2. Materials and Methods
2.1. Study Design
This was a prospective, single-center, observational cohort study conducted at a high-volume structural heart disease center. The primary study cohort comprised consecutive adult patients with symptomatic structural degeneration of a previously implanted surgical bioprosthetic aortic valve who underwent transfemoral valve-in-valve (ViV) transcatheter aortic valve implantation (TAVI) with the MyVal transcatheter heart valve between July 2022 and June 2025. The objectives of the present analysis were to evaluate procedural success, early safety, serial hemodynamic performance, and 1-year clinical outcomes after MyVal ViV-TAVI, with prespecified analyses focused on residual transvalvular gradients, anatomy-based subgroup effects, and an exploratory internal device-platform comparison.
All cases were evaluated by a multidisciplinary Heart Team that included interventional cardiologists, cardiac surgeons, cardiac imaging specialists, and cardiac anesthesiologists. Treatment decisions were based on clinical status, bioprosthetic failure mechanism, anatomical feasibility, procedural risk, and overall comorbidity burden in accordance with contemporary guideline-based practice. The study was conducted in accordance with the Declaration of Helsinki and was approved by the local institutional ethics committee. Written informed consent for the procedure and institutional use of clinical data was obtained according to local policy.
2.2. Patient Population
Eligible patients were adults with symptomatic dysfunction of a surgical bioprosthetic aortic valve who were treated with transfemoral ViV-TAVI using the MyVal platform during the study period. Structural valve degeneration was established using integrated clinical and imaging assessment and was classified according to the predominant failure mechanism as stenosis, regurgitation, or mixed dysfunction. The primary analysis was restricted to the consecutive MyVal-treated transfemoral ViV-TAVI cohort. Non-MyVal ViV procedures were not included in the primary cohort analysis, but an internal ACURATE neo2 cohort was used separately for the exploratory matched comparative analysis.
Patients were not excluded from the primary MyVal cohort on the basis of small surgical valve size, coronary obstruction-risk anatomy, or anticipated need for adjunctive procedural strategies such as coronary protection, chimney stenting, or surgical valve fracture, because these features were intrinsic to the real-world anatomical complexity that this study sought to evaluate.
2.3. Preprocedural Assessment and Anatomical Planning
All patients underwent structured preprocedural clinical and imaging evaluation, including transthoracic echocardiography and invasive coronary angiography when clinically indicated. Contrast-enhanced electrocardiographically gated cardiac computed tomography was used for ViV planning whenever feasible and clinically appropriate. Preprocedural imaging was used to characterize aortic root anatomy, identify the failed surgical bioprosthesis, assess coronary ostial height and sinus/sinotubular junction dimensions, and inform procedural planning for transcatheter valve sizing, implantation strategy, and coronary protection when necessary.
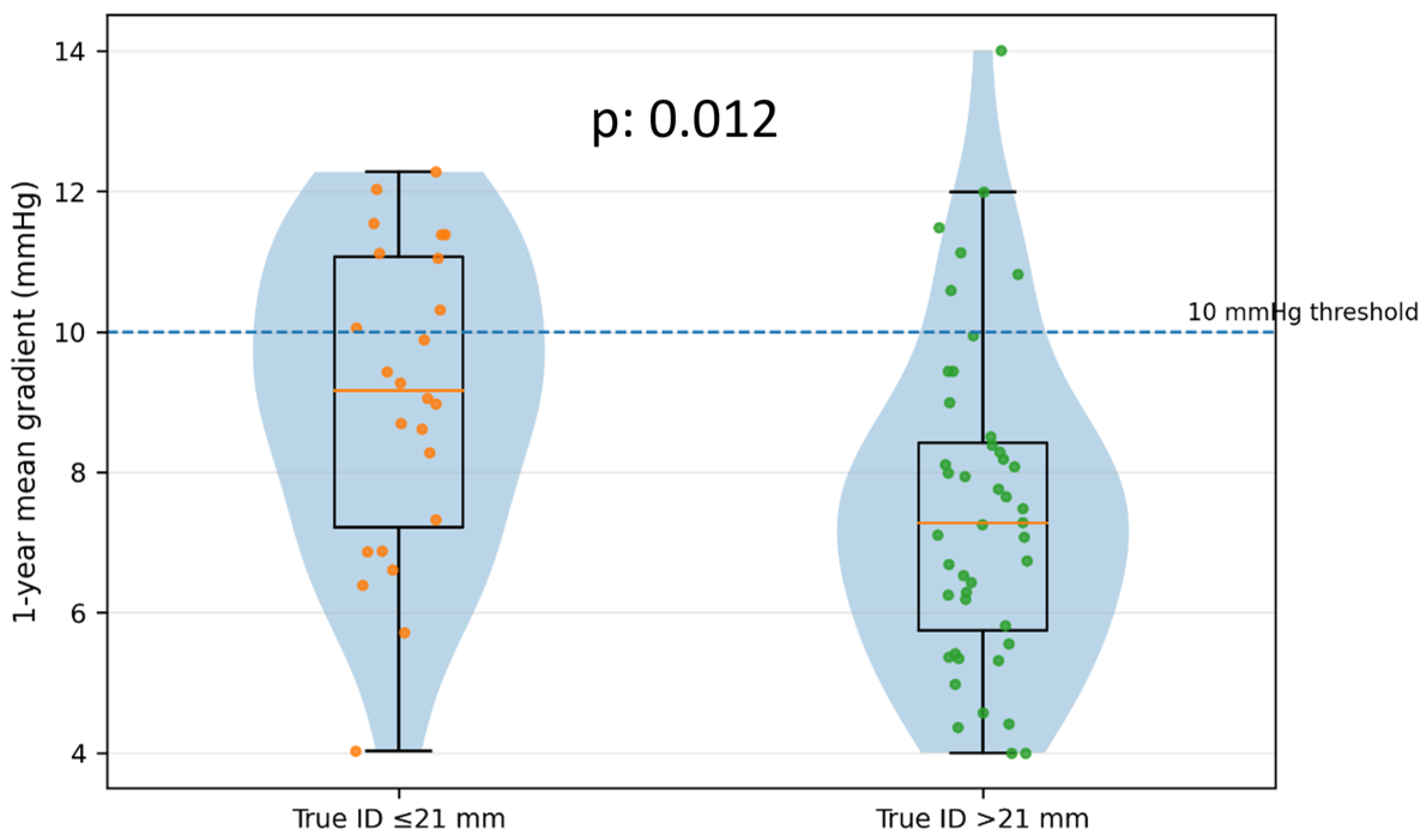
Surgical valve true internal diameter (true ID) was determined from the known surgical prosthesis model and labeled size using manufacturer specifications and standard ViV planning references, with CT-based anatomical confirmation when available. For prespecified subgroup analyses, small surgical valves were defined as true ID ≤21 mm.
2.4. Procedure
All procedures were performed via transfemoral access under fluoroscopic guidance according to institutional practice. The anesthesia strategy, intraprocedural echocardiographic support, vascular access management, and pacing strategy were selected according to case complexity and operator judgment. Valve sizing, implantation depth, and overall deployment strategy were based on preprocedural imaging, surgical valve identification, and anatomical risk assessment.
The procedural approach was anatomy-driven and allowed selective use of adjunctive techniques when indicated, including balloon predilation, postdilation, coronary protection, chimney stenting, and surgical valve fracture. These adjunctive strategies were used at operator discretion to optimize valve expansion, minimize coronary risk, and reduce residual hemodynamic burden in anatomically constrained ViV procedures. Procedural variables prospectively recorded included procedure duration, fluoroscopy time, contrast volume, use of adjunctive techniques, and intraprocedural complications. Because preservation of future coronary access formed part of the procedural planning framework, implantation orientation was optimized whenever feasible on the basis of anatomical considerations and device positioning strategy.
2.5. Follow-Up and Data Collection
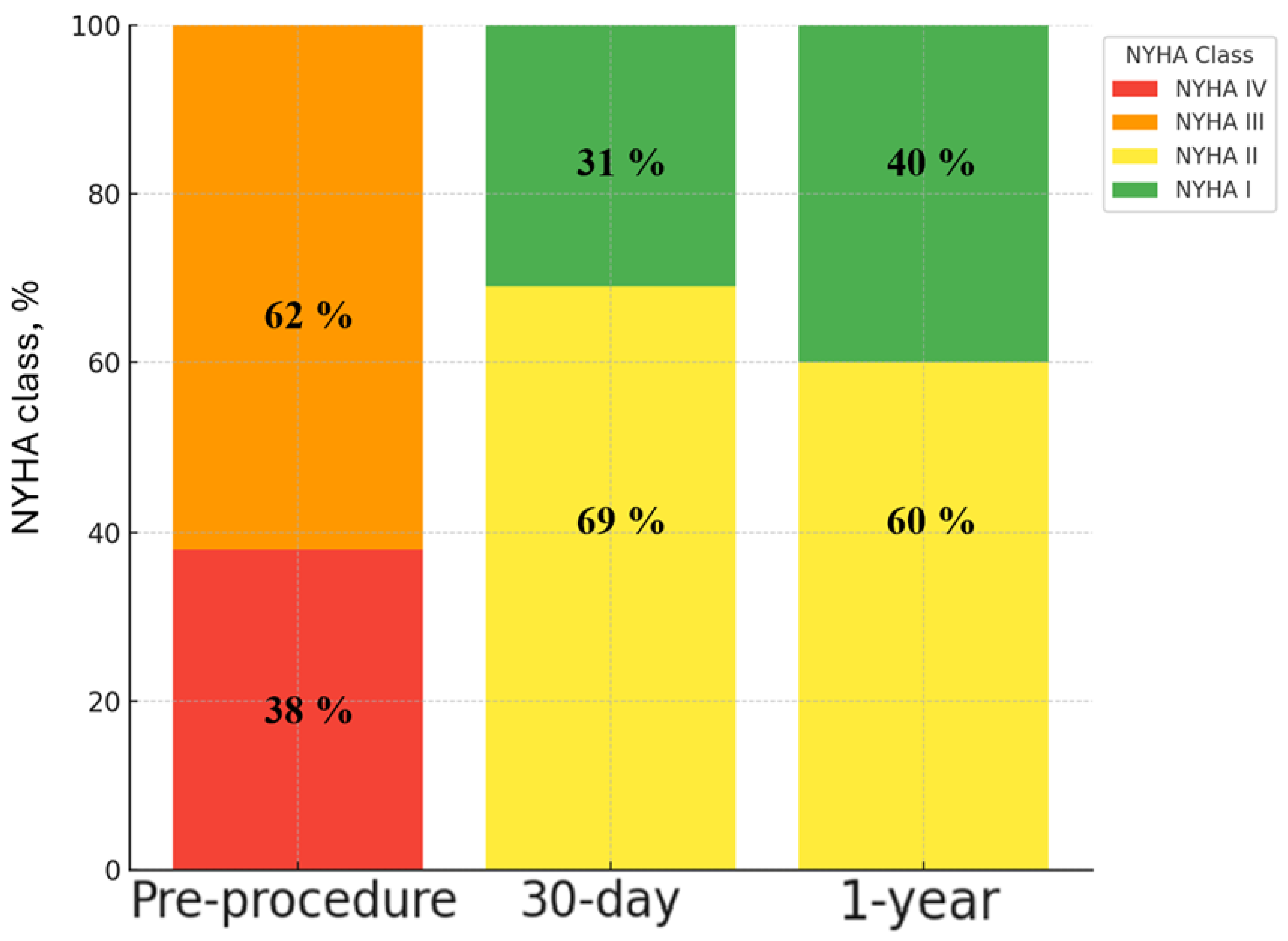
Clinical and echocardiographic follow-up were prospectively scheduled at baseline, early post-procedure, 30 days, and 1 year. Collected variables included baseline demographics, comorbidities, surgical valve characteristics, procedural details, in-hospital events, 30-day outcomes, survival status, heart failure hospitalization, repeat coronary angiography and/or percutaneous coronary intervention after ViV-TAVI, and New York Heart Association (NYHA) functional class at follow-up. Follow-up data were obtained from institutional records, outpatient assessments, and structured follow-up documentation.
2.6. Echocardiographic Assessment
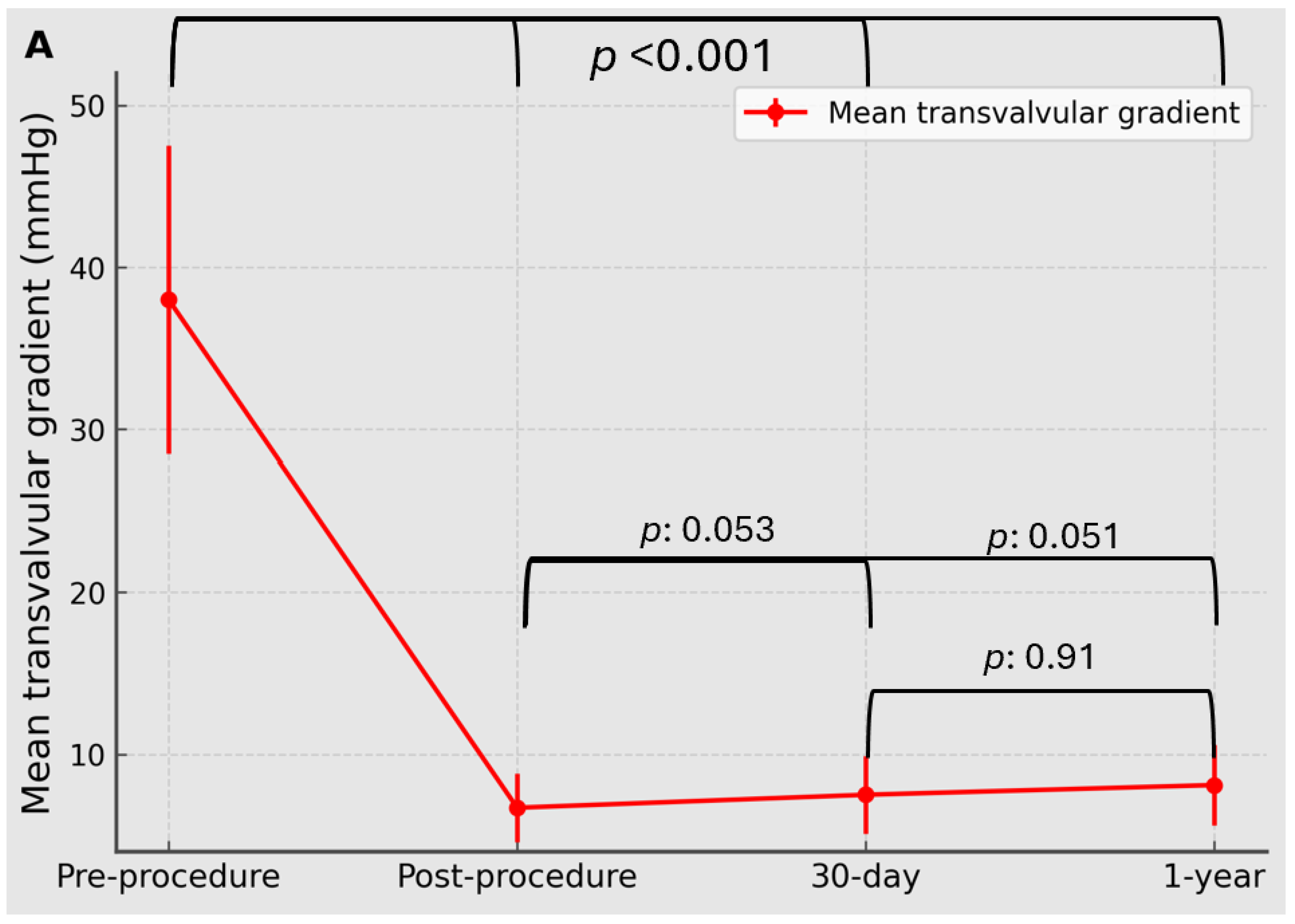
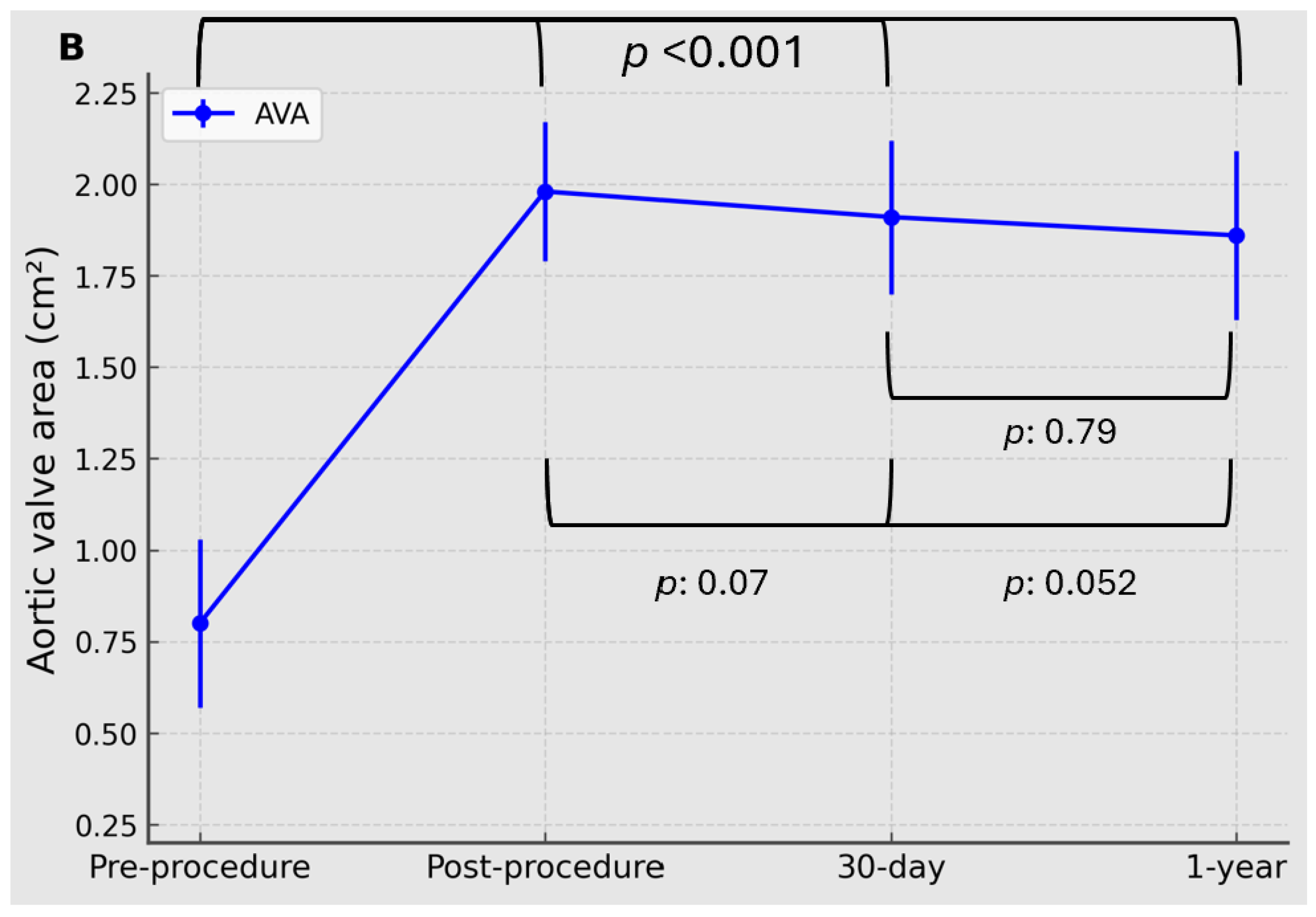
Transthoracic echocardiography was performed according to routine institutional standards and interpreted by experienced echocardiographers using standard multiparametric prosthetic valve assessment principles. For the present analysis, the prespecified core hemodynamic variables were mean transvalvular gradient and aortic valve area (AVA).
Mean transvalvular gradient was derived from continuous-wave Doppler interrogation of the aortic prosthesis using standard Bernoulli-based methods. AVA was calculated by the continuity equation. Measurements were recorded at baseline, post-procedure, 30 days, and 1 year. At 1 year, valve competence was additionally described by grading total aortic regurgitation and paravalvular leak (PVL), and the proportions with greater-than-mild regurgitation or PVL were reported descriptively. For echocardiographic follow-up endpoints, percentages were calculated using the number of patients with available echocardiography at the relevant time point. This available-case approach also applied to subgroup analyses of 1-year echocardiographic outcomes.
2.7. Outcomes and Endpoint Definitions
Procedural and early clinical outcomes were assessed using Valve Academic Research Consortium-3 (VARC-3) definitions where applicable. Technical success was reported according to VARC-3 criteria [
20]. Early safety at 30 days was summarized using a VARC-3-based composite, and the individual 30-day adverse events reported in the present manuscript included all-cause mortality, stroke, myocardial infarction, major vascular complications, major or life-threatening bleeding, acute kidney injury stage 2-3, new permanent pacemaker implantation, valve dysfunction requiring repeat procedure, and valve-related reintervention.
The primary hemodynamic effectiveness analyses focused on serial changes in mean transvalvular gradient and AVA from baseline to post-procedure, 30 days, and 1 year. A prespecified residual hemodynamic endpoint for risk-factor analysis was mean transvalvular gradient ≥10 mmHg at 1-year echocardiography.
Clinical follow-up outcomes included all-cause mortality, heart failure hospitalization, endocarditis, valve thrombosis, valve-related reintervention, and NYHA functional class. Post-ViV coronary access preservation was assessed pragmatically as the feasibility of selective coronary angiography and/or PCI in cases in which coronary access was attempted during follow-up.
2.8. Prespecified Subgroup Analyses
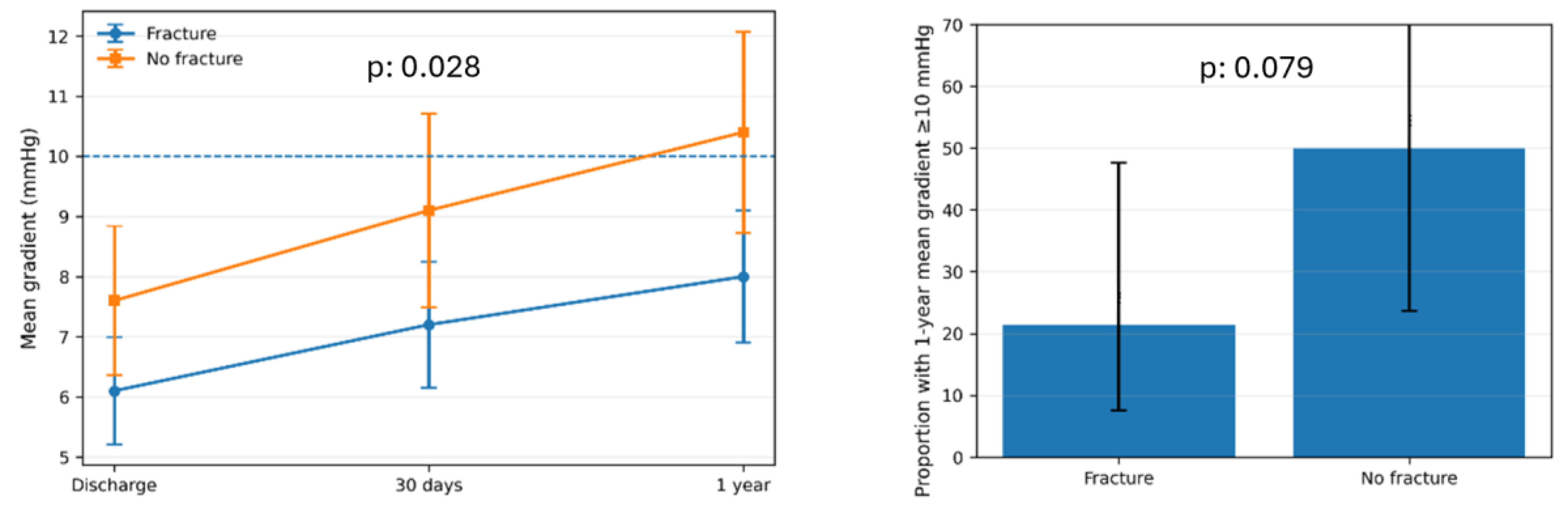
Two prespecified subgroup analyses were performed to evaluate anatomy- and strategy-related determinants of residual hemodynamic burden. First, the primary MyVal cohort was stratified according to surgical valve true ID (≤21 mm vs >21 mm) to examine differences in serial gradients, 1-year echocardiographic valve performance, and clinical outcomes. Second, within the small-valve subgroup (true ID ≤21 mm), outcomes were compared between patients treated with surgical valve fracture and those treated without fracture to explore the hemodynamic impact of fracture-enabled optimization in constrained anatomies.
Given the limited subgroup sample sizes and event counts, these analyses were considered mechanistic and hypothesis-generating. Emphasis was therefore placed on effect size, directionality, and internal consistency across related hemodynamic endpoints rather than on isolated nominal p-values alone.
2.9. Exploratory Internal Device-Platform Comparison
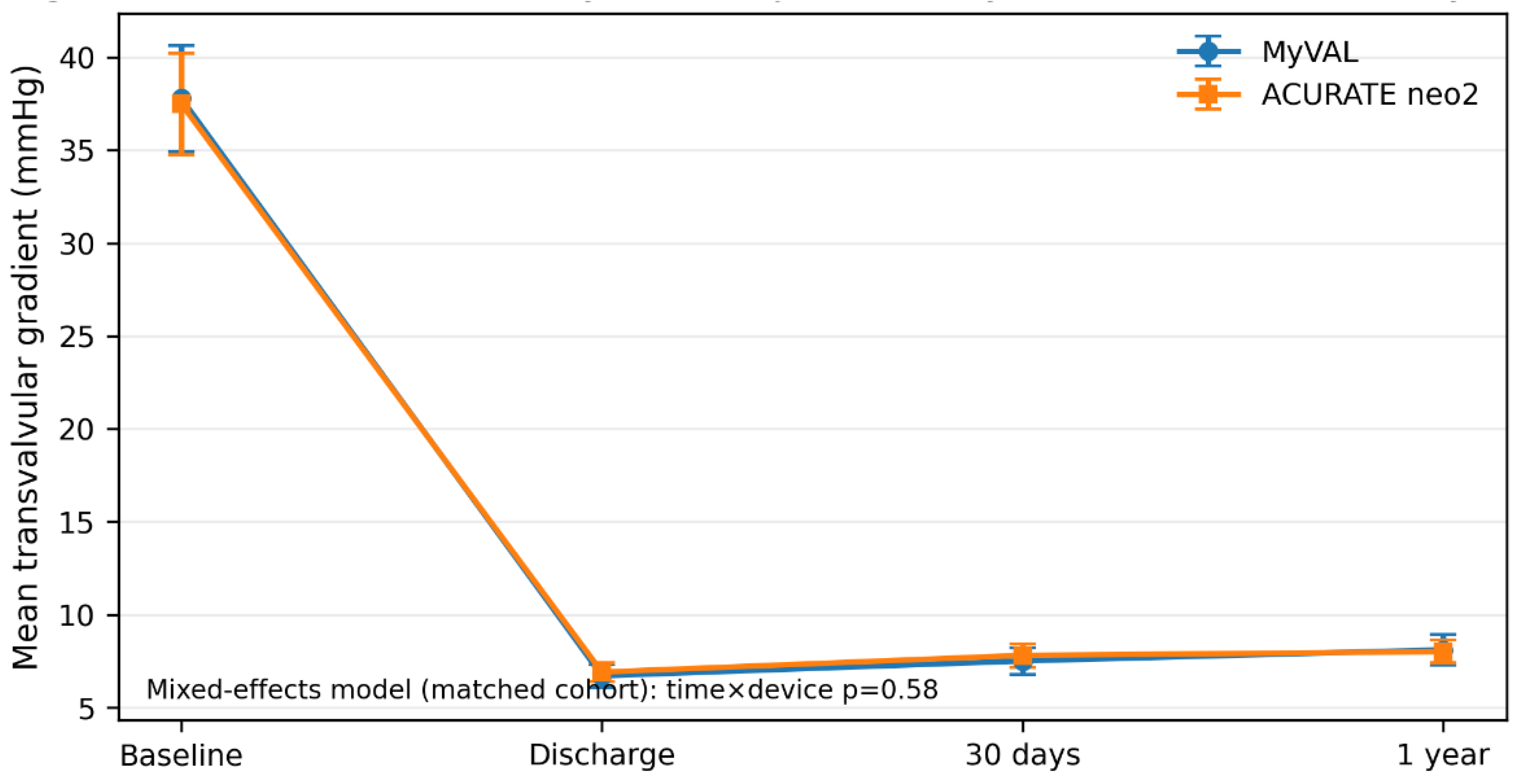
To contextualize MyVal performance within local ViV-TAVI practice, an exploratory internal comparative analysis was performed against a non-randomized institutional cohort of transfemoral ViV-TAVI procedures treated with ACURATE neo2. Because device selection was influenced by anatomy and treatment era, crude comparisons were considered susceptible to confounding by indication. Propensity score methods were therefore used to improve comparability between device groups before exploratory comparative analyses were performed. A detailed description of propensity score estimation, matching strategy, covariate balance assessment, and post-matching diagnostics is provided in the Supplementary Methods.
2.10. Statistical Analysis
All analyses were performed using R (R Foundation for Statistical Computing, Vienna, Austria). Continuous variables are reported as mean ± standard deviation or median with interquartile range, as appropriate, and categorical variables as counts and percentages. All statistical tests were two-sided, and a p-value <0.05 was considered statistically significant. Because subgroup and device-platform comparisons were exploratory, p-values from these analyses were interpreted descriptively and were not adjusted for multiple comparisons.
For between-group comparisons in the primary cohort and prespecified subgroup analyses, continuous variables were compared using Student’s t-test or Welch’s t-test when assumptions for parametric testing were judged acceptable and by Mann-Whitney U testing otherwise. Categorical variables were compared using the chi-square test or Fisher’s exact test, as appropriate according to cell counts.
Serial hemodynamic outcomes, including mean transvalvular gradient and AVA at baseline, post-procedure, 30 days, and 1 year, were analyzed using linear mixed-effects models with a patient-level random intercept to account for within-patient correlation across repeated measurements. Time was modeled as a categorical fixed effect. Overall time effects and prespecified pairwise comparisons between post-procedure, 30-day, and 1-year time points were derived from model-based marginal means.
Predictors of residual hemodynamic burden were evaluated using logistic regression for the endpoint of mean transvalvular gradient ≥10 mmHg at 1 year among patients with available 1-year echocardiography. Because of the limited number of events, Firth penalized logistic regression was used to reduce small-sample bias and instability related to sparse data or quasi-separation. Univariable models were followed by a prespecified multivariable model including surgical valve true ID as a continuous variable, baseline mean transvalvular gradient, failure mechanism category, and surgical valve fracture strategy. Odds ratios (ORs) with 95% confidence intervals (CIs) are reported, and the effect of true ID is expressed per 1-mm decrease. An exploratory true ID × fracture interaction was examined in a secondary model.
For the exploratory MyVal versus ACURATE neo2 comparison, propensity scores for treatment with MyVal were estimated using multivariable logistic regression including clinically relevant baseline, anatomical, and treatment-era variables associated with device selection and outcomes. One-to-one nearest-neighbor matching without replacement was performed on the logit of the propensity score using a caliper width of 0.2 standard deviations of the logit of the propensity score and restriction to the region of common support. Covariate balance after matching was assessed using standardized mean differences, with an absolute standardized mean difference <0.10 considered acceptable.
In the matched cohort, continuous fixed-time outcomes were analyzed using matched-pair methods and are reported as mean differences with 95% CIs. Binary fixed-time outcomes were analyzed using matched-data methods appropriate for paired samples, with effect estimates reported as ORs when estimable. Longitudinal matched hemodynamic trajectories were additionally examined using mixed-effects models including fixed effects for time, device platform, and the time-by-device interaction. Time-to-event outcomes during follow-up, including all-cause mortality, heart failure hospitalization, and the composite of all-cause mortality or heart failure hospitalization, were analyzed exploratorily using Cox proportional hazards models stratified by matched pair, with hazard ratios and 95% CIs reported. Given the low number of clinical follow-up events, these time-to-event estimates were considered exploratory and interpreted descriptively.
2.11. Missing Data
Missing data were handled using an analysis-specific available-data approach. Serial hemodynamic analyses were performed using linear mixed-effects models estimated with maximum likelihood, thereby incorporating all available repeated measurements without requiring complete follow-up at every time point under a missing-at-random assumption. Cross-sectional follow-up analyses, including the 1-year residual gradient endpoint and subgroup-specific 1-year echocardiographic comparisons, were conducted as available-case analyses using the corresponding time-point denominators. No imputation was performed.