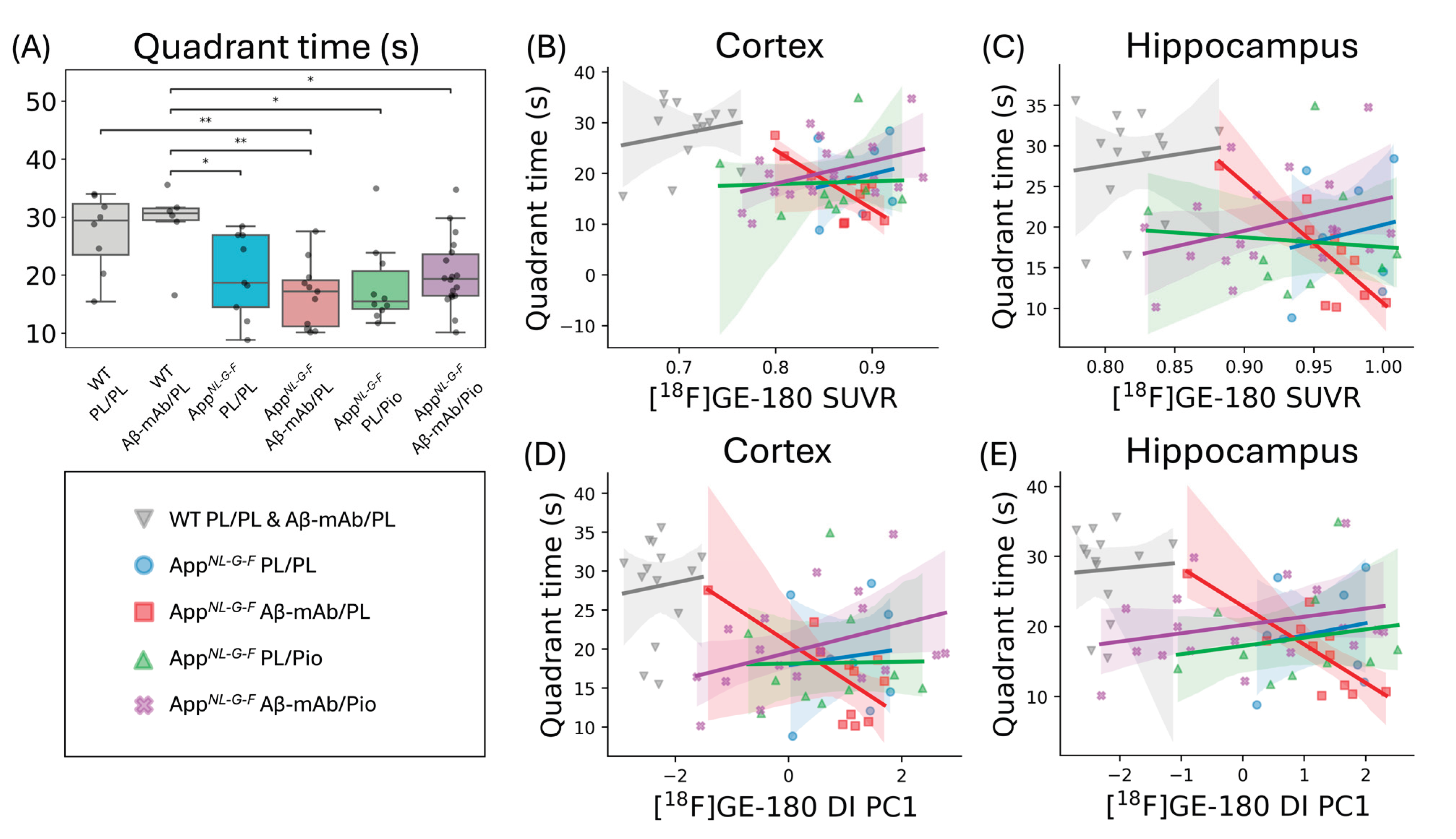
2.4. TSPO-PET Correlates with Behavioral Outcomes in AppNL-G-F Mice Treated with Aβ-mAb
3. Discussion
4. Materials and Methods
4.1. Experimental Design
4.1.1. Randomization
4.1.2. Blinding
4.2. Animals
4.3. Treatment
4.4. Behavioral Testing
4.5. Analysis of AD Signature Proteins
4.6. PET Imaging and Analysis
4.7. Individual Assessment of Microglia Desynchronization
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Aβ | β-amyloid |
| AD | Alzheimer’s disease |
| ANOVA | Analysis of variance |
| ApoE | Apolipoprotein E |
| ARIA | Amyloid-related imaging abnormalities |
| CAA | Cerebral amyloid angiopathy |
| CDS | Connectivity deviation score |
| DEA | Diethylamine buffer |
| DI | Desynchronization index |
| EHA | Entorhinal-hippocampus-amygdala region |
| FA | Formic acid |
| FBB | Florbetaben |
| FDR | False-discovery rate |
| ICC | Interregional correlation coefficient |
| ID | Injected dose |
| IQR | Interquartile range |
| LMEM | Linear mixed-effects model |
| mAb | Monoclonal antibody |
| MWM | Morris water maze |
| MWU | Mann-Whitney U-test |
| PBS | Phosphate-buffered saline |
| PC1 | First principal component |
| PET | Positron emission tomography |
| Pio | Pioglitazone |
| PL | Placebo |
| PPAR-γ | Peroxisome proliferator-activated receptor γ |
| RIPA | Radio-immuno-precipitation assay buffer |
| SUVR | Standardized uptake value ratio |
| TREM2 | Triggering receptor expressed on myeloid cells 2 |
| TSPO | 18 kDa translocator protein |
| VOI | Volume of interest |
| WT | Wild type |
| μPET | Small-animal PET |
References
- Gustavsson, A. Global estimates on the number of persons across the Alzheimer's disease continuum. Alzheimer's & Dementia 2023, 19, 658–670. [Google Scholar]
- Cummings, J. Anti-amyloid monoclonal antibodies for the treatment of Alzheimer’s disease. BioDrugs 2024, 38, 5–22. [Google Scholar] [CrossRef]
- Sperling, R.A. Amyloid-related imaging abnormalities in amyloid-modifying therapeutic trials: recommendations from the Alzheimer’s Association Research Roundtable Workgroup. Alzheimer's & Dementia 2011, 7, 367–385. [Google Scholar]
- Jeong, S.Y. Amyloid-related imaging abnormalities in the era of anti-amyloid beta monoclonal antibodies for Alzheimer’s disease: recent updates on clinical and imaging features and MRI monitoring. Korean Journal of Radiology 2024, 25, 726. [Google Scholar] [CrossRef]
- Zimmer, E.R. Tracking neuroinflammation in Alzheimer’s disease: the role of positron emission tomography imaging. Journal of neuroinflammation 2014, 11, 120. [Google Scholar] [CrossRef]
- Heneka, M.T. Neuroinflammation in Alzheimer's disease. The Lancet Neurology 2015, 14, 388–405. [Google Scholar] [CrossRef]
- Wilcock, D.M. Passive immunotherapy against Aβ in aged APP-transgenic mice reverses cognitive deficits and depletes parenchymal amyloid deposits in spite of increased vascular amyloid and microhemorrhage. Journal of neuroinflammation 2004, 1, 24. [Google Scholar] [CrossRef]
- Wilcock, D.M. Deglycosylated anti-amyloid-β antibodies eliminate cognitive deficits and reduce parenchymal amyloid with minimal vascular consequences in aged amyloid precursor protein transgenic mice. Journal of Neuroscience 2006, 26, 5340–5346. [Google Scholar] [CrossRef]
- Taylor, X. Amyloid-β (Aβ) immunotherapy induced microhemorrhages are associated with activated perivascular macrophages and peripheral monocyte recruitment in Alzheimer’s disease mice. Molecular Neurodegeneration 2023, 18, 59. [Google Scholar] [CrossRef]
- Guilarte, T.R. Imaging neuroinflammation with TSPO: A new perspective on the cellular sources and subcellular localization. Pharmacology & therapeutics 2022, 234, 108048. [Google Scholar]
- Nutma, E. Translocator protein is a marker of activated microglia in rodent models but not human neurodegenerative diseases. Nature Communications 2023, 14, 5247. [Google Scholar] [CrossRef]
- Sacher, C. Longitudinal PET monitoring of amyloidosis and microglial activation in a second-generation amyloid-β mouse model. Journal of Nuclear Medicine 2019, 60, 1787–1793. [Google Scholar] [CrossRef]
- Van Camp, N. TSPO imaging in animal models of brain diseases. European Journal of Nuclear Medicine and Molecular Imaging 2021, 49, 77–109. [Google Scholar] [CrossRef] [PubMed]
- Heneka, M.T. Acute treatment with the PPARγ agonist pioglitazone and ibuprofen reduces glial inflammation and Aβ1–42 levels in APPV717I transgenic mice. Brain 2005, 128, 1442–1453. [Google Scholar] [CrossRef]
- Yamanaka, M. PPARγ/RXRα-induced and CD36-mediated microglial amyloid-β phagocytosis results in cognitive improvement in amyloid precursor protein/presenilin 1 mice. Journal of Neuroscience 2012, 32, 17321–17331. [Google Scholar] [CrossRef] [PubMed]
- Blume, T. Chronic PPARγ stimulation shifts amyloidosis to higher fibrillarity but improves cognition. Frontiers in aging neuroscience 2022, 14, 854031. [Google Scholar] [CrossRef]
- Biechele, G. Pre-therapeutic microglia activation and sex determine therapy effects of chronic immunomodulation. Theranostics 2021, 11, 8964. [Google Scholar] [CrossRef]
- Zatcepin, A. Regional desynchronization of microglial activity is associated with cognitive decline in Alzheimer’s disease. Molecular neurodegeneration 2024, 19, 64. [Google Scholar] [CrossRef] [PubMed]
- Sevigny, J. The antibody aducanumab reduces Aβ plaques in Alzheimer’s disease. Nature 2016, 537, 50–56. [Google Scholar] [CrossRef]
- LeVine, H., III; Walker, L.C. What amyloid ligands can tell us about molecular polymorphism and disease. Neurobiology of aging 2016, 42, 205–212. [Google Scholar] [CrossRef]
- Sabri, O. Florbetaben PET imaging to detect amyloid beta plaques in Alzheimer's disease: phase 3 study. Alzheimer's & dementia 2015, 11, 964–974. [Google Scholar]
- Bales, K.R. Apolipoprotein E is essential for amyloid deposition in the APP V717F transgenic mouse model of Alzheimer's disease. Proceedings of the National Academy of Sciences 1999, 96, 15233–15238. [Google Scholar] [CrossRef]
- Holtzman, D.M. Apolipoprotein E isoform-dependent amyloid deposition and neuritic degeneration in a mouse model of Alzheimer's disease. Proceedings of the National Academy of Sciences 2000, 97, 2892–2897. [Google Scholar] [CrossRef]
- Abrahamson, E.E. Development of a PET radioligand selective for cerebral amyloid angiopathy. Nuclear medicine and biology 2021, 92, 85–96. [Google Scholar] [CrossRef]
- Geldmacher, D.S. A randomized pilot clinical trial of the safety of pioglitazone in treatment of patients with Alzheimer disease. Archives of neurology 2011, 68, 45–50. [Google Scholar] [CrossRef]
- Burns, D.K. Safety and efficacy of pioglitazone for the delay of cognitive impairment in people at risk of Alzheimer's disease (TOMMORROW): a prognostic biomarker study and a phase 3, randomised, double-blind, placebo-controlled trial. The Lancet Neurology 2021, 20, 537–547. [Google Scholar] [CrossRef]
- Lebovitz, H.E. Thiazolidinediones: the forgotten diabetes medications. Current diabetes reports 2019, 19, 151. [Google Scholar] [CrossRef]
- Viscoli, C.M. Pioglitazone and risk for bone fracture: safety data from a randomized clinical trial. The Journal of Clinical Endocrinology & Metabolism 2017, 102, 914–922. [Google Scholar]
- Van Dyck, C.H. Lecanemab in early Alzheimer’s disease. New England Journal of Medicine 2023, 388, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Sims, J.R. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. Jama 2023, 330, 512–527. [Google Scholar] [CrossRef]
- Golde, T.E. Disease-modifying therapies for Alzheimer's disease: more questions than answers. Neurotherapeutics 2022, 19, 209–227. [Google Scholar] [CrossRef]
- Cummings, J.L. Maximizing the benefit and managing the risk of anti-amyloid monoclonal antibody therapy for Alzheimer's disease: strategies and research directions. Neurotherapeutics 2025, 22, e00570. [Google Scholar] [CrossRef]
- Biechele, G. Associations between sex, body mass index and the individual microglial response in Alzheimer’s disease. Journal of Neuroinflammation 2024, 21, 30. [Google Scholar] [CrossRef]
- Garland, E.F. The microglial translocator protein (TSPO) in Alzheimer’s disease reflects a phagocytic phenotype. Acta Neuropathologica 2024, 148, 62. [Google Scholar] [CrossRef]
- Plowey, E.D. Alzheimer disease neuropathology in a patient previously treated with aducanumab. Acta neuropathologica 2022, 144, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Whyte, L.S. Reduction in open field activity in the absence of memory deficits in the AppNL− G− F knock-in mouse model of Alzheimer’s disease. Behavioural brain research 2018, 336, 177–181. [Google Scholar] [CrossRef]
- Mehla, J. Age-dependent behavioral and biochemical characterization of single APP knock-in mouse (APPNL-GF/NL-GF) model of Alzheimer's disease. Neurobiology of Aging 2019, 75, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Saito, T. Single App knock-in mouse models of Alzheimer's disease. Nature neuroscience 2014, 17, 661–663. [Google Scholar] [CrossRef]
- Paganetti, P.A. Amyloid precursor protein truncated at any of the γ-secretase sites is not cleaved to β-amyloid. Journal of neuroscience research 1996, 46, 283–293. [Google Scholar] [CrossRef]
- Parhizkar, S. Loss of TREM2 function increases amyloid seeding but reduces plaque-associated ApoE. Nat Neurosci Correction in Nat Neurosci 2024, 27, 1858–1859.. 2019, 22, 191–204. [Google Scholar] [CrossRef]
- Page, R.M. Generation of Abeta38 and Abeta42 is independently and differentially affected by familial Alzheimer disease-associated presenilin mutations and gamma-secretase modulation. J Biol Chem 2008, 283, 677–83. [Google Scholar] [CrossRef]
- Brendel, M. Glial activation and glucose metabolism in a transgenic amyloid mouse model: a triple-tracer PET study. Journal of Nuclear Medicine 2016, 57, 954–960. [Google Scholar] [CrossRef]
- Brendel, M. Cross-sectional comparison of small animal [18F]-florbetaben amyloid-PET between transgenic AD mouse models. PloS one 2015, 10, e0116678. [Google Scholar] [CrossRef]
- Overhoff, F. Automated spatial brain normalization and hindbrain white matter reference tissue give improved [18F]-florbetaben PET quantitation in Alzheimer's model mice. Frontiers in neuroscience 2016, 10, 45. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y. A three-dimensional digital atlas database of the adult C57BL/6J mouse brain by magnetic resonance microscopy. Neuroscience 2005, 135, 1203–1215. [Google Scholar] [CrossRef] [PubMed]
- Brett, M. nipy/nibabel: 5.3.1; Zenodo, 2024. [Google Scholar]
- Zatcepin, A. molecular-connectivity. 2025.
- Huber, M. Metabolic correlates of dopaminergic loss in dementia with Lewy bodies. Movement disorders 2020, 35, 595–605. [Google Scholar] [CrossRef]
- Shapiro, S.S.; Wilk, M.B. An analysis of variance test for normality (complete samples). Biometrika 1965, 52, 591–611. [Google Scholar] [CrossRef]
- Mann, H.B.; Whitney, D.R. On a test of whether one of two random variables is stochastically larger than the other. The annals of mathematical statistics 1947, 50–60. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of the Royal statistical society: series B (Methodological) 1995, 57, 289–300. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



