Submitted:
03 April 2026
Posted:
08 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Cadmium Exposure Limits and Toxicity Thresholds
2.1. Food and Environmental Safety Standards
2.1. Toxicity Threshold Definition
2.2. Official Dietary Exposure Guidelines
2.2.1. JECFA/TDI
2.2.2. EFSA/RfD
2.2.3. US FDA/TRV
2.2.4. ATSDR/MRL
2.3. Other Exposure Limits and Thresholds Derived for Oral Cd
2.4. Cadmium Inhalational Exposure Limits for Workers
2.5. Summary on Official Cd Exposure Limits
3. Cd Excretion Threshold Levels Based on Disease Endpoints
3.1. Excretion of Low-Molecular Weight Proteins: Tubular Proteinuria
3.2. From NOAEL to BMDL
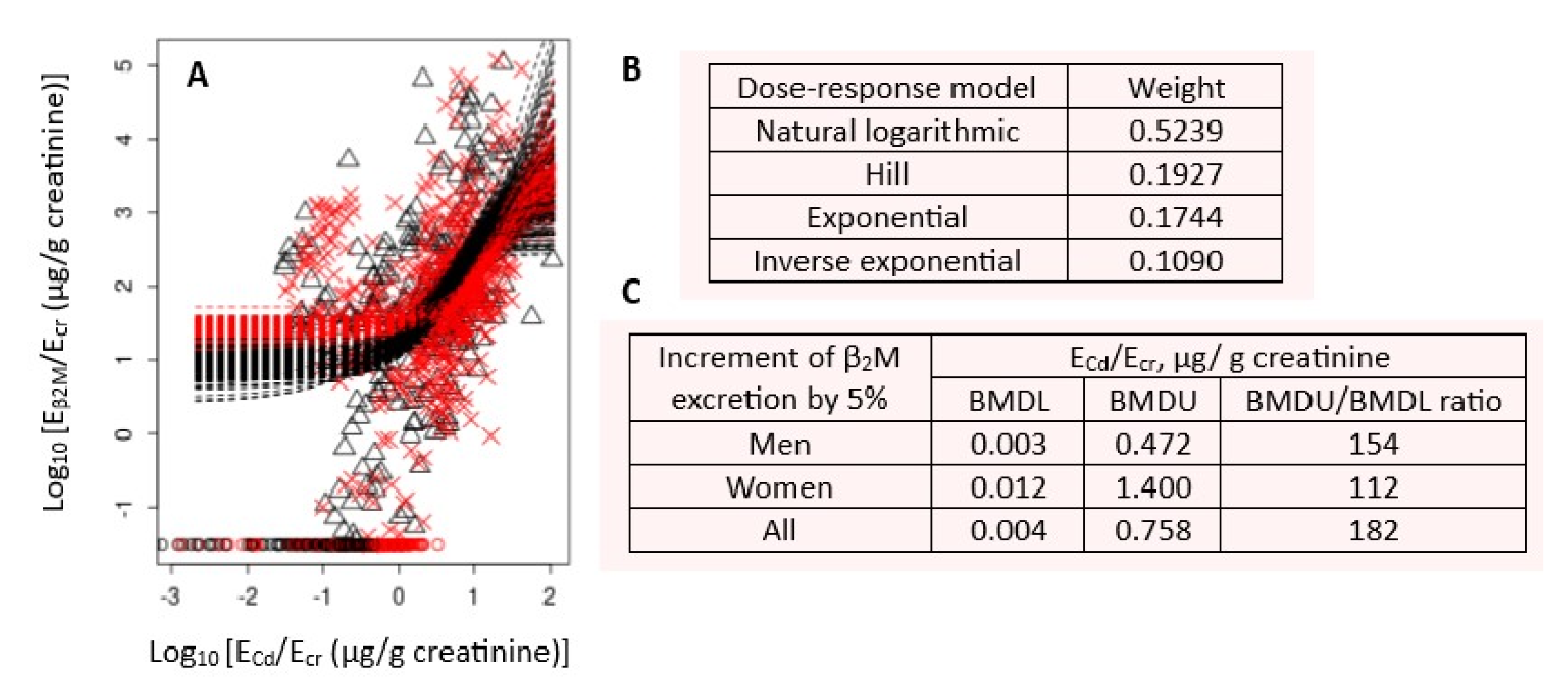
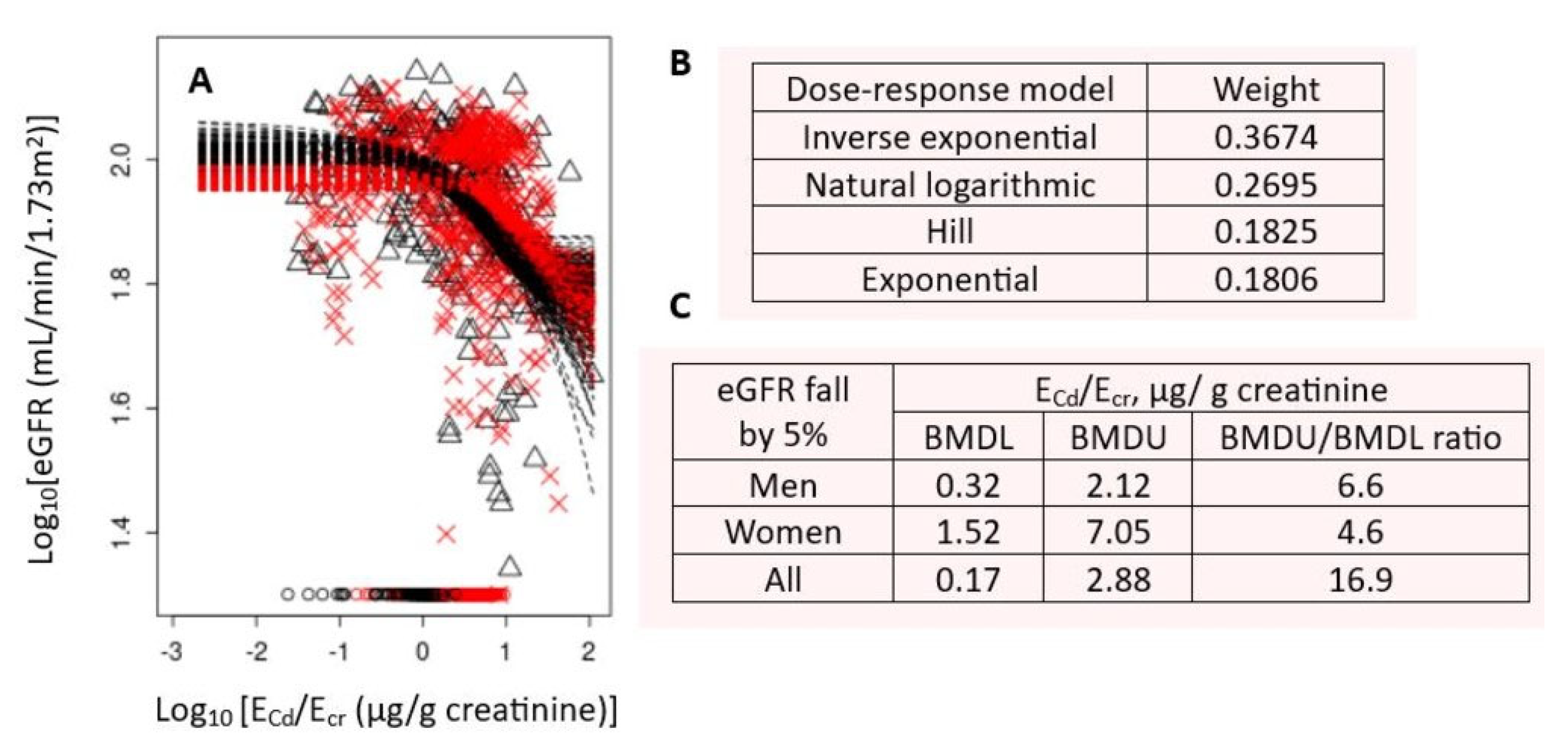
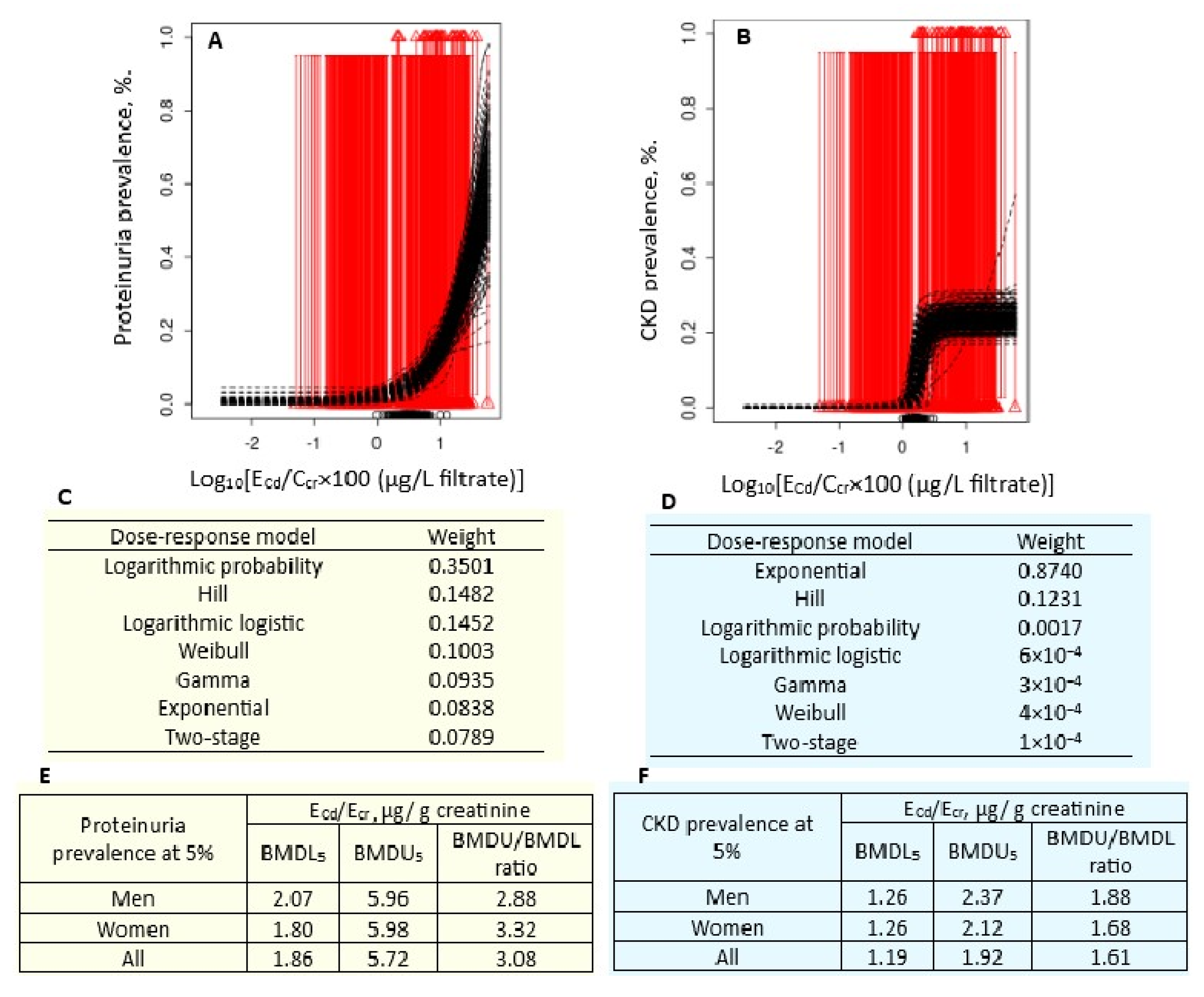
3.3. Multiple Mathematic Dose-Response Models: The Akaike Information Criterion (AIC)
3.3.1. Comparing Cd excretion benchmarks for the β2M and other endpoints
3.3.2. Comparing Benchmark Cd Excretion Rates Derived from Human Population Data
3.4. Summary on Cd Toxicity Thresholds for the Kidney Target
4. Mechanism-Based Strategies to Mitigate Cd Toxicity
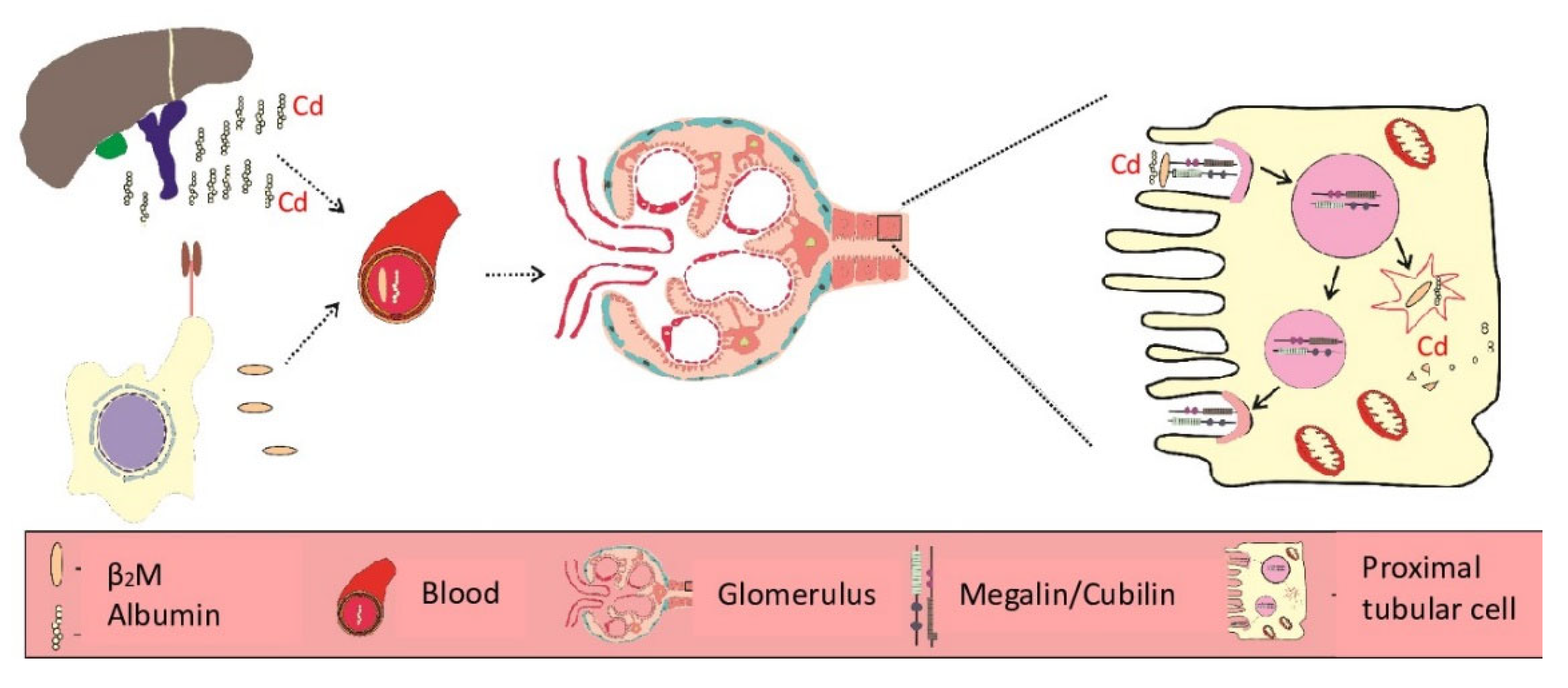
4.1. Why Most Acquired Cadmium Accumulates in the Proximal Tubules?
4.2. Cd-Induced Albuminuria: Glomerular or Tubular Cause
4.3. Albuminuria/Proteinuria and CKD
4.4. CKD and Its Progression Toward Kidney Failure
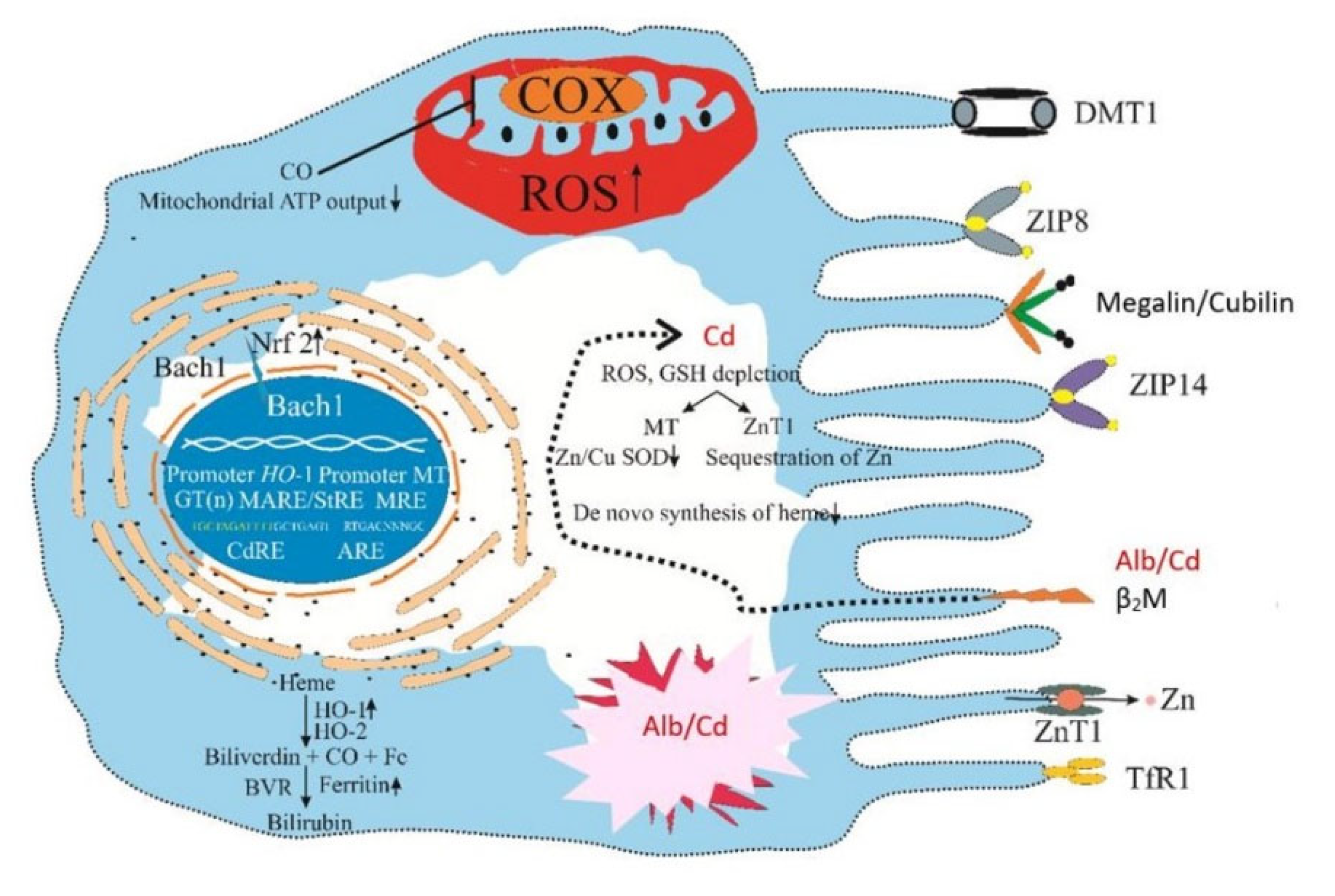
4.5. Cd and Cellular Stress Responses
4.5.1. Cd-Induced Upregulation of the MT Gene
4.5.2. Cd-Induced Upregulation of the HO-1 Gene
4.6. Can Exogenous HO-1 Inducers Mitigate Cd-Induced Oxidative Stress?
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Satarug, S.; Phelps, K.R. Cadmium Exposure and Toxicity. In Metal Toxicology Handbook, Ch 14; Bagchi, D., Bagchi, M., Eds.; CRC Press: New York, NY, USA, 2020; pp. 219–272. [Google Scholar]
- Satarug, S.; Vesey, D.A.; Gobe, G.C.; Phelps, K.R. Estimation of health risks associated with dietary cadmium exposure. Arch. Toxicol. 2023, 97, 329–358. [Google Scholar] [CrossRef]
- Hsu, P.; Gamlat, C.J.; Felicia Wu, F. Assessing Causal Evidence Beyond Correlation: Linking Cadmium Exposure to Disease, Using Bradford Hill Framework. Expos. Health 2026, 18, 19. [Google Scholar] [CrossRef]
- Satarug, S. Hypertension in People Exposed to Environmental Cadmium: Roles for 20-Hydroxyeicosatetraenoic Acid in the Kidney. J. Xenobiot. 2025, 15, 122. [Google Scholar] [CrossRef] [PubMed]
- Lichtler, R.; Cowley, M. Environmental Contaminants, Iron Deficiency, and Iron-Deficiency Anemia: A Review of the Liter-ature. Scientifica 2025, 2025, 5007983. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Lin, H.; Zhang, M.; Meng, J.; Hu, L.; Yu, B. Urine Cadmium as a Risk Factor for Osteoporosis and Osteopenia: A Meta-Analysis. Front. Med. 2021, 8, 648902. [Google Scholar] [CrossRef] [PubMed]
- Ougier, E.; Fiore, K.; Rousselle, C.; Assunção, R.; Martins, C.; Buekers, J. Burden of osteoporosis and costs associated with human biomonitored cadmium exposure in three European countries: France, Spain and Belgium. Int. J. Hyg. Environ. Health 2021, 234, 113747. [Google Scholar] [CrossRef]
- Pouillot, R.; Santillana Farakos, S.; Van Doren, J.M. Modeling the risk of low bone mass and osteoporosis as a function of urinary cadmium in U.S adults aged 50–79 years. Environ. Res. 2022, 212, 113315. [Google Scholar] [CrossRef]
- Filippini, T.; Wise, L.A.; Vinceti, M. Cadmium exposure and risk of diabetes and prediabetes: A systematic review and dose-response meta-analysis. Environ. Int. 2022, 158, 106920. [Google Scholar] [CrossRef]
- Satarug, S. Is Environmental Cadmium Exposure Causally Related to Diabetes and Obesity? Cells 2024, 13, 83. [Google Scholar] [CrossRef]
- Doccioli, C.; Sera, F.; Francavilla, A.; Cupisti, A.; Biggeri, A. Association of cadmium environmental exposure with chronic kidney disease: A systematic review and meta-analysis. Sci. Total Environ. 2024, 906, 167165. [Google Scholar] [CrossRef]
- Satarug, S. Is Chronic Kidney Disease Due to Cadmium Exposure Inevitable and Can It Be Reversed? Biomedicines 2024, 12, 718. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Perrais, M.; Golshayan, D.; Wuerzner, G.; Vaucher, J.; Thomas, A.; Marques-Vidal, P. Association between urinary heavy metal/trace element concentrations and kidney function: A prospective study. Clin. Kidney J. 2024, 18, sfae378. [Google Scholar] [CrossRef] [PubMed]
- Elinder, C.G.; Lind, B.; Kjellström, T.; Linnman, L.; Friberg, L. Cadmium in kidney cortex, liver, and pancreas from Swedish autopsies. Estimation of biological half time in kidney cortex, considering calorie intake and smoking habits. Arch. Environ. Health 1976, 31, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Suwazono, Y.; Kido, T.; Nakagawa, H.; Nishijo, M.; Honda, R.; Kobayashi, E.; Dochi, M.; Nogawa, K. Biological half-life of cadmium in the urine of inhabitants after cessation of cadmium exposure. Biomarkers 2009, 14, 77–81. [Google Scholar] [CrossRef]
- Ishizaki, M.; Suwazono, Y.; Kido, T.; Nishijo, M.; Honda, R.; Kobayashi, E.; Nogawa, K.; Nakagawa, H. Estimation of biological half-life of urinary cadmium in inhabitants after cessation of environmental cadmium pollution using a mixed linear model. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2015, 32, 1273–1276. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Ruangyuttikarn, W.; Nishijo, M.; Gobe, G.C.; Phelps, K.R. The Source and Pathophysiologic Significance of Excreted Cadmium. Toxics 2019, 7, 55. [Google Scholar] [CrossRef]
- Akerstrom, M.; Barregard, L.; Lundh, T.; Sallsten, G. The relationship between cadmium in kidney and cadmium in urine and blood in an environmentally exposed population. Toxicol. Appl. Pharmacol. 2013, 268, 286–293. [Google Scholar] [CrossRef]
- Satarug, S. Antioxidative Function of Zinc and Its Protection Against the Onset and Progression of Kidney Disease Due to Cadmium. Biomolecules 2025, 15, 183. [Google Scholar] [CrossRef]
- Cirovic, A.; Milovanovic, P.; Satarug, S. Cadmium, Iron Deficiency Anemia and Hypophosphatemic Osteomalacia Due to Intravenous Iron Supplementation. Biomedicines 2026, 14, 292. [Google Scholar] [CrossRef]
- Wong, C.; Roberts, S.M.; Saab, I.N. Review of regulatory reference values and background levels for heavy metals in the human diet. Regul. Toxicol. Pharmacol. 2022, 130, 105122. [Google Scholar] [CrossRef]
- Satarug, S. Challenges in Toxicological Risk Assessment of Environmental Cadmium Exposure. Toxics 2025, 13, 404. [Google Scholar] [CrossRef] [PubMed]
- Costa, L.G.; Aschner, M.; Vitalone, A.; Syversen, T.; Soldin, O.P. Developmental neuropathology of environmental agents. Annu. Rev. Pharmacol. Toxicol. 2004, 44, 87–110. [Google Scholar] [CrossRef] [PubMed]
- Hauptman, M.; Woolf, A.D. Childhood Ingestions of Environmental Toxins: What Are the Risks? Pediatr. Ann. 2017, 46, e466–e471. [Google Scholar] [CrossRef] [PubMed]
- Vogt, R.; Bennett, D.; Cassady, D.; Frost, J.; Ritz, B.; Hertz-Picciotto, I. Cancer and non-cancer health effects from food con-taminant exposures for children and adults in California: A risk assessment. Environ. Health 2012, 11, 83. [Google Scholar] [CrossRef]
- Flannery, B.M.; Schaefer, H.R.; Middleton, K.B. A scoping review of infant and children health effects associated with cadmium exposure. Regul. Toxicol. Pharmacol. 2022, 131, 105155. [Google Scholar] [CrossRef]
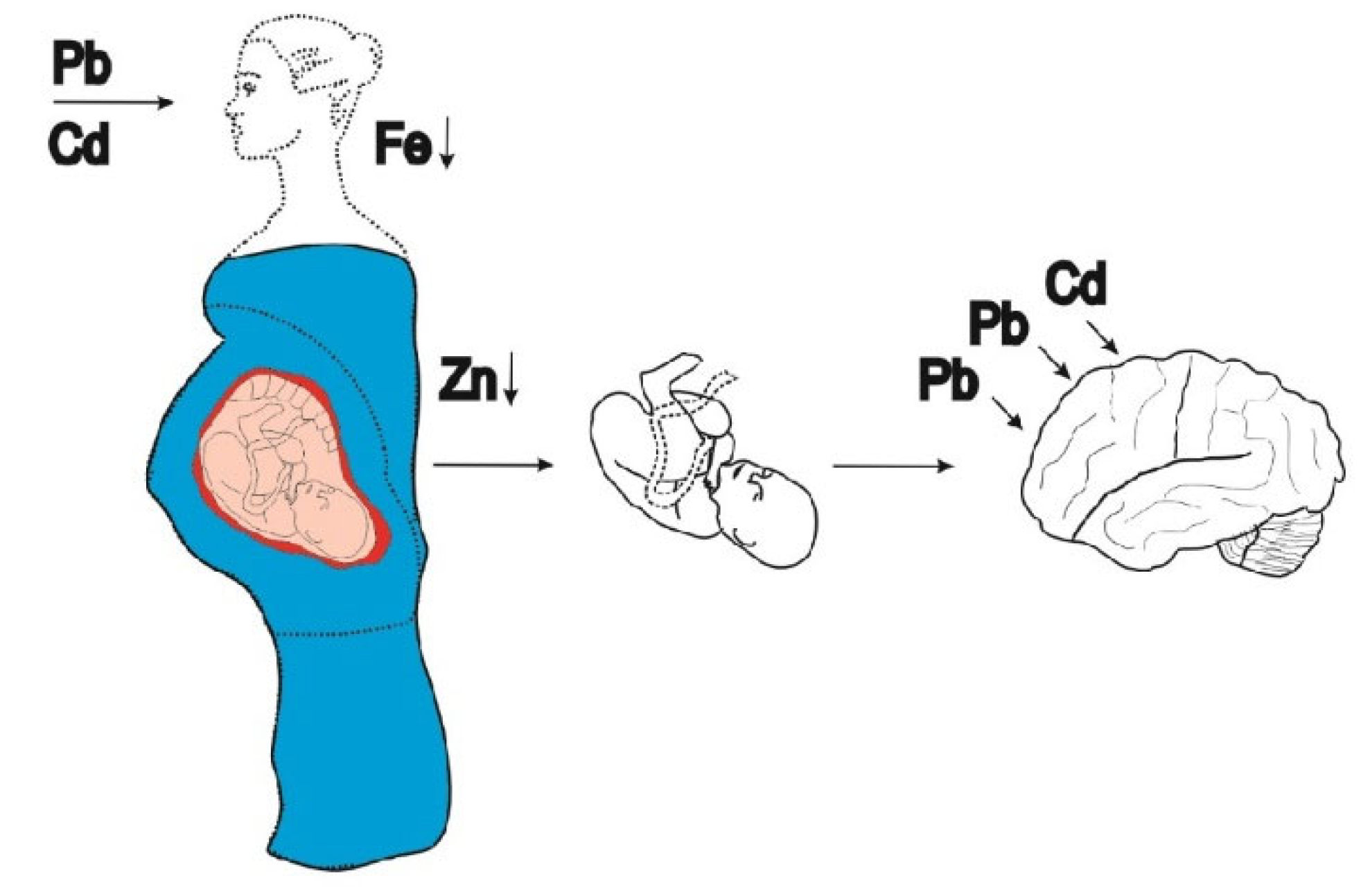
- Chen, Z; Myers, R; Wei, T; Bind, E; Kassim, P; Wang, G; Ji, Y; Hong, X; Caruso, D; Bartell, T; Gong, Y; Strickland, P; Navas-Acien, A; Guallar, E; Wang, X. Placental transfer and concentrations of cadmium, mercury, lead, and selenium in mothers, newborns, and young children. J. Expo. Sci. Environ. Epidemiol. 2014, 24, 537–544. [Google Scholar] [CrossRef]
- Kippler, M.; Hoque, A.M.; Raqib, R.; Ohrvik, H.; Ekström, E.C.; Vahter, M. Accumulation of cadmium in human placenta interacts with the transport of micronutrients to the fetus. Toxicol. Lett. 2010, 192, 162–168. [Google Scholar] [CrossRef]
- Huang, S.; Kuang, J.; Zhou, F.; Jia, Q.; Lu, Q.; Feng, C.; Yang, W.; Fan, G. The association between prenatal cadmium exposure and birth weight: A systematic review and meta-analysis of available evidence. Environ. Pollut. 2019, 251, 699–707. [Google Scholar] [CrossRef]
- Amegah, A.K.; Sewor, C.; Jaakkola, J.J.K. Cadmium exposure and risk of adverse pregnancy and birth outcomes: a systematic review and dose-response meta-analysis of cohort and cohort-based case-control studies. J. Expo. Sci. Environ. Epidemiol. 2021, 31, 299–317. [Google Scholar] [CrossRef]
- Kippler, M.; Lönnerdal, B.; Goessler, W.; Ekström, E.C.; Arifeen, S.E.; Vahter, M. Cadmium interacts with the transport of essential micronutrients in the mammary gland—A study in rural Bangladeshi women. Toxicology 2009, 257, 64–69. [Google Scholar] [CrossRef]
- Olszowski, T.; Baranowska-Bosiacka, I.; Rębacz-Maron, E.; Gutowska, I.; Jamioł, D.; Prokopowicz, A.; Goschorska, M.; Chlubek, D. Cadmium Concentration in Mother’s Blood, Milk, and Newborn’s Blood and Its Correlation with Fatty Acids, Anthropometric Characteristics, and Mother’s Smoking Status. Biol. Trace. Elem. Res. 2016, 174, 8–20. [Google Scholar] [CrossRef]
- Rebelo, F.M.; Caldas, E.D. Arsenic, lead, mercury and cadmium: Toxicity, levels in breast milk and the risks for breastfed infants. Environ. Res. 2016, 151, 671–688. [Google Scholar] [CrossRef] [PubMed]
- Kippler, M.; Hossain, M.B.; Lindh, C.; Moore, S.E.; Kabir, I.; Vahter, M.; Broberg, K. Early life low-level cadmium exposure is positively associated with increased oxidative stress. Environ. Res. 2012, 112, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Gustin, K.; Tofail, F.; Vahter, M.; Kippler, M. Cadmium exposure and cognitive abilities and behavior at 10 years of age: A prospective cohort study. Environ. Int. 2018, 113, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Iwai-Shimada, M.; Nakayama, S.F.; Isobe, T.; Kobayashi, Y.; Tatsuta, N.; Taniguchi, Y.; Sekiyama, M.; Michikawa, T.; Yamazaki, S.; et al. Association of prenatal exposure to cadmium with neurodevelopment in children at 2 years of age: The Japan Environment and Children’s Study. Environ Int. 2021, 156, 106762. [Google Scholar] [CrossRef]
- Hou, D.; Jia, X.; Wang, L.; McGrath, S.P.; Zhu, Y.G.; Hu, Q.; Zhao, F.J.; Bank, M.S.; O’Connor, D.; Nriagu, J. Global soil pollution by toxic metals threatens agriculture and human health. Science 2025, 388, 316–321. [Google Scholar] [CrossRef]
- Slob, W.; Moerbeek, M.; Rauniomaa, E.; Piersma, A.H. A statistical evaluation of toxicity study designs for the estimation of the benchmark dose in continuous endpoints. Toxicol. Sci. 2005, 84, 167–185. [Google Scholar] [CrossRef]
- Slob, W.; Setzer, R.W. Shape and steepness of toxicological dose-response relationships of continuous endpoints. Crit. Rev. Toxicol. 2014, 44, 270–297. [Google Scholar] [CrossRef]
- Slob, W. A general theory of effect size, and its consequences for defining the benchmark response (BMR) for continuous endpoints. Crit. Rev. Toxicol. 2017, 47, 342–351. [Google Scholar] [CrossRef]
- Satarug, S. Benchmark dose modeling to define permissible exposure levels for environmental cadmium. J. Environ. Expo. Assess. 2025, 4, 28. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Buha Đorđević, A. Use of Urinary β2-Microglobulin in the Assessment of the Health Risk from Environmental Cadmium Exposure. Appl. Sci. 2025, 15, 11757. [Google Scholar] [CrossRef]
- Faroon, O.; Keith, S.; Mumtaz, M.; Ruiz, P. Minimal Risk Level Derivation for Cadmium: Acute and Intermediate Duration Exposures. J. Exp. Clin. Toxicol. 2017, 1, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Moffett, D.B.; Mumtaz, M.M.; Sullivan, D.W., Jr.; Whittaker, M.H. Chapter 13, General Considerations of Dose-Effect and Dose-Response Relationships. In Handbook on the Toxicology of Metals;Volume I: General Considerations, 5th ed.; Nordberg, G., Costa, M., Eds.; Academic Press: Cambridge, MA, USA, 2022; pp. 299–317. [Google Scholar]
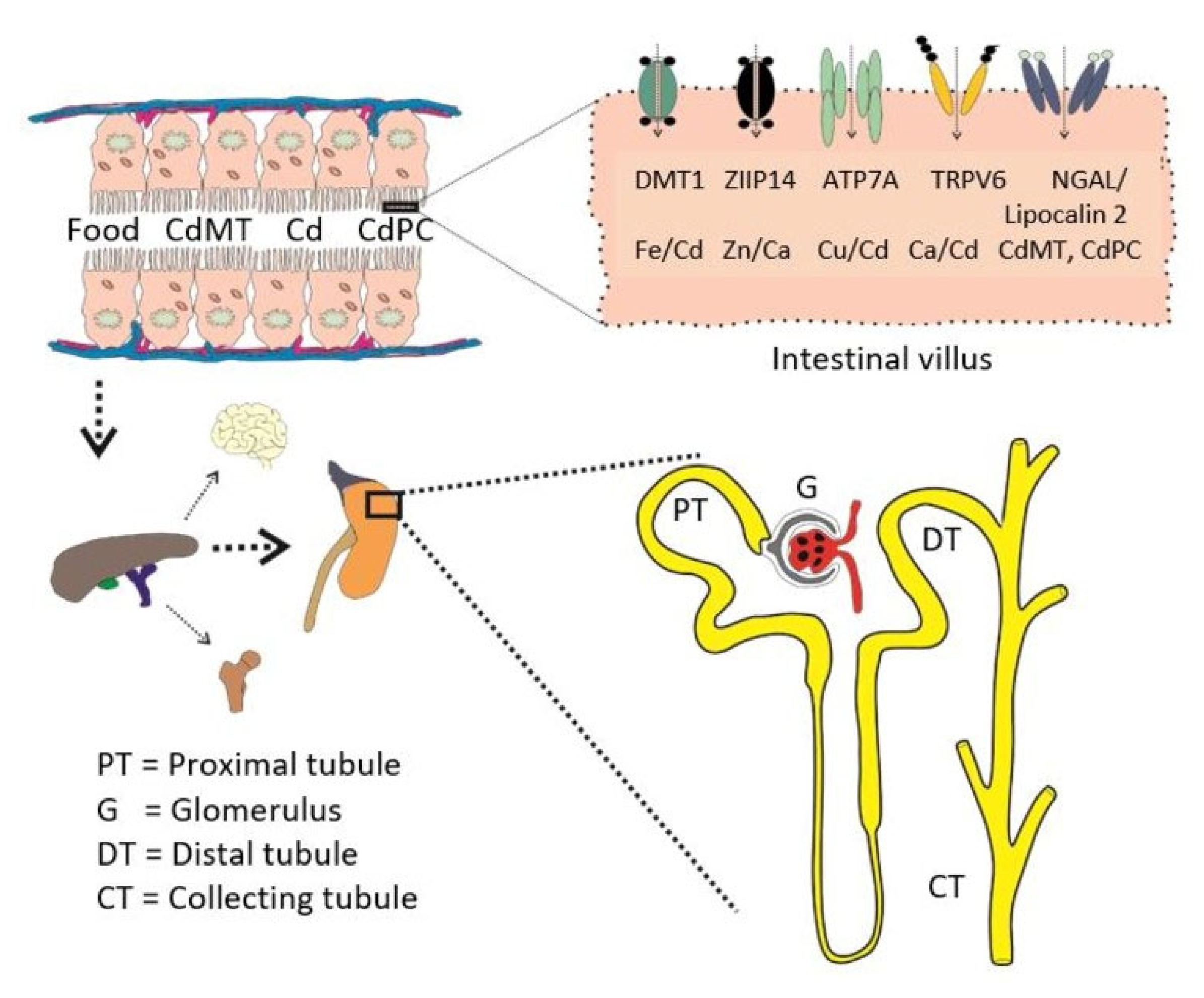
- Park, J.D.; Cherrington, N.J.; Klaassen, C.D. Intestinal absorption of cadmium is associated with divalent metal transporter 1 in rats. Toxicol. Sci. 2002, 68, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Aydemir, T.B.; Cousins, R.J. The multiple faces of the metal transporter ZIP14 (SLC39A14). J. Nutr. 2018, 148, 174–184. [Google Scholar] [CrossRef]
- Nebert, D.W. Comparing gene expression during cadmium uptake and distribution: Untreated versus oral Cd-treated wild-type and ZIP14 knockout mice. Toxicol. Sci. 2015, 143, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Ohta, H.; Ohba, K. Involvement of metal transporters in the intestinal uptake of cadmium. J. Toxicol. Sci. 2020, 45, 539–548. [Google Scholar] [CrossRef]
- Kovacs, G.; Danko, T.; Bergeron, M.J.; Balazs, B.; Suzuki, Y.; Zsembery, A.; Hediger, M.A. Heavy metal cations permeate the TRPV6 epithelial cation channel. Cell Calcium 2011, 49, 43–55. [Google Scholar] [CrossRef]
- Kovacs, G.; Montalbetti, N.; Franz, M.C.; Graeter, S.; Simonin, A.; Hediger, M.A. Human TRPV5 and TRPV6: Key players in cadmium and zinc toxicity. Cell Calcium 2013, 54, 276–286. [Google Scholar] [CrossRef]
- Fujita, Y.; el Belbasi, H.I.; Min, K.S.; Onosaka, S.; Okada, Y.; Matsumoto, Y.; Mutoh, N.; Tanaka, K. Fate of cadmium bound to phytochelatin in rats. Res. Commun. Chem. Pathol. Pharmacol. 1993, 82, 357–365. [Google Scholar]
- Langelueddecke, C.; Lee, W.K.; Thévenod, F. Differential transcytosis and toxicity of the hNGAL receptor ligands cadmium-metallothionein and cadmium-phytochelatin in colon-like Caco-2 cells: Implications for in vivo cadmium toxicity. Toxicol. Lett. 2014, 226, 228–235. [Google Scholar] [CrossRef]
- Codex Alimentarius, CODEX STAN 193–1995, General Standard for Contaminants and Toxins in Food and Feed. Available online: http://www.fao.org/fileadmin/user_upload/livestockgov/documents/1_CXS_193e.pdf (accessed on 17 March 2026).
- European Food Safety Authority (EFSA). Scientific opinion: Cadmium in food. 2009. Available online: https://efsa.onlinelibrary.wiley.com/doi/pdf/10.2903/j.efsa.2009.980 (accessed on 17 March 2026).
- Boon, P.E.; Pustjens, A.M.; Te Biesebeek, J.D.; Brust, G.M.H.; Castenmiller, J.J.M. Dietary intake and risk assessment of elements for 1- and 2-year-old children in the Netherlands. Food Chem. Toxicol. 2022, 161, 112810. [Google Scholar] [CrossRef]
- Vasco, E.; Dias, M.G.; Oliveira, L. The first harmonised total diet study in Portugal: Arsenic, cadmium and lead exposure assessment. Chemosphere 2025, 372, 144003. [Google Scholar] [CrossRef] [PubMed]
- Minh, N.G.; Hough, R.L.; Thuy, L.T.; Nyberg, Y.; Mai, L.B.; Vinh, N.C.; Khai, N.M.; Oborn, I. Assessing dietary exposure to cadmium in a metal recycling community in Vietnam: age and gender aspects. Sci. Total Environ. 2012, 416, 164–171. [Google Scholar] [CrossRef]
- Horiguchi, H.; Oguma, E.; Sasaki, S.; Miyamoto, K.; Hosoi, Y.; Ono, A.; Kayama, F. Exposure Assessment of Cadmium in Female Farmers in Cadmium-Polluted Areas in Northern Japan. Toxics 2020, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Zhang, Y.; Su, J.; Bai, Z.; Li, T.; Wu, Y. Maximum cadmium limits establishment strategy based on the dietary exposure estimation: an example from Chinese populations and subgroups. Environ. Sci. Pollut. Res. Int. 2018, 25, 18762–18771. [Google Scholar] [CrossRef] [PubMed]
- Nogawa, K.; Sakurai, M.; Ishizaki, M.; Kido, T.; Nakagawa, H.; Suwazono, Y. Threshold limit values of the cadmium concentration in rice in the development of itai-itai disease using benchmark dose analysis. J. Appl. Toxicol. 2017, 37, 962–966. [Google Scholar] [CrossRef]
- Joint FAO/WHO Expert Committee on Food Additives. Evaluation of Certain Food Additives and Contaminants (Forty-First Report of the Joint FAO/WHO Expert Committee on Food Additives); WHO Technical Report Series No. 837; World Health Organization: Geneva, Switzerland, 1993; Available online: https://www.who.int/publications/i/item/9241208376. (accessed on 17 March 2026).
- Joint FAO/WHO Expert Committee on Food Additives. Meeting. 73th & World Health Organization (2011). Safety evaluation of certain food additives and contaminants: prepared by the Seventy-third meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA); World Health Organization. World Health Organization; Available online: https://iris.who.int/handle/10665/44521 (accessed on 17 March 2026).
- European Food Safety Authority (EFSA). Scientific opinion: Statement on tolerable weekly intake for cadmium. 2011. Available online: https://efsa.onlinelibrary.wiley.com/doi/pdf/10.2903/j.efsa.2011.1975 (accessed on 17 March 2026).
- Schaefer, H.R.; Flannery, B.M.; Crosby, L.M.; Pouillot, R.; Farakos, S.M.S.; Van Doren, J.M.; Dennis, S.; Fitzpatrick, S.; Mid-dleton, K. Reassessment of the cadmium toxicological reference value for use in human health assessments of foods. Regul. Toxicol. Pharmacol. 2023, 144, 105487. [Google Scholar] [CrossRef]
- Nishijo, M.; Nogawa, K.; Suwazono, Y.; Kido, T.; Sakurai, M.; Nakagawa, H. Lifetime Cadmium Exposure and Mortality for Renal Diseases in Residents of the Cadmium-Polluted Kakehashi River Basin in Japan. Toxics 2020, 8, 81. [Google Scholar] [CrossRef]
- Qing, Y.; Li, Y.; Cai, X.; He, W.; Liu, S.; Ji, Y.; Jiang, M.; Yang, L.; Wang, J.; Ping, S.; Chen, Y.; Luo, Y.; Li, Y. Assessment of cadmium concentrations in foodstuffs and dietary exposure risk across China: a metadata analysis. Expos. Health 2023, 15, 951–61. [Google Scholar] [CrossRef]
- Hoffman-Pennesi, D.; Winfield, S.; Gavelek, A.; Santillana Farakos, S.M.; Spungen, J. Infants’ and young children’s dietary exposures to lead and cadmium: FDA total diet study 2018-2020. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2024, 41, 1454–1479. [Google Scholar] [CrossRef]
- Flannery, B.M.; Dolan, L.C.; Hoffman-Pennesi, D.; Gavelek, A.; Jones, O.E.; Kanwal, R.; Wolpert, B.; Gensheimer, K.; Dennis, S.; Fitzpatrick, S.U.S. U.S. Food and Drug Administration’s interim reference levels for dietary lead exposure in children and women of childbearing age. Regul. Toxicol. Pharmacol. 2020, 110, 104516. [Google Scholar] [CrossRef] [PubMed]
- Dolan, L.C.; Flannery, B.M.; Hoffman-Pennesi, D.; Gavelek, A.; Jones, O.E.; Kanwal, R.; Wolpert, B.; Gensheimer, K.; Dennis, S.; Fitzpatrick, S. A review of the evidence to support interim reference level for dietary lead exposure in adults. Regul. Toxicol. Pharmacol. 2020, 111, 104579. [Google Scholar] [CrossRef] [PubMed]
- Brzóska, M. M.; Moniuszko-Jakoniuk, J. Disorders in bone metabolism of female rats chronically exposed to cadmium. Toxicol. Appl. Pharmacol. 2005, 202, 68–83. [Google Scholar] [CrossRef] [PubMed]
- Brzóska, M. M.; Moniuszko-Jakoniuk, J. Bone metabolism of male rats chronically exposed to cadmium. Toxicol. Appl. Pharmacol. 2005, 207, 195–211. [Google Scholar] [CrossRef]
- Brzóska, M. M.; Moniuszko-Jakoniuk, J. Effect of low-level lifetime exposure to cadmium on calciotropic hormones in aged female rats. Arch. Toxicol. 2005, 79, 636–46. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services; National Institutes of Health. NTP, NIH Publication 95–3388: NTP Technical Report on Toxicity Studies of Cadmium Oxide (CAS No. 1306–19-0) Administered by Inhalation to F344/N Rats and B6C3F1 Mice. National Toxicology Program: Research Triangle Park, NC, USA, 1995.
- Suwazono, Y.; Sand, S.; Vahter, M.; Skerfving, S.; Lidfeldt, J.; Akesson, A. Benchmark dose for cadmium-induced osteoporosis in women. Toxicol Lett. 2010, 197, 123–127. [Google Scholar] [CrossRef]
- Leconte, S.; Rousselle, C.; Bodin, L.; Clinard, F.; Carne, G. Refinement of health-based guidance values for cadmium in the French population based on modelling. Toxicol. Lett. 2021, 340, 43–51. [Google Scholar] [CrossRef]
- Qing, Y.; Yang, J.; Chen, Y.; Shi, C.; Zhang, Q.; Ning, Z.; Yu, Y.; Li, Y. Urinary cadmium in relation to bone damage: Cadmium exposure threshold dose and health-based guidance value estimation. Ecotoxicol. Environ. Saf. 2021, 226, 112824. [Google Scholar] [CrossRef]
- Qing, Y.; Yang, J.; Zhu, Y.; Li, Y.; Zheng, W.; Wu, M.; He, G. Dose-response evaluation of urinary cadmium and kidney injury biomarkers in Chinese residents and dietary limit standards. Environ. Health 2021, 20, 75. [Google Scholar] [CrossRef]
- Wu, X.; Wei, S.; Wei, Y.; Guo, B.; Yang, M.; Zhao, D.; Liu, X.; Cai, X. The reference dose for subchronic exposure of pigs to cadmium leading to early renal damage by benchmark dose method. Toxicol. Sci. 2012, 128, 524–531. [Google Scholar] [CrossRef]
- Committee for Recommendation of Occupational Exposure Limits; Japan Society for Occupational Health. Occupational exposure limits for acetaldehyde, 2-bromopropane, glyphosate, manganese and inorganic manganese compounds, and zinc oxide nanoparticle, and the biological exposure indices for cadmium and cadmium compounds and ethylbenzene, and carcinogenicity, occupational sensitizer, and reproductive toxicant classifications. J. Occup. Health 2021, 63, e12294. [Google Scholar]
- Nogawa, K.; Suwazono, Y.; Watanabe, Y.; Elinder, C. G. Estimation of benchmark dose of cumulative cadmium exposure for renal tubular effect. Int. J. Environ. Res. Publ. Health 2021, 18, 5177. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, K.; Iwasawa, S.; Yoshioka, N.; Suzuki, S.; Hashimoto, I.; Ukita, S.; Toshima, G.; Nagashima, K.; Takebayashi, T.; Tsunoda, M. Increased risk of proximal tubular dysfunction due to occupational cadmium exposure: a survival analysis study. J. Occup. Health 2025, 67, uiaf016. [Google Scholar] [CrossRef] [PubMed]
- Choi, W. J.; Kang, S. K.; Ham, S.; Chung, W.; Kim, A. J.; Kang, M. Chronic cadmium intoxication and renal injury among workers of a small-scale silver soldering company. Saf. Health. Work. 2020, 11, 235–40. [Google Scholar] [CrossRef] [PubMed]
- Roy, A.; Saha, T.; Sahoo, J.; Das, A. Hypophosphatemic osteomalacia due to cadmium toxicity in silverware industry: A curious case of aches and pains. J. Fam. Med. Prim. Care 2024, 13, 2516–2519. [Google Scholar] [CrossRef] [PubMed]
- Giri, S.; Roy, A.; Kumar, A.; Ghosh, S.; Bhunia, A.; Patra, S. Cadmium toxicity-related metabolic bone disease: A clinical conundrum of five cases. Osteoporos. Int. 2025; Online ahead of print. [Google Scholar] [CrossRef]
- Akin, I.; Yazihan, N. Can cadmium toxicity be a driving force for iron deficiency anemia—a systematic review and meta-analysis. Rev. Environ. Health 2026. [Google Scholar] [CrossRef]
- Kido, S.; Fujihara, M.; Nomura, K.; Sasaki, S.; Mukai, R.; Ohnishi, R.; Kaneko, I.; Segawa, H.; Tatsumi, S.; Izumi, H.; et al. Molecular mechanisms of cadmium-induced fibroblast growth factor 23 upregulation in osteoblast-like cells. Toxicol. Sci. 2014, 139, 301–316. [Google Scholar] [CrossRef]
- Aranami, F.; Segawa, H.; Furutani, J.; Kuwahara, S.; Tominaga, R.; Hanabusa, E.; Tatsumi, S.; Kido, S.; Ito, M.; Miyamoto, K. Fibroblast growth factor 23 mediates the phosphaturic actions of cadmium. J. Med. Investig. 2010, 57, 95–108. [Google Scholar] [CrossRef]
- Kunioka, C.T.; Manso, M.C.; Carvalho, M. Association between Environmental Cadmium Exposure and Osteoporosis Risk in Postmenopausal Women: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2022, 20, 485. [Google Scholar] [CrossRef]
- Woo, H. D.; Chiu, W. A.; Jo, S.; Kim, J. Benchmark dose for urinary cadmium based on a marker of renal dysfunction: a meta-analysis. PLoS One 2015, 10, e0126680. [Google Scholar] [CrossRef]
- Thévenod, F.; Lee, W.K. Challenging the current dogma of chronic Cd nephrotoxicity: myths and facts. Arch. Toxicol. 2026. [Google Scholar] [CrossRef] [PubMed]
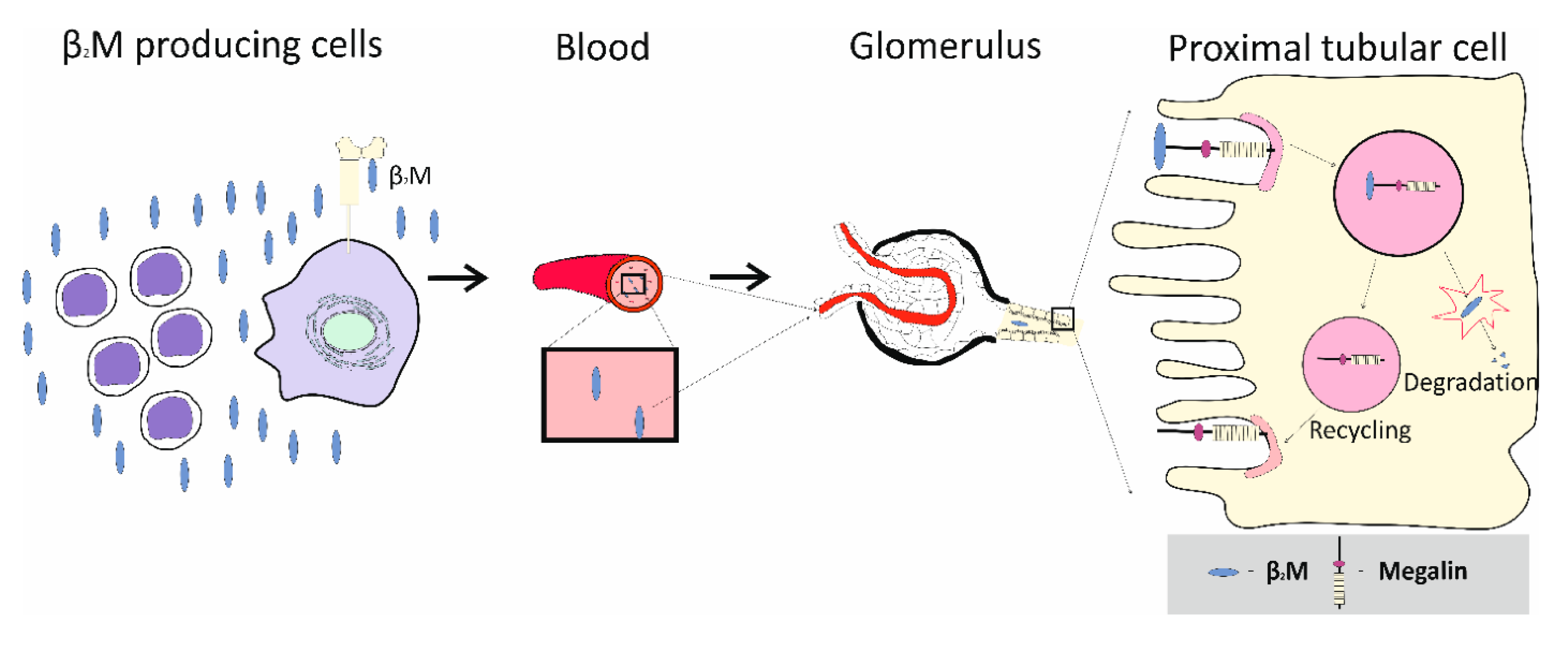
- Argyropoulos, C.P.; Chen, S.S.; Ng, Y.-H.; Roumelioti, M.-E.; Shaffi, K.; Singh, P.P.; Tzamaloukas, A.H. Rediscovering Beta-2 Microglobulin As a Biomarker across the Spectrum of Kidney Diseases. Front. Med. 2017, 4, 73. [Google Scholar] [CrossRef]
- Phelps, K. R.; Yimthiang, S.; Pouyfung, P.; Khamphaya, T.; Vesey, D. A.; Satarug, S. Homeostasis of β2-microglobulin in diabetics and non-diabetics with modest cadmium intoxication. J. Environ. Expo. Assess. 2025, 4, 23. [Google Scholar] [CrossRef]
- Callan, A.; Hinwood, A.; Devine, A. Metals in commonly eaten groceries in Western Australia: A market basket survey and dietary assessment. Food Addit. Contam. Part. A Chem. Anal. Control Expo. Risk Assess. 2014, 31, 1968–1981. [Google Scholar] [CrossRef] [PubMed]
- Crump, K.S. A new method for determining allowable daily intakes. Fundam. Appl. Toxicol. 1984, 4, 854–871. [Google Scholar] [CrossRef]
- Gaylor, D.; Ryan, L.; Krewski, D.; Zhu, Y. Procedures for calculating benchmark doses for health risk assessment. Regul. Toxicol. Pharmacol. 1998, 28, 150–164. [Google Scholar] [CrossRef]
- EFSA Scientific Committee. Update: Use of the benchmark dose approach in risk assessment. EFSA J. 2017, 15, 4658. [Google Scholar] [CrossRef]
- Swaddiwudhipong, W.; Nguntra, P.; Kaewnate, Y.; Mahasakpan, P.; Limpatanachote, P.; Aunjai, T.; Jeekeeree, W.; Punta, B.; Funkhiew, T.; Phopueng, I. Human health effects from cadmium exposure: Comparison between persons living in cadmium-contaminated and non-contaminated areas in northwestern Thailand. Southeast Asian J. Trop. Med. Public Health 2015, 46, 133–142. [Google Scholar]
- Suwatvitayakorn, P.; Ko, M.S.; Kim, K.W.; Chanpiwat, P. Human health risk assessment of cadmium exposure through rice consumption in cadmium-contaminated areas of the Mae Tao sub-district, Tak, Thailand. Environ. Geochem. Health 2020, 42, 2331–2344. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Nishijo, M.; Ruangyuttikarn, W.; Gobe, G.C. The inverse association of glomerular function and urinary β2-MG excretion and its implications for cadmium health risk assessment. Environ. Res. 2019, 173, 40–47. [Google Scholar] [CrossRef]
- Satarug, S.; Ruangyuttikarn, W.; Nishijo, M.; Ruiz, P. Urinary Cadmium Threshold to Prevent Kidney Disease Development. Toxics 2018, 6, 26. [Google Scholar] [CrossRef]
- Satarug, S.; Đorđević, A.B.; Yimthiang, S.; Vesey, D.A.; Gobe, G.C. The NOAEL Equivalent of Environmental Cadmium Exposure Associated with GFR Reduction and Chronic Kidney Disease. Toxics 2022, 10, 614. [Google Scholar] [CrossRef]
- Wang, X.; Wang, Y.; Feng, L.; Tong, Y.; Chen, Z.; Ying, S.; Chen, T.; Li, T.; Xia, H.; Jiang, Z.; et al. Application of the benchmark dose (BMD) method to identify thresholds of cadmium-induced renal effects in non-polluted areas in China. PLoS ONE 2016, 11, e0161240. [Google Scholar] [CrossRef]
- Suwazono, Y.; Nogawa, K.; Uetani, M.; Miura, K.; Sakata, K.; Okayama, A.; Ueshima, H.; Stamler, J.; Nakagawa, H. Application of hybrid approach for estimating the benchmark dose of urinary cadmium for adverse renal effects in the general population of Japan. J. Appl. Toxicol. 2011, 31, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Vesey, D.A.; Gobe, G.C.; Đorđević, A.B. The Validity of Benchmark Dose Limit Analysis for Estimating Permissible Accumulation of Cadmium. Int. J. Environ. Res. Public Health 2022, 19, 15697. [Google Scholar] [CrossRef] [PubMed]
- Sand, S.; Filipsson, A.F.; Victorin, K. Evaluation of the benchmark dose method for dichotomous data: Model dependence and model selection. Regul. Toxicol. Pharmacol. 2002, 36, 184–197. [Google Scholar] [CrossRef] [PubMed]
- Filipsson, A. F.; Sand, S.; Nilsson, J.; Victorin, K. The benchmark dose method—review of available models, and recommendations for application in health risk assessment. Crit. Rev. Toxicol. 2003, 33, 505–42. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Đorđević, A.B. The NOAEL equivalent for the cumulative body burden of cadmium: Focus on proteinuria as an endpoint. J. Environ. Expo. Assess. 2024, 3, 26. [Google Scholar] [CrossRef]
- Xie, S.; Perrais, M.; Golshayan, D.; Wuerzner, G.; Vaucher, J.; Thomas, A.; Marques-Vidal, P. Association between urinary heavy metal/trace element concentrations and kidney function: A prospective study. Clin. Kidney J. 2024, 18, sfae378. [Google Scholar] [CrossRef]
- Makhammajanov, Z.; Gaipov, A.; Myngbay, A.; Bukasov, R.; Aljofan, M.; Kanbay, M. Tubular toxicity of proteinuria and the progression of chronic kidney disease. Nephrol. Dial. Transplant. 2024, 39, 589–599. [Google Scholar] [CrossRef]
- Liu, D.; Lv, L.L. New Understanding on the Role of Proteinuria in Progression of Chronic Kidney Disease. Adv. Exp. Med. Biol. 2019, 1165, 487–500. [Google Scholar]
- Sharma, S.; Smyth, B. From Proteinuria to Fibrosis: An Update on Pathophysiology and Treatment Options. Kidney Blood Press. Res. 2021, 46, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Cirovic, A.; Denic, A.; Clarke, B.L.; Vassallo, R.; Cirovic, A.; Landry, G.M. A hypoxia-driven occurrence of chronic kidney disease and osteoporosis in COPD individuals: New insights into environmental cadmium exposure. Toxicol. 2022, 482, 153355. [Google Scholar] [CrossRef]
- Cirovic, A.; Cirovic, A. Factors moderating cadmium bioavailability: Key considerations for comparing blood cadmium levels between groups. Food Chem. Toxicol. 2024, 191, 114865. [Google Scholar] [CrossRef] [PubMed]
- Cirovic, A.; Milovanovic, P. Can adjustment for iron, zinc, and calcium levels clarify the relationship between blood cadmium and bone mineral density? Osteoporos. Int. 2025, 36, 1489–1490. [Google Scholar] [CrossRef] [PubMed]
- Fujishiro, H.; Ikeue, Y.; Himeno, S.; Sumi, D. Cadmium Uptake into Rat Osteoblast UMR-106 Cells is Mediated via Multiple Pathways. Biol. Trace Elem. Res. 2025; Online ahead of print. [Google Scholar] [CrossRef]
- Nielsen, R.; Christensen, E.I.; Birn, H. Megalin and cubilin in proximal tubule protein reabsorption: From experimental models to human disease. Kidney Int. 2016, 89, 58–67. [Google Scholar] [CrossRef]
- Smith, C.P.; Lee, W.K.; Haley, M.; Poulsen, S.B.; Thévenod, F.; Fenton, R.A. Proximal tubule transferrin uptake is modulated by cellular iron and mediated by apical membrane megalin-cubilin complex and transferrin receptor 1. J Biol Chem. 2019, 294, 7025–7036. [Google Scholar] [CrossRef]
- Thévenod, F.; Herbrechter, R.; Schlabs, C.; Pethe, A.; Lee, W.K.; Wolff, N.A.; Roussa, E. Role of the SLC22A17/lipocalin-2 receptor in renal endocytosis of proteins/metalloproteins: a focus on iron- and cadmium-binding proteins. Am. J. Physiol. Renal Physiol. 2023, 325, F564–F577. [Google Scholar] [CrossRef]
- Fels, J.; Scharner, B.; Zarbock, R.; Zavala Guevara, I.P.; Lee, W.-K.; Barbier, O.C.; Thévenod, F. Cadmium Complexed with β2-Microglubulin, Albumin and Lipocalin-2 rather than Metallothionein Cause Megalin:Cubilin Dependent Toxicity of the Renal Proximal Tubule. Int. J. Mol. Sci. 2019, 20, 2379. [Google Scholar] [CrossRef]
- Zavala-Guevara, I.P.; Ortega-Romero, M.S.; Narváez-Morales, J.; Jacobo-Estrada, T.L.; Lee, W.-K.; Arreola-Mendoza, L.; Thévenod, F.; Barbier, O.C. Increased Endocytosis of Cadmium-Metallothionein through the 24p3 Receptor in an In Vivo Model with Reduced Proximal Tubular Activity. Int. J. Mol. Sci. 2021, 22, 7262. [Google Scholar] [CrossRef]
- Fujishiro, H.; Sumino, M.; Sumi, D.; Umemoto, H.; Tsuneyama, K.; Matsukawa, T.; Yokoyama, K.; Himeno, S. Spatial localization of cadmium and metallothionein in the kidneys of mice at the early phase of cadmium accumulation. J. Toxicol. Sci. 2022, 47, 507–517. [Google Scholar] [CrossRef]
- Boonprasert, K.; Satarug, S.; Morais, C.; Gobe, G.C.; Johnson, D.W.; Na-Bangchang, K.; Vesey, D.A. The stress response of human proximal tubule cells to cadmium involves up-regulation of haemoxygenase 1 and metallothionein but not cytochrome P450 enzymes. Toxicol. Lett. 2016, 249, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Molitoris, B.A.; Sandoval, R.M.; Yadav, S.P.S.; Wagner, M.C. Albumin uptake and processing by the proximal tubule: Physiological, pathological, and therapeutic implications. Physiol. Rev. 2022, 102, 1625–1667. [Google Scholar] [CrossRef] [PubMed]
- Comper, W.D.; Vuchkova, J.; McCarthy, K.J. New insights into proteinuria/albuminuria. Front. Physiol. 2022, 13, 991756. [Google Scholar] [CrossRef] [PubMed]
- Castrop, H.; Schießl, I.M. Novel routes of albumin passage across the glomerular filtration barrier. Acta Physiol. 2017, 219, 544–553. [Google Scholar] [CrossRef]
- Gburek, J.; Konopska, B.; Gołąb, K. Renal handling of albumin-from early findings to current concepts. Int. J. Mol. Sci. 2021, 22, 5809. [Google Scholar] [CrossRef]
- Bernard, A.; Lauwerys, R.; Ouled Amor, A. Loss of glomerular polyanion correlated with albuminuria in experimental cadmium nephropathy. Arch. Toxicol. 1992, 66, 272–278. [Google Scholar] [CrossRef]
- Cardenas, A.; Bernard, A.M.; Lauwerys, R.R. Disturbance of sialic acid metabolism by chronic cadmium exposure and its relation to proteinuria. Toxicol. Appl. Pharmacol. 1991, 108, 547–558. [Google Scholar] [CrossRef]
- Li, L.; Dong, F.; Xu, D.; Du, L.; Yan, S.; Hu, H.; Lobe, C.G.; Yi, F.; Kapron, C.M.; Liu, J. Short-term, low-dose cadmium exposure induces hyperpermeability in human renal glomerular endothelial cells. J. Appl. Toxicol. 2016, 36, 257–265. [Google Scholar] [CrossRef]
- Li, Z.; Jiang, L.; Tao, T.; Su, W.; Guo, Y.; Yu, H.; Qin, J. Assessment of cadmium-induced nephrotoxicity using a kidney-on-a-chip device. Toxicol. Res. 2017, 6, 372–380. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Gobe, G.C.; Phelps, K.R. The pathogenesis of albuminuria in cadmium nephropathy. Curr. Res. Toxicol. 2023, 6, 100140. [Google Scholar] [CrossRef]
- Zhang, K.; Long, M.; Dong, W.; Li, J.; Wang, X.; Liu, W.; Huang, Q.; Ping, Y.; Zou, H.; Song, R.; et al. Cadmium Induces Kidney Iron Deficiency and Chronic Kidney Injury by Interfering with the Iron Metabolism in Rats. Int. J. Mol. Sci. 2024, 25, 763. [Google Scholar] [CrossRef] [PubMed]
- Wolf, C.; Strenziok, R.; Kyriakopoulos, A. Elevated metallothionein-bound cadmium concentrations in urine from bladder carcinoma patients, investigated by size exclusion chromatography-inductively coupled plasma mass spectrometry. Anal. Chim. Acta 2009, 631, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Sabolić, I.; Škarica, M.; Ljubojević, M.; Breljak, D.; Herak-Kramberger, C.M.; Crljen, V.; Ljubešić, N. Expression and immunolocalization of metallothioneins MT1, MT2 and MT3 in rat nephron. J. Trace Elem. Med. Biol. 2018, 46, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.K.; Probst, S.; Scharner, B.; Deba, T.; Dahdouh, F.; Thévenod, F. Distinct concentration-dependent oxidative stress profiles by cadmium in a rat kidney proximal tubule cell line. Arch. Toxicol. 2024, 98, 1043–1059. [Google Scholar] [CrossRef]
- Lee, W.K.; Thévenod, F. Cell organelles as targets of mammalian cadmium toxicity. Arch. Toxicol. 2020, 94, 1017–1049. [Google Scholar] [CrossRef]
- Fujiwara, Y.; Lee, J.Y.; Tokumoto, M.; Satoh, M. Cadmium renal toxicity via apoptotic pathways. Biol. Pharm. Bull. 2012, 35, 1892–1897. [Google Scholar] [CrossRef]
- Lenoir, O.; Tharaux, P.L.; Huber, T.B. Autophagy in kidney disease and aging: Lessons from rodent models. Kidney Int. 2016, 90, 950–964. [Google Scholar] [CrossRef]
- Liang, S.; Wu, Y.S.; Li, D.Y.; Tang, J.X.; Liu, H.F. Autophagy and Renal Fibrosis. Aging Dis. 2022, 13, 712–731. [Google Scholar] [CrossRef]
- Dong, P.F.; Liu, T.B.; Chen, K.; Li, D.; Li, Y.; Lian, C.Y.; Wang, Z.Y.; Wang, L. Cadmium targeting transcription factor EB to inhibit autophagy-lysosome function contributes to acute kidney injury. J. Adv. Res. 2025, 72, 653–669. [Google Scholar] [CrossRef]
- Okubo, A.; Nakashima, A.; Doi, S.; Doi, T.; Ueno, T.; Maeda, K.; Tamura, R.; Yamane, K.; Masaki, T. High-normal albuminuria is strongly associated with incident chronic kidney disease in a nondiabetic population with normal range of albuminuria and normal kidney function. Clin. Exp. Nephrol. 2020, 24, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jiang, R.; Shi, Z.; Sui, Y.; Cheng, J.; Suda, M.; Niimi, M.; Gao, K.; Fan, J.; Yao, J. Oxidized Albumin Induces Renal Tubular Cell Death and Promotes the Progression of Renal Diseases Through Ferroptosis. Int. J. Mol. Sci. 2025, 26, 5924. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Gu, X. Emerging roles of proximal tubular endocytosis in renal fibrosis. Front. Cell Dev. Biol. 2023, 11, 1235716. [Google Scholar] [CrossRef] [PubMed]
- Thijssen, S.; Lambrichts, I.; Maringwa, J.; Van Kerkhove, E. Changes in expression of fibrotic markers and histopathological alterations in kidneys of mice chronically exposed to low and high Cd doses. Toxicol. 2007, 238, 200–210. [Google Scholar] [CrossRef]
- Liang, L.; Huang, K.; Yuan, W.; Liu, L.; Zou, F.; Wang, G. Dysregulations of miR-503-5p and Wnt/β-catenin pathway coordinate in mediating cadmium-induced kidney fibrosis. Ecotoxicol. Environ. Saf. 2021, 224, 112667. [Google Scholar] [CrossRef]
- Gobe, G.C.; Mott, S.A.; de Jonge, M.; Hoy, W.E. Heavy metal imaging in fibrotic human kidney tissue using the synchrotron X-ray fluorescence microprobe. Transl. Androl. Urol. 2019, 8, S184–S191. [Google Scholar] [CrossRef]
- Barregard, L.; Sallsten, G.; Lundh, T.; Mölne, J. Low-level exposure to lead, cadmium and mercury, and histopathological findings in kidney biopsies. Environ. Res. 2022, 211, 113119. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Gobe, G.C.; Yimthiang, S.; Buha Đorđević, A. Health Risk in a Geographic Area of Thailand with Endemic Cadmium Contamination: Focus on Albuminuria. Toxics 2023, 11, 68. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Gobe, G.C. Cadmium-Induced Proteinuria: Mechanistic Insights from Dose–Effect Analyses. Int. J. Mol. Sci. 2023, 24, 1893. [Google Scholar] [CrossRef]
- Cirovic, A.; Cirovic, A. Iron deficiency as a promoter of cadmium-induced cardiotoxicity. Eur. Heart J. 2023, 44, 2639–2640. [Google Scholar] [CrossRef]
- Cirovic, A.; Starcevic, A.; Ivanovski, A.; Bogicevic, D.; Orisakwe, O.E.; Cirovic, A. Mechanisms Underlying Iron Deficiency-Induced Cardiac Disorders: Implications for Treatment. Discov. Med. 2025, 37, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Foreman, K.J.; Marquez, N.; Dolgert, A.; Fukutaki, K.; Fullman, N.; McGaughey, M.; Pletcher, M.A.; Smith, A.E.; Tang, K.; Yuan, C.W.; et al. Forecasting life expectancy, years of life lost, and all-cause and cause-specific mortality for 250 causes of death: Reference and alternative scenarios for 2016-40 for 195 countries and territories. Lancet 2018, 392, 2052–2090. [Google Scholar] [CrossRef] [PubMed]
- GBD 2021 Forecasting Collaborators. Burden of disease scenarios for 204 countries and territories, 2022–2050: A forecasting analysis for the Global Burden of Disease Study 2021. Lancet 2024, 403, 2204–2256. [Google Scholar] [CrossRef] [PubMed]
- Demchenkov, E.L.; Nagdalian, A.A.; Budkevich, R.O.; Oboturova, N.P.; Okolelova, A.I. Usage of atomic force microscopy for detection of the damaging effect of CdCl2 on red blood cells membrane. Ecotoxicol. Environ. Saf. 2021, 208, 111683. [Google Scholar] [CrossRef]
- Thévenod, F.; Lee, W.K.; Garrick, M.D. Iron and cadmium entry into renal mitochondria: Physiological and toxicological implications. Front. Cell Dev. Biol. 2020, 8, 848. [Google Scholar] [CrossRef]
- Branca, J.J.V.; Pacini, A.; Gulisano, M.; Taddei, N.; Fiorillo, C.; Becatti, M. Cadmium-induced cytotoxicity: Effects on mitochondrial electron transport chain. Front. Cell Dev. Biol. 2020, 8, 604377. [Google Scholar] [CrossRef]
- Tavakolpournegari, A.; Moosavi, S.S.; Matinahmadi, A.; Zayani, Z.; Bidooki, S.H. Exploring How Reactive Oxygen Species Contribute to Cancer via Oxidative Stress. Stresses 2025, 5, 69. [Google Scholar] [CrossRef]
- Krężel, A.; Maret, W. The functions of metamorphic metallothioneins in zinc and copper metabolism. Int. J. Mol. Sci. 2017, 18, 1237. [Google Scholar] [CrossRef]
- Krężel, A.; Maret, W. The bioinorganic chemistry of mammalian metallothioneins. Chem. Rev. 2021, 121, 14594–14648. [Google Scholar] [CrossRef]
- Boonprasert, K.; Ruengweerayut, R.; Aunpad, R.; Satarug, S.; Na-Bangchang, K. Expression of metallothionein isoforms in peripheral blood leukocytes from Thai population residing in cadmium-contaminated areas. Environ. Toxicol. Pharmacol. 2012, 34, 935–940. [Google Scholar] [CrossRef]
- Hennigar, S.R.; Kelley, A.M.; McClung, J.P. Metallothionein and zinc transporter expression in circulating human blood cells as biomarkers of zinc status: A systematic review. Adv. Nutr. 2016, 7, 735–746. [Google Scholar] [CrossRef] [PubMed]
- Garrett, S.H.; Sens, M.A.; Todd, J.H.; Somji, S.; Sens, D.A. Expression of MT-3 protein in the human kidney. Toxicol. Lett. 1999, 105, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Vašák, M.; Meloni, G. Mammalian metallothionein-3: New functional and structural insights. Int. J. Mol. Sci. 2017, 18, 1117. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.-J.; Hung, C.-H.; Wang, C.-W.; Tu, H.-P.; Li, C.-H.; Tsai, C.-C.; Lin, W.-Y.; Chen, S.-C.; Kuo, C.-H. Associations among Heavy Metals and Proteinuria and Chronic Kidney Disease. Diagnostics 2021, 11, 282. [Google Scholar] [CrossRef]
- Sun, D.Q.; Zhong, M.Y.; Zhang, J.H.; Tang, H.; Hu, B.; Shen, J.Q.; Yan, F.; Xu, X.Y.; Chen, K.; Targher, G.; et al. Oxidized-LDL aggravates renal injury via tubular cuproptosis. Cell Signal. 2025, 132, 111839. [Google Scholar] [CrossRef]
- Misra, R.R.; Hochadel, J.F.; Smith, G.T.; Cook, J.C.; Waalkes, M.P.; Wink, D.A. Evidence that nitric oxide enhances cadmium toxicity by displacing the metal from metallothionein. Chem. Res. Toxicol. 1996, 9, 326–332. [Google Scholar] [CrossRef]
- Satarug, S.; Baker, J.R.; Reilly, P.E.; Esumi, H.; Moore, M.R. Evidence for a synergistic interaction between cadmium and endotoxin toxicity and for nitric oxide and cadmium displacement of metals in the kidney. Nitric Oxide 2000, 4, 431–440. [Google Scholar] [CrossRef]
- Zhu, J.; Meeusen, J.; Krezoski, S.; Petering, D.H. Reactivity of Zn-, Cd-, and apo-metallothionein with nitric oxide compounds: In vitro and cellular comparison. Chem. Res. Toxicol. 2010, 23, 422–431. [Google Scholar] [CrossRef]
- Petering, D.H. Reactions of the Zn proteome with Cd2+ and other xenobiotics: Trafficking and toxicity. Chem. Res. Toxicol. 2017, 30, 189–202. [Google Scholar] [CrossRef]
- Petering, D.H.; Mahim, A. Proteomic High Affinity Zn2+ Trafficking: Where Does Metallothionein Fit in? Int. J. Mol. Sci. 2017, 18, 1289. [Google Scholar] [CrossRef]
- Hübner, C.; Haase, H. Interactions of zinc- and redox-signaling pathways. Redox Biol. 2021, 41, 101916. [Google Scholar] [CrossRef]
- Polykretis, P.; Cencetti, F.; Donati, C.; Luchinat, E.; Banci, L. Cadmium effects on superoxide dismutase 1 in human cells revealed by NMR. Redox Biol. 2019, 21, 101102. [Google Scholar] [CrossRef]
- Satarug, S.; Wisedpanichkij, R.; Takeda, K.; Li, B.; Na-Bangchang, K.; Moore, M.R.; Shibahara, S. Prostaglandin D2 induces heme oxygenase-1 mRNA expression through the DP2 receptor. Biochem. Biophys. Res. Commun. 2008, 377, 878–883. [Google Scholar] [CrossRef]
- Takeda, T.A.; Mu, A.; Tai, T.T.; Kitajima, S.; Taketani, S. Continuous de novo biosynthesis of haem and its rapid turnover to bilirubin are necessary for cytoprotection against cell damage. Sci. Rep. 2015, 5, 10488. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Ishizawa, S.; Sato, M.; Yoshida, T.; Shibahara, S. Identification of a cis-acting element that is responsible for cadmium-mediated induction of the human heme oxygenase gene. J. Biol. Chem. 1994, 269, 22858–22867. [Google Scholar] [CrossRef] [PubMed]
- Stewart, D.; Killeen, E.; Naquin, R.; Alam, S.; Alam, J. Degradation of transcription factor Nrf2 via the ubiquitin-proteasome pathway and stabilization by cadmium. J. Biol. Chem. 2003, 278, 2396–2402. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Tashiro, S.; Sun, J.; Doi, H.; Satomi, S.; Igarashi, K. Cadmium induces nuclear export of Bach1, a transcriptional repressor of heme oxygenase-1 gene. J. Biol. Chem. 2003, 278, 49246–49253. [Google Scholar] [CrossRef]
- Simmons, S.O.; Fan, C.Y.; Yeoman, K.; Wakefield, J.; Ramabhadran, R. NRF2 Oxidative Stress Induced by Heavy Metals is Cell Type Dependent. Curr. Chem. Genomics 2011, 5, 1–12. [Google Scholar] [CrossRef]
- Lin, J.P.; Vitek, L.; Schwertner, H.A. Serum bilirubin and genes controlling bilirubin concentrations as biomarkers for cardiovascular disease. Clin. Chem. 2010, 56, 1535–1543. [Google Scholar] [CrossRef]
- Zhao, C.; Yu, D.; He, Z.; Bao, L.; Feng, L.; Chen, L.; Liu, Z.; Hu, X.; Zhang, N.; Wang, T.; et al. Endoplasmic reticulum stress-mediated autophagy activation is involved in cadmium-induced ferroptosis of renal tubular epithelial cells. Free Radic. Biol. Med. 2021, 175, 236–248. [Google Scholar] [CrossRef]
- Deng, P.; Li, J.; Lu, Y.; Hao, R.; He, M.; Li, M.; Tan, M.; Gao, P.; Wang, L.; Hong, H.; et al. Chronic cadmium exposure triggered ferroptosis by perturbing the STEAP3-mediated glutathione redox balance linked to altered metabolomic signatures in humans. Sci. Total Environ. 2023, 905, 167039. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.T.; Liu, T.B.; Li, Y.; Wang, Z.Y.; Lian, C.Y.; Wang, L. HO-1 activation contributes to cadmium-induced ferroptosis in renal tubular epithelial cells via increasing the labile iron pool and promoting mitochondrial ROS generation. Chem. Biol. Interact. 2024, 399, 111152. [Google Scholar] [CrossRef] [PubMed]
- Hahn, D.; Shin, S.H.; Bae, J.S. Natural antioxidant and anti-inflammatory compounds in foodstuff or medicinal herbs inducing heme oxygenase-1 expression. Antioxidants 2020, 9, 1191. [Google Scholar] [CrossRef] [PubMed]
- Colacino, J.A.; Arthur, A.E.; Ferguson, K.K.; Rozek, L.S. Dietary antioxidant and anti-inflammatory intake modifies the effect of cadmium exposure on markers of systemic inflammation and oxidative stress. Environ. Res. 2014, 131, 6–12. [Google Scholar] [CrossRef]
- Joshi, S.; Hashmi, S.; Shah, S.; Kalantar-Zadeh, K. Plant-based diets for prevention and management of chronic kidney disease. Curr. Opin. Nephrol. Hypertens. 2020, 29, 16–21. [Google Scholar] [CrossRef]
- Joshi, S; McMacken, M; Kalantar-Zadeh, K. Plant-Based Diets for Kidney Disease: A Guide for Clinicians. Am. J. Kidney Dis. 2021, 77, 287–296. [Google Scholar] [CrossRef]
- Kim, H.; Rebholz, C.M. Plant-based diets for kidney disease prevention and treatment. Curr. Opin. Nephrol. Hypertens. 2024, 33, 593–602. [Google Scholar] [CrossRef]
- Tamaru, E.; Kokubu, D.; Ushida, Y.; Itoh, K. Nrf2 induction potency of plant-derived compounds determined using an antioxidant response element luciferase reporter and conventional NAD(P)H-quinone acceptor oxidoreductase 1 activity assay. BMC Res Notes 2024, 17, 373. [Google Scholar] [CrossRef]
- Shi, P.; Yan, H.; Fan, X.; Xi, S. A benchmark dose analysis for urinary cadmium and type 2 diabetes mellitus. Environ. Pollut. 2021, 273, 116519. [Google Scholar] [CrossRef]
| Toxicity target/Endpoint | Exposure limit/Threshold | Reference |
|---|---|---|
| Kidneys/β2M excretion ≥ 300 µg/g creatinine | TDI of 0.83 μg/kg b.w./d/ 5.24 μg/g creatinine |
JECFA [61,62] |
| Kidneys/β2M excretion ≥ 300 µg/g creatinine. | RfD of 0.36 μg/kg b.w./d 1 μg/g creatinine |
EFSA [63] |
| Kidneys and bones/ β2M excretion and bone mineral density |
TRV of 0.21−0.36 μg/kg b.w./d 0.5 μg/g creatinine |
US FDA [64] |
| Bones/Bone mineral density | MRLof 0.5 µg/kg b.w./day for an intermediate exposure duration (15−365 days) | ATSDR [43] |
| Lungs/Alveolar histiocytic infiltration and focal inflammation in alveolar septa | MRLof 0.03 μg CdO/m3 for an acute exposure duration between 1 and 14 days | ATSDR [43] |
| Method/Endpoint | Cd excretion benchmark | Country/ Reference |
|---|---|---|
| Conventional BMD RBP, β2M, and NAG |
For men, BMDL5 (BMDL10) values for Cd excretion rates with abnormal excretion of RBP, β2M, and NAG were 0.89 (1.59), 0.62 (1.30), 0.49 (1.04) μg/g cr., respectively Corresponding BMDL5 (BMDL10) values for Cd excretion rates in women were 0.76 (1.53), 0.64 (1.34), 0.65 (1.37) μg/g cr. |
China, Wang et al. [102]. |
| Conventional BMD β2M |
Respective BMD values of Cd excretion rates with abnormal β2M excretion in men and women were 0.6–1.2 and 0.6–2.3 µg g cr. [102]. | Japan, Suwazono et al. [103] |
| Advanced BMD β2M, NAG |
Cd excretion benchmarks at 5% increase in NAG excretion in men and women were 0.060 and 0.069 µg/g cr., respectively. BMDL10 value of Cd excretion rate at 10% prevalence of β2M excretion rates ≥ 300 μg/g cr. were 0.469 and 0.733 µg/g cr. in men and women, respectively. |
Thailand, Satarug et al. [104] |
| Advanced BMD Total protein, eGFR |
Cd excretion benchmark at 5% (10%) increase in protein excretion was 0.054 (0.114) µg/g cr. BMDL5 (BMD10) value of Cd excretion at 5% (10%) prevalence of CKD was 1.19 (1.35) µg/g cr. BMDL5 (BMDL10) value of Cd excretion at 5% prevalence of proteinuria was 1.86 (4.47) µg/g cr. |
Thailand, Satarug et al. [105] |
| PTC Status | Protein | Filtration Rate |
Excretion Rate |
Catabolic Rate |
Transcytosis Rate |
|---|---|---|---|---|---|
| Normal | Albumin | 60 g/d | 20 mg/d | 2.980 g/d | 57 g/d |
| β2M | 300 mg/d | 100 μg/d | 299.9 mg/d | 0 | |
| Cd-intoxicated | Albumin | 60 g/d | 50 g/d | 2.950 g/d | 57 g/d |
| β2M | 300 mg/d | 1000 μg/d | 299 mg/d | 0 |
| Metal | Kidney (HEK293T) |
Liver (HepG2) |
Breast (MCF7) |
Brain (A172) |
Lung (A549) |
|---|---|---|---|---|---|
| Cd | 0.907 | 0.954 | 11 | 6.03 | 54.7 |
| As | 1.88 | 16.5 | 9.05 | 15.9 | 207 |
| Hg | 2.82 | 19.5 | 6.35 | 6.18 | NR |
| Pb | # | 426 | # | # | NR |
| Ag | 11.8 | 2.52 | 4.73 | 5.54 | NR |
| Au | 76.1 | 169 7 | 40 | 146 | NR |
| Zn | 84.8 | 249 | 256 | 100 | NR |
| Cu | 281 | 455 | 295 | 136 | 392 |
| Co | 484 | 185 | 532 | NR | NR |
| Fe | # | # | NR | 239 | NR |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




