Submitted:
01 April 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Baseline Clinical, Surgical, and Biochemical Characteristics According to POAF Status
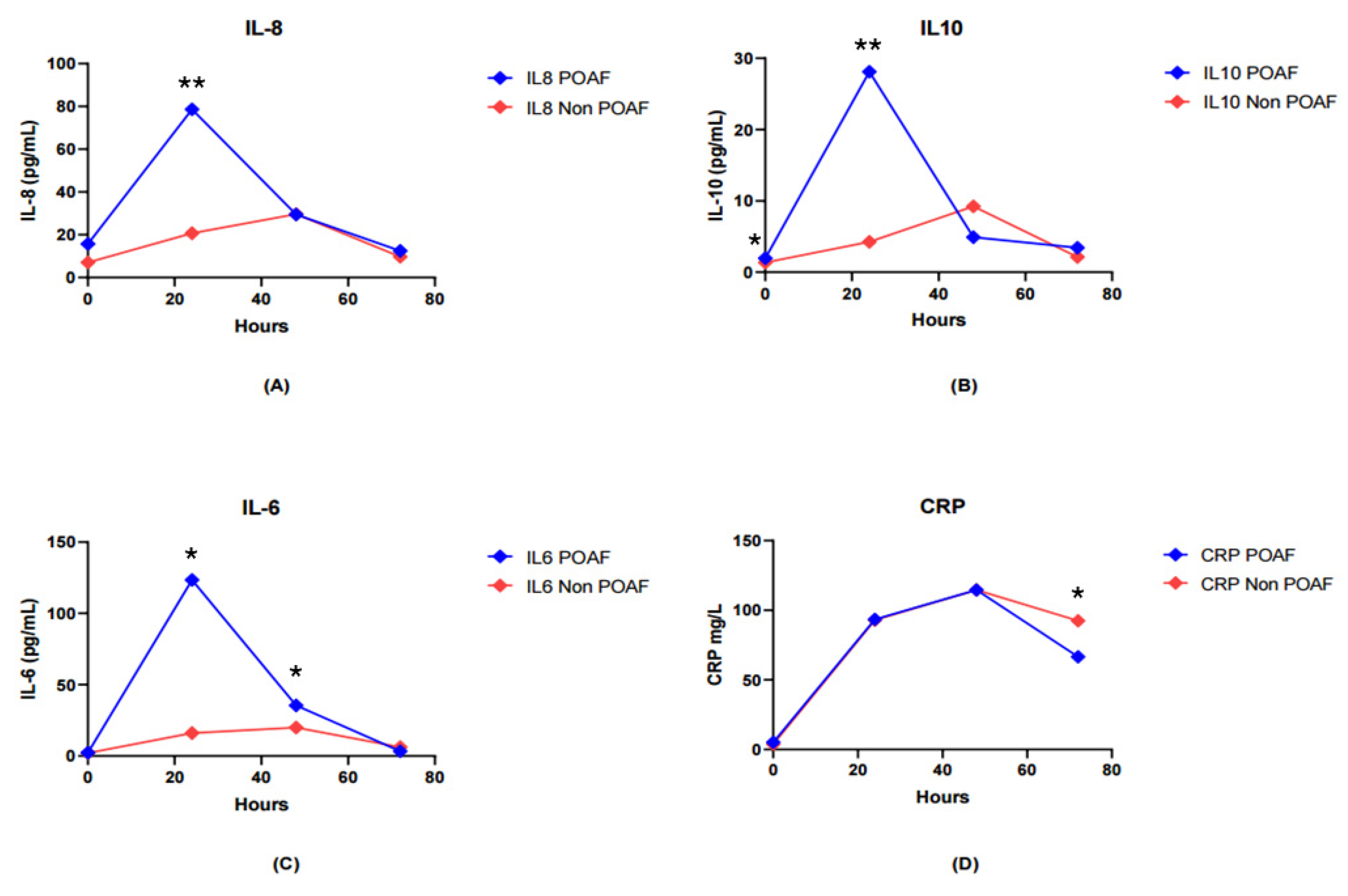
2.2. Inflammatory Biomarkers: Cytokines and C-Reactive Protein
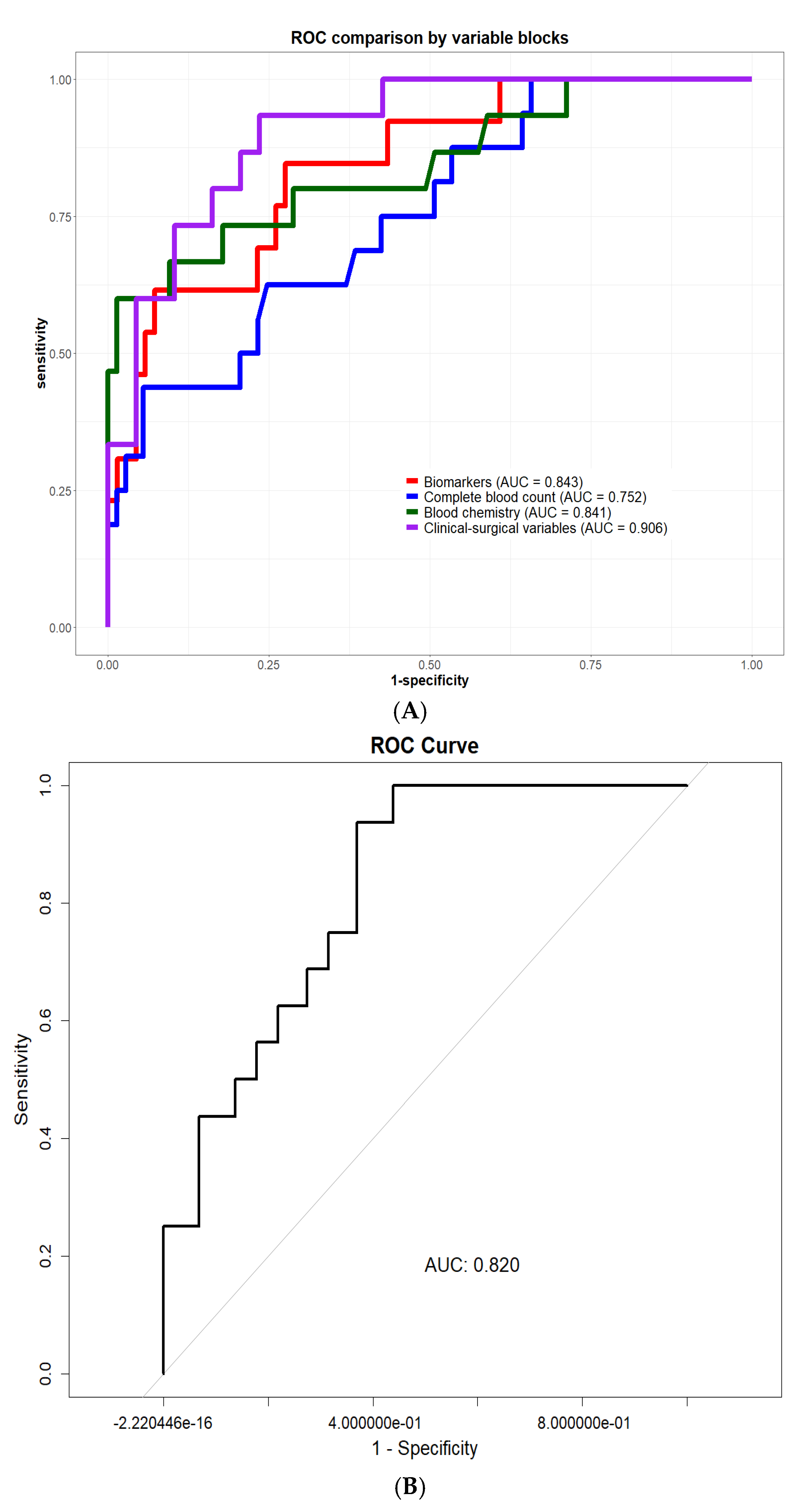
2.3. Clinical-Surgical and Biomarker-Based Predictive Models for POAF
3. Discussion
3.1. Contribution of Comorbidities to POAF Development
3.2. Impact of Clinical Risk Scores and Postoperative Hemodynamic Status
3.3. Serum Inflammatory Biomarkers Across Perioperative Time-Points of Cardiac Surgery with Cardiopulmonary Bypass
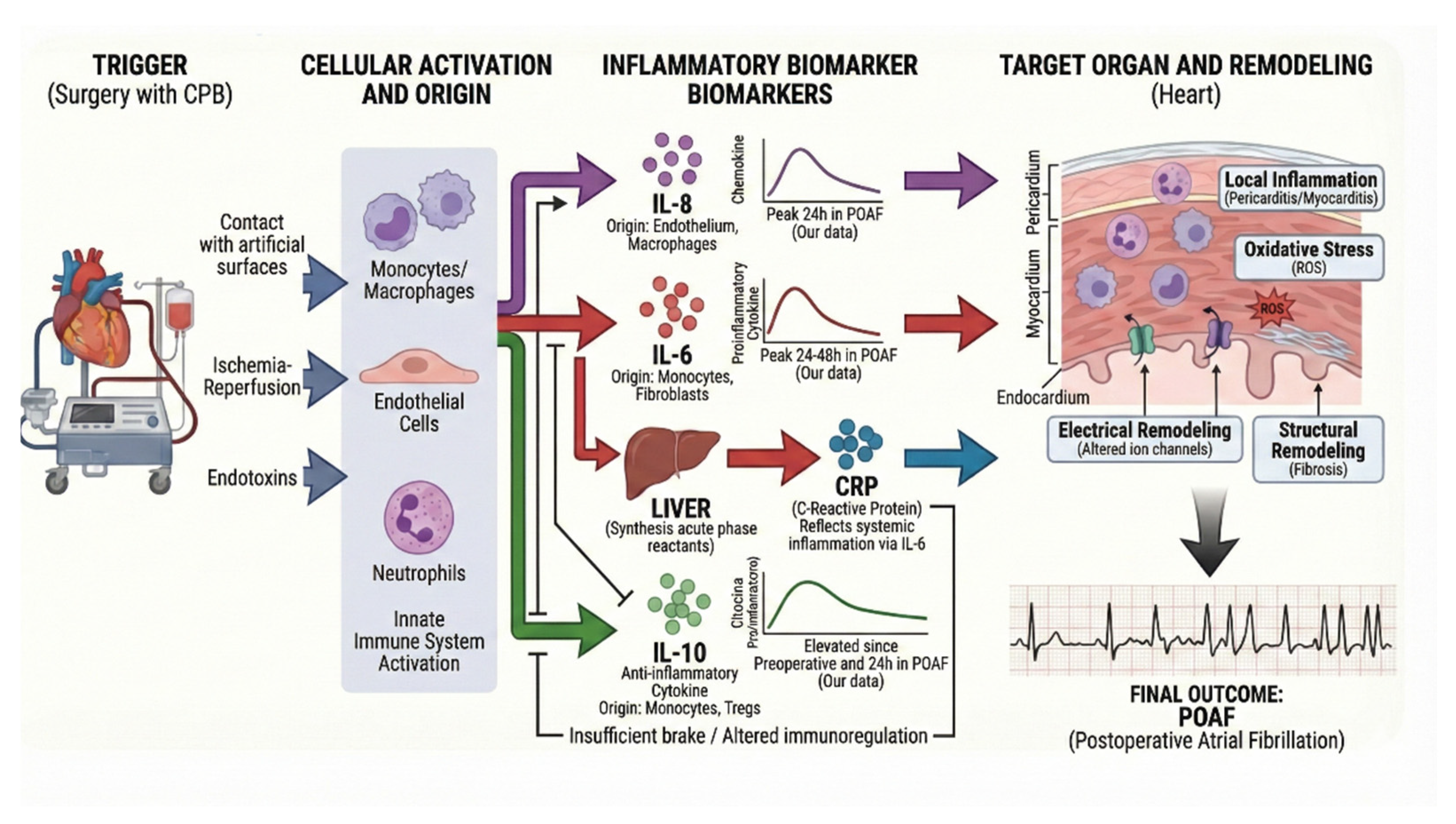
3.4. Systemic Inflammatory Response and Inflammatory Biomarkers Profile in the Physiopathology of POAF
3.5. Integrative Synthesis: Proposal of a Multidimensional Risk Model for POAF Development
3.5. Clinical Application of the Predictive Model
4. Materials and Methods
4.1. Study Design and Population
4.2. Inclusion and Exclusion Criteria
4.3. Clinical, Laboratory, and Surgical Data Collection
4.4. Blood Sampling and Biomarker Analysis
4.5. Outcome Definition and Follow-Up
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACT | Activated Clotting Time |
| AMI | Acute Myocardial Infarction |
| AUC | Area Under the Curve |
| BMI | Body Mass Index |
| CABG | Coronary Artery Bypass Grafting |
| CCI | Chronic Cardiac Insufficiency |
| CCS | Chronic Coronary Syndrome |
| CPB | Cardiopulmonary Bypass |
| CRP | C-Reactive Protein |
| CVDs | Cardiovascular Diseases |
| EuroSCORE II | European System for Cardiac Operative Risk Evaluation II |
| IL-6 | Interleukin-6 |
| IL-8 | Interleukin-8 |
| IL-10 | Interleukin-10 |
| IQR | Interquartile Range |
| LVEF | Left Ventricular Ejection Fraction |
| POAF | Postoperative Atrial Fibrillation |
| ROC | Receiver Operating Characteristic |
| SAH | Systemic Arterial Hypertension |
| STS score | Society of Thoracic Surgeons score |
| T2DM | Type 2 Diabetes Mellitus |
References
- Tsao, CW; Aday, AW; Almarzooq, ZI; Alonso, A; Beaton, AZ; Bittencourt, MS; et al. Heart Disease and Stroke Statistics—2022 Update: A Report From the American Heart Association. Circulation 2022, 145(8). [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Arias, AG; Bobadilla-Serrano, ME; Dimas-Altamirano, B; Gómez-Ortega, M; González-González, G. Enfermedad cardiovascular: primera causa de morbilidad en un hospital de tercer nivel. Rev Mex Cardiol 2016, 27(S3), 98–102. [Google Scholar]
- Filardo, G; Damiano, RJ; Ailawadi, G; Thourani, VH; Pollock, BD; Sass, DM; et al. Epidemiology of new-onset atrial fibrillation following coronary artery bypass graft surgery. Heart;PubMed 2018, 104(12), 985–92. [Google Scholar] [CrossRef] [PubMed]
- Fleet, H; Pilcher, D; Bellomo, R; Coulson, TG. Predicting atrial fibrillation after cardiac surgery: a scoping review of associated factors and systematic review of existing prediction models. Perfusion 2023, 38(1), 92–108. [Google Scholar] [CrossRef]
- Gaudino, M; Di Franco, A; Rong, LQ; Piccini, J; Mack, M. Postoperative atrial fibrillation: from mechanisms to treatment. In Eur Heart J; PubMed Central, 21 Mar 2023; Volume 44, 12, pp. 1020–39. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Shah, SM; Shah, J; Lakey, SM; Garg, P; Ripley, DP. Pathophysiology, emerging techniques for the assessment and novel treatment of aortic stenosis. Open Heart;PubMed 2023, 10(1). [Google Scholar] [CrossRef]
- Muehlschlegel, JD; Burrage, PS; Ngai, JY; Prutkin, JM; Huang, CC; Xu, X; et al. Society of Cardiovascular Anesthesiologists/European Association of Cardiothoracic Anaesthetists Practice Advisory for the Management of Perioperative Atrial Fibrillation in Patients Undergoing Cardiac Surgery. Anesth Analg. PubMed. 2019, 128(1), 33–42. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y; Li, D; Yu, D; Liang, Q; Chen, G; Li, F; et al. Comprehensive Analysis of Hemophilia A (CAHEA): Towards Full Characterization of the F8 Gene Variants by Long-Read Sequencing. Thromb Haemost PubMed. 2023. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, Y; Peighambari, MM; Naghshbandi, S; Samiei, N; Ghavidel, AA; Dehghani, MR; et al. Postoperative Atrial Fibrillation Following Cardiac Surgery: From Pathogenesis to Potential Therapies. Am J Cardiovasc Drugs PubMed. 2020, 20(1), 19–49. [Google Scholar] [CrossRef] [PubMed]
- Dobrev, D; Aguilar, M; Heijman, J; Guichard, JB; Nattel, S. Postoperative atrial fibrillation: mechanisms, manifestations and management. Nat Rev Cardiol PubMed. 2019, 16(7), 417–36. [Google Scholar] [CrossRef] [PubMed]
- Burrage, PS; Low, YH; Campbell, NG; O’Brien, B. New-Onset Atrial Fibrillation in Adult Patients After Cardiac Surgery. In Curr Anesthesiol Rep.; PubMed Central, Jun 2019; Volume 9, 2, pp. 174–93. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Giacinto, O; Satriano, U; Nenna, A; Spadaccio, C; Lusini, M; Mastroianni, C; et al. Inflammatory Response and Endothelial Dysfunction Following Cardiopulmonary Bypass: Pathophysiology and Pharmacological Targets. Recent Pat Inflamm Allergy Drug Discov. PubMed. 2019, 13(2), 158–73. [Google Scholar] [CrossRef] [PubMed]
- Zakkar, M; Guida, G; Suleiman, MS; Angelini, GD. Cardiopulmonary bypass and oxidative stress. In Oxid Med Cell Longev.; PubMed Central, 2015; Volume 2015, p. 189863. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Packer, M. Characterization, Pathogenesis, and Clinical Implications of Inflammation-Related Atrial Myopathy as an Important Cause of Atrial Fibrillation. In J Am Heart Assoc.; PubMed Central, 7 Apr 2020; Volume 9, 7, p. e015343. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Akintoye, E; Sellke, F; Marchioli, R; Tavazzi, L; Mozaffarian, D. Factors associated with postoperative atrial fibrillation and other adverse events after cardiac surgery. The Journal of Thoracic and Cardiovascular Surgery 2018, 155(1), 242–251.e10. [Google Scholar] [CrossRef] [PubMed]
- Boriani, G; Valenti, AC; Vitolo, M. Biomarkers in atrial fibrillation: a constant search for simplicity, practicality, and cost-effectiveness. Kardiol Pol. 2021, 79(3), 243–5. [Google Scholar] [CrossRef] [PubMed]
- Wu, ZK; Laurikka, J; Vikman, S; Nieminen, R; Moilanen, E; Tarkka, MR. High postoperative interleukin-8 levels related to atrial fibrillation in patients undergoing coronary artery bypass surgery. World J Surg;PubMed 2008, 32(12), 2643–9. [Google Scholar] [CrossRef] [PubMed]
- Anatoĺevna, RO; Veniaminovich, FO; Mikhaylovich, KS. Predictors of new-onset atrial fibrillation in elderly patients with coronary artery disease after coronary artery bypass graft. J Geriatr Cardiol. 2016, 13(5), 444–9. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Xu, S; Zhang, J; Liu, J; Ye, J; Xu, Y; Wang, Z; et al. The role of interleukin-10 family members in cardiovascular diseases. International Immunopharmacology 2021, 94, 107475. [Google Scholar] [CrossRef]
- Sproston, NR; Ashworth, JJ. Role of C-Reactive Protein at Sites of Inflammation and Infection. Front Immunol 2018, 9. [Google Scholar] [CrossRef]
- Olesen, OJ; Vinding, NE; Østergaard, L; Butt, JH; Gislason, GH; Torp-Pedersen, C; et al. C-reactive protein after coronary artery bypass graft surgery and its relationship with postoperative atrial fibrillation. EP Europace 2020, 22(8), 1182–8. [Google Scholar] [CrossRef]
- Canbaz, S; Erbas, H; Huseyin, S; Duran, E. The role of inflammation in atrial fibrillation following open heart surgery. J Int Med Res. PubMed. 2008, 36(5), 1070–6. [Google Scholar] [CrossRef] [PubMed]
- Kota, R.; Gemelli, M.; Dimagli, A.; Suleiman, M.-S.; Moscarelli, M.; Dong, T.; Angelini, G.; Fudulu, D.P. Patterns of Cytokine Release and Association with New Onset of Post-Cardiac Surgery Atrial Fibrillation. Front. Surg. Correction in Front. Surg. 2024, 11, 1320769. 2023, 10, 1205396. [Google Scholar] [CrossRef]
- Bowdish, ME; Bagiella, E; Giustino, G; Atluri, P; Alexander, JH; Thourani, VH; et al. Prospective Study of Risk Factors for Postoperative Atrial Fibrillation After Cardiac Surgery. In J Surg Res.;PubMed; PubMed Central, Feb 2024; Volume 294, pp. 262–8. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Higa; Maesato; Ishigaki; Suenari, Et. A. Diabetes and Endocrine Disorders (Hyperthyroidism/Hypothyroidism) as Risk Factors for Atrial Fibrillation. Cardiac Electrophysiology Clinics 2021. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y; Dedkov, EI; Teplitsky, D; Weltman, NY; Pol, CJ; Rajagopalan, V; et al. Both hypothyroidism and hyperthyroidism increase atrial fibrillation inducibility in rats. Circ Arrhythm Electrophysiol 2013, 6(5), 952–9. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dell’Aquila, M; Rossi, CS; Caldonazo, T; Cancelli, G; Harik, L; Soletti, GJ; et al. Subclinical hypothyroidism and clinical outcomes after cardiac surgery: A systematic review and meta-analysis. JTCVS Open 2024, 18, 64–79. [Google Scholar] [CrossRef]
- Nso, N; Bookani, KR; Metzl, M; Radparvar, F. Role of inflammation in atrial fibrillation: A comprehensive review of current knowledge. J Arrhythm. 2021, 37(1), 1–10. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Perić, V; Golubović, M; Stošić, M; Milić, D; Lazović, L; Stojanović, D; et al. Echocardiographic Predictors of Postoperative Atrial Fibrillation After Cardiac Surgery: Assessing Atrial Mechanics for Risk Stratification. In J Cardiovasc Dev Dis.; PubMed Central, 17 Apr 2025; Volume 12, 4. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Karakasis, P; Pamporis, K; Theofilis, P; Milaras, N; Vlachakis, PK; Grigoriou, K; et al. Inflammasome Signaling in Cardiac Arrhythmias: Linking Inflammation, Fibrosis, and Electrical Remodeling. International Journal of Molecular Sciences 2025, 26(13), 5954. [Google Scholar] [CrossRef]
- Spicer, S; Hasheminia, A; Kandi, S; Abu-Omar, Y; Fernández, ÁL; El-Diasty, M. Cardiac and pericardial inflammatory changes and post-cardiac surgery atrial fibrillation. Cytokine & Growth Factor Reviews 2025, 85, 158–64. [Google Scholar] [CrossRef]
- Weymann, A; Popov, AF; Sabashnikov, A; Ali-Hasan-Al-Saegh, S; Ryazanov, M; Tse, G; et al. Baseline and postoperative levels of C-reactive protein and interleukins as inflammatory predictors of atrial fibrillation following cardiac surgery: a systematic review and meta-analysis. Kardiol Pol. 2018, 76(2), 440–51. [Google Scholar] [CrossRef] [PubMed]
- Huang, M; Huiskes, FG; de Groot, NMS; Brundel, BJJM. The Role of Immune Cells Driving Electropathology and Atrial Fibrillation. In Cells; PubMed Central, 8 Feb 2024; Volume 13, 4. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Fatehi Hassanabad, A; Schoettler, FI; Kent, WDT; Adams, CA; Holloway, DD; Ali, IS; et al. Cardiac surgery elicits pericardial inflammatory responses that are distinct compared with postcardiopulmonary bypass systemic inflammation. JTCVS Open;Precision Therapy in Lung Cancer 2023, 16, 389–400. [Google Scholar] [CrossRef]
- Yan, Y; Yu, Z; Lu, J; Jin, P; Tang, Z; Hu, Y. Predictive values profiling of interleukin-2, interleukin-8, tumor necrosis factor-α, procalcitonin, and C-reactive protein in critical gastrointestinal cancer patients. J Gastrointest Oncol.;PubMed 2021, 12(4), 1398–406. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Saetang, M; Wasinwong, W; Oofuvong, M; Tanasansutthiporn, J; Rattanapittayaporn, L; Petsakul, S; et al. Effect of Combined Vitamin C and Thiamine Therapy on Myocardial and Inflammatory Markers in Cardiac Surgery: A Randomized Controlled Clinical Trial. Nutrients 2025, 17(6), 1006. [Google Scholar] [CrossRef] [PubMed Central]
- Hindricks, G; Potpara, T; Dagres, N; Arbelo, E; Bax, JJ; Blomström-Lundqvist, C; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur Heart J PubMed. 2021, 42(5), 373–498. [Google Scholar] [CrossRef] [PubMed]
- Steyerberg, EW; Harrell, FE. Prediction models need appropriate internal, internal-external, and external validation. J Clin Epidemiol. 2016, 69, 245–7. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Collins, GS; Reitsma, JB; Altman, DG; Moons, KGM. Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis (TRIPOD): The TRIPOD Statement. Ann Intern Med. 2015, 162(1), 55–63. [Google Scholar] [CrossRef] [PubMed]
| Variable | POAF Group (n=16) |
Non POAF Group (n=73) |
p-Value |
|---|---|---|---|
| Anthropometric data | |||
| Age (years)b | 67 (50-76) | 65 (50-78) | 0.684 |
| Malea | 10 (62.5%) | 51 (69.9) | 0.574 |
| Femalea | 6 (37.5) | 22 (30.1) | |
| BMI (kg/m2) b | 26.8 (19.7-31.9) | 27.1 (19.71-39.0) | 0.589 |
| Comorbidities | |||
| Previous myocardial infarctiona | 4 (25) | 20 (27.4) | 0.872 |
| Type 2 diabetes mellitusa | 6 (37.5) | 37 (50.7) | 0.339 |
| Systemic arterial hypertensiona | 13 (81.3) | 52 (71.3) | 0.474 |
| Obesitya | 4 (25) | 20 (27.4) | 0.847 |
| Dyslipidemiaa | 3 (18.75) | 24 (32.9) | 0.266 |
| Hypothyroidism a | 5 (31.3) | 4(5.5) | 0.0018 |
| Variable | POAF Group (n=16) |
Non POAF Group (n=73) |
p-Value | |
|---|---|---|---|---|
| Surgical data | ||||
| Cardiopulmonary bypass time (min) b | 123 (73-217) | 120 (64-255) | 0.712 | |
| Aortic cross-clamp time (min)b | 91 (60-193) | 92 (24-202) | 0.634 | |
| Initial ACT (s)b | 111.50 (79-143) | 118 (76-171) | 0.352 | |
| Final ACT (s)b | 115 (103-138) | 116 (84-167) | 0.881 | |
| Number of graftsa | 0 | 9 (56.3) | 31 (42.5) | - |
| 1 | 3 (18.8) | 6 (8.2) | ||
| 2 | - | 8 (11.0) | ||
| 3 | 4 (25) | 15 (20.5) | ||
| 4 | - | 12 (16.4) | ||
| 5 | - | 1 (1.4) | ||
| Number of repaired valvesa | 0 | 4 (25%) | 33 (45.2%) | - |
| 1 | 10 (62.5%) | 32 (43.8%) | ||
| 2 | 1 (6.3%) | 6 (8.25%) | ||
| 3 | 1 (6.3%) | 1 (2.7%) | ||
| Pre-CPB blood loss (mL)b | 160 (15-300) | 200 (20-400) | 0.286 | |
| Intra-CPB blood loss (mL)b | 132.5 (0-400) | 100 (0-640) | 0.430 | |
| Post-CPB blood loss (mL)b | 325 (165-615) | 300 (o-800) | 0.348 | |
| EuroSCORE II (%)b | 1.36 (0.50-8.84) | 1.09 (0.50-11.37) | 0.168 | |
| Left ventricular ejection fraction (LVEF, %)b | 53 (20-73) | 55 (24-86) | 0.608 | |
| STS score (%)b | 1.70 (0.19-16) | 0.94 (0.22-6.61) | 0.032* | |
| Blood count and biochemical parameters | ||||
| Hemoglobin (g/dL), preop b | 13.75 (10.20-16.6) | 14.20 (8.80-18.80) | 0.175 | |
| Hemoglobin (g/dL), 24 h b | 9.80 (6.20-12.10) | 10.8 (5.60-15) | 0.007** | |
| Hematocrit (%), preop b | 39.35 (31-50) | 41 (26.8-58.1) | 0.148 | |
| Hematocrit (%), 24 h b | 30 (21.1-42) | 32.75 (24.3-51.4) | 0.061 | |
| Platelets (×103/µL), preop b | 202 (136-352) | 210 (37-471) | 0.560 | |
| Platelets (×103/µL), 24 hc | 182 (124-301) | 181 (75-345) | 0.638 | |
| Leukocytes (×103/µL), preop b | 6.5 (3.50-9.25) | 6.8 (2.5-17.20) | 0.361 | |
| Leukocytes (×103/µL), 24 hb | 11.6 (7.87-23.70) | 12.30 (1.60-23) | 0.795 | |
| Glucose (mg/dL), preop b | 96 (75-156) | 101 (75-171) | 0.426 | |
| Glucose (mg/dL), 24 hb | 176 (85-589) | 148 (75-324) | 0.478 | |
| Urea (mg/dL), preop b | 29 (20-65) | 31 (17-66) | 0.915 | |
| Urea (mg/dL), 24 hb | 38 (21.6-95.9) | 34 (16.8-93) | 0.566 | |
| Creatinine (mg/dL), preop b | 0.85 (0.50-1.36) | 0.80 (0.51-1.40) | 0.940 | |
| Creatinine (mg/dL), 24 hb | 0.85 (0.60-1.33) | 0.9 (0.51-2.30) | 0.696 | |
| Variable | POAF Group (n=16) |
Non POAF Group (n=73) |
p-Value |
|---|---|---|---|
| Proinflammatory interleukins | |||
| Preoperative IL-8 (pg/mL)a | 6.73 (3.6-143.3) | 5.35 (0.5-34.3) | 0.205 |
| Postoperative IL-8 at 24 h (pg/mL)a | 21.85 (5.3-512.1) | 11.02 (1.8-127.3) | 0.010** |
| Postoperative IL-8 at 48 h (pg/mL)a | 10.09 (5.0-169.3) | 9.2 (0.36-539.2) | 0.171 |
| IL-8 at discharge (pg/mL)a | 11.37 (1.1-39.6) | 7.34 (0-34) | 0.338 |
| Preoperative IL-10 (pg/mL)a | 1.10 (0-8.19) | 0.43 (0-8.95) | 0.029* |
| Postoperative IL-10 at 24 h (pg/mL)a | 7.33 (0.19-194.0) | 2.13 (0-67.39) | 0.003** |
| Postoperative IL-10 at 48 h (pg/mL)a | 1.52 (0-21.97) | 1.56 (0-328.7) | 0.389 |
| IL-10 at discharge (pg/mL) a | 1.2 (0-14.4) | 0.65 (0-66.92) | 0.109 |
| Preoperative IL-6 (pg/mL)a | 0.98 (0-16.73) | 0.35 (0-16.72) | 0.174 |
| Postoperative IL-6 at 24 h (pg/mL)a | 12.44 (1.44-1102) | 4.97 (0-158.3) | 0.012* |
| Postoperative IL-6 at 48 h (pg/mL)a | 13.56 (0.11-131.5) | 2.42 (0-202.5) | 0.038* |
| IL-6 at discharge (pg/mL)a | 1.64 (0-8.46) | 0.90 (0-81.0) | 0.657 |
| Preoperative CRP (mg/L)a | 1.75 (0.75-50) | 1.92 (0.68-31.94) | 0.894 |
| Postoperative CRP at 24 h (mg/L)a | 10.8 (5.65-123.51) | 105.4 (1.88-125) | 0.957 |
| Postoperative CRP at 48 h (mg/L) a | 124 (37.15-127.6) | 122.8 (1.85-128.7) | 0.761 |
| CRP at discharge (mg/L) a | 67.90 (6.99-126.1) | 106.6 (2.27-128.91) | 0.050* |
| Variable | Odds Ratio (OR) | 95% Confidence Interval | p-Value |
|---|---|---|---|
| STS score (%) | 1.32 | 1.05–1.72 | 0.021 |
| IL-6 at 24 h (pg/mL) | 1.08 | 1.01–1.15 | 0.032 |
| Magnesium at 24 h (mEq/L) | 0.64 | 0.42–0.95 | 0.028 |
| Model | Apparent AUC | Mean Optimism | Optimism-Corrected AUC |
|---|---|---|---|
| Multivariable POAF prediction model | 0.82 | 0.046 | 0.77 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




