Submitted:
02 April 2026
Posted:
03 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
The Enduring Puzzle of Skull Pneumatization
Historical Context and Reappraisal
What Has Been Missing: The Novel Integration
2. Materials and Methods
Anatomical Background
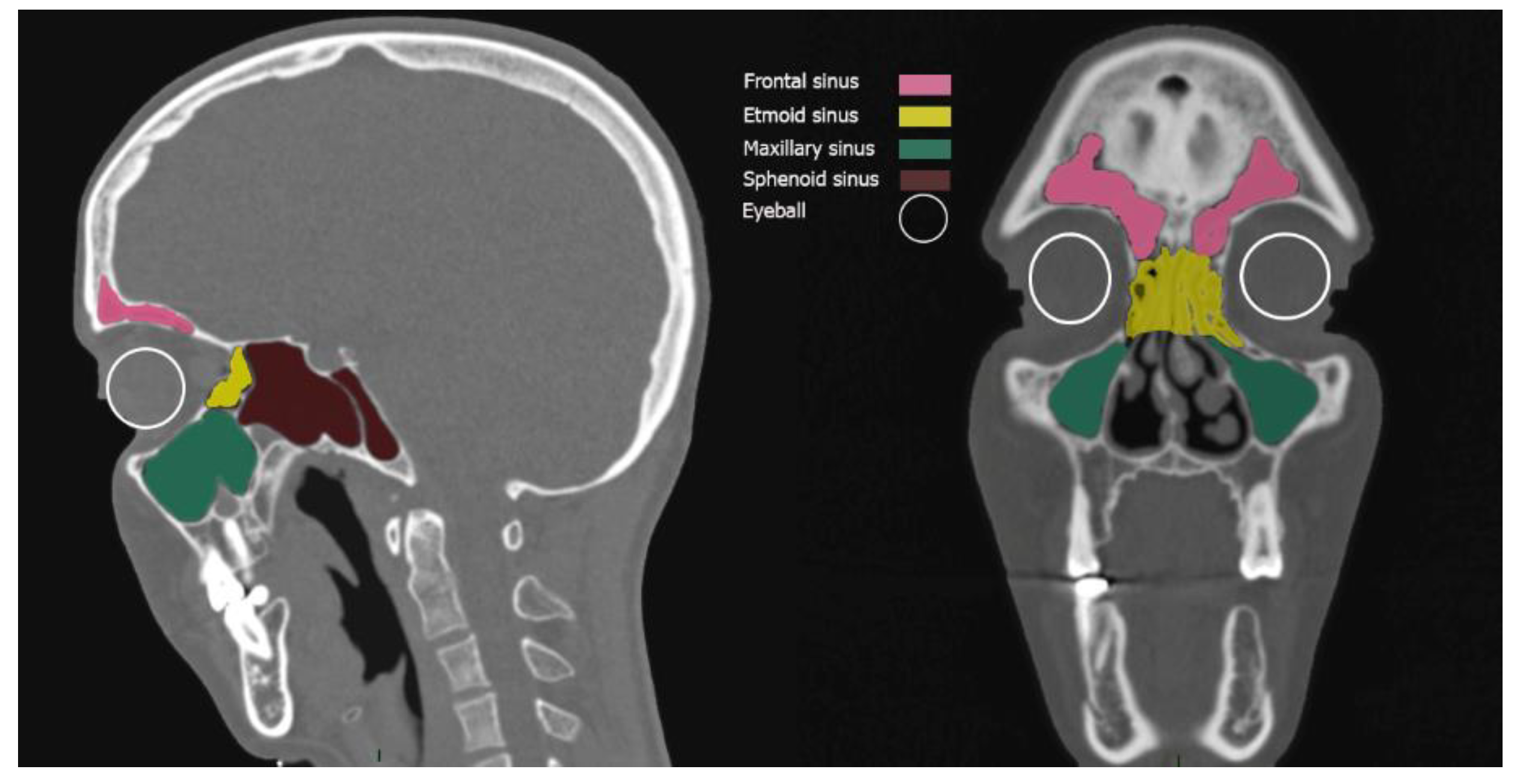
Paranasal Sinuses: The Periorbital Thermal Envelope
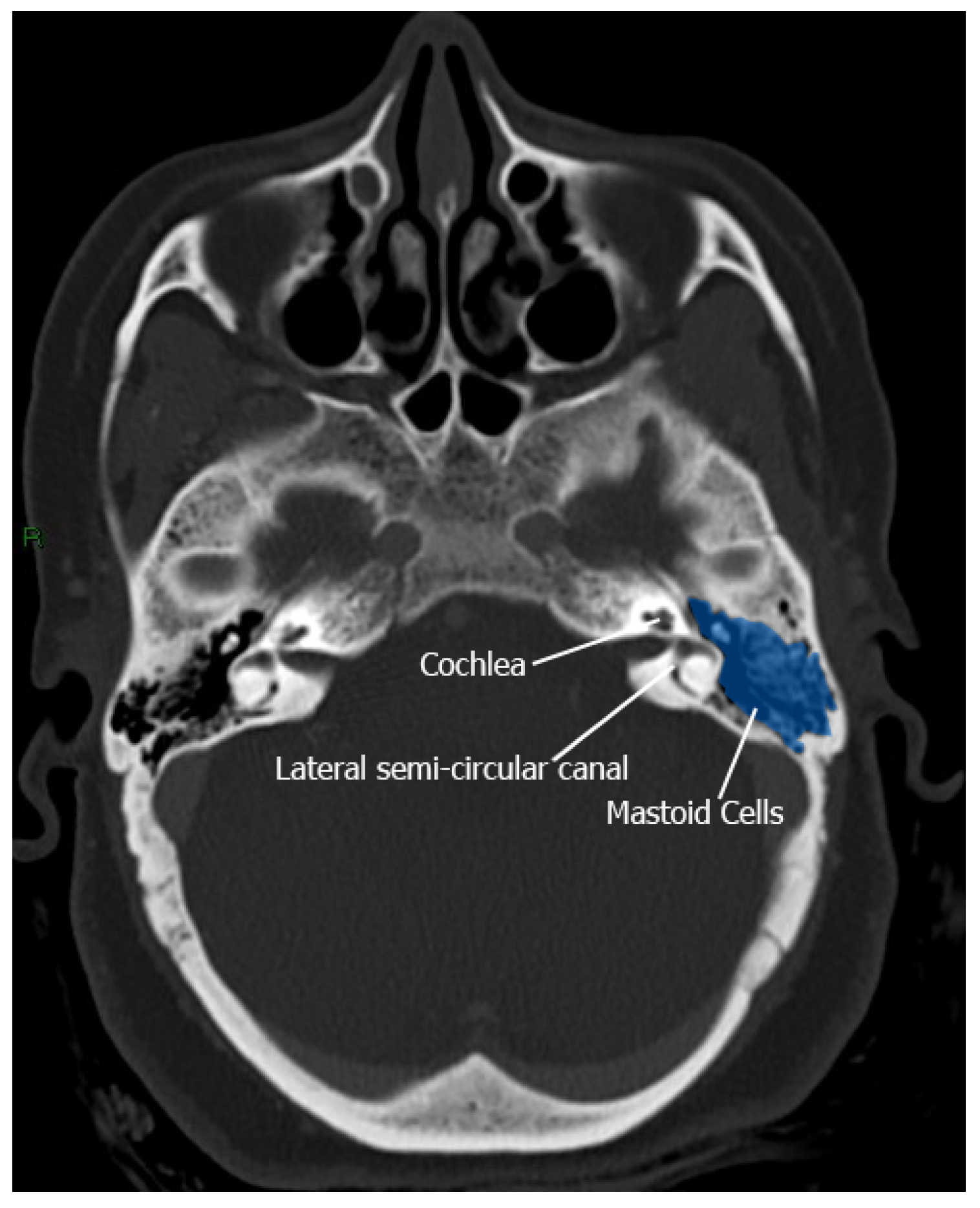
Mastoid Air Cells: The Labyrinthine Thermal Shield
Target Structures: Vitreous and Endolymph
3. Results
Proposed Mechanism: Three-Component Architecture
Component 1: Passive Thermal Insulation
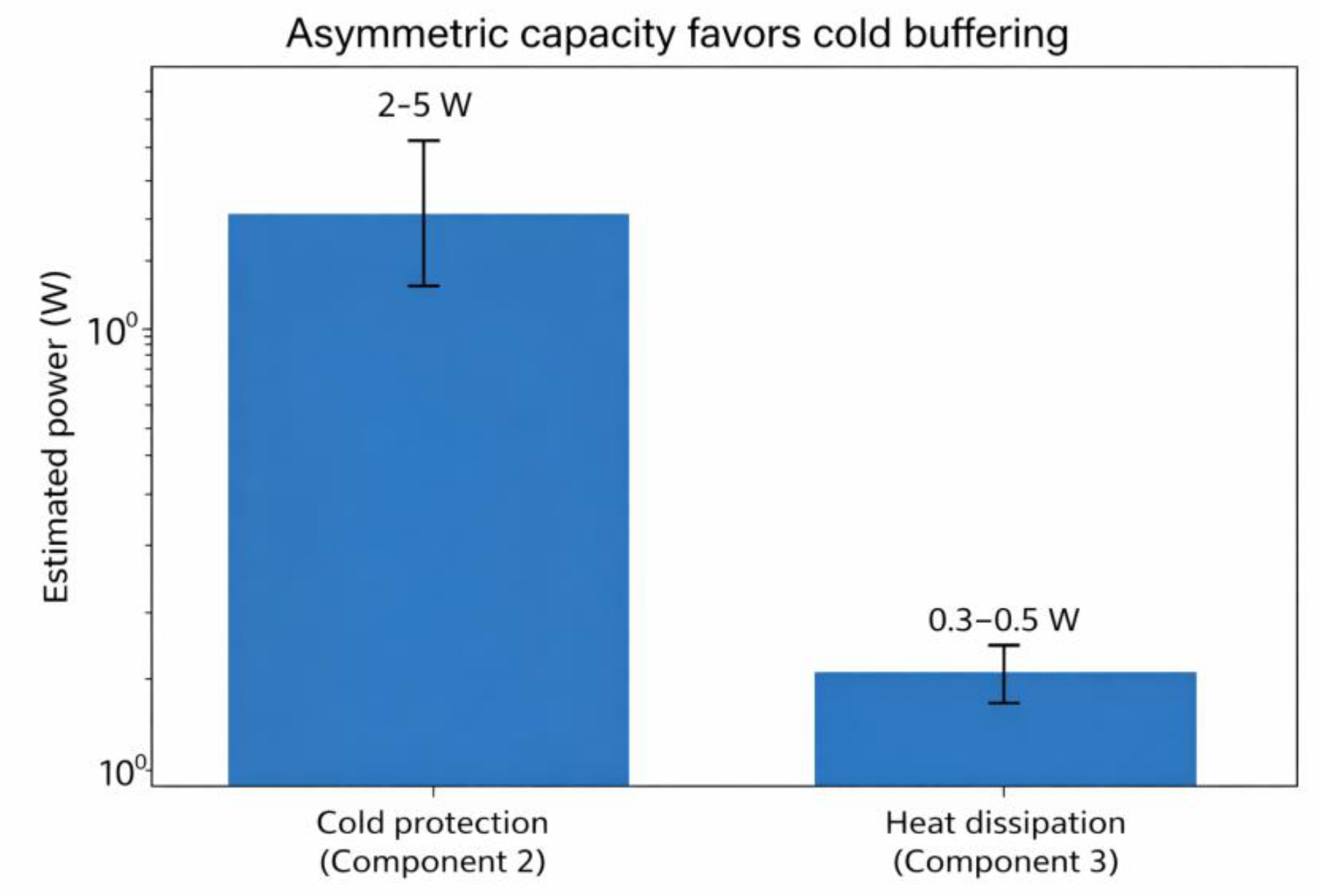
Component 2: Active Cold Protection
Component 3: Active Heat Dissipation
Asymmetric Architecture: Thermodynamic Rationale
4. Discussion
Supporting Evidence
Clinical Evidence: Post-Mastoidectomy Thermal Sensitivity
Physiological Evidence: Selective Brain Cooling Studies
Anatomical Evidence: Climate Correlation Reinterpreted
Why Sensory Organs Rather than the Brain?
Ocular Implications
Testable Predictions
Prediction 1: Structural-Thermal Correlation
Prediction 2: Directional Asymmetry
Prediction 3: Autonomic Modulation
Prediction 4: Clinical Phenotype
Prediction 5: Comparative Anatomy
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Ethics Approval
Data Availability
Running Title
Conflicts of Interest
References
- Proetz, AW. Applied Physiology of the Nose, 2nd ed.; Annals Publishing Company: St. Louis, 1953. [Google Scholar]
- Koertvelyessy, T. Relationships between the frontal sinus and climatic conditions. Am J Phys Anthropol. 1972, 37(2), 161–172. [Google Scholar] [CrossRef] [PubMed]
- Negus, VE. The Comparative Anatomy and Physiology of the Nose and Paranasal Sinuses; E. & S. Livingstone Ltd: Edinburgh, 1958. [Google Scholar]
- Rae, TC; Koppe, T; Stringer, CB. The Neanderthal face is not cold adapted. J Hum Evol. 2011, 60(2), 234–239. [Google Scholar] [CrossRef] [PubMed]
- Magnuson, B. Functions of the mastoid cell system: auto-regulation of temperature and gas pressure. Acta Otolaryngol. 2003, 123(2), 122–129. [Google Scholar] [CrossRef] [PubMed]
- Gallup, AC; Hack, GD. Human paranasal sinuses and selective brain cooling: A ventilation system activated by yawning? Med Hypotheses 2011, 77(6), 970–973. [Google Scholar] [CrossRef] [PubMed]
- Švagan, M; Battelino, S; Gros, A; et al. Evaluating the sequelae of mastoidectomy for acute mastoiditis: Long-term follow-up study of mastoid function. J Clin Med. 2025, 14(19), 6689. [Google Scholar] [CrossRef] [PubMed]
- Standring, S (Ed.) Gray’s Anatomy: The Anatomical Basis of Clinical Practice, 42nd ed.; Elsevier: Edinburgh, 2021. [Google Scholar]
- Bluestone, CD; Klein, JO. Otitis Media in Infants and Children, 4th ed.; BC Decker Inc: Hamilton, 2007. [Google Scholar]
- Baloh, RW; Honrubia, V; Kerber, KA. Baloh and Honrubia’s Clinical Neurophysiology of the Vestibular System, 4th ed.; Oxford University Press: New York, 2011. [Google Scholar]
- Duck, FA. Physical Properties of Tissues: A Comprehensive Reference Book; Academic Press: San Diego, 1990. [Google Scholar]
- Cengel, YA; Ghajar, AJ. Heat and Mass Transfer: Fundamentals and Applications, 5th ed.; McGraw-Hill: New York, 2015. [Google Scholar]
- Eccles, R. Nasal airflow in health and disease. Acta Otolaryngol. 2000, 120(5), 580–595. [Google Scholar] [CrossRef] [PubMed]
- Baker, MA. A brain-cooling system in mammals. Sci Am. 1979, 240(5), 130–139. [Google Scholar] [CrossRef] [PubMed]
- Koehler, RC; Traystman, RJ; Bhardwaj, A. Rapid selective cerebral cooling with extracorporeal circulation. Crit Care Med. 2009, 37, S291–S296. [Google Scholar]
- Morrison, SF; Nakamura, K. Central mechanisms for thermoregulation. Annu Rev Physiol. 2019, 81, 285–308. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



