Submitted:
02 April 2026
Posted:
03 April 2026
You are already at the latest version
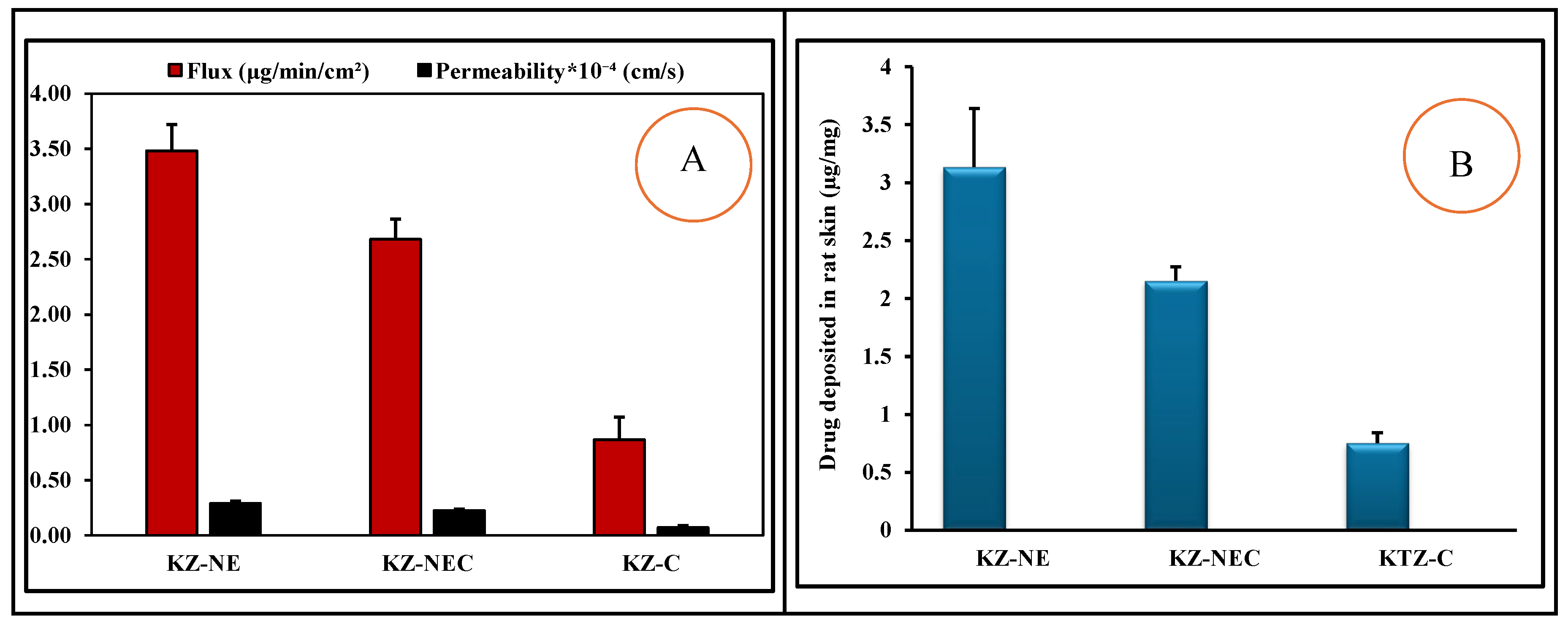
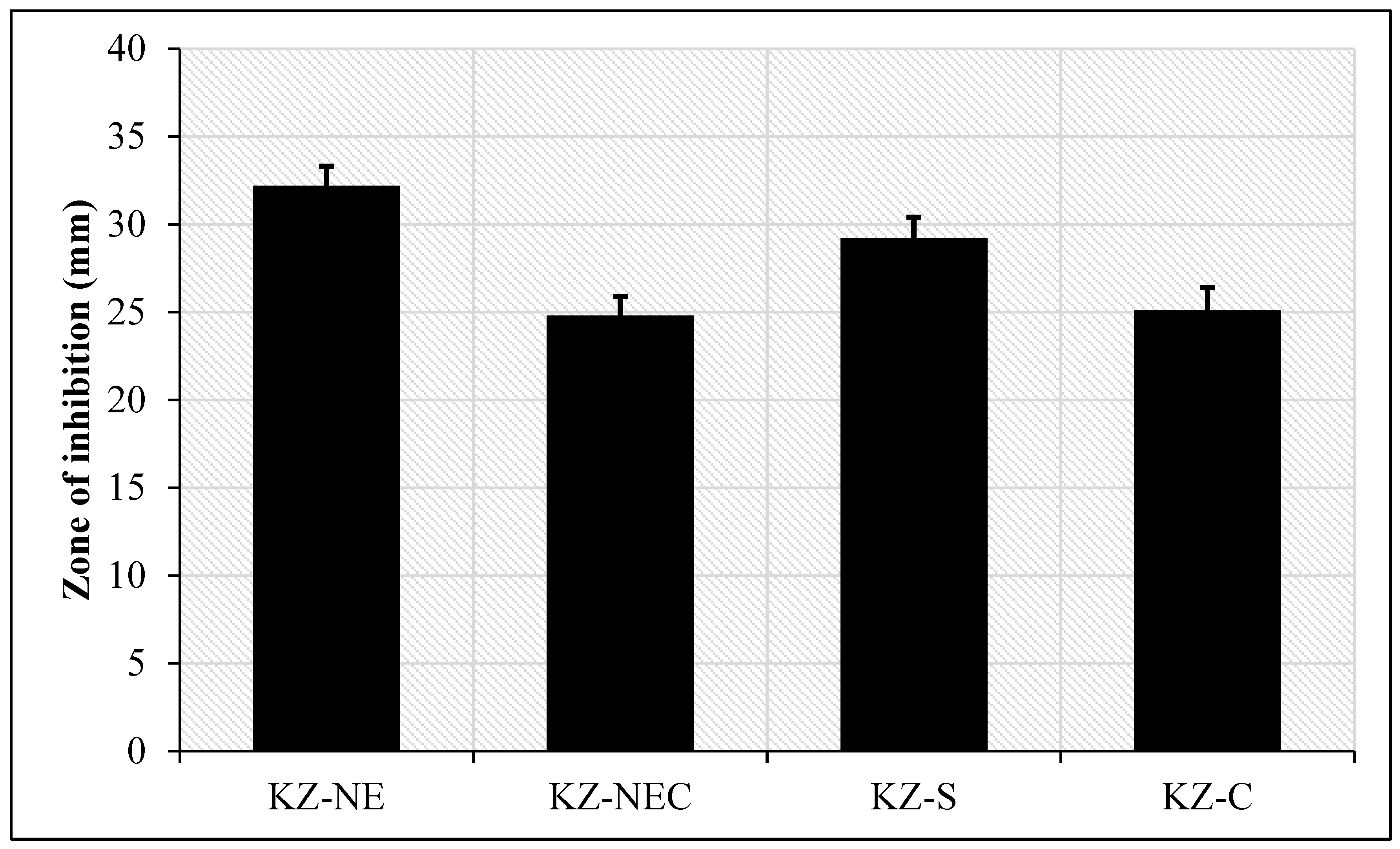
Abstract
Background/Objective: The introduction of Ketoconazole (KZ, Nizoral®) in 1977 by Janssen Pharmaceutica marked a significant milestone in medical mycology as the first broad-spectrum oral antifungal agent. However, KZ is a highly lipophilic compound, presenting significant challenges in the development of efficient topical formulations. Moreover, oral KZ has undergone labeling revisions and market withdrawal due to serious hepatic side effects. This study aimed to design, optimize, and evaluate KZ-loaded nanoemulsions (NEs; KZ-NEs) as a delivery platform that could improve skin bioavailability and antifungal activity. Methods: Optimized KZ-NEs were converted to a mucoadhesive formulation (KZ-NEC) by the addition of Carbopol® 940 NF to enhance the adherence of the formulations to the skin surface. NEs were evaluated concerning physical appearance, globule size, polydispersity index, zeta potential, pH, viscosity, and drug content. Optimized KZ-NE and lead KZ-NEC formulations were further evaluated for in vitro release, ex vivo skin permeation and deposition, skin irritation, and in vivo studies. Results: In vitro release studies revealed that nanocarrier systems provided a sustained release of KZ over 24 hours. The ex vivo permeability coefficients of KZ from the optimized KZ-NE and lead KZ-NEC formulations were approximately 4 and 3-fold greater than that achieved with the marketed cream formulation, respectively. In addition, the Cmax of the lead KZ-NEC formulation (14.4±1.1 μg/mL) was significantly higher (p<0.05) compared with the marketed cream formulation (10.5±0.5 μg/mL). Moreover, in vitro antifungal susceptibility testing showed that KZ demonstrated improved antifungal efficacy when incorporated into the NE and NEC formulations. Neither of the NE-based formulations caused any alterations in skin color or morphology during the 24-hour visual observation period. Both NE-based formulations were stable for 90 days (the last time-point tested) at three different storage conditions. Conclusions: NE-based formulation could serve as an effective topical delivery platform for KZ and could improve therapeutic outcomes for patients with topical fungal infections.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Animals
3. Methods
3.1. Analytical Method
3.2. Screening Oils
3.3. Saturation Solubility in the Candidate Oil
3.4. Experimental Design
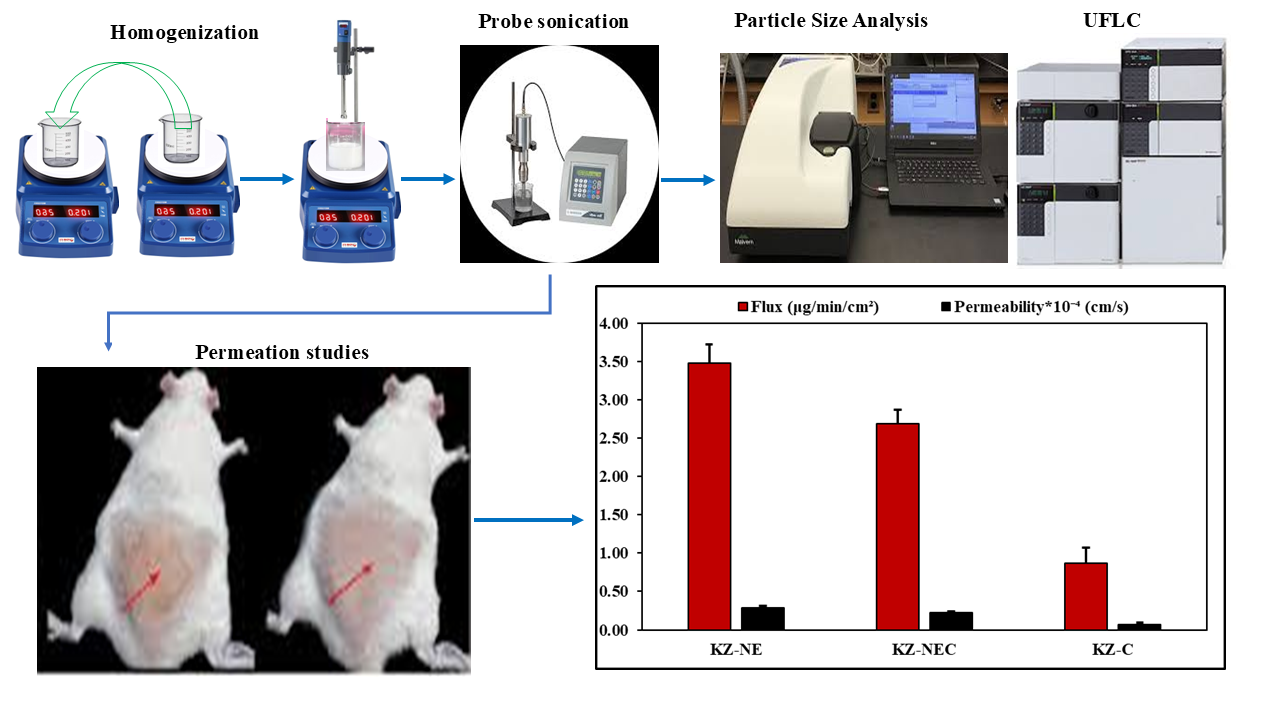
3.5. Preparation KZ-NEs
3.6. Preparation of the Mucoadhesive KZ-NE (KZ-NEC)
3.7. Preparation of KZ Solution (KZ-S)
3.8. Control Formulation (Cream, KZ-C)
3.9. Characterization of KZ-NEs and KZ-NEC
4. KZ Content
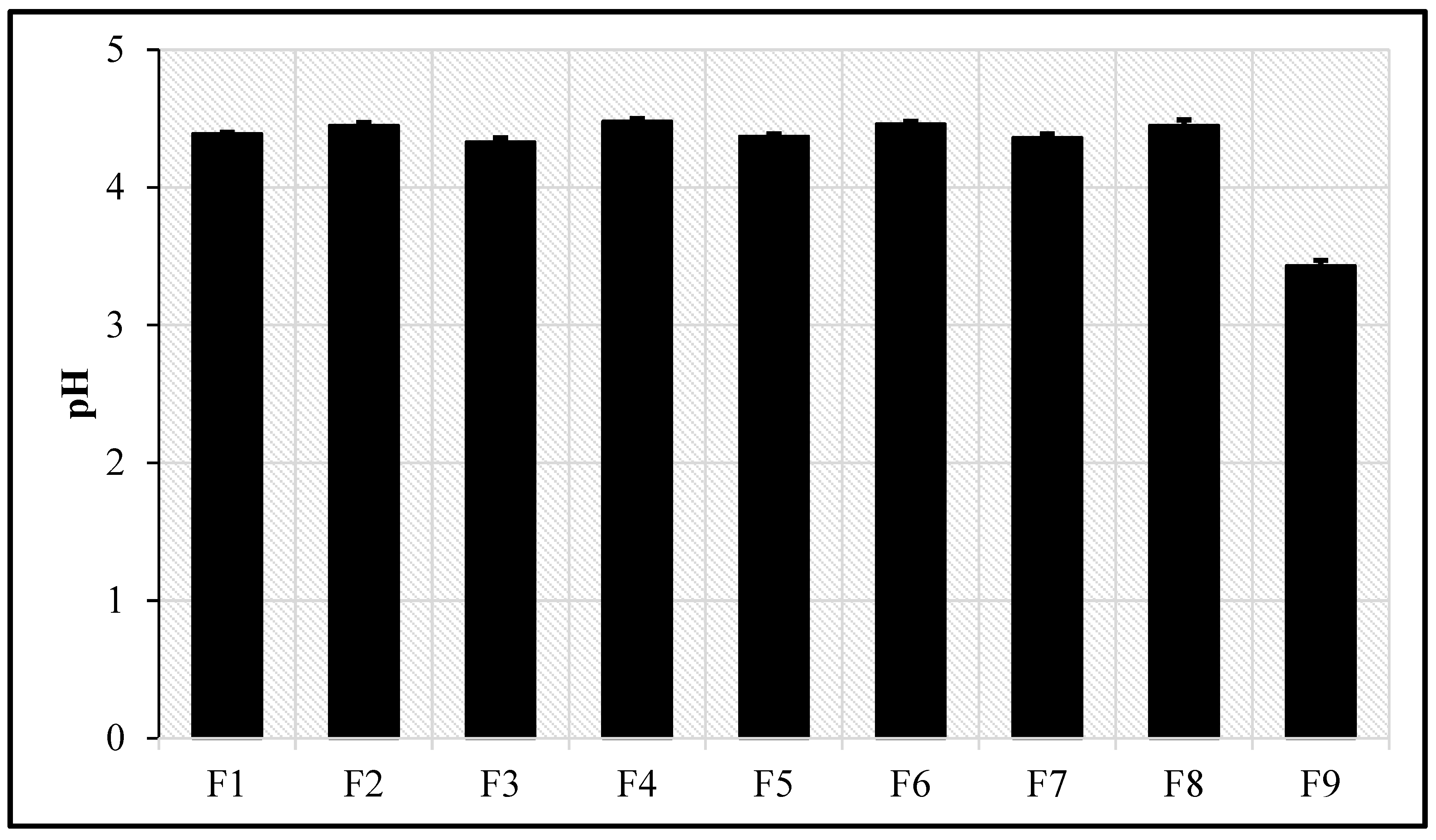
4.1. pH Measurement
4.2. Viscosity Measurement
4.3. In vitro Release Testing
4.4. Ex Vivo Permeation
- Qn was calculated using the following equation:
- Jss of KZ was calculated using the following equation:
- The transdermal permeability coefficient was calculated by the following equation:
4.5. Ex Vivo Deposition Study
4.6. In Vivo Studies
4.7. Quantification of KZ During In vivo Studies
4.8. Skin Irritation Studies
4.9. In Vitro Antifungal Activity
4.1. Physicochemical Stability Studies
4.11. Statistical Analysis
5. Results and Discussion
5.1. Screening of Oil Excipients
5.2. Saturation Solubility Studies
5.3. Factorial Design
5.4. Statistical Analysis of GS
5.5. Statistical Analysis of ZP
5.6. Optimization
5.7. Preparation of KZ-NE and KZ-NEC
| Formulation | Oleic acid (% w/v) |
Span® 80 (% w/v) | Tween® 80 (% w/v) | Ketoconazole (% w/v) |
Carbopol® 940 (% w/v) |
Water up to (mL) |
| F1 | 3.0 | 0.5 | 2.0 | 1.0 | - | 10 |
| F2 | 5.0 | 0.5 | 2.0 | 1.0 | - | 10 |
| F3 | 3.0 | 1.0 | 2.0 | 1.0 | - | 10 |
| F4 | 5.0 | 1.0 | 2.0 | 1.0 | - | 10 |
| F5 | 3.0 | 0.5 | 3.0 | 1.0 | - | 10 |
| F6 | 5.0 | 0.5 | 3.0 | 1.0 | - | 10 |
| F7 | 3.0 | 1.0 | 3.0 | 1.0 | - | 10 |
| F8 | 5.0 | 1.0 | 3.0 | 1.0 | - | 10 |
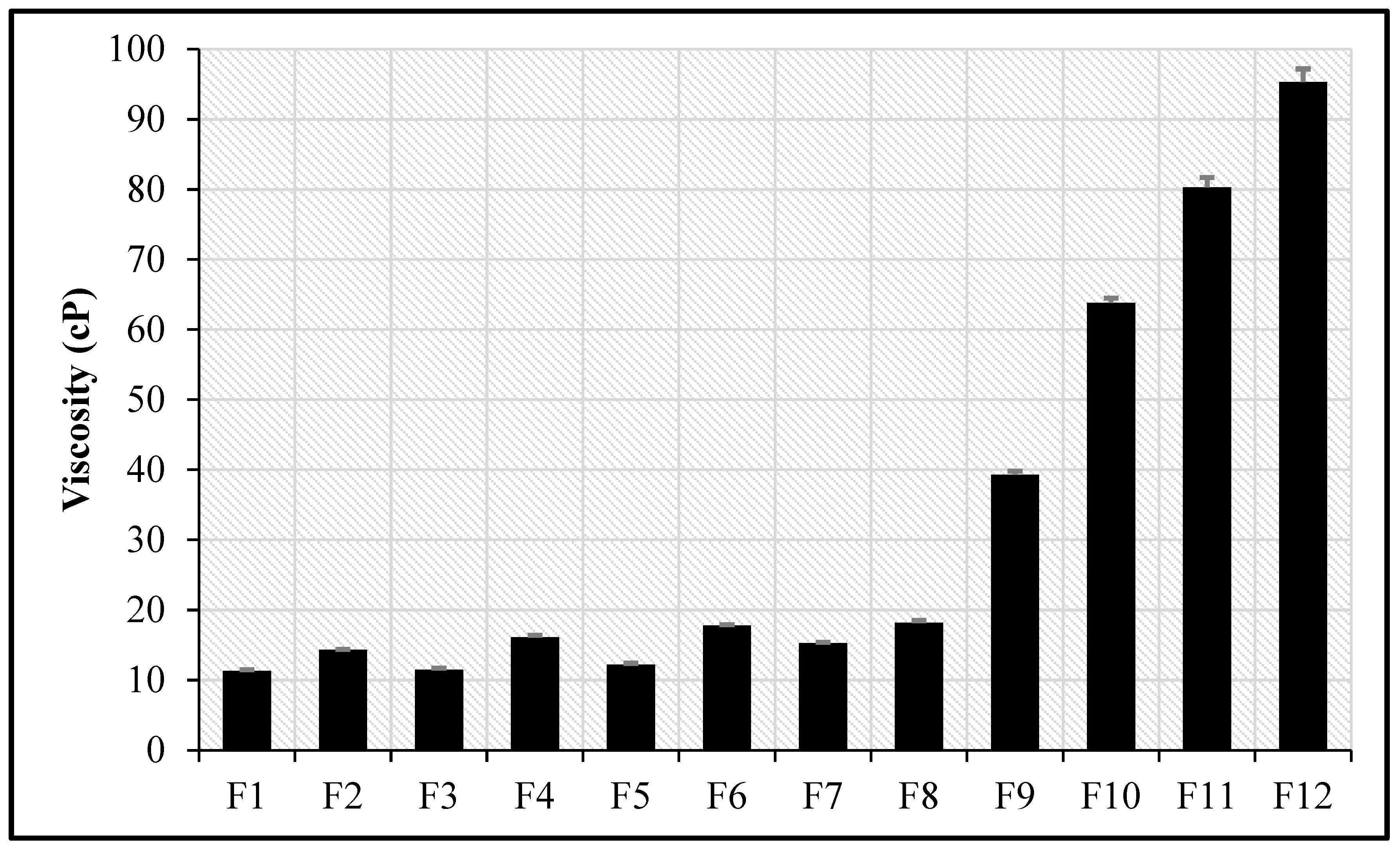
| F9 | 5.0 | 0.5 | 2.0 | 1.0 | 0.5 | 10 |
| F10 | 5.0 | 0.5 | 2.0 | 1.0 | 1.0 | 10 |
| F11 | 5.0 | 0.5 | 2.0 | 1.0 | 2.0 | 10 |
| F12 | 5.0 | 0.5 | 2.0 | 1.0 | 3.0 | 10 |
5.8. Viscosity Measurement
5.9. Measurement of GS, PDI, and ZP
5.10. Drug Content
5.11. pH Measurement
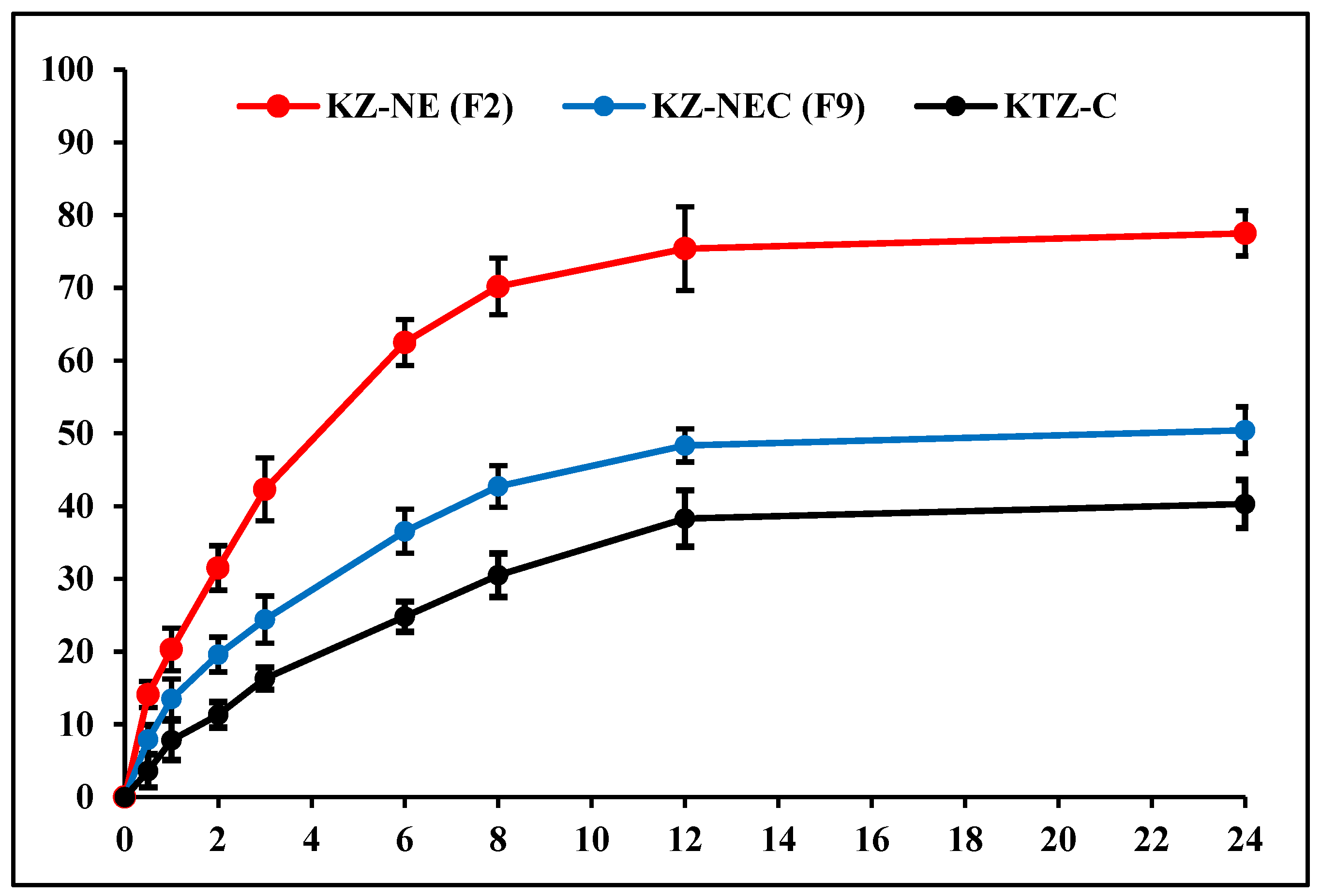
5.12. In vitro Release Testing
5.13. Ex vivo Permeation and Deposition Testing
5.14. In Vivo Studies
5.15. Skin Irritation Studies
5.16. In Vitro Antifungal Activity
5.17. Physicochemical Stability
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Akhtar, N.; Verma, A.; Pathak, K. Topical Delivery of Drugs for the Effective Treatment of Fungal Infections of Skin. Curr. Pharm. Des. 2015, 21, 2892–2913. [Google Scholar] [CrossRef] [PubMed]
- Ameen, M. Epidemiology of Superficial Fungal Infections. Clin. Dermatol. 2010, 28, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Havlickova, B.; Czaika, V.A.; Friedrich, M. Epidemiological Trends in Skin Mycoses Worldwide. Mycoses 2008, 51, 2–15. [Google Scholar] [CrossRef] [PubMed]
- Hay, R. Superficial Fungal Infections. Medicine (Baltimore) 2013, 41, 716–718. [Google Scholar] [CrossRef]
- Gupta, A.K.; Lyons, D.C.A. The Rise and Fall of Oral Ketoconazole. J. Cutan. Med. Surg. 2015, 19, 352–357. [Google Scholar] [CrossRef]
- Choi, F.D.; Juhasz, M.L.W.; Atanaskova Mesinkovska, N. Topical Ketoconazole: A Systematic Review of Current Dermatological Applications and Future Developments. J. Dermatol. Treat. 2019, 30, 760–771. [Google Scholar] [CrossRef]
- Dixon, D.; Shadomy, S.; Shadomy, H.J.; Espinel-Ingroff, A.; Kerkering, T.M. Comparison of the in Vitro Antifungal Activities of Miconazole and a New Imidazole, R41, 400. J. Infect. Dis. 1978, 138, 245–248. [Google Scholar] [CrossRef]
- Odds, F.C.; Milne, L.J.R.; Gentles, J.C.; Ball, E.H. The Activity in Vitro and in Vivo of a New Imidazole Antifungal, Ketoconazole. J. Antimicrob. Chemother. 1980, 6, 97–104. [Google Scholar] [CrossRef]
- Heeres, J.; Backx, L.J.J.; Mostmans, J.H.; Van Cutsem, J. Antimycotic Imidazoles. Part 4. Synthesis and Antifungal Activity of Ketoconazole, a New Potent Orally Active Broad-Spectrum Antifungal Agent. J. Med. Chem. 1979, 22, 1003–1005. [Google Scholar] [CrossRef]
- Thienpont, D.; Van Cutsem, J.; Van Gerven, F.; Heeres, J.; Janssen, P.A.J. Ketoconazole — a New Broad Spectrum Orally Active Antimycotic. Experientia 1979, 35, 606–607. [Google Scholar] [CrossRef]
- Tsume, Y.; Igawa, N.; Drelich, A.J.; Amidon, G.E.; Amidon, G.L. The Combination of GIS and Biphasic to Better Predict In Vivo Dissolution of BCS Class IIb Drugs, Ketoconazole and Raloxifene. J. Pharm. Sci. 2018, 107, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Ramzan, M.; Gourion-Arsiquaud, S.; Hussain, A.; Gulati, J.S.; Zhang, Q.; Trehan, S.; Puri, V.; Michniak-Kohn, B.; Kaur, I.P. In Vitro Release, Ex Vivo Penetration, and in Vivo Dermatokinetics of Ketoconazole-Loaded Solid Lipid Nanoparticles for Topical Delivery. Drug Deliv. Transl. Res. 2022, 12, 1659–1683. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.; Wang, J.; Ma, M.; Tan, F.; Li, N. Skin Targeted Lipid Vesicles as Novel Nano-Carrier of Ketoconazole: Characterization, in Vitro and in Vivo Evaluation. J. Mater. Sci. Mater. Med. 2015, 26, 175. [Google Scholar] [CrossRef] [PubMed]
- Youssef, A.A.A.; Cai, C.; Dudhipala, N.; Majumdar, S. Design of Topical Ocular Ciprofloxacin Nanoemulsion for the Management of Bacterial Keratitis. Pharm. Basel Switz. 2021, 14, 210. [Google Scholar] [CrossRef]
- Ghasemiyeh, P.; Mohammadi-Samani, S. Potential of Nanoparticles as Permeation Enhancers and Targeted Delivery Options for Skin: Advantages and Disadvantages. Drug Des. Devel. Ther. 2020, 14, 3271–3289. [Google Scholar] [CrossRef]
- Rai, V.K.; Mishra, N.; Yadav, K.S.; Yadav, N.P. Nanoemulsion as Pharmaceutical Carrier for Dermal and Transdermal Drug Delivery: Formulation Development, Stability Issues, Basic Considerations and Applications. J. Controlled Release 2018, 270, 203–225. [Google Scholar] [CrossRef]
- Joshi, M.; Butola, B.; Saha, K. Advances in Topical Drug Delivery System: Micro to Nanofibrous Structures. J. Nanosci. Nanotechnol. 2014, 14, 853–867. [Google Scholar] [CrossRef]
- Dudhipala, N.; Youssef, A.A.A.; Banala, N. Colloidal Lipid Nanodispersion Enriched Hydrogel of Antifungal Agent for Management of Fungal Infections: Comparative in-Vitro, Ex-Vivo and in-Vivo Evaluation for Oral and Topical Application. Chem. Phys. Lipids 2020, 233, 104981. [Google Scholar] [CrossRef]
- Joshi, P.H.; Youssef, A.A.A.; Ghonge, M.; Varner, C.; Tripathi, S.; Dudhipala, N.; Majumdar, S. Gatifloxacin Loaded Nano Lipid Carriers for the Management of Bacterial Conjunctivitis. Antibiotics 2023, 12, 1318. [Google Scholar] [CrossRef]
- A., N.; Kovooru, L.; Behera, A.K.; Kumar, K.P.P.; Srivastava, P. A Critical Review of Synthesis Procedures, Applications and Future Potential of Nanoemulsions. Adv. Colloid Interface Sci. 2021, 287, 102318. [Google Scholar] [CrossRef]
- Gurpreet, K.; Singh, S.K. Review of Nanoemulsion Formulation and Characterization Techniques. Indian J. Pharm. Sci. 2018, 80, 781–789. [Google Scholar] [CrossRef]
- Dudhipala, N.; Ay, A.A. Amelioration of Ketoconazole in Lipid Nanoparticles for Enhanced Antifungal Activity and Bioavailability through Oral Administration for Management of Fungal Infections. Chem. Phys. Lipids 2020, 232, 104953. [Google Scholar] [CrossRef] [PubMed]
- Youssef, A.A.A.; Thakkar, R.; Senapati, S.; Joshi, P.H.; Dudhipala, N.; Majumdar, S. Design of Topical Moxifloxacin Mucoadhesive Nanoemulsion for the Management of Ocular Bacterial Infections. Pharmaceutics 2022, 14, 1246. [Google Scholar] [CrossRef] [PubMed]
- Sadozai, S.K.; Khan, S.A.; Baseer, A.; Ullah, R.; Zeb, A.; Schneider, M. In Vitro, Ex Vivo, and in Vivo Evaluation of Nanoparticle-Based Topical Formulation against Candida Albicans Infection. Front. Pharmacol. 2022, 13, 909851. [Google Scholar] [CrossRef]
- Sadozai, S.K.; Khan, S.A.; Baseer, A.; Ullah, R.; Zeb, A.; Schneider, M. In Vitro, Ex Vivo, and in Vivo Evaluation of Nanoparticle-Based Topical Formulation against Candida Albicans Infection. Front. Pharmacol. 2022, 13, 909851. [Google Scholar] [CrossRef]
- Bajad, S.; Johri, R.K.; Singh, K.; Singh, J.; Bedi, K.L. Simple High-Performance Liquid Chromatography Method for the Simultaneous Determination of Ketoconazole and Piperine in Rat Plasma and Hepatocyte Culture. J. Chromatogr. A 2002, 949, 43–47. [Google Scholar] [CrossRef]
- Zhu, W.; Guo, C.; Yu, A.; Gao, Y.; Cao, F.; Zhai, G. Microemulsion-Based Hydrogel Formulation of Penciclovir for Topical Delivery. Int. J. Pharm. 2009, 378, 152–158. [Google Scholar] [CrossRef]
- Azeem, A.; Rizwan, M.; Ahmad, F.J.; Iqbal, Z.; Khar, R.K.; Aqil, M.; Talegaonkar, S. Nanoemulsion Components Screening and Selection: A Technical Note. AAPS PharmSciTech 2009, 10, 69–76. [Google Scholar] [CrossRef]
- Lawrence, M.J.; Rees, G.D. Microemulsion-Based Media as Novel Drug Delivery Systems. Adv. Drug Deliv. Rev. 2000, 33. [Google Scholar] [CrossRef]
- Narang, A.; Delmarre, D.; Gao, D. Stable Drug Encapsulation in Micelles and Microemulsions. Int. J. Pharm. 2007, 345, 9–25. [Google Scholar] [CrossRef]
- Sita, V G; Vavia, P. Bromocriptine Nanoemulsion-Loaded Transdermal Gel: Optimization Using Factorial Design, In Vitro and In Vivo Evaluation. AAPS PharmSciTech 2020, 21, 80. [Google Scholar] [CrossRef] [PubMed]
- Kovacevic, A.; Savic, S.; Vuleta, G.; Müller, R.H.; Keck, C.M. Polyhydroxy Surfactants for the Formulation of Lipid Nanoparticles (SLN and NLC): Effects on Size, Physical Stability and Particle Matrix Structure. Int. J. Pharm. 2011, 406, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Kundu, P.; Kumar, V.; Mishra, I.M. Modeling the Steady-Shear Rheological Behavior of Dilute to Highly Concentrated Oil-in-Water (o/w) Emulsions: Effect of Temperature, Oil Volume Fraction and Anionic Surfactant Concentration. J. Pet. Sci. Eng. 2015, 129, 189–204. [Google Scholar] [CrossRef]
- Onaizi, S.A. Effect of Oil/Water Ratio on Rheological Behavior, Droplet Size, Zeta Potential, Long-Term Stability, and Acid-Induced Demulsification of Crude Oil/Water Nanoemulsions. J. Pet. Sci. Eng. 2021, 109857. [Google Scholar] [CrossRef]
- Ashrafizadeh, S.N.; Motaee, E.; Hoshyargar, V. Emulsification of Heavy Crude Oil in Water by Natural Surfactants. J. Pet. Sci. Eng. 2012, 86–87, 137–143. [Google Scholar] [CrossRef]
- Abdullah, N.; Jufri, M.; Mun’im, A.; Saputri, F. Optimization of Nanoemulsion in Chemix 7.00 Program : Using Isopropyl Myristate as Oil Phase; 2021. [Google Scholar]
- Effendy, I.; Maibach, H.I. Surfactants and Experimental Irritant Contact Dermatitis. Contact Dermatitis 1995, 33, 217–225. [Google Scholar] [CrossRef]
- Sweeney, C.; Dudhipala, N.; Thakkar, R.; Mehraj, T.; Marathe, S.; Gul, W.; ElSohly, M.A.; Murphy, B.; Majumdar, S. Impact of Mucoadhesive Agent Inclusion on the Intraocular Pressure Lowering Profile of Δ9-Tetrahydrocannabinol-Valine-Hemisuccinate Loaded Nanoemulsions in New Zealand White Rabbits. Int. J. Pharm. 2022, 616, 121564. [Google Scholar] [CrossRef]
- Binder, L.; Mazál, J.; Petz, R.; Klang, V.; Valenta, C. The Role of Viscosity on Skin Penetration from Cellulose Ether-Based Hydrogels. Skin Res. Technol. 2019, 25, 725–734. [Google Scholar] [CrossRef]
- Giri, S.; Markandeywar, T.S.; Irfan, Z.; Manna, S. Gellan Gum and Sodium Alginate In-Situ Gel of Monocaprin for Effective Corneal Permeation. Food Hydrocoll. Health 2023, 4, 100156. [Google Scholar] [CrossRef]
- Sanjana, A.; Ahmed, M.G.; Bh, J.G. Preparation and Evaluation of In-Situ Gels Containing Hydrocortisone for the Treatment of Aphthous Ulcer. J. Oral Biol. Craniofacial Res. 2021, 11, 269–276. [Google Scholar] [CrossRef]
- Su, R.; Fan, W.; Yu, Q.; Dong, X.; Qi, J.; Zhu, Q.; Zhao, W.; Wu, W.; Chen, Z.; Li, Y.; et al. Size-Dependent Penetration of Nanoemulsions into Epidermis and Hair Follicles: Implications for Transdermal Delivery and Immunization. Oncotarget 2017, 8, 38214–38226. [Google Scholar] [CrossRef]
- Lin, L.; Gu, Y.; Cui, H. Moringa Oil/Chitosan Nanoparticles Embedded Gelatin Nanofibers for Food Packaging against Listeria Monocytogenes and Staphylococcus Aureus on Cheese. Food Packag. Shelf Life 2019, 19, 86–93. [Google Scholar] [CrossRef]
- Mitri, K.; Shegokar, R.; Gohla, S.; Anselmi, C.; Müller, R.H. Lipid Nanocarriers for Dermal Delivery of Lutein: Preparation, Characterization, Stability and Performance. Int. J. Pharm. 2011, 414, 267–275. [Google Scholar] [CrossRef]
- Senapati, S.; Youssef, A.A.A.; Sweeney, C.; Cai, C.; Dudhipala, N.; Majumdar, S. Cannabidiol Loaded Topical Ophthalmic Nanoemulsion Lowers Intraocular Pressure in Normotensive Dutch-Belted Rabbits. Pharmaceutics 2022, 14, 2585. [Google Scholar] [CrossRef] [PubMed]
- Lukić, M.; Pantelić, I.; Savić, S.D. Towards Optimal pH of the Skin and Topical Formulations: From the Current State of the Art to Tailored Products. Cosmetics 2021, 8, 69. [Google Scholar] [CrossRef]
- Akhter, S.; Anwar, M.; Siddiqui, M.A.; Ahmad, I.; Ahmad, J.; Ahmad, M.Z.; Bhatnagar, A.; Ahmad, F.J. Improving the Topical Ocular Pharmacokinetics of an Immunosuppressant Agent with Mucoadhesive Nanoemulsions: Formulation Development, in-Vitro and in-Vivo Studies. Colloids Surf. B Biointerfaces 2016, 148, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Shete, A.; Murthy, S.; Korpale, S.; Yadav, A.; Sajane, S.; Sakhare, S.; Doijad, R. Cocrystals of Itraconazole with Amino Acids: Screening, Synthesis, Solid State Characterization, in Vitro Drug Release and Antifungal Activity. J. Drug Deliv. Sci. Technol. 2015, 28, 46–55. [Google Scholar] [CrossRef]
- Shettar, A.; Shankar, V.K.; Ajjarapu, S.; Kulkarni, V.I.; Repka, M.A.; Murthy, S.N. Development and Characterization of Novel Topical Oil/PEG Creams of Voriconazole for the Treatment of Fungal Infections. J. Drug Deliv. Sci. Technol. 2021, 66, 102928. [Google Scholar] [CrossRef]
- Shaker, D.S.; Ishak, R.A.H.; Ghoneim, A.; Elhuoni, M.A. Nanoemulsion: A Review on Mechanisms for the Transdermal Delivery of Hydrophobic and Hydrophilic Drugs. Sci. Pharm. 2019, 87, 17. [Google Scholar] [CrossRef]
- Jiang, S.J.; Zhou, X.J. Examination of the Mechanism of Oleic Acid-Induced Percutaneous Penetration Enhancement: An Ultrastructural Study. Biol. Pharm. Bull. 2003, 26, 66–68. [Google Scholar] [CrossRef]
- Benson, H. Transdermal Drug Delivery: Penetration Enhancement Techniques. Curr. Drug Deliv. 2005, 2, 23–33. [Google Scholar] [CrossRef]
- Kogan, A.; Garti, N. Microemulsions as Transdermal Drug Delivery Vehicles. Adv. Colloid Interface Sci. 2006, 123–126, 369–385. [Google Scholar] [CrossRef]
- Khurana, S.; Jain, N.K.; Bedi, P.M.S. Nanoemulsion Based Gel for Transdermal Delivery of Meloxicam: Physico-Chemical, Mechanistic Investigation. Life Sci. 2013, 92, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Liu, S.; Gu, Y.; Tang, X.; Wang, T.; Wu, J.; Liu, J. Development of Sulconazole-Loaded Nanoemulsions for Enhancement of Transdermal Permeation and Antifungal Activity. Int. J. Nanomedicine 2019, Volume 14, 3955–3966. [Google Scholar] [CrossRef] [PubMed]
- Komaiko, J.S.; McClements, D.J. Formation of Food-Grade Nanoemulsions Using Low-Energy Preparation Methods: A Review of Available Methods. Compr. Rev. Food Sci. Food Saf. 2016, 15, 331–352. [Google Scholar] [CrossRef] [PubMed]
- Mishra, N.; Yadav, K.S.; Rai, V.K.; Yadav, N.P. Polysaccharide Encrusted Multilayered Nano-Colloidal System of Andrographolide for Improved Hepatoprotection. Aaps Pharmscitech 2017, 18, 381–392. [Google Scholar] [CrossRef]
- Santos, J.; Calero, N.; Trujillo-Cayado, L.A.; Martín-Piñero, M.J.; Muñoz, J. Processing and Formulation Optimization of Mandarin Essential Oil-Loaded Emulsions Developed by Microfluidization. Materials 2020, 13, 3486. [Google Scholar] [CrossRef]
- GRIFFIN, W.C. Calculation of HLB Values of Non-Ionic Surfactants. J Soc Cosmet Chem 1954, 5, 249–256. [Google Scholar]

| Factors | Coded levels | |
| Independent Variables | Level –1 | Level +1 |
| A: Oil (% w/v) | 3.0 | 5.0 |
| B: Span® 80 (% w/v) | 0.5 | 1.0 |
| C: Tween® 80 (% w/v) | 2.0 | 3.0 |
| Dependent Variables | ||
| Y1: Globule size (GS) | ||
| Y2: Zeta potential (ZP) | ||
| Run |
Assigned independent variables |
Actual independent variables | Response | |||||
| A | B | C | Oil (Oleic acid, % w/v) |
Span® 80 (% w/v) |
Tween® 80 (% w/v) |
GS (nm) | ZP (mV) | |
| 1 | -1 | -1 | -1 | 3 | 0.5 | 2 | 109.5±0.5 | -24.9±0.9 |
| 2 | +1 | -1 | -1 | 5 | 0.5 | 2 | 141.6±3.2 | -28.5±0.3 |
| 3 | -1 | +1 | -1 | 3 | 1 | 2 | 135.0±4.9 | -25.6±1.4 |
| 4 | +1 | +1 | -1 | 5 | 1 | 2 | 171.9±5.8 | -27.7±0.5 |
| 5 | -1 | -1 | +1 | 3 | 0.5 | 3 | 96.7±2.9 | -23.5±0.9 |
| 6 | +1 | -1 | +1 | 5 | 0.5 | 3 | 130.2±3.8 | -27.2±1.0 |
| 7 | -1 | +1 | +1 | 3 | 1 | 3 | 116.2±3.4 | -24.0±0.7 |
| 8 | +1 | +1 | +1 | 5 | 1 | 3 | 155.8±7.2 | -28.1±0.9 |
| Oil | Solubility | Oil | Solubility |
| Soybean oil | (✓) | CapryolTM 90 | (x) |
| Cottonseed oil | (x) | Maisine® CC | (x) |
| Sesame oil | (x) | Olive oil | (✓) |
| Castor oil | (✓) | Lauroglycol™ 90 | (x) |
| Isopropyl myristate | (x) | Labrafac™ lipophile WL 1349 | (x) |
| Maisine® CC | (x) | Oleic acid | (✓) |
| Miglyol® 829 | (x) | Captex® 200 | (x) |
| Labrasol® | (x) | Captex® 355 EP | (x) |
| Response | F value | p-value | R2 | Predicted R2 | Adjusted R2 | SD |
| Y1: GS | 173.33 | 0.0001 | 0.992 | 0.971 | 0.987 | 2.81 |
| Y2: ZP | 38.53 | 0.0008 | 0.865 | 0.761 | 0.843 | 0.78 |
| Variable | Goal | Lower limit | Upper limit |
| Oleic acid | In range | 3.0% w/v | 5.0% w/v |
| Span® 80 | In range | 0.5% w/v | 0.5% w/v |
| Tween® 80 | In range | 2.0% w/v | 2.0% w/v |
| GS | In range | 95.0 nm | 500 nm |
| ZP | Target (-30.0 mV) | -27.0 mV | -32.0 mV |
| Response | Predicted value | Results of validation trials | 95% CI (Low) | 95% CI (High) | ||
| GS (nm) | 144.7 | 148.3 | 139.6 | 147.9 | 133.5 | 154.1 |
| ZP (mV) | -27.9 | -28.7 | -27.9 | -28.4 | -26.3 | -29.3 |
| Inactive Ingredient | Topical Dosage Form | Maximum Potency per unit dose (% w/w) | Maximum Daily Exposure (MDE, mg) |
| Precirol® ATO 5 | NA | 100* | |
| Oleic acid | Cream | 25 | |
| Gel, Metered | 88 | ||
| Solution | 7.4 | ||
| Span® 80 | Cream | 3.5 | |
| Cream, Augmented | 0.2 | ||
| Emulsion | 2.5 | ||
| Gel | 1 | ||
| Lotion | 7 | ||
| Ointment | NA | ||
| Spray | 0.25 | ||
| Tween® 80 | Aerosol, Foam | 0.98 | |
| Cream | 4 | ||
| Emulsion | 2.5 | ||
| Gel | 8.5 | ||
| Lotion | 15 | ||
| Ointment | 0.1 | ||
| Carbopol® 940 NF | Cream | 103 | |
| Cream, Augmented | 20 | ||
| Emulsion | 6 | ||
| Gel | 85 | ||
| Lotion | 300 | ||
| Ointment, Augmented | 23 |
| Formulations | Zero Order | First Order | Higuchi | Korsmeyer-Peppas | |
| M0-M = k.t | ln M = k.t | M0-M = k.t1/2 | log (M0-M) = n log t + log k | ||
| R2 | R2 | R2 | R2 | n | |
| Optimized NE (F2) | 0.1758 | 0.9076 | 0.8666 | 0.9987 | 0.612 |
| Lead NEC (F9) | 0.2968 | 0.6637 | 0.9091 | 0.9382 | 0.390 |
| KZ-C | 0.5662 | 0.7559 | 0.9437 | 0.9374 | 0.474 |
| Where Mo represents the initial drug content at time to, and M represents the drug content remaining at time t; Zero-order model: % drug released vs time; First order model: Amount drug remaining vs time; Higuchi model: % drug released vs square root of time; Korsmeyer–Peppas model: log % drug released vs log time. | |||||
| Group | Formulation | Erythema Score | Edema Score | ||||
| 6 h | 12 h | 24 h | 6 h | 12 h | 24 h | ||
| I | Negative Control | 0 | 0 | 0 | 0 | 0 | 0 |
| II | KZ-NE | 0 | 0 | 0 | 0 | 0 | 0 |
| III | KZ-NEC | 0 | 0 | 0 | 0 | 0 | 0 |
| IV | KZ-C | 0 | 0 | 1 | 0 | 0 | 0 |
| V | Formalin (0.8% w/v) | 0 | 1 | 2 | 1 | 2 | 3 |
|
Erythema scale: 0 = none, 1 = slight, 2 = well-defined, 3 = moderate, and 4 = scar formation. Edema scale: 0 = none, 1 = slight, 2 = well-defined, 3 = moderate, and 4 = severe. | |||||||
| Day | GS (nm) | PDI | ZP (mV) | pH | Viscosity (cP) | Drug content (%) | |
| Storage at 5±3°C | |||||||
| F2 | 0 | 138.5±3.8 | 0.12±0.01 | -28.9±0.4 | 4.45±0.03 | NA | 98.6±0.4 |
| 90 | 139.1±2.5 | 0.13±0.01 | -39.5±0.4 | 4.42±0.03 | 97.5±2.7 | ||
| Storage at 30±2°C | |||||||
| F2 | 0 | 139.9±3.4 | 0.11±0.02 | -28.5±0.5 | 4.43±0.02 | 14.5±0.1 | 98.8±2.0 |
| 90 | 142.4±3.0 | 0.13±0.03 | -29.1±0.7 | 4.44±0.02 | 14.8±0.2 | 99.3±1.9 | |
| Storage at 40±2°C | |||||||
| F2 | 0 | 142.4±5.2 | 0.12±0.04 | -31.2±1.0 | 4.42±0.03 | NA | 99.2±1.2 |
| 90 | 138.5±2.5 | 0.12±0.02 | -30.1±1.1 | 4.43±0.03 | 97.9±2.3 | ||
| Storage at 5±3°C | |||||||
| F9 | 0 | 206.3±2.9 | 0.29±0.03 | -38.2±0.7 | 3.43±0.02 | NA | 100.2±3.8 |
| 90 | 208.6±4.1 | 0.28±0.02 | -37.2±0.3 | 3.44±0.02 | 99.2±2.4 | ||
| Storage at 30±2°C | |||||||
| F9 | 0 | 201.8±2.9 | 0.27±0.03 | -39.4±1.2 | 3.46±0.03 | 38.8±1.5 | 98.8±1.8 |
| 90 | 204.1±4.7 | 0.29±0.02 | -38.8±1.0 | 3.47±0.02 | 40.2±1.1 | 99.0±1.7 | |
| Storage at 40±2°C | |||||||
| F9 | 0 | 207.3±4.1 | 0.26±0.04 | -37.3±1.5 | 3.38±0.02 | NA | 99.5±2.2 |
| 90 | 203.5±2.5 | 0.27±0.03 | -39.2±0.5 | 3.45±0.01 | 97.9±2.5 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




