Submitted:
07 April 2026
Posted:
08 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
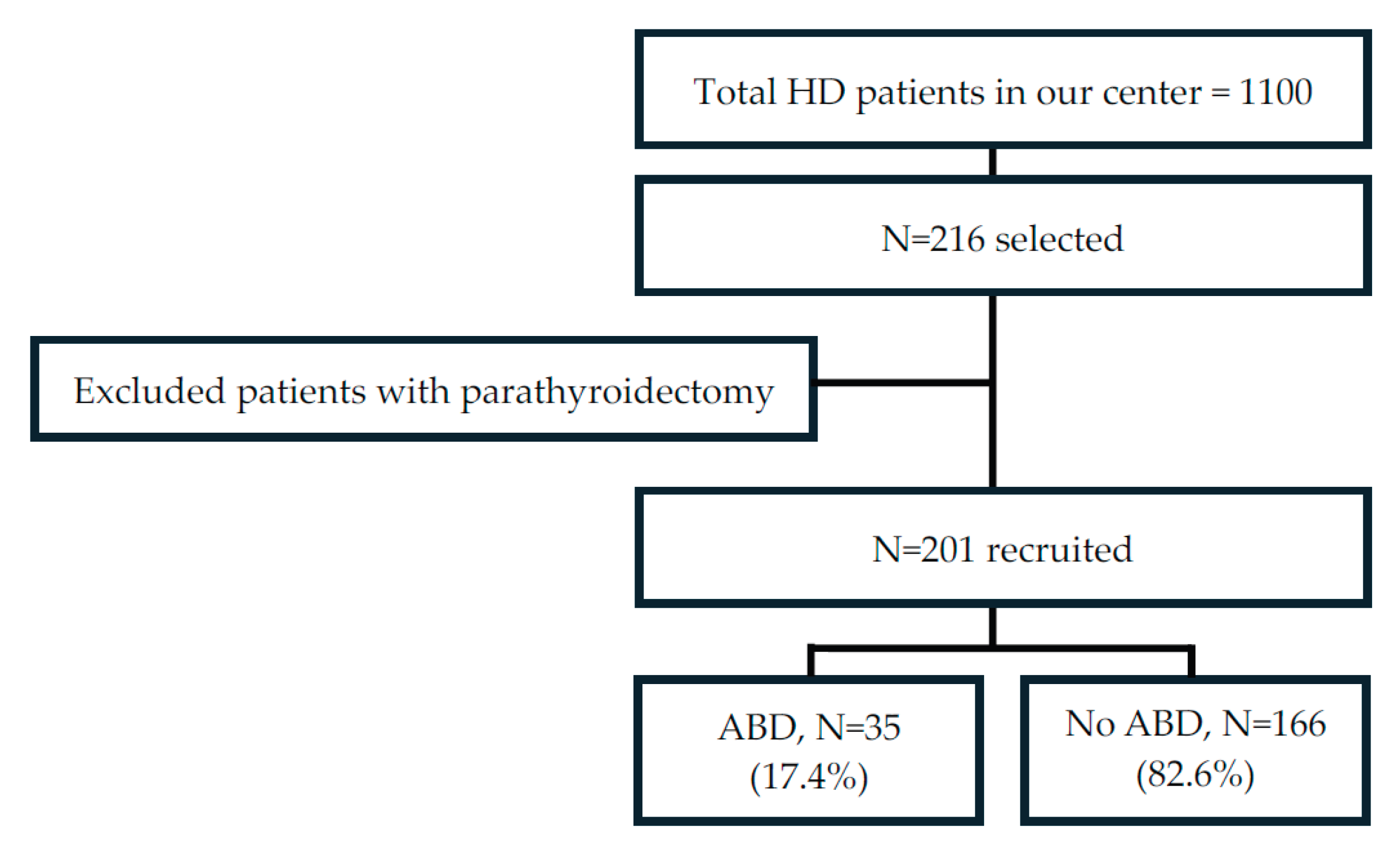
2.1. Study Design
2.2. Inclusion and Exclusion Criteria
2.3. Data Collection
2.4. Statistical Analysis
3. Results
Abbreviations: HD, hemodialysis; ABD, adynamic bone disease
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Deng, L.; et al. Global, regional, and national burden of chronic kidney disease and its underlying etiologies from 1990 to 2021: a systematic analysis for the Global Burden of Disease Study 2021. BMC Public Health 2025, 25, 636. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; et al. Temporal trends in prevalence and mortality for chronic kidney disease in China from 1990 to 2019: an analysis of the Global Burden of Disease Study 2019. Clin Kidney J 2023, 16, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Collaboration, G.B.D.C.K.D., Global, regional, and national burden of chronic kidney disease, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [CrossRef] [PubMed]
- Huijben, J.A.; et al. Increasing numbers and improved overall survival of patients on kidney replacement therapy over the last decade in Europe: an ERA Registry study. Nephrol Dial Transplant 2023, 38, 1027–1040. [Google Scholar] [CrossRef] [PubMed]
- MacRae, C.; et al. Comorbidity in chronic kidney disease: a large cross-sectional study of prevalence in Scottish primary care. Br J Gen Pract 2021, 71, e243–e249. [Google Scholar] [CrossRef] [PubMed]
- London, G.M.; et al. Arterial calcifications and bone histomorphometry in end-stage renal disease. J Am Soc Nephrol 2004, 15, 1943–51. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.T.; et al. Worldwide Epidemiology of Diabetes-Related End-Stage Renal Disease, 2000-2015. Diabetes Care 2021, 44, 89–97. [Google Scholar] [CrossRef]
- Sanches, C.P., A.G.D. Vianna, and F.C. Barreto. The impact of type 2 diabetes on bone metabolism. Diabetol Metab Syndr 2017, 9, 85. [CrossRef]
- Goodman, W.G.; et al. Development of adynamic bone in patients with secondary hyperparathyroidism after intermittent calcitriol therapy. Kidney Int 1994, 46, 1160–6. [Google Scholar] [CrossRef]
- Andress, D.L. Adynamic bone in patients with chronic kidney disease. Kidney International 2008, 73, 1345–1354. [Google Scholar] [CrossRef] [PubMed]
- Sprague, S.M.; et al. Diagnostic Accuracy of Bone Turnover Markers and Bone Histology in Patients With CKD Treated by Dialysis. Am J Kidney Dis 2016, 67, 559–66. [Google Scholar] [CrossRef] [PubMed]
- Bover, J.; et al. Adynamic bone disease: from bone to vessels in chronic kidney disease. Semin Nephrol 2014, 34, 626–40. [Google Scholar] [CrossRef] [PubMed]
- Malluche, H.H., H.W. Mawad, and M.C. Monier-Faugere. Renal osteodystrophy in the first decade of the new millennium: analysis of 630 bone biopsies in black and white patients. J Bone Miner Res 2011, 26, 1368–76. [CrossRef] [PubMed]
- Coco, M.; Rush, H. Increased incidence of hip fractures in dialysis patients with low serum parathyroid hormone. Am J Kidney Dis 2000, 36, 1115–21. [Google Scholar] [CrossRef]
- Naves-Diaz, M.; et al. Calcium, phosphorus, PTH and death rates in a large sample of dialysis patients from Latin America. The CORES Study. Nephrol Dial Transplant 2011, 26, 1938–47. [Google Scholar] [CrossRef] [PubMed]
- R Geethanjali, M.G., V Jayaprakash, E Ramprasad, A Manikandan, M Rakchna. Prevalence and biochemical profile of adynamic bone disease in a hemodialysis population. Biomedicine 2023, 43, 582–586. [CrossRef]
- Dalle Carbonare, L.; et al. Bone Biopsy for Histomorphometry in Chronic Kidney Disease (CKD): State-of-the-Art and New Perspectives. J Clin Med 2021, 10. [Google Scholar] [CrossRef] [PubMed]
| Variables | Overall (N=201) |
Adynamic bone disease (N = 35) |
No adynamic bone disease (N = 166) |
|
|---|---|---|---|---|
| Age, mean (SD), years | 64.50 (12.50) | 68.70 (12.50) | 63.60 (12.40) | |
| Gender, N (%) | ||||
| Male | 123.00 (61.20) | 21.00 (60.00) | 102.00 (61.40) | |
| Female | 78.00 (38.80) | 14.00 (40.00) | 64.00 (38.60) | |
| Dialysis vintage, median (IQR), months | 54.00 (30, 80) | 41.00 (18, 78) | 56.00 (31, 90) | |
| Etiology of kidney failure, N (%) | ||||
| Diabetes mellitus | 124.00 (61.70) | 25.00 (71.40) | 99.00 (59.60) | |
| Hypertension | 34.00 (16.90) | 5.00 (14.30) | 29.00 (17.50) | |
| Chronic glomerulonephritis | 35.00 (17.40) | 3.00 (8.60) | 32.00 (19.30) | |
| Others | 8.00 (4.00) | 2.00 (5.70) | 6.00 (3.60) | |
| Diabetes mellitus, N (%) | ||||
| Yes | 144.00 (71.60) | 29.00 (82.90) | 115.00 (69.30) | |
| No | 57.00 (28.40) | 6.00 (17.10) | 51.00 (30.70) | |
| Dialysate calcium, N (%), mmol/L | ||||
| Normal (1.5) | 111.00 (55.20) | 22.00 (62.90) | 89.00 (53.60) | |
| Low (1.25) | 90.00 (44.80) | 13.00 (37.10) | 77.00 (46.40) | |
| Serum calcium, mean (SD), mmol/L | 2.27 (0.14) | 2.30 (0.13) | 2.27 (0.14) | |
| Serum phosphate, mean (SD), mmol/L | 1.56 (0.42) | 1.47 (0.37) | 1.57 (0.43) | |
| Serum alkaline phosphatase, mean (SD), U/L | 143.73 (159.33) | 100.90 (113.91) | 152.76 (166.22) | |
| Serum albumin, mean (SD), g/L | 39.25 (4.60) | 38.91 (5.08) | 39.32 (4.50) | |
| Calcium-based phosphate binder use, N (%) | ||||
| Yes | 160.00 (79.6) | 30.00 (85.70) | 130.00 (78.30) | |
| No | 41.00 (20.4) | 5.00 (14.30) | 36.00 (21.70) | |
| Non-calcium-based phosphate binder use, N (%) | ||||
| Yes | 50.00 (24.9) | 1.00 (2.90) | 49.00 (29.50) | |
| No | 151.00 (75.1) | 34.00 (97.10) | 117.00 (70.50) | |
| Calcitriol or vitamin D analogue use, N (%) | ||||
| Yes | 119.00 (59.2) | 10.00 (28.60) | 109.00 (65.70) | |
| No | 82.00 (40.8) | 25.00 (71.40) | 57.00 (34.30) | |
| Calcimimetic use, N (%) | ||||
| Yes | 22.00 (11.00) | 0 (0) | 22.00 (13.30) | |
| No | 179.00 (89.00) | 35.00 (100.00) | 144.00 (86.70) |
| Variables | Univariable model | |
|---|---|---|
| Crude OR (95% CI) | p value* | |
| Age | 1.04 (1.01-1.07) | 0.03 |
| Gender | ||
| Male | 0.94 (0.45-1.98) | 0.87 |
| Female | Reference | |
| Etiology of kidney failure | ||
| Diabetes mellitus | Reference | |
| Hypertension | 0.68 (0.24-1.94) | 0.47 |
| Chronic glomerulonephritis | 0.37 (0.11-1.31) | 0.12 |
| Others | 1.32 (0.25-6.94) | 0.74 |
| Dialysis vintage | 1.00 (0.99-1.01) | 0.55 |
| Diabetes mellitus | ||
| Yes | 2.14 (0.84-5.48) | 0.11 |
| No | Reference | |
| Dialysate calcium, mmol/L | ||
| Normal (1.5) | Reference | |
| Low (1.25) | 0.68 (0.32-1.45) | 0.32 |
| Serum calcium, in 0.1mmol/L | 1.21 (0.93-1.57) | 0.16 |
| Serum phosphate, mmol/L) | 0.54 (0.21-1.37) | 0.19 |
| Serum alkaline phosphatase, U/L | 0.99 (0.98-1.00) | 0.038 |
| Serum albumin, g/L | 0.98 (0.91-1.06) | 0.64 |
| Calcium-based phosphate binder use | ||
| Yes | 1.66 (0.60-4.59) | 0.34 |
| No | Reference | |
| Non-calcium-based phosphate binder use | ||
| Yes | 0.07 (0.00-0.52) | 0.01 |
| No | Reference | |
| Calcitriol or vitamin D analogue use | ||
| Yes | 0.21 (0.09-0.47) | <0.001 |
| No | Reference | |
| Variables | Multivariable model | |
|---|---|---|
| Adjusted OR (95% CI) | p value* | |
| Age | 1.02 (0.99-1.06) | 0.22 |
| Diabetes mellitus | ||
| Yes | 2.32 (0.80-6.69) | 0.12 |
| No | Reference | |
| Dialysate calcium, mmol/L | ||
| Normal (1.5) | Reference | |
| Low (1.25) | 0.55 (0.23-1.33) | 0.18 |
| Serum calcium, in 0.1mmol/L | 1.68 (1.15-2.45) | 0.007 |
| Serum alkaline phosphatase, U/L | 1.00 (0.99-1.00) | 0.40 |
| Non-calcium-based phosphate binder use | ||
| Yes | 0.05 (0.00-0.47) | 0.008 |
| No | Reference | |
| Calcitriol or vitamin D analogue use | ||
| Yes | 0.20 (0.08-0.50) | <0.001 |
| No | Reference | |
| Complications | Adynamic bone disease (N = 35) |
No adynamic bone disease (N = 166) |
p value |
|---|---|---|---|
| Ischemic heart disease, N (%) | |||
| Yes | 21.00 (60.00) | 77.00 (46.39) | 0.143 |
| No | 14.00 (40.00) | 89.00 (53.61) | |
| Peripheral vascular disease, N (%) | |||
| Yes | 5.00 (14.29) | 29.00 (17.47) | 0.648 |
| No | 30.00 (85.71) | 137.00 (82.53) | |
| Stroke, N (%) | |||
| Yes | 6.00 (17.14) | 29.00 (17.47) | 0.963 |
| No | 29.00 (82.86) | 137.00 (82.53) | |
| Fracture, N (%) | |||
| Yes | 4.00 (11.43) | 30.00 (18.07) | 0.341 |
| No | 31.00 (88.57) | 136.00 (81.93) | |
| Mortality , N (%) | |||
| Yes | 3.00 (8.57) | 10.00 (6.02) | 0.578 |
| No | 32.00 (91.43) | 156.00 (93.98) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




