Submitted:
03 April 2026
Posted:
07 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. NSAIDS
2.1. Brief Pharmacology Notes
2.2. Adverse Effects and Prudent Use
2.2.1. Cardiovascular Risk
2.2.2. Gastrointestinal Risks
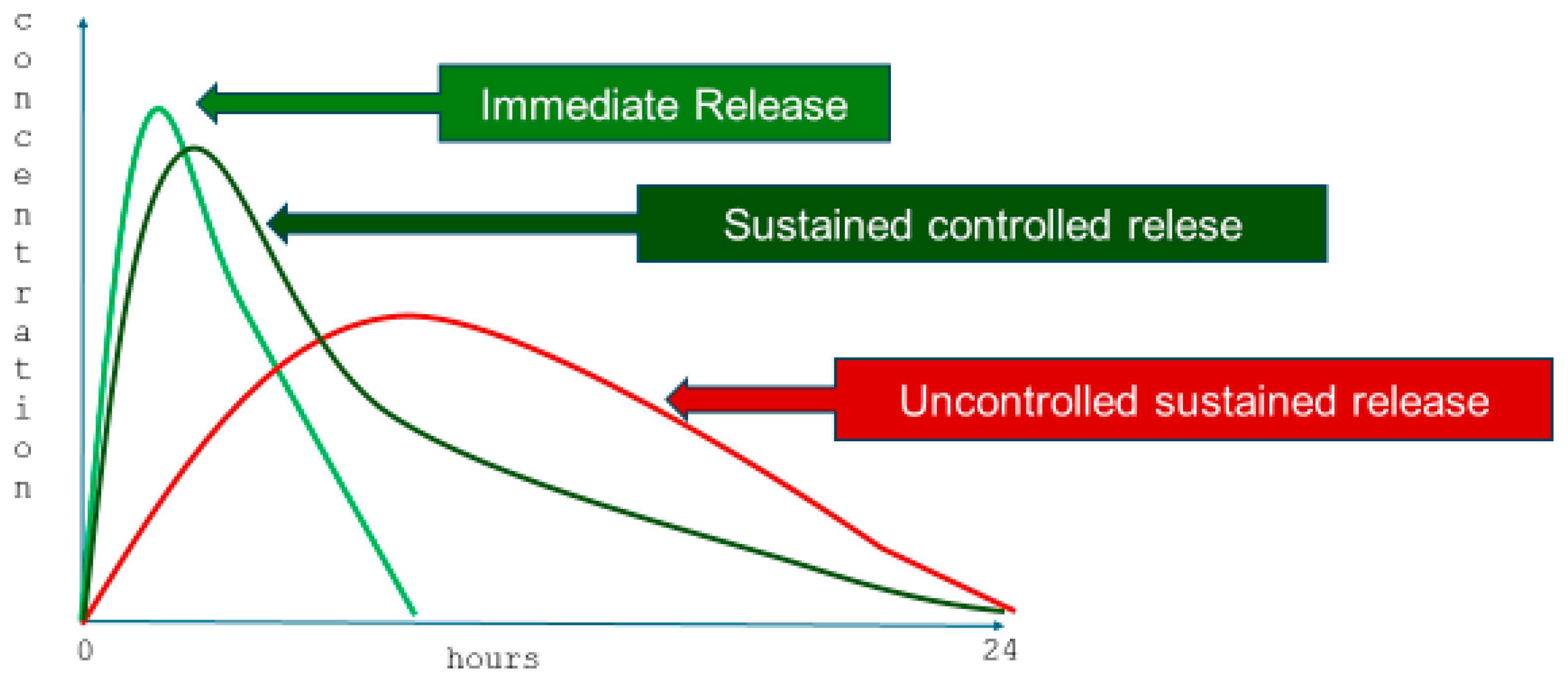
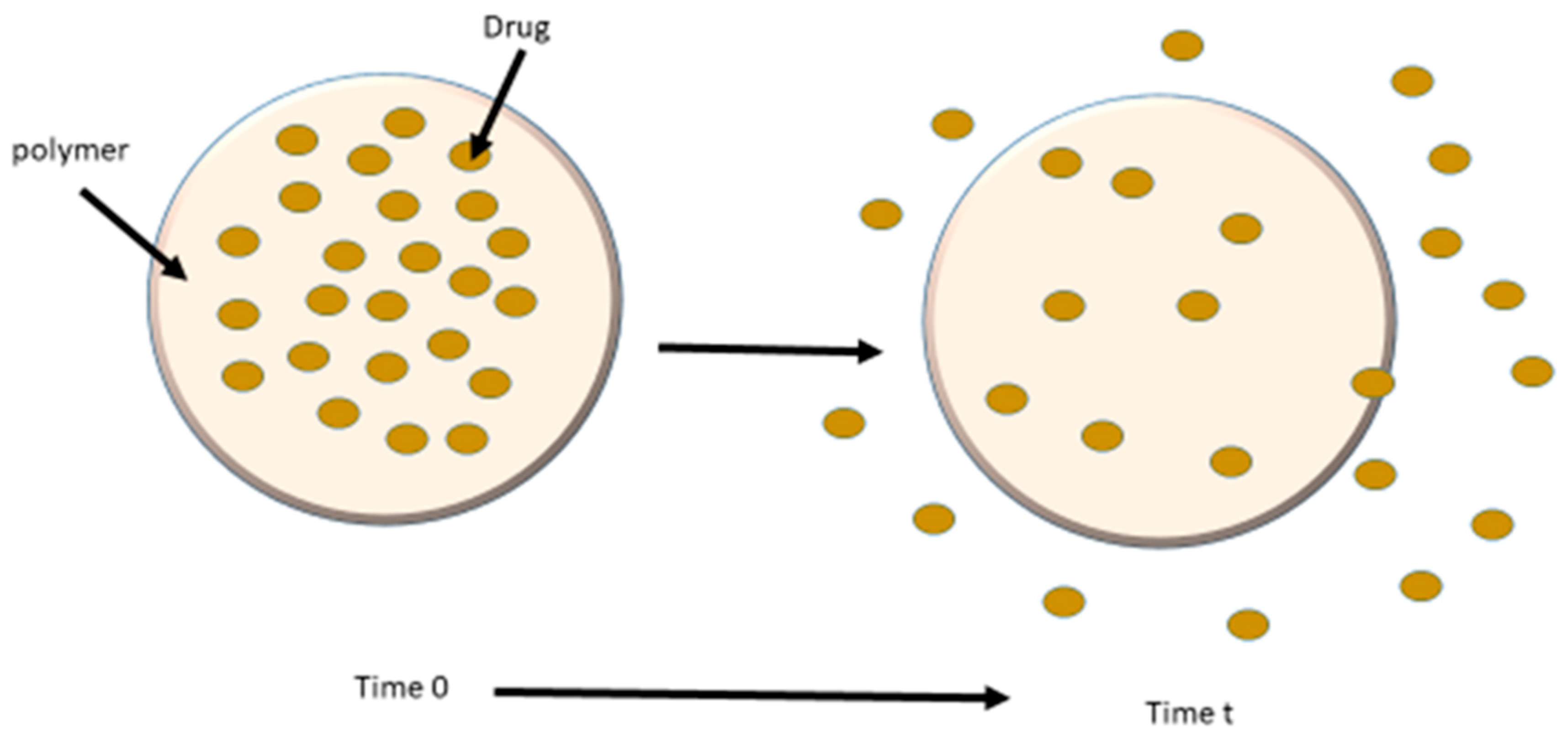
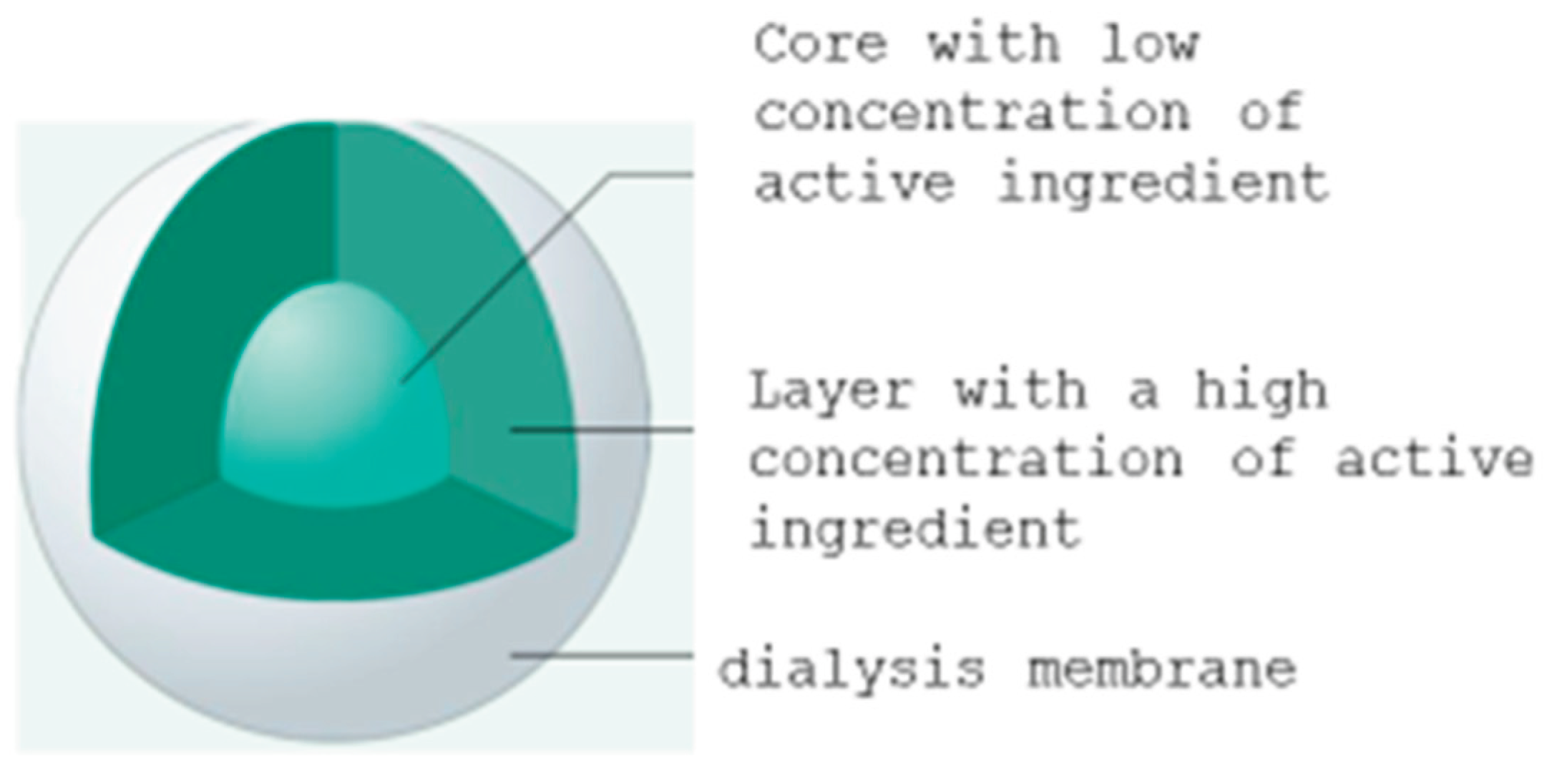
2.3. Sustained Release Formulations
3. Clinical Setting
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| API | Active pharmaceutical ingredient |
| ANMAR | National Association of Rheumatic Patients |
| APMARR | National Association of People with Rheumatological and Rare Diseases |
| COX | Cyclooxygenase |
| CR | Controlled release |
| GDP | Gross Domestic Product |
| IASP | International Association for the Study of Pain |
| KLS | Ketoprofen lysine salt |
| LBP | Low Back Pain |
| NSAIDs | Nonsteroidal anti-inflammatory drugs |
| OA | Osteoarthritis |
| PG | Prostaglandin |
| PR | Prolonged release |
| RA | Rheumatoid arthritis |
| SD | Standard deviation |
| SEM | Standard error of mean |
| SIA-ARTI | Italian Society of Anesthesia, Analgesia, Resuscitation and Intensive Care |
| SIMFER | Italian Society of Physical and Rehabilitation Medicine |
| SIOT | Italian Society of Orthopedics and Traumatology |
| Tlag | Time to onset in blood |
| Tmax | Time to peak |
References
- Raja, SN; Carr, DB; Cohen, M.; et al. The revised International Association for the Study of Pain definition of pain: concepts, challenges, and compromises. Pain 2020, 161, 1976–1982. [Google Scholar] [CrossRef]
- Dahlhamer, J; Lucas, J; Zelaya, C.; et al. Prevalence of Chronic Pain and High-Impact Chronic Pain Among Adults - United States, 2016. MMWR Morb Mortal Wkly Rep 2018, 67, 1001–1006. [Google Scholar] [CrossRef]
- Fayaz, A; Croft, P; Langford, RM.; et al. Prevalence of chronic pain in the UK: a systematic review and meta-analysis of population studies. BMJ Open 2016, 6. Epub ahead of print. [Google Scholar] [CrossRef]
- Coluzzi, F; Di Martino, A. ‘Pain Prehabilitation’ in Major Joint Surgery: The Way Forward to Improve Outcomes and Prevent Pain Chronicity. J Clin Med 2025, 14. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- St. Sauver, JL; Warner, DO; Yawn, BP.; et al. Why patients visit their doctors: assessing the most prevalent conditions in a defined American population. Mayo Clin Proc 2013, 88, 56–67. [Google Scholar] [CrossRef]
- Murray, CJL; Abraham, J; Ali, MK.; et al. The state of US health, 1990-2010: burden of diseases, injuries, and risk factors. JAMA 2013, 310, 591–608. [Google Scholar] [CrossRef] [PubMed]
- Mabarak, D; Khan, ST; Elmenawi, KA.; et al. Chronic Pain Diagnosis Before Total Knee Arthroplasty Leads to Higher Readmission Risk, Lower Patient-Reported Outcome Measures, and Dissatisfaction at One Year: An Analysis of 13,894 Patients. Journal of Arthroplasty 2025. Epub ahead of print. [Google Scholar] [CrossRef]
- Stubhaug, A; Hansen, JL; Hallberg, S.; et al. The costs of chronic pain-Long-term estimates. Eur J Pain 2024, 28, 960–977. [Google Scholar] [CrossRef]
- Lipnik-Stangelj, M. Mediators of inflammation as targets for chronic pain treatment. Mediators Inflamm. 2013. Epub ahead of print 2013. [CrossRef]
- Tal, M. A Role for Inflammation in Chronic Pain. Curr Rev Pain 1999, 3, 440–446. [Google Scholar] [CrossRef]
- Fang, XX; Zhai, MN; Zhu, M.; et al. Inflammation in pathogenesis of chronic pain: Foe and friend. Mol Pain 2023, 19. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Hochberg, MC; Altman, RD; April, KT.; et al. American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res (Hoboken) 2012, 64, 465–474. [Google Scholar] [CrossRef]
- Jordan, KM; Arden, NK; Doherty, M.; et al. EULAR Recommendations 2003: An evidence-based approach to the management of knee osteoarthritis: Report of a Task Force of the Standing Committee for International Clinical Studies Including Therapeutic Trials (ESCISIT). Ann Rheum Dis 2003, 62, 1145–1155. [Google Scholar] [CrossRef] [PubMed]
- Machado, GC; Maher, CG; Ferreira, PH.; et al. Efficacy and safety of paracetamol for spinal pain and osteoarthritis: systematic review and meta-analysis of randomised placebo-controlled trials. BMJ 2015, 350. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Lee, C; Straus, WL; Balshaw, R.; et al. A comparison of the efficacy and safety of nonsteroidal antiinflammatory agents versus acetaminophen in the treatment of osteoarthritis: a meta-analysis. Arthritis Rheum 2004, 51, 746–754. [Google Scholar] [CrossRef]
- Graham, GG; Day, RO; Graudins, A.; et al. FDA proposals to limit the hepatotoxicity of paracetamol (acetaminophen): are they reasonable? Inflammopharmacology 2010, 18, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Sudano, I; Flammer, AJ; Périat, D.; et al. Acetaminophen increases blood pressure in patients with coronary artery disease. Circulation 2010, 122, 1789–1796. [Google Scholar] [CrossRef]
- Marcum, ZA; Duncan, NA; Makris, UE. Pharmacotherapies in Geriatric Chronic Pain Management. Clin Geriatr Med 2016, 32, 705–724. [Google Scholar] [CrossRef]
- Da Costa, BR; Pereira, T V.; Saadat, P.; et al. Effectiveness and safety of non-steroidal anti-inflammatory drugs and opioid treatment for knee and hip osteoarthritis: network meta-analysis. BMJ 2021, 375. Epub ahead of print. [Google Scholar] [CrossRef]
- Shareef, J; Sridhar, SB; Saeed, ZM.; et al. Evaluation of Potential Drug-Drug Interactions, Polypharmacy, and Prescribing Patterns of NSAIDs Among the Older Adults in a Secondary Care Setting. Clin Interv Aging 2025, 20, 1875–1894. [Google Scholar] [CrossRef]
- Regi, JK; Lalwani, K; Pawar, S. Comparative trends in the usage of nonsteroidal anti-inflammatory drugs: Self-administration versus prescription. MGM Journal of Medical Sciences 2024, 11, 139–145. [Google Scholar] [CrossRef]
- Gunaydin, C; Bilge, SS. Effects of Nonsteroidal Anti-Inflammatory Drugs at the Molecular Level. Eurasian J Med 2018, 50, 116–121. [Google Scholar] [CrossRef]
- Kulesza, A; Paczek, L; Burdzinska, A. The Role of COX-2 and PGE2 in the Regulation of Immunomodulation and Other Functions of Mesenchymal Stromal Cells. Biomedicines 2023, 11. Epub ahead of print. [Google Scholar] [CrossRef]
- Scarpignato, C; Lanas, A; Blandizzi, C.; et al. Safe prescribing of non-steroidal anti-inflammatory drugs in patients with osteoarthritis--an expert consensus addressing benefits as well as gastrointestinal and cardiovascular risks. BMC Med 2015, 13. Epub ahead of print. [Google Scholar] [CrossRef]
- Davies, EA; O’Mahony, MS. Adverse drug reactions in special populations - the elderly. Br J Clin Pharmacol 2015, 80, 796–807. [Google Scholar] [CrossRef]
- Griffin, MR. Epidemiology of nonsteroidal anti-inflammatory drug-associated gastrointestinal injury. American Journal of Medicine 1998, 104. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Antman, EM; Bennett, JS; Daugherty, A.; et al. Use of nonsteroidal antiinflammatory drugs: an update for clinicians: a scientific statement from the American Heart Association. Circulation 2007, 115, 1634–1642. [Google Scholar] [CrossRef] [PubMed]
- Sondergaard, KB; Weeke, P; Wissenberg, M.; et al. Non-steroidal anti-inflammatory drug use is associated with increased risk of out-of-hospital cardiac arrest: a nationwide case-time-control study. Eur Heart J Cardiovasc Pharmacother 2017, 3, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Helin-Salmivaara, A; Virtanen, A; Vesalainen, R.; et al. NSAID use and the risk of hospitalization for first myocardial infarction in the general population: a nationwide case-control study from Finland. Eur Heart J 2006, 27, 1657–1663. [Google Scholar] [CrossRef]
- Masclee, GMC; Straatman, H; Arfè, A.; et al. Risk of acute myocardial infarction during use of individual NSAIDs: A nested case-control study from the SOS project. PLoS One 2018, 13. Epub ahead of print. [Google Scholar] [CrossRef]
- Baigent, C; Bhala, N; Emberson, J.; et al. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: Meta-analyses of individual participant data from randomised trials. The Lancet 2013, 382, 769–779. [Google Scholar]
- Hopkins, S; Yang, V; Liew, DFL. Choosing a nonsteroidal anti-inflammatory drug for pain. Aust Prescr 2025, 48, 139–144. [Google Scholar] [CrossRef]
- Rockwell, MS; Oyese, EG; Singh, E.; et al. Scoping review of interventions to de-implement potentially harmful non-steroidal anti-inflammatory drugs (NSAIDs) in healthcare settings. BMJ Open 2024, 14. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Brandolini, L; d’Angelo, M; Antonosante, A.; et al. Differential protein modulation by ketoprofen and ibuprofen underlines different cellular response by gastric epithelium. J Cell Physiol 2018, 233, 2304–2312. [Google Scholar] [CrossRef]
- Novelli, R; Aramini, A; Boccella, S.; et al. Ketoprofen lysine salt has a better gastrointestinal and renal tolerability than ketoprofen acid: A comparative tolerability study in the Beagle dog. Biomedicine and Pharmacotherapy 2022, 153. Epub ahead of print. [Google Scholar] [CrossRef]
- De Abajo, FJ; Montero, D; Madurga, M.; et al. Acute and clinically relevant drug-induced liver injury: a population-based case-control study. Br J Clin Pharmacol 2004, 58, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Jung, SH; Lee, W; Park, SH.; et al. Diclofenac impairs autophagic flux via oxidative stress and lysosomal dysfunction: Implications for hepatotoxicity. Redox Biol 2020, 37. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Biasi, G; Canova, N; Palazzini, E.; et al. Comparative pharmacokinetic study of a single dose of two prolonged-release formulations of diclofenac in healthy subjects. Current Therapeutic Research 1998, 59, 785–792. [Google Scholar] [CrossRef]
- Fincato, G; MPF; ACScapinelli. Pharmacokinetic comparison of 320 mg of 2 different oral capsule formulations of ketoprofen-lysine salt in healthy-volunteers.10.4. Adv Ther 1993, 10, 182–188. [Google Scholar]
- Jiménez-Sánchez, S; Jiménez-García, R; Hernández-Barrera, V.; et al. Has the Prevalence of Invalidating Musculoskeletal Pain Changed Over the Last 15 Years (1993-2006)? A Spanish Population-Based Survey. Journal of Pain 2010, 11, 612–620. [Google Scholar] [CrossRef]
- Alves, B; Tavares, I; Pozza, DH. Biomarkers and Breakdowns: Neuroinflammatory Drivers Linking Sleep Disorders and Chronic Pain. Biomedicines 2026, 14, 116. [Google Scholar] [CrossRef] [PubMed]
- Oostinga, D; Steverink, JG; van Wijck, AJM.; et al. An understanding of bone pain: A narrative review. Bone 2020, 134. Epub ahead of print. [Google Scholar] [CrossRef]
- Anderson, DB; Shaheed, CA. Medications for Treating Low Back Pain in Adults. Evidence for the Use of Paracetamol, Opioids, Nonsteroidal Anti-inflammatories, Muscle Relaxants, Antibiotics, and Antidepressants: An Overview for Musculoskeletal Clinicians. J Orthop Sports Phys Ther 2022, 52, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Anderson, DB; De, Luca K; Jensen, RK.; et al. A critical appraisal of clinical practice guidelines for the treatment of lumbar spinal stenosis. Spine Journal 2021, 21, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Iolascon, G; Migliore, A; Beretta, G.; et al. Pain Management in Knee Osteoarthritis: Insights from an Exploratory Online Survey of Italian Patients and Physicians. Healthcare (Basel) 2024, 12. Epub ahead of print. [Google Scholar] [CrossRef]
- Price, AJ; Alvand, A; Troelsen, A.; et al. Knee replacement. The Lancet 2018, 392, 1672–1682. [Google Scholar] [CrossRef] [PubMed]
- Staab, JS; Kolb, AL; Tomlinson, RE.; et al. Emerging evidence that adaptive bone formation inhibition by non-steroidal anti-inflammatory drugs increases stress fracture risk. Exp Biol Med 2021, 246, 1104–1111. [Google Scholar] [CrossRef] [PubMed]
- Lee, M; Cha, JM. Real-World Bleeding Risk of Anticoagulant and Nonsteroidal Anti-inflammatory Drugs Combotherapy versus Anticoagulant Monotherapy. Gut Liver 2024, 18, 824–833. [Google Scholar] [CrossRef] [PubMed]
- US Department of Health and Human Services. Pain Management Best Practices Inter-Agency Task Force Report - Healthy People 2030 | odphp.health.gov. Available online: https://odphp.health.gov/healthypeople/tools-action/browse-evidence-based-resources/pain-management-best-practices-inter-agency-task-force-report (accessed on 27 February 2026).
- NICE. Overview | Chronic pain (primary and secondary) in over 16s: assessment of all chronic pain and management of chronic primary pain | Guidance | NICE. Available online: https://www.nice.org.uk/guidance/ng193 (accessed on 27 February 2026).
- Ho, KY; Gwee, KA; Cheng, YK.; et al. Nonsteroidal anti-inflammatory drugs in chronic pain: implications of new data for clinical practice. J Pain Res 2018, 11, 1937–1948. [Google Scholar] [CrossRef] [PubMed]
- Chou, R; McDonagh, MS; Nakamoto, E.; et al. Analgesics for Osteoarthritis: An Update of the 2006 Comparative Effectiveness Review [Internet] - PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/22091473/ (accessed on 27 February 2026).
- Bally, M; Dendukuri, N; Rich, B.; et al. Risk of acute myocardial infarction with NSAIDs in real world use: bayesian meta-analysis of individual patient data. BMJ 2017, 357. Epub ahead of print. [Google Scholar] [CrossRef]
- Bally, M; Beauchamp, ME; Abrahamowicz, M.; et al. Risk of acute myocardial infarction with real-world NSAIDs depends on dose and timing of exposure. Pharmacoepidemiol Drug Saf 2018, 27, 69–77. [Google Scholar]
- Drini, M. Peptic ulcer disease and non-steroidal anti-inflammatory drugs. Aust Prescr 2017, 40, 91–93. [Google Scholar] [CrossRef] [PubMed]
| NSAID |
Dose equivalence (mg) |
Dose Frequency (times daily) |
Max. daily dose (mg) |
| ASA (anti-inflammatory) |
650 | 4 to 6 | 4000 |
| Celecoxib* | 200 | 1 to 2 | 400 |
| Diclofenac | 50 | 2 to 3 | 150 |
| Etoricoxib* | 30 | once daily | 120 |
| Ibuprofen | 600 | 4 to 6 | 3200 |
| Indometacin | 75 | 2 | 200 |
| Ketoprofen lysine salt | 50 | 2 to 4 | 300 |
| Ketorolac | 20 | 2 | 40 |
| Naproxen | 500 | 2 | 1500 |
| Gastrointestinal risk (REF:29-31) |
Cardiovascular risk (REF: 23-27) |
|
| High risk | piroxicam ketorolac high-dose aspirin (when used as an anti-inflammatory) |
etoricoxib ketorolac diclofenac |
| Moderate risk | naproxen indometacin diclofenac ibuprofen |
ibuprofen meloxicam indometacin |
| Low risk | meloxicam etoricoxib (low dose) ketoprofen lisine salt celecoxib (lowest) |
naproxen piroxicam ketoprofen (lowest) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




