Submitted:
01 April 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
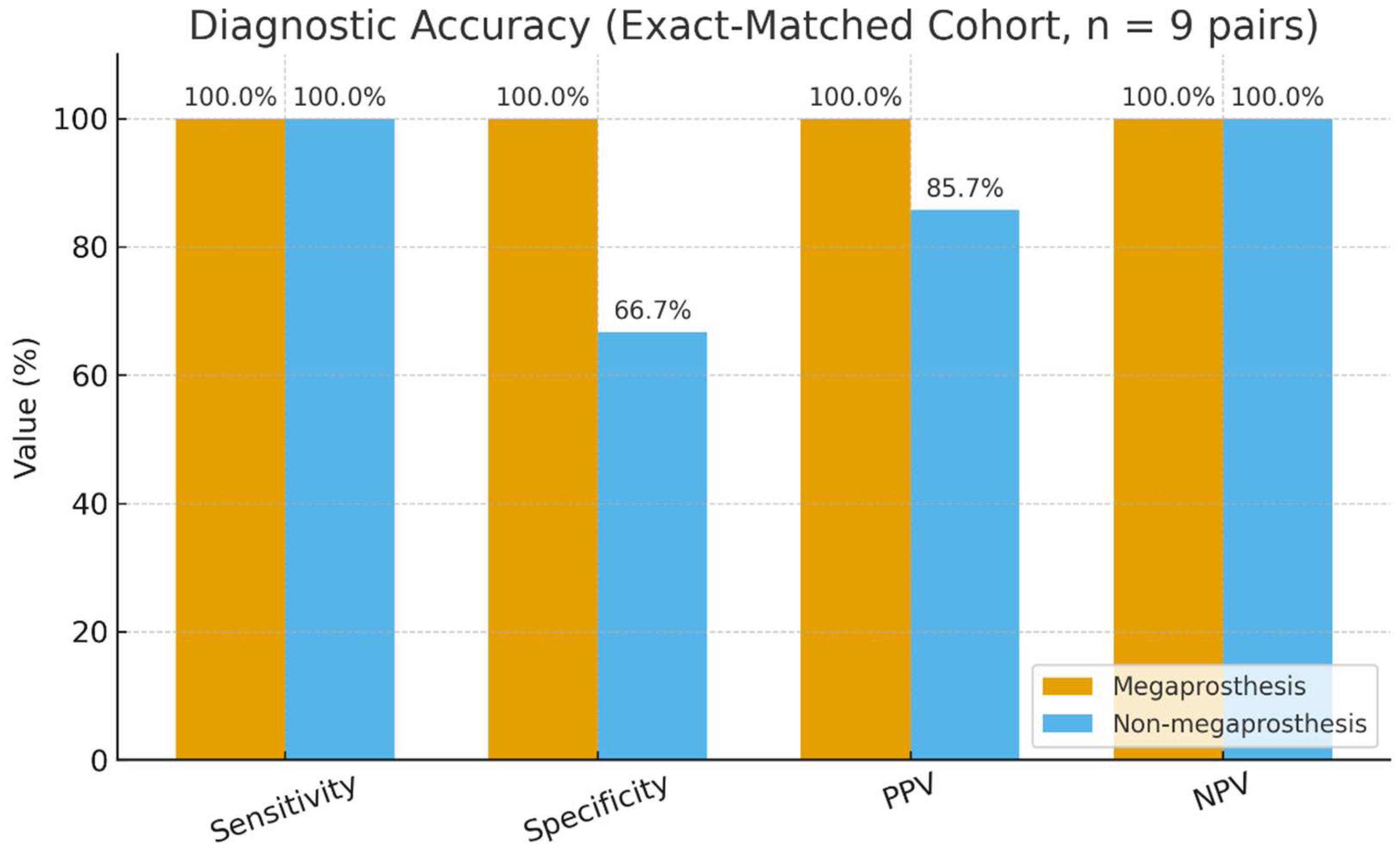
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jeys, L.; Grimer, R.; Carter, S.; Tillman, R. Periprosthetic Infection in Patients Treated for an Orthopaedic Oncological Condition. J. Bone Jt. Surg. 2005, 87, 842–849. [Google Scholar] [CrossRef]
- Karampikas, V.; Gavriil, P.; Goumenos, S.; Trikoupis, I.G.; Roustemis, A.G.; Altsitzioglou, P.; Kontogeorgakos, V.; Mavrogenis, A.F.; Papagelopoulos, P.J. Risk factors for peri-megaprosthetic joint infections in tumor surgery: A systematic review. SICOT-J 2024, 10, 19. [Google Scholar] [CrossRef] [PubMed]
- Roustemis, A.G.; Gavriil, P.; Goumenos, S.; Trikoupis, I.; Karampikas, V.; Koulouvaris, P.; Kontogeorgakos, V.; Savvidou, O.; Mavrogenis, A.F.; Papagelopoulos, P.J. Implant survival and risk factors for failure after proximal femoral megaprosthetic reconstruction. SICOT-J 2025, 11, 50. [Google Scholar] [CrossRef] [PubMed]
- Sukhonthamarn, K.; Tan, T.L.; Strony, J.; Brown, S.; Nazarian, D.; Parvizi, J. The Fate of Periprosthetic Joint Infection Following Megaprosthesis Reconstruction. JBJS Open Access 2021, 6. [Google Scholar] [CrossRef]
- Gonzalez, M.R.; Pretell-Mazzini, J.; Lozano-Calderon, S.A. Risk Factors and Management of Prosthetic Joint Infections in Megaprostheses—A Review of the Literature. Antibiotics 2023, 13, 25. [Google Scholar] [CrossRef]
- Gonzalez, M.R.; Kuthiala, R.S.; Newman, E.T.; Raskin, K.A.; Lozano-Calderon, S.A. What Is the Diagnostic Performance and Accuracy of Serum Inflammatory Biomarkers and Synovial Fluid Analysis in Megaprosthetic Periprosthetic Joint Infections? J. Arthroplast. 2025. [Google Scholar] [CrossRef]
- Lazarides, A.L.; Vovos, T.J.; Reddy, G.B.; Kildow, B.J.; Wellman, S.S.; Jiranek, W.A.; Seyler, T.M. Traditional Laboratory Markers Hold Low Diagnostic Utility for Immunosuppressed Patients With Periprosthetic Joint Infections. J. Arthroplast. 2019, 34, 1441–1445. [Google Scholar] [CrossRef]
- Antonios, J.K.; Lim, E.S.; Chang, Y.-H.H.; Bingham, J.S.; Clarke, H.D.; Spangehl, M.J.; Schwartz, A.J. The Fate of the Inconclusive Periprosthetic Joint Infection Workup and Reliability of Data Points. Orthopedics 2023, 46, E291–+. [Google Scholar] [CrossRef]
- Parvizi, J.; Tan, T.L.; Goswami, K.; Higuera, C.; Della Valle, C.; Chen, A.F.; Shohat, N. The 2018 Definition of Periprosthetic Hip and Knee Infection: An Evidence-Based and Validated Criteria. J. Arthroplast. 2018, 33, 1309–1314.e2. [Google Scholar] [CrossRef]
- Mavrogenis, A.F.; Papagelopoulos, P.J.; Coll-Mesa, L.; Pala, E.; Guerra, G.; Ruggieri, P. Infected Tumor Prostheses. Orthopedics 2011, 34, 991–998. [Google Scholar] [CrossRef]
- Ahmad, S.S.; Shaker, A.; Saffarini, M.; Chen, A.F.; Hirschmann, M.T.; Kohl, S. Accuracy of diagnostic tests for prosthetic joint infection: a systematic review. Knee Surgery, Sports Traumatol. Arthrosc. 2016, 24, 3064–3074. [Google Scholar] [CrossRef]
- Altsitzioglou, P.; Avgerinos, K.; Karampikas, V.; Gavriil, P.; Vlachos, A.; Soucacou, F.; Zafiris, I.; Kontogeorgakos, V.; Papagelopoulos, P.J.; Mavrogenis, A.F. Point of care testing for the diagnosis of periprosthetic joint infections: a review. SICOT-J 2024, 10, 24. [Google Scholar] [CrossRef]
- Carli, A.V.; Abdelbary, H.; Ahmadzai, N.; Cheng, W.; Shea, B.; Hutton, B.; Sniderman, J.; Sanders, B.S.P.; Esmaeilisaraji, L.; Skidmore, B.; et al. Diagnostic Accuracy of Serum, Synovial, and Tissue Testing for Chronic Periprosthetic Joint Infection After Hip and Knee Replacements. J. Bone Jt. Surg. 2019, 101, 635–649. [Google Scholar] [CrossRef] [PubMed]
- Alkadhem, M.F.; Jutte, P.C.; Wouthuyzen-Bakker, M.; Kobold, A.C.M. Analytical and clinical considerations of synovial fluid calprotectin in diagnosing periprosthetic joint infections. Crit. Rev. Clin. Lab. Sci. 2025, 62, 228–239. [Google Scholar] [CrossRef] [PubMed]
- Festa, E.; Ascione, T.; Di Gennaro, D.; De Mauro, D.; Mariconda, M.; Balato, G. Synovial calprotectin in prosthetic joint infection. A systematic review and meta-analysis of the literature. Arch. Orthop. Trauma Surg. 2024, 144, 5217–5227. [Google Scholar] [CrossRef] [PubMed]
- Hantouly, A.T.; Salameh, M.; Toubasi, A.A.; Salman, L.A.; Alzobi, O.; Ahmed, A.F.; Hameed, S.; Zikria, B.; Ahmed, G. Synovial fluid calprotectin in diagnosing periprosthetic joint infection: A meta-analysis. Int. Orthop. 2022, 46, 971–981. [Google Scholar] [CrossRef]
- Xing, J.; Li, J.; Yan, Z.; Li, Y.; Liu, X.; He, L.; Xu, T.; Wang, C.; Zhao, L.; Jie, K. Diagnostic accuracy of calprotectin in periprosthetic joint infection: a diagnostic meta-analysis. J. Orthop. Surg. Res. 2022, 17, 1–12. [Google Scholar] [CrossRef]
- Wouthuyzen-Bakker, M.; Ploegmakers, J.J.; Ottink, K.; Kampinga, G.A.; Wagenmakers-Huizenga, L.; Jutte, P.C.; Kobold, A.C. Synovial Calprotectin: An Inexpensive Biomarker to Exclude a Chronic Prosthetic Joint Infection. J. Arthroplast. 2018, 33, 1149–1153. [Google Scholar] [CrossRef]
- Peng, X.; Zhang, H.; Xin, P.; Bai, G.; Ge, Y.; Cai, M.; Wang, R.; Fan, Y.; Pang, Z. Synovial calprotectin for the diagnosis of periprosthetic joint infection: a diagnostic meta-analysis. J. Orthop. Surg. Res. 2022, 17, 1–12. [Google Scholar] [CrossRef]
- Grassi, M.; Salari, P.; Farinelli, L.; D’aNzeo, M.; Onori, N.; Gigante, A. Synovial Biomarkers to Detect Chronic Periprosthetic Joint Infection: A Pilot Study to Compare Calprotectin Rapid Test, Calprotectin ELISA Immunoassay and Leukocyte Esterase Test. J. Arthroplast. 2022, 37, 781–786. [Google Scholar] [CrossRef]
- Lazic, I.; Stephan, M.; Pohlig, F.; Langer, S.; VON Eisenhart-Rothe, R.; Suren, C. Synovial Calprotectin for Diagnosing Periprosthetic Joint Infection in Loose Hip and Knee Arthroplasties: A Prospective Cohort Study. Vivo 2023, 37, 1714–1720. [Google Scholar] [CrossRef]
- Cheok, T.; Smith, T.; Siddiquee, S.; Jennings, M.P.; Jayasekera, N.; Jaarsma, R.L. Synovial fluid calprotectin performs better than synovial fluid polymerase chain reaction and interleukin-6 in the diagnosis of periprosthetic joint infection. Bone Jt. J. 2022, 104-B, 311–320. [Google Scholar] [CrossRef]
- Tang, H.; Xu, J.; Yuan, W.; Wang, Y.; Yue, B.; Qu, X. Reliable Diagnostic Tests and Thresholds for Preoperative Diagnosis of Non-Inflammatory Arthritis Periprosthetic Joint Infection: A Meta-analysis and Systematic Review. Orthop. Surg. 2022, 14, 2822–2836. [Google Scholar] [CrossRef]
- Salari, P.; Grassi, M.; Cinti, B.; Onori, N.; Gigante, A. Synovial Fluid Calprotectin for the Preoperative Diagnosis of Chronic Periprosthetic Joint Infection. J. Arthroplast. 2020, 35, 534–537. [Google Scholar] [CrossRef]
- Vale, J.S.; Castelo, F.S.; Barros, B.S.; Ribau, A.C.; Carvalho, A.D.; Sousa, R.J. Synovial Fluid Biomarkers for the Diagnosis of Periprosthetic Joint Infection—A Systematic Review and Meta-Analysis of Their Diagnostic Accuracy According to Different Definitions. J. Arthroplast. 2023, 38, 2731–2738.e3. [Google Scholar] [CrossRef]
- Abildtrup, M.; Kingsley, G.H.; Scott, D.L. Calprotectin as a Biomarker for Rheumatoid Arthritis: A Systematic Review. J. Rheumatol. 2015, 42, 760–770. [Google Scholar] [CrossRef]
| Variable | Value |
| Age (years) | Mean ± SD: 52.9 ± 22.5; Median (IQR): 57.0 (34.0–72.0); Range: 16–83 |
| Sex (male) | 14 (70%) |
| Preoperative culture positive | 8 (40%) |
| Infection final status (infected) | 13 (65%) |
| CRP (mg/L) | Mean ± SD: 51.4 ± 85.1; Median (IQR): 9.23 (5.50–44.75); Range: 1.2–325.0 |
| ESR (mm/hr) | Mean ± SD: 48.0 ± 32.9; Median (IQR): 44.0 (20.0–70.0); Range: 8–120 |
| Synovial WBC (K/μL) | Mean ± SD: 11.0 ± 17.2; Median (IQR): 3.55 (1.25–8.35); Range: 0.1–64.0 |
| PMN (%) | Mean ± SD: 60.8 ± 23.9; Median (IQR): 64.0 (38.75–85.0); Range: 20–90 |
| D-dimer (ng/mL) | n = 2; Median (IQR): 2449.5 (1250.8–3648.3); Range: 52–4847 |
| Systemic WBC | Normal: 9 (45%); Elevated: 11 (55%) |
| Antibiotics prior to aspiration | 15 (75%) |
| Time from last surgery to aspiration (years) | Mean ± SD: 1.77 ± 2.26; Median (IQR): 1.0 (0.46–2.0); Range: 0.1–10.0 |
| Type of implant | Knee: 16 (80%); Hip: 3 (15%); Humerus: 1 (5%) |
| Surgical stage | Primary: 5 (25%); Revision: 15 (75%) |
| Sinus tract present | 5 (25%) |
| Metallosis / wear | Present: 6 (30%); Absent: 8 (40%); Not reported: 6 (30%) |
| Timing category | Early: 3; Delayed: 9; Late: 1; Aseptic: 7 |
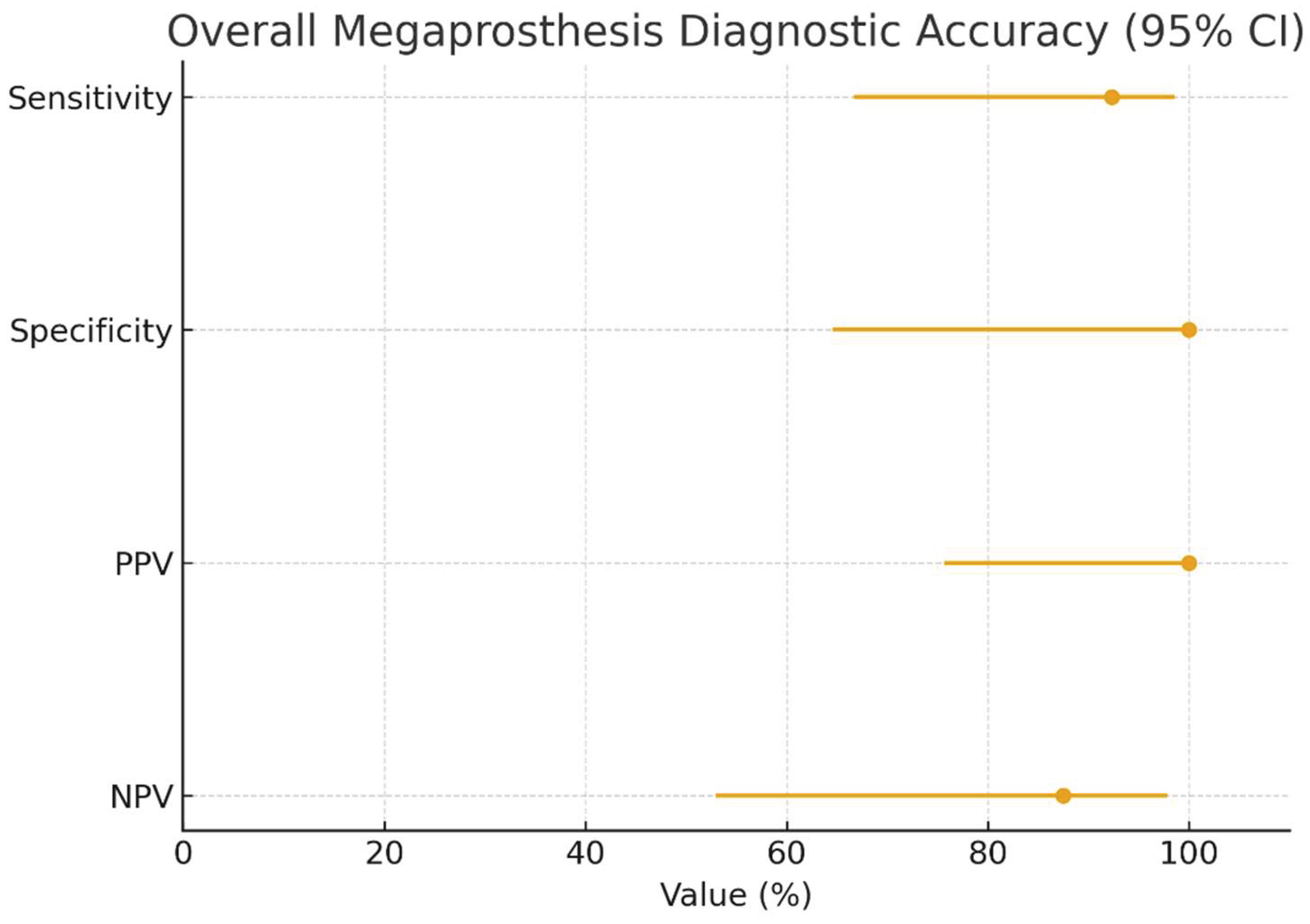
| Metric | Value (95% CI) |
| Sensitivity | 92.3% (66.7–98.6) |
| Specificity | 100% (64.6–100) |
| Positive Predictive Value (PPV) | 100% (75.7–100) |
| Negative Predictive Value (NPV) | 87.5% (52.9–97.8) |
| Positive Likelihood Ratio (LR+) | ∞ (no false positives observed) |
| Negative Likelihood Ratio (LR−) | 0.08 (0.01–0.51) |
| AUC (calprotectin, continuous) | 1.00 (perfect separation in dataset) |
| Microorganism Category | Subgroup | n (%) |
| Coagulase-negative staphylococci (CoNS) | S. epidermidis | 3 (23.1%) |
| Coagulase-negative staphylococci (CoNS) | S. haemolyticus | 3 (23.1%) |
| Coagulase-negative staphylococci (CoNS) | S. hominis | 2 (15.4%) |
| Staphylococcus aureus | S. aureus | 3 (23.1%) |
| Gram-negative organisms | Pseudomonas oryzihabitans | 1 (7.7%) |
| Gram-negative organisms | Pseudomonas aeruginosa | 1 (7.7%) |
| Gram-negative organisms | E. coli | 1 (7.7%) |
| Gram-negative organisms | Klebsiella pneumoniae | 1 (7.7%) |
| Gram-negative organisms | Proteus mirabilis | 1 (7.7%) |
| Gram-negative organisms | Achromobacter insolitus | 1 (7.7%) |
| Streptococci | Streptococcus spp | 1 (7.7%) |
| Fungi | Aspergillus niger | 1 (7.7%) |
| Polymicrobial infections | - | 4 (30.8%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




